 Found 196 hits with Last Name = 'rabe' and Initial = 's'
Found 196 hits with Last Name = 'rabe' and Initial = 's' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cathepsin E
(Homo sapiens (Human)) | BDBM50305527
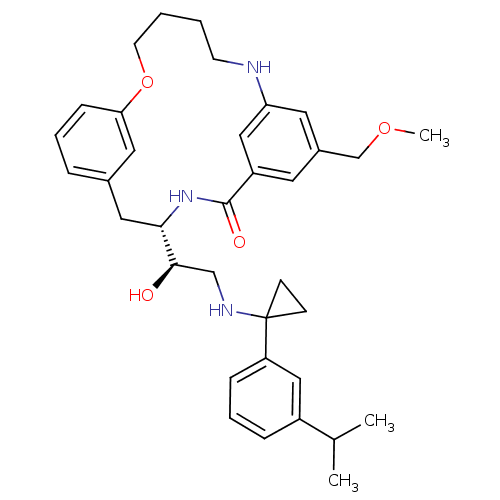
((4S)-4-[(1R)-1-hydroxy-2-({1-[3-(1-methylethyl)phe...)Show SMILES COCc1cc2NCCCCOc3cccc(C[C@H](NC(=O)c(c1)c2)[C@H](O)CNC1(CC1)c1cccc(c1)C(C)C)c3 |r| Show InChI InChI=1S/C35H45N3O4/c1-24(2)27-9-7-10-29(20-27)35(12-13-35)37-22-33(39)32-19-25-8-6-11-31(18-25)42-15-5-4-14-36-30-17-26(23-41-3)16-28(21-30)34(40)38-32/h6-11,16-18,20-21,24,32-33,36-37,39H,4-5,12-15,19,22-23H2,1-3H3,(H,38,40)/t32-,33+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin E |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus (HCV)) | BDBM50300504

(CHEMBL574455 | N-({3-[(5S)-5-tert-butyl-1-[(4-fluo...)Show SMILES CN(Cc1cccc2C(NS(=O)(=O)c12)=C1C(=O)[C@@H](N(Cc2ccc(F)cc2)C1=O)C(C)(C)C)S(N)(=O)=O |r,w:14.16| Show InChI InChI=1S/C24H27FN4O6S2/c1-24(2,3)22-20(30)18(23(31)29(22)12-14-8-10-16(25)11-9-14)19-17-7-5-6-15(13-28(4)37(26,34)35)21(17)36(32,33)27-19/h5-11,22,27H,12-13H2,1-4H3,(H2,26,34,35)/t22-/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Palo Alto LLC
Curated by ChEMBL
| Assay Description
Inhibition of HSV1b con1 NS5B polymerase assessed as [3H]UTP incorporation into acid insoluble RNA product |
Bioorg Med Chem Lett 19: 5652-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.08.022
BindingDB Entry DOI: 10.7270/Q2N016K0 |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50294218
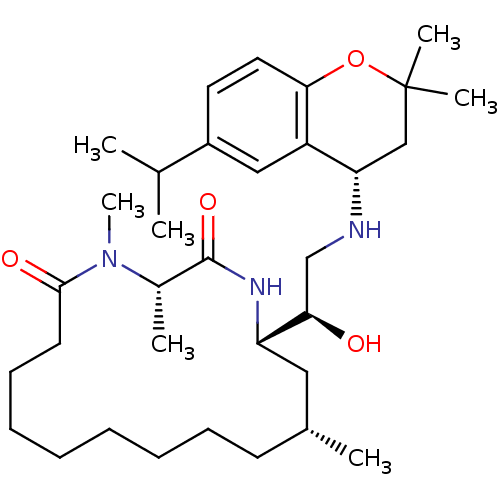
((3S,14R,16S)-16-((R)-1-hydroxy-2-((S)-6-isopropyl-...)Show SMILES CC(C)c1ccc2OC(C)(C)C[C@H](NC[C@@H](O)[C@@H]3C[C@H](C)CCCCCCCCC(=O)N(C)[C@@H](C)C(=O)N3)c2c1 |r| Show InChI InChI=1S/C33H55N3O4/c1-22(2)25-16-17-30-26(19-25)28(20-33(5,6)40-30)34-21-29(37)27-18-23(3)14-12-10-8-9-11-13-15-31(38)36(7)24(4)32(39)35-27/h16-17,19,22-24,27-29,34,37H,8-15,18,20-21H2,1-7H3,(H,35,39)/t23-,24+,27+,28+,29-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin D |
