 Found 182 hits with Last Name = 'munroe' and Initial = 'j'
Found 182 hits with Last Name = 'munroe' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Genome polyprotein
(Human rhinovirus B) | BDBM50137724
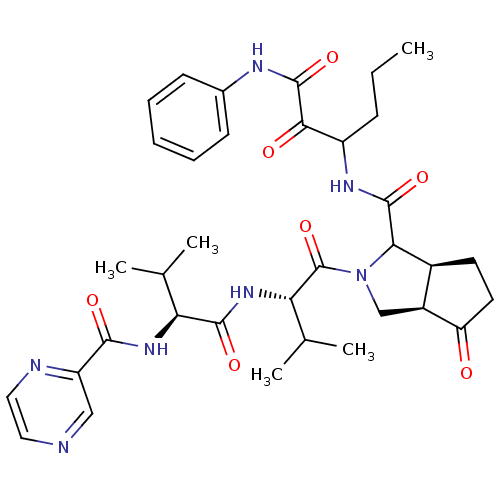
((3aR,5S)-2-((S)-3-Methyl-2-{(S)-3-methyl-2-[(pyraz...)Show SMILES CCCC(NC(=O)C1[C@H]2CCC(=O)[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)C)C(=O)C(=O)Nc1ccccc1 Show InChI InChI=1S/C35H45N7O7/c1-6-10-24(30(44)34(48)38-21-11-8-7-9-12-21)39-33(47)29-22-13-14-26(43)23(22)18-42(29)35(49)28(20(4)5)41-32(46)27(19(2)3)40-31(45)25-17-36-15-16-37-25/h7-9,11-12,15-17,19-20,22-24,27-29H,6,10,13-14,18H2,1-5H3,(H,38,48)(H,39,47)(H,40,45)(H,41,46)/t22-,23-,24?,27-,28-,29?/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards Protease using PNA assay in rats |
Bioorg Med Chem Lett 14: 251-6 (2003)
BindingDB Entry DOI: 10.7270/Q24B30R9 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50152750
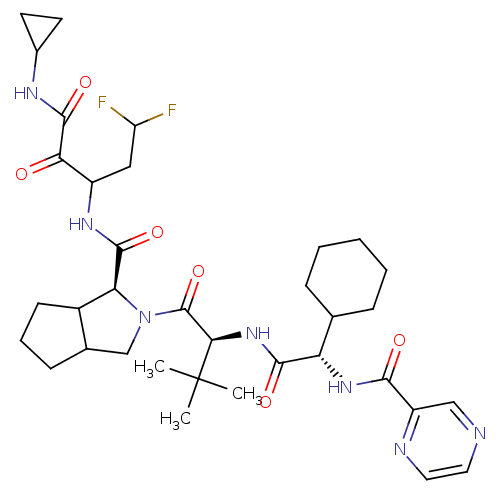
(2-((S)-2-{(S)-2-Cyclohexyl-2-[(pyrazine-2-carbonyl...)Show SMILES CC(C)(C)[C@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C1CCCCC1)C(=O)N1CC2CCCC2[C@H]1C(=O)NC(CC(F)F)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C35H49F2N7O6/c1-35(2,3)29(43-31(47)26(19-8-5-4-6-9-19)42-30(46)24-17-38-14-15-39-24)34(50)44-18-20-10-7-11-22(20)27(44)32(48)41-23(16-25(36)37)28(45)33(49)40-21-12-13-21/h14-15,17,19-23,25-27,29H,4-13,16,18H2,1-3H3,(H,40,49)(H,41,48)(H,42,46)(H,43,47)/t20?,22?,23?,26-,27-,29+/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Inhibition of HCV NS3 protease in the pNA based inhibition assay |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50137721

(2-(3-{[(1S,5S,6R)-2-((S)-3-Methyl-2-{(S)-3-methyl-...)Show SMILES CCCC(NC(=O)[C@@H]1[C@H]2CCC[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)C)C(=O)C(=O)N[C@@H](Cc1ccccc1)C(O)=O Show InChI InChI=1S/C38H51N7O8/c1-6-11-26(32(46)36(50)42-27(38(52)53)18-23-12-8-7-9-13-23)41-35(49)31-25-15-10-14-24(25)20-45(31)37(51)30(22(4)5)44-34(48)29(21(2)3)43-33(47)28-19-39-16-17-40-28/h7-9,12-13,16-17,19,21-22,24-27,29-31H,6,10-11,14-15,18,20H2,1-5H3,(H,41,49)(H,42,50)(H,43,47)(H,44,48)(H,52,53)/t24-,25-,26?,27-,29-,30-,31-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 68 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards Protease using PNA assay in rats |
Bioorg Med Chem Lett 14: 251-6 (2003)
BindingDB Entry DOI: 10.7270/Q24B30R9 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50152754
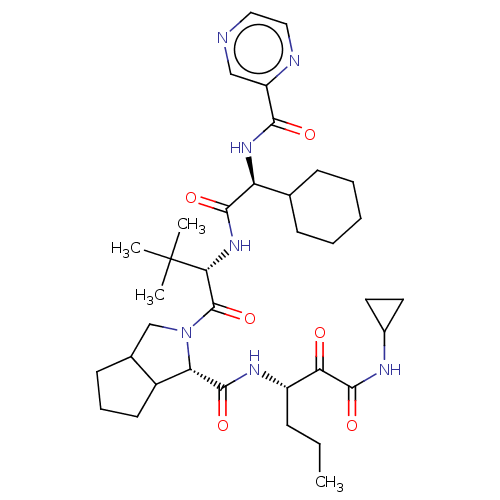
(2-((S)-2-{(S)-2-Cyclohexyl-2-[(pyrazine-2-carbonyl...)Show SMILES CCC[C@H](NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C1CCCCC1)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C36H53N7O6/c1-5-10-25(29(44)34(48)39-23-15-16-23)40-33(47)28-24-14-9-13-22(24)20-43(28)35(49)30(36(2,3)4)42-32(46)27(21-11-7-6-8-12-21)41-31(45)26-19-37-17-18-38-26/h17-19,21-25,27-28,30H,5-16,20H2,1-4H3,(H,39,48)(H,40,47)(H,41,45)(H,42,46)/t22?,24?,25?,27-,28-,30+/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Inhibition of HCV NS3 protease in the pNA based inhibition assay |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50137733
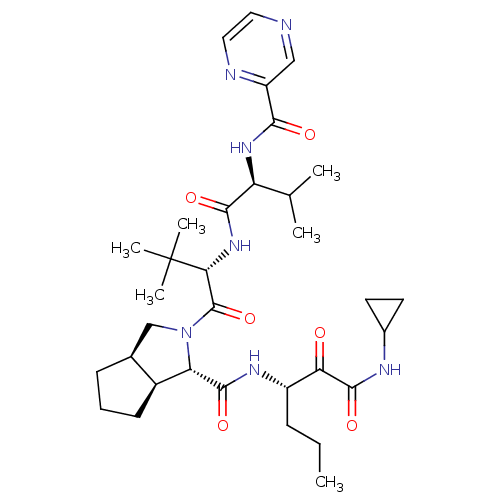
((1S,5S,6R)-2-((S)-3,3-Dimethyl-2-{(S)-3-methyl-2-[...)Show SMILES CCC[C@H](NC(=O)[C@@H]1[C@H]2CCC[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C33H49N7O6/c1-7-9-22(26(41)31(45)36-20-12-13-20)37-30(44)25-21-11-8-10-19(21)17-40(25)32(46)27(33(4,5)6)39-29(43)24(18(2)3)38-28(42)23-16-34-14-15-35-23/h14-16,18-22,24-25,27H,7-13,17H2,1-6H3,(H,36,45)(H,37,44)(H,38,42)(H,39,43)/t19-,21-,22-,24-,25-,27+/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Inhibition of HCV NS3 protease in the pNA based inhibition assay |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50152753

((S)-2-((S)-3,3-Dimethyl-2-{(S)-3-methyl-2-[(pyrazi...)Show SMILES CCC[C@H](NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)[C@@H](C)CC)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C34H51N7O6/c1-7-10-23(27(42)32(46)37-21-13-14-21)38-31(45)26-22-12-9-11-20(22)18-41(26)33(47)28(34(4,5)6)40-30(44)25(19(3)8-2)39-29(43)24-17-35-15-16-36-24/h15-17,19-23,25-26,28H,7-14,18H2,1-6H3,(H,37,46)(H,38,45)(H,39,43)(H,40,44)/t19?,20?,22?,23?,25-,26-,28+/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Inhibition of HCV NS3 protease in the pNA based inhibition assay |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50152748
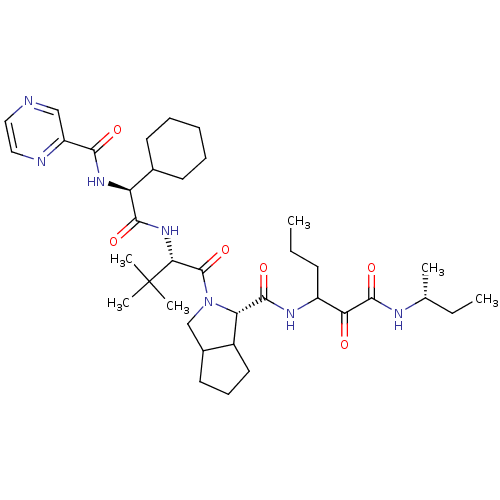
((S)-2-((S)-2-{(S)-2-Cyclohexyl-2-[(pyrazine-2-carb...)Show SMILES CCCC(NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C1CCCCC1)C(C)(C)C)C(=O)C(=O)N[C@H](C)CC Show InChI InChI=1S/C37H57N7O6/c1-7-13-26(30(45)35(49)40-22(3)8-2)41-34(48)29-25-17-12-16-24(25)21-44(29)36(50)31(37(4,5)6)43-33(47)28(23-14-10-9-11-15-23)42-32(46)27-20-38-18-19-39-27/h18-20,22-26,28-29,31H,7-17,21H2,1-6H3,(H,40,49)(H,41,48)(H,42,46)(H,43,47)/t22-,24?,25?,26?,28+,29+,31-/m1/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Inhibition of HCV NS3 protease in the pNA based inhibition assay |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50137713
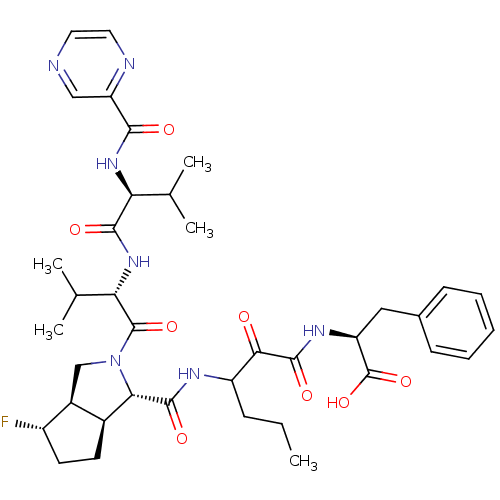
((S)-2-((S)-3-{[(1S,5S,6R)-4-Fluoro-2-((S)-3-methyl...)Show SMILES CCCC(NC(=O)[C@@H]1[C@H]2CC[C@H](F)[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)C)C(=O)C(=O)N[C@@H](Cc1ccccc1)C(O)=O Show InChI InChI=1S/C38H50FN7O8/c1-6-10-26(32(47)36(51)43-27(38(53)54)17-22-11-8-7-9-12-22)42-35(50)31-23-13-14-25(39)24(23)19-46(31)37(52)30(21(4)5)45-34(49)29(20(2)3)44-33(48)28-18-40-15-16-41-28/h7-9,11-12,15-16,18,20-21,23-27,29-31H,6,10,13-14,17,19H2,1-5H3,(H,42,50)(H,43,51)(H,44,48)(H,45,49)(H,53,54)/t23-,24-,25-,26?,27-,29-,30-,31-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 82 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards Protease using PNA assay in rats |
Bioorg Med Chem Lett 14: 251-6 (2003)
BindingDB Entry DOI: 10.7270/Q24B30R9 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50137718
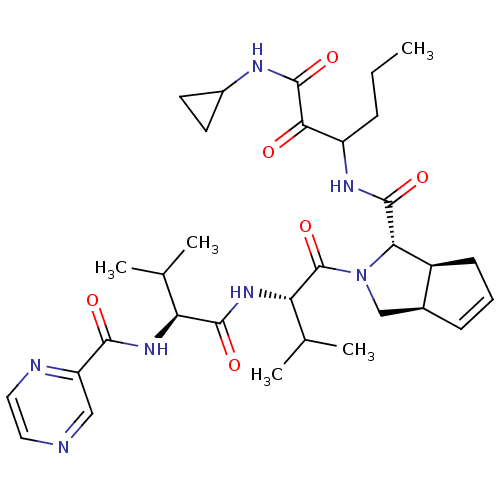
((1S,5S,6R)-2-((S)-3-Methyl-2-{(S)-3-methyl-2-[(pyr...)Show SMILES CCCC(NC(=O)[C@@H]1[C@H]2CC=C[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)C)C(=O)C(=O)NC1CC1 |c:10| Show InChI InChI=1S/C32H45N7O6/c1-6-8-22(27(40)31(44)35-20-11-12-20)36-30(43)26-21-10-7-9-19(21)16-39(26)32(45)25(18(4)5)38-29(42)24(17(2)3)37-28(41)23-15-33-13-14-34-23/h7,9,13-15,17-22,24-26H,6,8,10-12,16H2,1-5H3,(H,35,44)(H,36,43)(H,37,41)(H,38,42)/t19-,21-,22?,24-,25-,26-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards Protease using PNA assay in rats |
Bioorg Med Chem Lett 14: 251-6 (2003)
BindingDB Entry DOI: 10.7270/Q24B30R9 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50152751

(2-((S)-2-{(S)-2-Cyclohexyl-2-[((R)-pyrazine-2-carb...)Show SMILES CCCC(NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C1CCCCC1)C(C)(C)C)C(=O)C(=O)N[C@H](C)c1ccccc1 Show InChI InChI=1S/C41H57N7O6/c1-6-14-30(34(49)39(53)44-25(2)26-15-9-7-10-16-26)45-38(52)33-29-20-13-19-28(29)24-48(33)40(54)35(41(3,4)5)47-37(51)32(27-17-11-8-12-18-27)46-36(50)31-23-42-21-22-43-31/h7,9-10,15-16,21-23,25,27-30,32-33,35H,6,8,11-14,17-20,24H2,1-5H3,(H,44,53)(H,45,52)(H,46,50)(H,47,51)/t25-,28?,29?,30?,32+,33+,35-/m1/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Inhibition of HCV NS3 protease in the pNA based inhibition assay |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50137720