Bioorg Med Chem Lett 19: 1366-70 (2009)
Article DOI: 10.1016/j.bmcl.2009.01.055
BindingDB Entry DOI: 10.7270/Q2SB45S3 |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305544
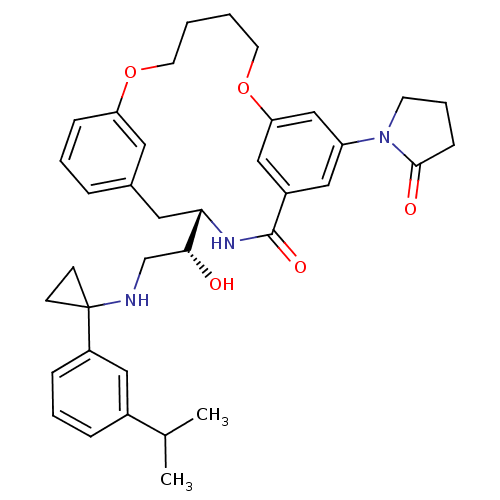
((S)-4-{(R)-1-Hydroxy-2-[1-(3-isopropyl-phenyl)-cyc...)Show SMILES CC(C)c1cccc(c1)C1(CC1)NC[C@@H](O)[C@@H]1Cc2cccc(OCCCCOc3cc(cc(c3)C(=O)N1)N1CCCC1=O)c2 |r| Show InChI InChI=1S/C37H45N3O5/c1-25(2)27-9-6-10-29(20-27)37(13-14-37)38-24-34(41)33-19-26-8-5-11-31(18-26)44-16-3-4-17-45-32-22-28(36(43)39-33)21-30(23-32)40-15-7-12-35(40)42/h5-6,8-11,18,20-23,25,33-34,38,41H,3-4,7,12-17,19,24H2,1-2H3,(H,39,43)/t33-,34+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 expressed in Escherichia coli |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50305527

((4S)-4-[(1R)-1-hydroxy-2-({1-[3-(1-methylethyl)phe...)Show SMILES COCc1cc2NCCCCOc3cccc(C[C@H](NC(=O)c(c1)c2)[C@H](O)CNC1(CC1)c1cccc(c1)C(C)C)c3 |r| Show InChI InChI=1S/C35H45N3O4/c1-24(2)27-9-7-10-29(20-27)35(12-13-35)37-22-33(39)32-19-25-8-6-11-31(18-25)42-15-5-4-14-36-30-17-26(23-41-3)16-28(21-30)34(40)38-32/h6-11,16-18,20-21,24,32-33,36-37,39H,4-5,12-15,19,22-23H2,1-3H3,(H,38,40)/t32-,33+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin D |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305542
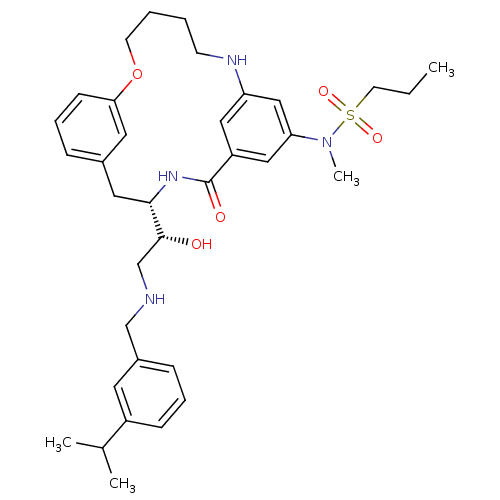
(CHEMBL595016 | Propane-1-sulfonic acid{(S)-4-[(R)-...)Show SMILES CCCS(=O)(=O)N(C)c1cc2NCCCCOc3cccc(C[C@H](NC(=O)c(c2)c1)[C@H](O)CNCc1cccc(c1)C(C)C)c3 |r| Show InChI InChI=1S/C35H48N4O5S/c1-5-16-45(42,43)39(4)31-21-29-20-30(22-31)37-14-6-7-15-44-32-13-9-10-26(18-32)19-33(38-35(29)41)34(40)24-36-23-27-11-8-12-28(17-27)25(2)3/h8-13,17-18,20-22,25,33-34,36-37,40H,5-7,14-16,19,23-24H2,1-4H3,(H,38,41)/t33-,34+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 expressed in Escherichia coli |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305531