((1S,3aR,6aS)-2-((S)-3-Methyl-2-{(S)-3-methyl-2-[(p...)Show SMILES CCCC(NC(=O)[C@@H]1[C@H]2CCC[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C32H47N7O6/c1-6-8-22(27(40)31(44)35-20-11-12-20)36-30(43)26-21-10-7-9-19(21)16-39(26)32(45)25(18(4)5)38-29(42)24(17(2)3)37-28(41)23-15-33-13-14-34-23/h13-15,17-22,24-26H,6-12,16H2,1-5H3,(H,35,44)(H,36,43)(H,37,41)(H,38,42)/t19-,21-,22?,24-,25-,26-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 123 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards Protease using PNA assay in rats |
Bioorg Med Chem Lett 14: 251-6 (2003)
BindingDB Entry DOI: 10.7270/Q24B30R9 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50152755
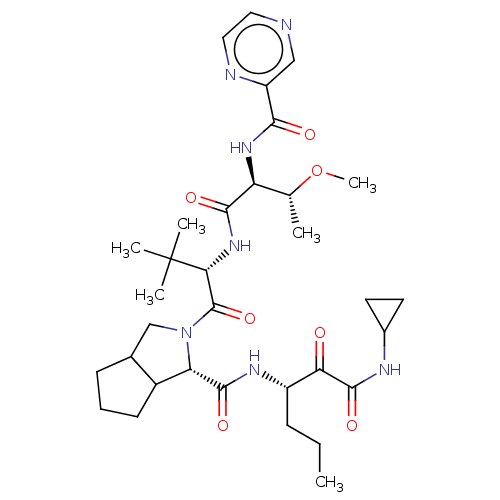
(2-((S)-2-{(S)-3-Methoxy-2-[(pyrazine-2-carbonyl)-a...)Show SMILES CCC[C@H](NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)[C@@H](C)OC)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C33H49N7O7/c1-7-9-22(26(41)31(45)36-20-12-13-20)37-30(44)25-21-11-8-10-19(21)17-40(25)32(46)27(33(3,4)5)39-29(43)24(18(2)47-6)38-28(42)23-16-34-14-15-35-23/h14-16,18-22,24-25,27H,7-13,17H2,1-6H3,(H,36,45)(H,37,44)(H,38,42)(H,39,43)/t18?,19?,21?,22?,24-,25-,27+/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Inhibition of HCV NS3 protease in the pNA based inhibition assay |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50152752
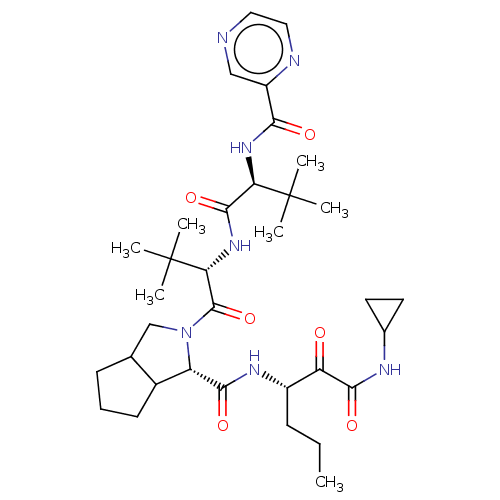
((S)-2-((S)-2-{(S)-3,3-Dimethyl-2-[(pyrazine-2-carb...)Show SMILES CCC[C@H](NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)(C)C)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C34H51N7O6/c1-8-10-22(25(42)30(45)37-20-13-14-20)38-29(44)24-21-12-9-11-19(21)18-41(24)32(47)27(34(5,6)7)40-31(46)26(33(2,3)4)39-28(43)23-17-35-15-16-36-23/h15-17,19-22,24,26-27H,8-14,18H2,1-7H3,(H,37,45)(H,38,44)(H,39,43)(H,40,46)/t19?,21?,22?,24-,26+,27+/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Inhibition of HCV NS3 protease in the pNA based inhibition assay |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50137722
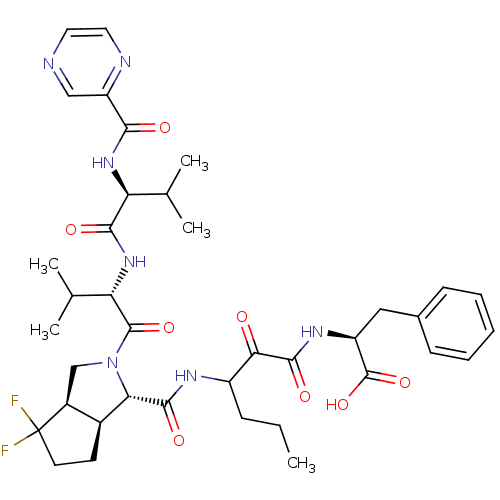
((S)-2-(3-{[(1S,5S,6R)-4,4-Difluoro-2-((S)-3-methyl...)Show SMILES CCCC(NC(=O)[C@@H]1[C@H]2CCC(F)(F)[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)C)C(=O)C(=O)N[C@@H](Cc1ccccc1)C(O)=O Show InChI InChI=1S/C38H49F2N7O8/c1-6-10-25(31(48)35(52)44-26(37(54)55)17-22-11-8-7-9-12-22)43-34(51)30-23-13-14-38(39,40)24(23)19-47(30)36(53)29(21(4)5)46-33(50)28(20(2)3)45-32(49)27-18-41-15-16-42-27/h7-9,11-12,15-16,18,20-21,23-26,28-30H,6,10,13-14,17,19H2,1-5H3,(H,43,51)(H,44,52)(H,45,49)(H,46,50)(H,54,55)/t23-,24-,25?,26-,28-,29-,30-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 176 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards Protease using PNA assay in rats |
Bioorg Med Chem Lett 14: 251-6 (2003)
BindingDB Entry DOI: 10.7270/Q24B30R9 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50152758
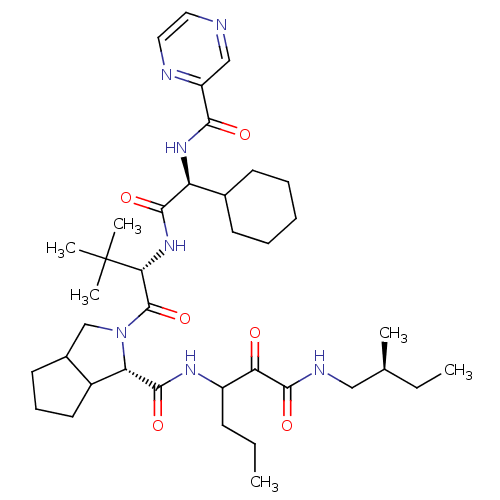
((S)-2-((S)-2-{(S)-2-Cyclohexyl-2-[(pyrazine-2-carb...)Show SMILES CCCC(NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C1CCCCC1)C(C)(C)C)C(=O)C(=O)NC[C@@H](C)CC Show InChI InChI=1S/C38H59N7O6/c1-7-13-27(31(46)36(50)41-20-23(3)8-2)42-35(49)30-26-17-12-16-25(26)22-45(30)37(51)32(38(4,5)6)44-34(48)29(24-14-10-9-11-15-24)43-33(47)28-21-39-18-19-40-28/h18-19,21,23-27,29-30,32H,7-17,20,22H2,1-6H3,(H,41,50)(H,42,49)(H,43,47)(H,44,48)/t23-,25?,26?,27?,29-,30-,32+/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Inhibition of HCV NS3 protease in the pNA based inhibition assay |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50137717
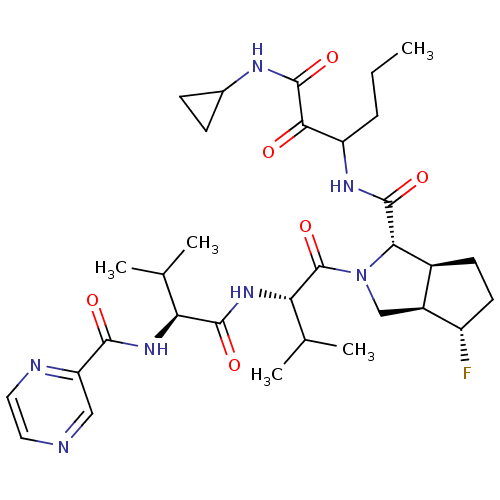
((1S,5S,6R)-4-Fluoro-2-((S)-3-methyl-2-{(S)-3-methy...)Show SMILES CCCC(NC(=O)[C@@H]1[C@H]2CC[C@H](F)[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C32H46FN7O6/c1-6-7-22(27(41)31(45)36-18-8-9-18)37-30(44)26-19-10-11-21(33)20(19)15-40(26)32(46)25(17(4)5)39-29(43)24(16(2)3)38-28(42)23-14-34-12-13-35-23/h12-14,16-22,24-26H,6-11,15H2,1-5H3,(H,36,45)(H,37,44)(H,38,42)(H,39,43)/t19-,20-,21-,22?,24-,25-,26-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards Protease using PNA assay in rats |
Bioorg Med Chem Lett 14: 251-6 (2003)
BindingDB Entry DOI: 10.7270/Q24B30R9 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50137727
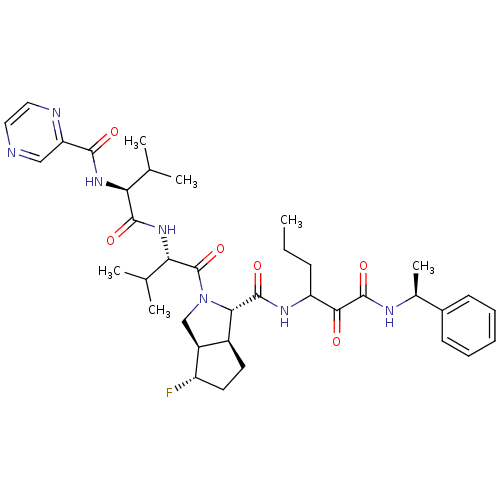
((1S,5S,6R)-4-Fluoro-2-((S)-3-methyl-2-{(S)-3-methy...)Show SMILES CCCC(NC(=O)[C@@H]1[C@H]2CC[C@H](F)[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)C)C(=O)C(=O)N[C@@H](C)c1ccccc1 Show InChI InChI=1S/C37H50FN7O6/c1-7-11-27(32(46)36(50)41-22(6)23-12-9-8-10-13-23)42-35(49)31-24-14-15-26(38)25(24)19-45(31)37(51)30(21(4)5)44-34(48)29(20(2)3)43-33(47)28-18-39-16-17-40-28/h8-10,12-13,16-18,20-22,24-27,29-31H,7,11,14-15,19H2,1-6H3,(H,41,50)(H,42,49)(H,43,47)(H,44,48)/t22-,24-,25-,26-,27?,29-,30-,31-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 315 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards Protease using PNA assay in rats |
Bioorg Med Chem Lett 14: 251-6 (2003)
BindingDB Entry DOI: 10.7270/Q24B30R9 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50137723
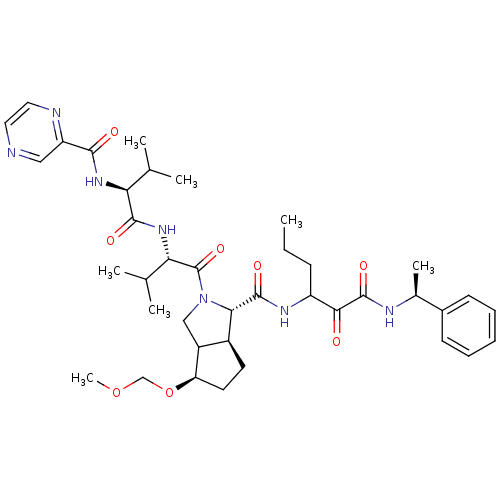
((1S,5S)-4-Methoxymethoxy-2-((S)-3-methyl-2-{(S)-3-...)Show SMILES CCCC(NC(=O)[C@@H]1[C@H]2CC[C@@H](OCOC)C2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)C)C(=O)C(=O)N[C@@H](C)c1ccccc1 Show InChI InChI=1S/C39H55N7O8/c1-8-12-28(34(47)38(51)42-24(6)25-13-10-9-11-14-25)43-37(50)33-26-15-16-30(54-21-53-7)27(26)20-46(33)39(52)32(23(4)5)45-36(49)31(22(2)3)44-35(48)29-19-40-17-18-41-29/h9-11,13-14,17-19,22-24,26-28,30-33H,8,12,15-16,20-21H2,1-7H3,(H,42,51)(H,43,50)(H,44,48)(H,45,49)/t24-,26-,27?,28?,30+,31-,32-,33-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 332 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Cytotoxic activity against Protease in rat liver Huh-7 cells |