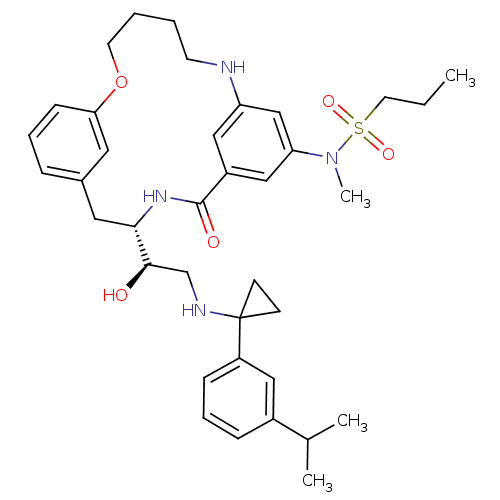
(CHEMBL595136 | Propane-1-sulfonic acid((S)-4-{(R)-...)Show SMILES CCCS(=O)(=O)N(C)c1cc2NCCCCOc3cccc(C[C@H](NC(=O)c(c2)c1)[C@H](O)CNC1(CC1)c1cccc(c1)C(C)C)c3 |r| Show InChI InChI=1S/C37H50N4O5S/c1-5-18-47(44,45)41(4)32-23-29-22-31(24-32)38-16-6-7-17-46-33-13-8-10-27(19-33)20-34(40-36(29)43)35(42)25-39-37(14-15-37)30-12-9-11-28(21-30)26(2)3/h8-13,19,21-24,26,34-35,38-39,42H,5-7,14-18,20,25H2,1-4H3,(H,40,43)/t34-,35+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 expressed in Escherichia coli |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50294218

((3S,14R,16S)-16-((R)-1-hydroxy-2-((S)-6-isopropyl-...)Show SMILES CC(C)c1ccc2OC(C)(C)C[C@H](NC[C@@H](O)[C@@H]3C[C@H](C)CCCCCCCCC(=O)N(C)[C@@H](C)C(=O)N3)c2c1 |r| Show InChI InChI=1S/C33H55N3O4/c1-22(2)25-16-17-30-26(19-25)28(20-33(5,6)40-30)34-21-29(37)27-18-23(3)14-12-10-8-9-11-13-15-31(38)36(7)24(4)32(39)35-27/h16-17,19,22-24,27-29,34,37H,8-15,18,20-21H2,1-7H3,(H,35,39)/t23-,24+,27+,28+,29-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 |
Bioorg Med Chem Lett 19: 1366-70 (2009)
Article DOI: 10.1016/j.bmcl.2009.01.055
BindingDB Entry DOI: 10.7270/Q2SB45S3 |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50305536
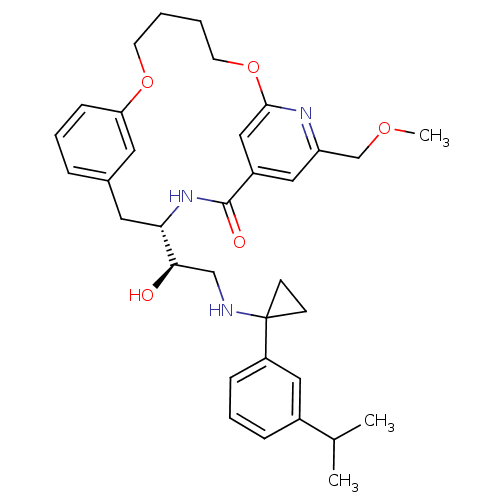
((S)-4-{(R)-1-Hydroxy-2-[1-(3-isopropyl-phenyl)-cyc...)Show SMILES COCc1cc2cc(OCCCCOc3cccc(C[C@H](NC2=O)[C@H](O)CNC2(CC2)c2cccc(c2)C(C)C)c3)n1 |r| Show InChI InChI=1S/C34H43N3O5/c1-23(2)25-9-7-10-27(18-25)34(12-13-34)35-21-31(38)30-17-24-8-6-11-29(16-24)41-14-4-5-15-42-32-20-26(33(39)37-30)19-28(36-32)22-40-3/h6-11,16,18-20,23,30-31,35,38H,4-5,12-15,17,21-22H2,1-3H3,(H,37,39)/t30-,31+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin D |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus (HCV)) | BDBM50300503