Bioorg Med Chem Lett 14: 251-6 (2003)
BindingDB Entry DOI: 10.7270/Q24B30R9 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50137719
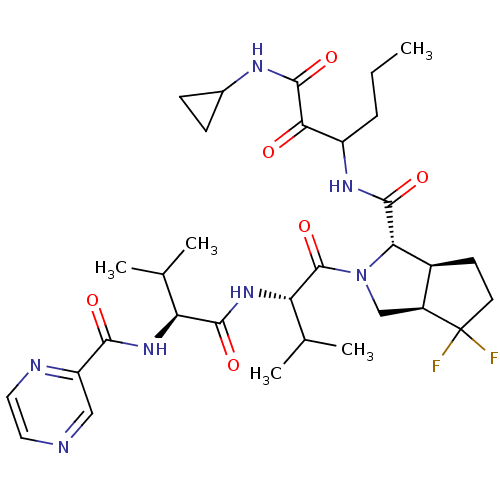
((1S,5S,6R)-4,4-Difluoro-2-((S)-3-methyl-2-{(S)-3-m...)Show SMILES CCCC(NC(=O)[C@@H]1[C@H]2CCC(F)(F)[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C32H45F2N7O6/c1-6-7-21(26(42)30(46)37-18-8-9-18)38-29(45)25-19-10-11-32(33,34)20(19)15-41(25)31(47)24(17(4)5)40-28(44)23(16(2)3)39-27(43)22-14-35-12-13-36-22/h12-14,16-21,23-25H,6-11,15H2,1-5H3,(H,37,46)(H,38,45)(H,39,43)(H,40,44)/t19-,20-,21?,23-,24-,25-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards Protease using PNA assay in rats |
Bioorg Med Chem Lett 14: 251-6 (2003)
BindingDB Entry DOI: 10.7270/Q24B30R9 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50137725
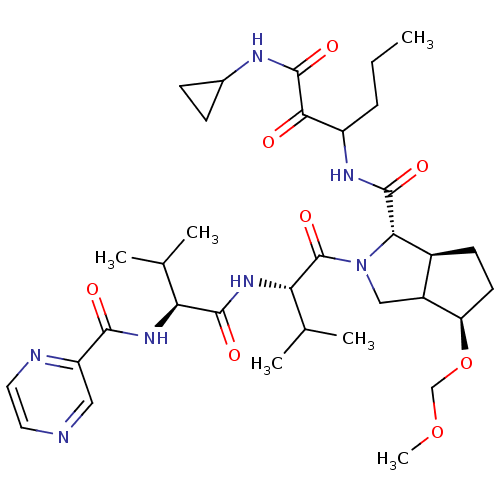
((1S,5S)-4-Methoxymethoxy-2-((S)-3-methyl-2-{(S)-3-...)Show SMILES CCCC(NC(=O)[C@@H]1[C@H]2CC[C@@H](OCOC)C2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C34H51N7O8/c1-7-8-23(29(42)33(46)37-20-9-10-20)38-32(45)28-21-11-12-25(49-17-48-6)22(21)16-41(28)34(47)27(19(4)5)40-31(44)26(18(2)3)39-30(43)24-15-35-13-14-36-24/h13-15,18-23,25-28H,7-12,16-17H2,1-6H3,(H,37,46)(H,38,45)(H,39,43)(H,40,44)/t21-,22?,23?,25+,26-,27-,28-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 415 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Cytotoxic activity against Protease in rat liver Huh-7 cells |
Bioorg Med Chem Lett 14: 251-6 (2003)
BindingDB Entry DOI: 10.7270/Q24B30R9 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50137728
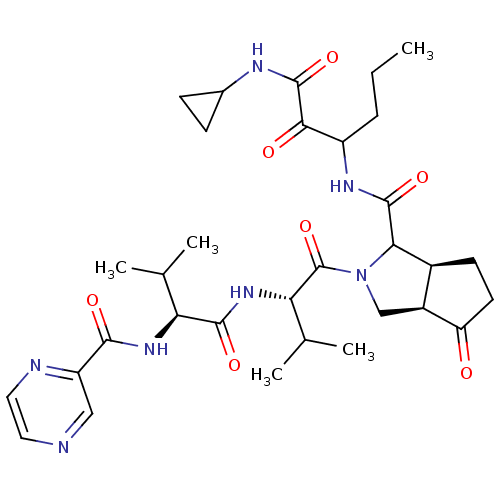
((5S,6R)-2-((S)-3-Methyl-2-{(S)-3-methyl-2-[(pyrazi...)Show SMILES CCCC(NC(=O)C1[C@H]2CCC(=O)[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C32H45N7O7/c1-6-7-21(27(41)31(45)35-18-8-9-18)36-30(44)26-19-10-11-23(40)20(19)15-39(26)32(46)25(17(4)5)38-29(43)24(16(2)3)37-28(42)22-14-33-12-13-34-22/h12-14,16-21,24-26H,6-11,15H2,1-5H3,(H,35,45)(H,36,44)(H,37,42)(H,38,43)/t19-,20-,21?,24-,25-,26?/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Cytotoxic activity against Protease in rat liver Huh-7 cells |
Bioorg Med Chem Lett 14: 251-6 (2003)
BindingDB Entry DOI: 10.7270/Q24B30R9 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50137714
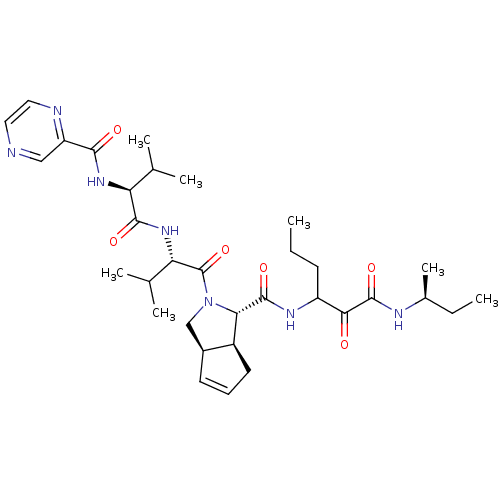
((1S,5S,6R)-2-((S)-3-Methyl-2-{(S)-3-methyl-2-[(pyr...)Show SMILES CCCC(NC(=O)[C@@H]1[C@H]2CC=C[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)C)C(=O)C(=O)N[C@@H](C)CC |c:10| Show InChI InChI=1S/C33H49N7O6/c1-8-11-23(28(41)32(45)36-20(7)9-2)37-31(44)27-22-13-10-12-21(22)17-40(27)33(46)26(19(5)6)39-30(43)25(18(3)4)38-29(42)24-16-34-14-15-35-24/h10,12,14-16,18-23,25-27H,8-9,11,13,17H2,1-7H3,(H,36,45)(H,37,44)(H,38,42)(H,39,43)/t20-,21-,22-,23?,25-,26-,27-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 625 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards Protease using PNA assay in rats |
Bioorg Med Chem Lett 14: 251-6 (2003)
BindingDB Entry DOI: 10.7270/Q24B30R9 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50152756
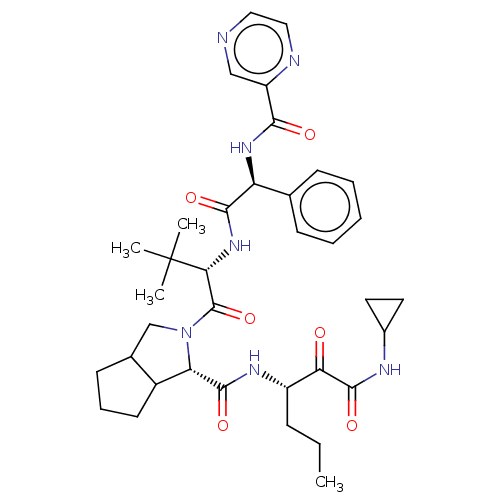
(2-((S)-3,3-Dimethyl-2-{(S)-2-phenyl-2-[(pyrazine-2...)Show SMILES CCC[C@H](NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)c1ccccc1)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C36H47N7O6/c1-5-10-25(29(44)34(48)39-23-15-16-23)40-33(47)28-24-14-9-13-22(24)20-43(28)35(49)30(36(2,3)4)42-32(46)27(21-11-7-6-8-12-21)41-31(45)26-19-37-17-18-38-26/h6-8,11-12,17-19,22-25,27-28,30H,5,9-10,13-16,20H2,1-4H3,(H,39,48)(H,40,47)(H,41,45)(H,42,46)/t22?,24?,25?,27-,28-,30+/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 650 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Inhibition of HCV NS3 protease in the pNA based inhibition assay |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus) | BDBM50152757
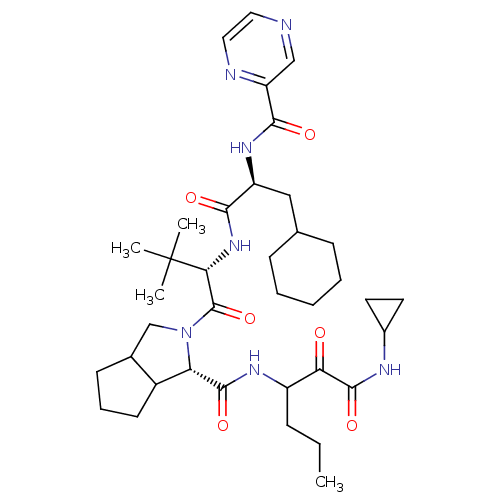
((S)-2-((S)-2-{(S)-3-Cyclohexyl-2-[(pyrazine-2-carb...)Show SMILES CCCC(NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@H](CC1CCCCC1)NC(=O)c1cnccn1)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C37H55N7O6/c1-5-10-26(30(45)35(49)40-24-15-16-24)41-34(48)29-25-14-9-13-23(25)21-44(29)36(50)31(37(2,3)4)43-32(46)27(19-22-11-7-6-8-12-22)42-33(47)28-20-38-17-18-39-28/h17-18,20,22-27,29,31H,5-16,19,21H2,1-4H3,(H,40,49)(H,41,48)(H,42,47)(H,43,46)/t23?,25?,26?,27-,29-,31+/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 650 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Inhibition of HCV NS3 protease in the pNA based inhibition assay |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50137716
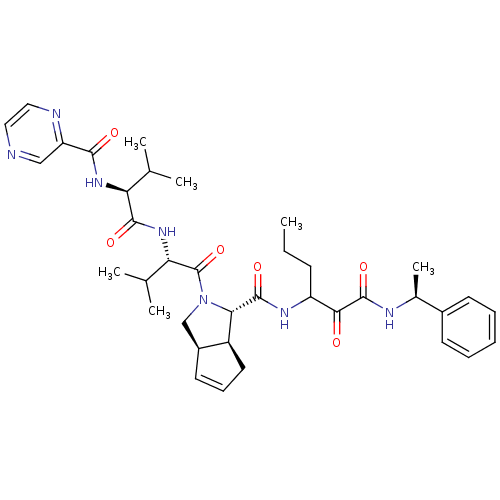
((1S,5S,6R)-2-((S)-3-Methyl-2-{(S)-3-methyl-2-[(pyr...)Show SMILES CCCC(NC(=O)[C@@H]1[C@H]2CC=C[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)C)C(=O)C(=O)N[C@@H](C)c1ccccc1 |c:10| Show InChI InChI=1S/C37H49N7O6/c1-7-12-27(32(45)36(49)40-23(6)24-13-9-8-10-14-24)41-35(48)31-26-16-11-15-25(26)20-44(31)37(50)30(22(4)5)43-34(47)29(21(2)3)42-33(46)28-19-38-17-18-39-28/h8-11,13-15,17-19,21-23,25-27,29-31H,7,12,16,20H2,1-6H3,(H,40,49)(H,41,48)(H,42,46)(H,43,47)/t23-,25-,26-,27?,29-,30-,31-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 880 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards Protease using PNA assay in rats |
Bioorg Med Chem Lett 14: 251-6 (2003)
BindingDB Entry DOI: 10.7270/Q24B30R9 |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50137733

((1S,5S,6R)-2-((S)-3,3-Dimethyl-2-{(S)-3-methyl-2-[...)Show SMILES CCC[C@H](NC(=O)[C@@H]1[C@H]2CCC[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C33H49N7O6/c1-7-9-22(26(41)31(45)36-20-12-13-20)37-30(44)25-21-11-8-10-19(21)17-40(25)32(46)27(33(4,5)6)39-29(43)24(18(2)3)38-28(42)23-16-34-14-15-35-23/h14-16,18-22,24-25,27H,7-13,17H2,1-6H3,(H,36,45)(H,37,44)(H,38,42)(H,39,43)/t19-,21-,22-,24-,25-,27+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human cathepsin B |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50137726
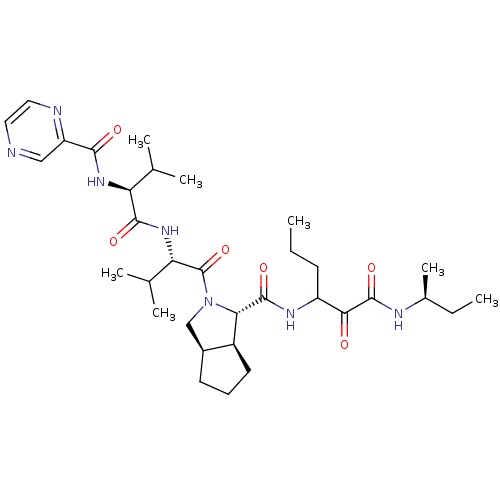
((1S,5S,6R)-2-((S)-3-Methyl-2-{(S)-3-methyl-2-[(pyr...)Show SMILES CCCC(NC(=O)[C@@H]1[C@H]2CCC[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)C)C(=O)C(=O)N[C@@H](C)CC Show InChI InChI=1S/C33H51N7O6/c1-8-11-23(28(41)32(45)36-20(7)9-2)37-31(44)27-22-13-10-12-21(22)17-40(27)33(46)26(19(5)6)39-30(43)25(18(3)4)38-29(42)24-16-34-14-15-35-24/h14-16,18-23,25-27H,8-13,17H2,1-7H3,(H,36,45)(H,37,44)(H,38,42)(H,39,43)/t20-,21-,22-,23?,25-,26-,27-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards Protease using PNA assay in rats |
Bioorg Med Chem Lett 14: 251-6 (2003)
BindingDB Entry DOI: 10.7270/Q24B30R9 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50137715
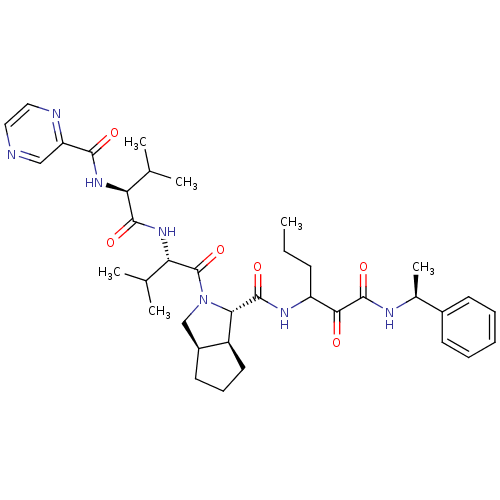
((1S,5S,6R)-2-((S)-3-Methyl-2-{(S)-3-methyl-2-[(pyr...)Show SMILES CCCC(NC(=O)[C@@H]1[C@H]2CCC[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)C)C(=O)C(=O)N[C@@H](C)c1ccccc1 Show InChI InChI=1S/C37H51N7O6/c1-7-12-27(32(45)36(49)40-23(6)24-13-9-8-10-14-24)41-35(48)31-26-16-11-15-25(26)20-44(31)37(50)30(22(4)5)43-34(47)29(21(2)3)42-33(46)28-19-38-17-18-39-28/h8-10,13-14,17-19,21-23,25-27,29-31H,7,11-12,15-16,20H2,1-6H3,(H,40,49)(H,41,48)(H,42,46)(H,43,47)/t23-,25-,26-,27?,29-,30-,31-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards Protease using PNA assay in rats |
Bioorg Med Chem Lett 14: 251-6 (2003)
BindingDB Entry DOI: 10.7270/Q24B30R9 |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50152754