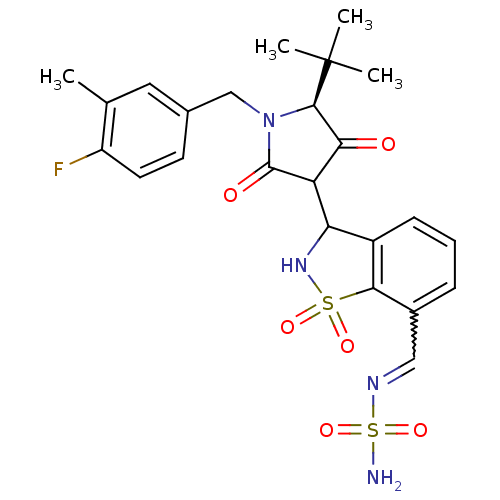
(CHEMBL582995 | N-({3-[(5S)-5-tert-butyl-1-[(4-fluo...)Show SMILES Cc1cc(CN2[C@H](C(=O)C(C3NS(=O)(=O)c4c3cccc4C=NS(N)(=O)=O)C2=O)C(C)(C)C)ccc1F |r,w:21.22| Show InChI InChI=1S/C24H27FN4O6S2/c1-13-10-14(8-9-17(13)25)12-29-22(24(2,3)4)20(30)18(23(29)31)19-16-7-5-6-15(11-27-37(26,34)35)21(16)36(32,33)28-19/h5-11,18-19,22,28H,12H2,1-4H3,(H2,26,34,35)/t18?,19?,22-/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Palo Alto LLC
Curated by ChEMBL
| Assay Description
Inhibition of HSV1b con1 NS5B polymerase assessed as [3H]UTP incorporation into acid insoluble RNA product |
Bioorg Med Chem Lett 19: 5652-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.08.022
BindingDB Entry DOI: 10.7270/Q2N016K0 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus (HCV)) | BDBM50300502

(CHEMBL572682 | N-({3-[(5S)-5-tert-butyl-1-[(4-fluo...)Show SMILES CC(C)(C)[C@@H]1N(Cc2ccc(F)cc2)C(=O)C(C2NS(=O)(=O)c3c2cccc3C=NS(N)(=O)=O)C1=O |r,w:28.30| Show InChI InChI=1S/C23H25FN4O6S2/c1-23(2,3)21-19(29)17(22(30)28(21)12-13-7-9-15(24)10-8-13)18-16-6-4-5-14(11-26-36(25,33)34)20(16)35(31,32)27-18/h4-11,17-18,21,27H,12H2,1-3H3,(H2,25,33,34)/t17?,18?,21-/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Palo Alto LLC
Curated by ChEMBL
| Assay Description
Inhibition of HSV1b con1 NS5B polymerase assessed as [3H]UTP incorporation into acid insoluble RNA product |
Bioorg Med Chem Lett 19: 5652-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.08.022
BindingDB Entry DOI: 10.7270/Q2N016K0 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus (HCV)) | BDBM50300497
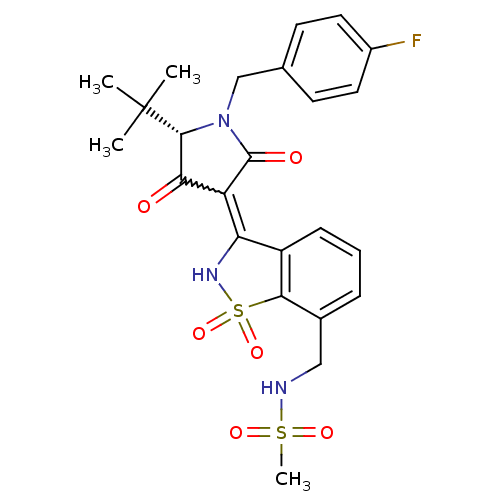