(2-((S)-2-{(S)-2-Cyclohexyl-2-[(pyrazine-2-carbonyl...)Show SMILES CCC[C@H](NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C1CCCCC1)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C36H53N7O6/c1-5-10-25(29(44)34(48)39-23-15-16-23)40-33(47)28-24-14-9-13-22(24)20-43(28)35(49)30(36(2,3)4)42-32(46)27(21-11-7-6-8-12-21)41-31(45)26-19-37-17-18-38-26/h17-19,21-25,27-28,30H,5-16,20H2,1-4H3,(H,39,48)(H,40,47)(H,41,45)(H,42,46)/t22?,24?,25?,27-,28-,30+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human cathepsin B |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50152752

((S)-2-((S)-2-{(S)-3,3-Dimethyl-2-[(pyrazine-2-carb...)Show SMILES CCC[C@H](NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)(C)C)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C34H51N7O6/c1-8-10-22(25(42)30(45)37-20-13-14-20)38-29(44)24-21-12-9-11-19(21)18-41(24)32(47)27(34(5,6)7)40-31(46)26(33(2,3)4)39-28(43)23-17-35-15-16-36-23/h15-17,19-22,24,26-27H,8-14,18H2,1-7H3,(H,37,45)(H,38,44)(H,39,43)(H,40,46)/t19?,21?,22?,24-,26+,27+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human cathepsin B |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50152755

(2-((S)-2-{(S)-3-Methoxy-2-[(pyrazine-2-carbonyl)-a...)Show SMILES CCC[C@H](NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)[C@@H](C)OC)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C33H49N7O7/c1-7-9-22(26(41)31(45)36-20-12-13-20)37-30(44)25-21-11-8-10-19(21)17-40(25)32(46)27(33(3,4)5)39-29(43)24(18(2)47-6)38-28(42)23-16-34-14-15-35-23/h14-16,18-22,24-25,27H,7-13,17H2,1-6H3,(H,36,45)(H,37,44)(H,38,42)(H,39,43)/t18?,19?,21?,22?,24-,25-,27+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human cathepsin B |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50152753