(CHEMBL578433 | N-({3-[(5S)-5-tert-butyl-1-(4-fluor...)Show SMILES CC(C)(C)[C@@H]1N(Cc2ccc(F)cc2)C(=O)C(C1=O)=C1NS(=O)(=O)c2c1cccc2CNS(C)(=O)=O |r,w:16.17| Show InChI InChI=1S/C24H26FN3O6S2/c1-24(2,3)22-20(29)18(23(30)28(22)13-14-8-10-16(25)11-9-14)19-17-7-5-6-15(12-26-35(4,31)32)21(17)36(33,34)27-19/h5-11,22,26-27H,12-13H2,1-4H3/t22-/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Palo Alto LLC
Curated by ChEMBL
| Assay Description
Inhibition of HSV1b con1 NS5B polymerase assessed as [3H]UTP incorporation into acid insoluble RNA product |
Bioorg Med Chem Lett 19: 5652-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.08.022
BindingDB Entry DOI: 10.7270/Q2N016K0 |
More data for this
Ligand-Target Pair | |
Cathepsin E
(Homo sapiens (Human)) | BDBM50305537
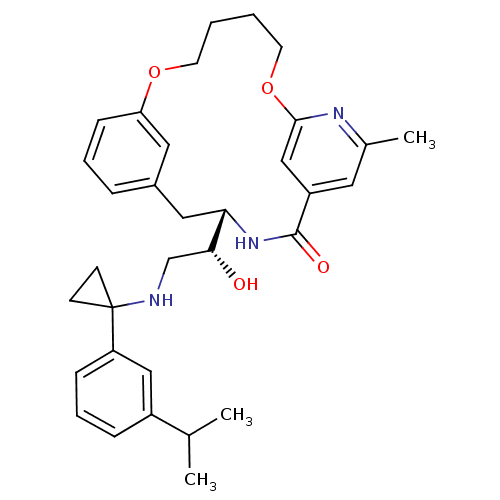
((S)-4-{(R)-1-Hydroxy-2-[1-(3-isopropyl-phenyl)-cyc...)Show SMILES CC(C)c1cccc(c1)C1(CC1)NC[C@@H](O)[C@@H]1Cc2cccc(OCCCCOc3cc(cc(C)n3)C(=O)N1)c2 |r| Show InChI InChI=1S/C33H41N3O4/c1-22(2)25-9-7-10-27(19-25)33(12-13-33)34-21-30(37)29-18-24-8-6-11-28(17-24)39-14-4-5-15-40-31-20-26(32(38)36-29)16-23(3)35-31/h6-11,16-17,19-20,22,29-30,34,37H,4-5,12-15,18,21H2,1-3H3,(H,36,38)/t29-,30+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin E |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus (HCV)) | BDBM50300499
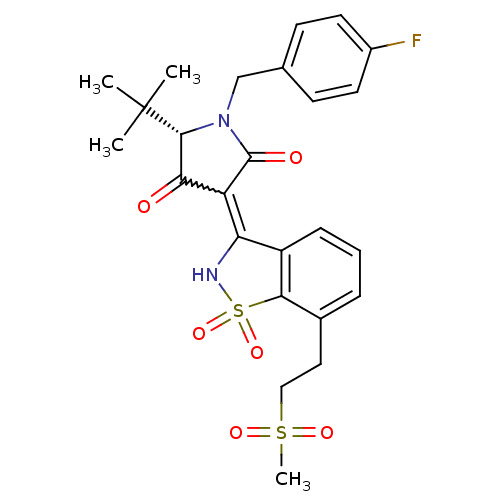
((S)-5-tert-Butyl-1-(4-fluoro-benzyl)-4-hydroxy-3-[...)Show SMILES CC(C)(C)[C@@H]1N(Cc2ccc(F)cc2)C(=O)C(C1=O)=C1NS(=O)(=O)c2c1cccc2CCS(C)(=O)=O |r,w:16.17| Show InChI InChI=1S/C25H27FN2O6S2/c1-25(2,3)23-21(29)19(24(30)28(23)14-15-8-10-17(26)11-9-15)20-18-7-5-6-16(12-13-35(4,31)32)22(18)36(33,34)27-20/h5-11,23,27H,12-14H2,1-4H3/t23-/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Palo Alto LLC
Curated by ChEMBL
| Assay Description
Inhibition of HSV1b con1 NS5B polymerase assessed as [3H]UTP incorporation into acid insoluble RNA product |
Bioorg Med Chem Lett 19: 5652-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.08.022