((S)-2-((S)-3,3-Dimethyl-2-{(S)-3-methyl-2-[(pyrazi...)Show SMILES CCC[C@H](NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)[C@@H](C)CC)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C34H51N7O6/c1-7-10-23(27(42)32(46)37-21-13-14-21)38-31(45)26-22-12-9-11-20(22)18-41(26)33(47)28(34(4,5)6)40-30(44)25(19(3)8-2)39-29(43)24-17-35-15-16-36-24/h15-17,19-23,25-26,28H,7-14,18H2,1-6H3,(H,37,46)(H,38,45)(H,39,43)(H,40,44)/t19?,20?,22?,23?,25-,26-,28+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human cathepsin B |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50152748

((S)-2-((S)-2-{(S)-2-Cyclohexyl-2-[(pyrazine-2-carb...)Show SMILES CCCC(NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C1CCCCC1)C(C)(C)C)C(=O)C(=O)N[C@H](C)CC Show InChI InChI=1S/C37H57N7O6/c1-7-13-26(30(45)35(49)40-22(3)8-2)41-34(48)29-25-17-12-16-24(25)21-44(29)36(50)31(37(4,5)6)43-33(47)28(23-14-10-9-11-15-23)42-32(46)27-20-38-18-19-39-27/h18-20,22-26,28-29,31H,7-17,21H2,1-6H3,(H,40,49)(H,41,48)(H,42,46)(H,43,47)/t22-,24?,25?,26?,28+,29+,31-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human cathepsin B |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Procathepsin L
(Homo sapiens (Human)) | BDBM50152753

((S)-2-((S)-3,3-Dimethyl-2-{(S)-3-methyl-2-[(pyrazi...)Show SMILES CCC[C@H](NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)[C@@H](C)CC)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C34H51N7O6/c1-7-10-23(27(42)32(46)37-21-13-14-21)38-31(45)26-22-12-9-11-20(22)18-41(26)33(47)28(34(4,5)6)40-30(44)25(19(3)8-2)39-29(43)24-17-35-15-16-36-24/h15-17,19-23,25-26,28H,7-14,18H2,1-6H3,(H,37,46)(H,38,45)(H,39,43)(H,40,44)/t19?,20?,22?,23?,25-,26-,28+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human cathepsin L |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50152750

(2-((S)-2-{(S)-2-Cyclohexyl-2-[(pyrazine-2-carbonyl...)Show SMILES CC(C)(C)[C@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C1CCCCC1)C(=O)N1CC2CCCC2[C@H]1C(=O)NC(CC(F)F)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C35H49F2N7O6/c1-35(2,3)29(43-31(47)26(19-8-5-4-6-9-19)42-30(46)24-17-38-14-15-39-24)34(50)44-18-20-10-7-11-22(20)27(44)32(48)41-23(16-25(36)37)28(45)33(49)40-21-12-13-21/h14-15,17,19-23,25-27,29H,4-13,16,18H2,1-3H3,(H,40,49)(H,41,48)(H,42,46)(H,43,47)/t20?,22?,23?,26-,27-,29+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human cathepsin B |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50152753

((S)-2-((S)-3,3-Dimethyl-2-{(S)-3-methyl-2-[(pyrazi...)Show SMILES CCC[C@H](NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)[C@@H](C)CC)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C34H51N7O6/c1-7-10-23(27(42)32(46)37-21-13-14-21)38-31(45)26-22-12-9-11-20(22)18-41(26)33(47)28(34(4,5)6)40-30(44)25(19(3)8-2)39-29(43)24-17-35-15-16-36-24/h15-17,19-23,25-26,28H,7-14,18H2,1-6H3,(H,37,46)(H,38,45)(H,39,43)(H,40,44)/t19?,20?,22?,23?,25-,26-,28+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human thrombin |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Plasminogen
(Homo sapiens (Human)) | BDBM50152751

(2-((S)-2-{(S)-2-Cyclohexyl-2-[((R)-pyrazine-2-carb...)Show SMILES CCCC(NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C1CCCCC1)C(C)(C)C)C(=O)C(=O)N[C@H](C)c1ccccc1 Show InChI InChI=1S/C41H57N7O6/c1-6-14-30(34(49)39(53)44-25(2)26-15-9-7-10-16-26)45-38(52)33-29-20-13-19-28(29)24-48(33)40(54)35(41(3,4)5)47-37(51)32(27-17-11-8-12-18-27)46-36(50)31-23-42-21-22-43-31/h7,9-10,15-16,21-23,25,27-30,32-33,35H,6,8,11-14,17-20,24H2,1-5H3,(H,44,53)(H,45,52)(H,46,50)(H,47,51)/t25-,28?,29?,30?,32+,33+,35-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human plasmin |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50152754

(2-((S)-2-{(S)-2-Cyclohexyl-2-[(pyrazine-2-carbonyl...)Show SMILES CCC[C@H](NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C1CCCCC1)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C36H53N7O6/c1-5-10-25(29(44)34(48)39-23-15-16-23)40-33(47)28-24-14-9-13-22(24)20-43(28)35(49)30(36(2,3)4)42-32(46)27(21-11-7-6-8-12-21)41-31(45)26-19-37-17-18-38-26/h17-19,21-25,27-28,30H,5-16,20H2,1-4H3,(H,39,48)(H,40,47)(H,41,45)(H,42,46)/t22?,24?,25?,27-,28-,30+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human thrombin |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50152751

(2-((S)-2-{(S)-2-Cyclohexyl-2-[((R)-pyrazine-2-carb...)Show SMILES CCCC(NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C1CCCCC1)C(C)(C)C)C(=O)C(=O)N[C@H](C)c1ccccc1 Show InChI InChI=1S/C41H57N7O6/c1-6-14-30(34(49)39(53)44-25(2)26-15-9-7-10-16-26)45-38(52)33-29-20-13-19-28(29)24-48(33)40(54)35(41(3,4)5)47-37(51)32(27-17-11-8-12-18-27)46-36(50)31-23-42-21-22-43-31/h7,9-10,15-16,21-23,25,27-30,32-33,35H,6,8,11-14,17-20,24H2,1-5H3,(H,44,53)(H,45,52)(H,46,50)(H,47,51)/t25-,28?,29?,30?,32+,33+,35-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human thrombin |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Chymotrypsinogen B
(Homo sapiens (Human)) | BDBM50137733

((1S,5S,6R)-2-((S)-3,3-Dimethyl-2-{(S)-3-methyl-2-[...)Show SMILES CCC[C@H](NC(=O)[C@@H]1[C@H]2CCC[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C33H49N7O6/c1-7-9-22(26(41)31(45)36-20-12-13-20)37-30(44)25-21-11-8-10-19(21)17-40(25)32(46)27(33(4,5)6)39-29(43)24(18(2)3)38-28(42)23-16-34-14-15-35-23/h14-16,18-22,24-25,27H,7-13,17H2,1-6H3,(H,36,45)(H,37,44)(H,38,42)(H,39,43)/t19-,21-,22-,24-,25-,27+/m0/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human Chymotrypsinogen |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Plasminogen
(Homo sapiens (Human)) | BDBM50137733

((1S,5S,6R)-2-((S)-3,3-Dimethyl-2-{(S)-3-methyl-2-[...)Show SMILES CCC[C@H](NC(=O)[C@@H]1[C@H]2CCC[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C33H49N7O6/c1-7-9-22(26(41)31(45)36-20-12-13-20)37-30(44)25-21-11-8-10-19(21)17-40(25)32(46)27(33(4,5)6)39-29(43)24(18(2)3)38-28(42)23-16-34-14-15-35-23/h14-16,18-22,24-25,27H,7-13,17H2,1-6H3,(H,36,45)(H,37,44)(H,38,42)(H,39,43)/t19-,21-,22-,24-,25-,27+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human plasmin |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50137733

((1S,5S,6R)-2-((S)-3,3-Dimethyl-2-{(S)-3-methyl-2-[...)Show SMILES CCC[C@H](NC(=O)[C@@H]1[C@H]2CCC[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C33H49N7O6/c1-7-9-22(26(41)31(45)36-20-12-13-20)37-30(44)25-21-11-8-10-19(21)17-40(25)32(46)27(33(4,5)6)39-29(43)24(18(2)3)38-28(42)23-16-34-14-15-35-23/h14-16,18-22,24-25,27H,7-13,17H2,1-6H3,(H,36,45)(H,37,44)(H,38,42)(H,39,43)/t19-,21-,22-,24-,25-,27+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human thrombin |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Trypsin
(Homo sapiens (Human)) | BDBM50137733

((1S,5S,6R)-2-((S)-3,3-Dimethyl-2-{(S)-3-methyl-2-[...)Show SMILES CCC[C@H](NC(=O)[C@@H]1[C@H]2CCC[C@H]2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)C)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C33H49N7O6/c1-7-9-22(26(41)31(45)36-20-12-13-20)37-30(44)25-21-11-8-10-19(21)17-40(25)32(46)27(33(4,5)6)39-29(43)24(18(2)3)38-28(42)23-16-34-14-15-35-23/h14-16,18-22,24-25,27H,7-13,17H2,1-6H3,(H,36,45)(H,37,44)(H,38,42)(H,39,43)/t19-,21-,22-,24-,25-,27+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human trypsin |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Trypsin
(Homo sapiens (Human)) | BDBM50152752

((S)-2-((S)-2-{(S)-3,3-Dimethyl-2-[(pyrazine-2-carb...)Show SMILES CCC[C@H](NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C(C)(C)C)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C34H51N7O6/c1-8-10-22(25(42)30(45)37-20-13-14-20)38-29(44)24-21-12-9-11-19(21)18-41(24)32(47)27(34(5,6)7)40-31(46)26(33(2,3)4)39-28(43)23-17-35-15-16-36-23/h15-17,19-22,24,26-27H,8-14,18H2,1-7H3,(H,37,45)(H,38,44)(H,39,43)(H,40,46)/t19?,21?,22?,24-,26+,27+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human trypsin |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Trypsin
(Homo sapiens (Human)) | BDBM50152754

(2-((S)-2-{(S)-2-Cyclohexyl-2-[(pyrazine-2-carbonyl...)Show SMILES CCC[C@H](NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C1CCCCC1)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C36H53N7O6/c1-5-10-25(29(44)34(48)39-23-15-16-23)40-33(47)28-24-14-9-13-22(24)20-43(28)35(49)30(36(2,3)4)42-32(46)27(21-11-7-6-8-12-21)41-31(45)26-19-37-17-18-38-26/h17-19,21-25,27-28,30H,5-16,20H2,1-4H3,(H,39,48)(H,40,47)(H,41,45)(H,42,46)/t22?,24?,25?,27-,28-,30+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human trypsin |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Trypsin
(Homo sapiens (Human)) | BDBM50152753

((S)-2-((S)-3,3-Dimethyl-2-{(S)-3-methyl-2-[(pyrazi...)Show SMILES CCC[C@H](NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)[C@@H](C)CC)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C34H51N7O6/c1-7-10-23(27(42)32(46)37-21-13-14-21)38-31(45)26-22-12-9-11-20(22)18-41(26)33(47)28(34(4,5)6)40-30(44)25(19(3)8-2)39-29(43)24-17-35-15-16-36-24/h15-17,19-23,25-26,28H,7-14,18H2,1-6H3,(H,37,46)(H,38,45)(H,39,43)(H,40,44)/t19?,20?,22?,23?,25-,26-,28+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human trypsin |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Trypsin
(Homo sapiens (Human)) | BDBM50152755

(2-((S)-2-{(S)-3-Methoxy-2-[(pyrazine-2-carbonyl)-a...)Show SMILES CCC[C@H](NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)[C@@H](C)OC)C(C)(C)C)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C33H49N7O7/c1-7-9-22(26(41)31(45)36-20-12-13-20)37-30(44)25-21-11-8-10-19(21)17-40(25)32(46)27(33(3,4)5)39-29(43)24(18(2)47-6)38-28(42)23-16-34-14-15-35-23/h14-16,18-22,24-25,27H,7-13,17H2,1-6H3,(H,36,45)(H,37,44)(H,38,42)(H,39,43)/t18?,19?,21?,22?,24-,25-,27+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human trypsin |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Trypsin
(Homo sapiens (Human)) | BDBM50152748

((S)-2-((S)-2-{(S)-2-Cyclohexyl-2-[(pyrazine-2-carb...)Show SMILES CCCC(NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C1CCCCC1)C(C)(C)C)C(=O)C(=O)N[C@H](C)CC Show InChI InChI=1S/C37H57N7O6/c1-7-13-26(30(45)35(49)40-22(3)8-2)41-34(48)29-25-17-12-16-24(25)21-44(29)36(50)31(37(4,5)6)43-33(47)28(23-14-10-9-11-15-23)42-32(46)27-20-38-18-19-39-27/h18-20,22-26,28-29,31H,7-17,21H2,1-6H3,(H,40,49)(H,41,48)(H,42,46)(H,43,47)/t22-,24?,25?,26?,28+,29+,31-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human trypsin |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Trypsin
(Homo sapiens (Human)) | BDBM50152751

(2-((S)-2-{(S)-2-Cyclohexyl-2-[((R)-pyrazine-2-carb...)Show SMILES CCCC(NC(=O)[C@@H]1C2CCCC2CN1C(=O)[C@@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C1CCCCC1)C(C)(C)C)C(=O)C(=O)N[C@H](C)c1ccccc1 Show InChI InChI=1S/C41H57N7O6/c1-6-14-30(34(49)39(53)44-25(2)26-15-9-7-10-16-26)45-38(52)33-29-20-13-19-28(29)24-48(33)40(54)35(41(3,4)5)47-37(51)32(27-17-11-8-12-18-27)46-36(50)31-23-42-21-22-43-31/h7,9-10,15-16,21-23,25,27-30,32-33,35H,6,8,11-14,17-20,24H2,1-5H3,(H,44,53)(H,45,52)(H,46,50)(H,47,51)/t25-,28?,29?,30?,32+,33+,35-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human trypsin |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |
Trypsin
(Homo sapiens (Human)) | BDBM50152750

(2-((S)-2-{(S)-2-Cyclohexyl-2-[(pyrazine-2-carbonyl...)Show SMILES CC(C)(C)[C@H](NC(=O)[C@@H](NC(=O)c1cnccn1)C1CCCCC1)C(=O)N1CC2CCCC2[C@H]1C(=O)NC(CC(F)F)C(=O)C(=O)NC1CC1 Show InChI InChI=1S/C35H49F2N7O6/c1-35(2,3)29(43-31(47)26(19-8-5-4-6-9-19)42-30(46)24-17-38-14-15-39-24)34(50)44-18-20-10-7-11-22(20)27(44)32(48)41-23(16-25(36)37)28(45)33(49)40-21-12-13-21/h14-15,17,19-23,25-27,29H,4-13,16,18H2,1-3H3,(H,40,49)(H,41,48)(H,42,46)(H,43,47)/t20?,22?,23?,26-,27-,29+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratory
Curated by ChEMBL
| Assay Description
Binding affinity towards human trypsin |
Bioorg Med Chem Lett 14: 5007-11 (2004)
Article DOI: 10.1016/j.bmcl.2004.07.007
BindingDB Entry DOI: 10.7270/Q29P314N |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data