BindingDB Entry DOI: 10.7270/Q2N016K0 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus (HCV)) | BDBM50300498
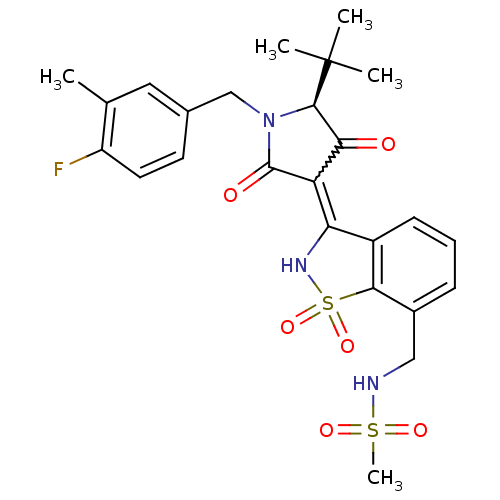
(CHEMBL575777 | N-{3-[(S)-5-tert-Butyl-1-(4-fluoro-...)Show SMILES Cc1cc(CN2[C@H](C(=O)C(C2=O)=C2NS(=O)(=O)c3c2cccc3CNS(C)(=O)=O)C(C)(C)C)ccc1F |r,w:9.8| Show InChI InChI=1S/C25H28FN3O6S2/c1-14-11-15(9-10-18(14)26)13-29-23(25(2,3)4)21(30)19(24(29)31)20-17-8-6-7-16(12-27-36(5,32)33)22(17)37(34,35)28-20/h6-11,23,27-28H,12-13H2,1-5H3/t23-/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Palo Alto LLC
Curated by ChEMBL
| Assay Description
Inhibition of HSV1b con1 NS5B polymerase assessed as [3H]UTP incorporation into acid insoluble RNA product |
Bioorg Med Chem Lett 19: 5652-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.08.022
BindingDB Entry DOI: 10.7270/Q2N016K0 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus (HCV)) | BDBM50300496

(CHEMBL577404 | N-{3-[(S)-5-tert-Butyl-1-(4-fluoro-...)Show SMILES CC(C)(C)[C@@H]1N(Cc2ccc(F)cc2)C(=O)C(C1=O)=C1NS(=O)(=O)c2c1cccc2NS(C)(=O)=O |r,w:16.17| Show InChI InChI=1S/C23H24FN3O6S2/c1-23(2,3)21-19(28)17(22(29)27(21)12-13-8-10-14(24)11-9-13)18-15-6-5-7-16(25-34(4,30)31)20(15)35(32,33)26-18/h5-11,21,25-26H,12H2,1-4H3/t21-/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Palo Alto LLC
Curated by ChEMBL
| Assay Description
Inhibition of HSV1b con1 NS5B polymerase assessed as [3H]UTP incorporation into acid insoluble RNA product |
Bioorg Med Chem Lett 19: 5652-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.08.022
BindingDB Entry DOI: 10.7270/Q2N016K0 |
More data for this
Ligand-Target Pair | |
Cathepsin E
(Homo sapiens (Human)) | BDBM50305536

((S)-4-{(R)-1-Hydroxy-2-[1-(3-isopropyl-phenyl)-cyc...)Show SMILES COCc1cc2cc(OCCCCOc3cccc(C[C@H](NC2=O)[C@H](O)CNC2(CC2)c2cccc(c2)C(C)C)c3)n1 |r| Show InChI InChI=1S/C34H43N3O5/c1-23(2)25-9-7-10-27(18-25)34(12-13-34)35-21-31(38)30-17-24-8-6-11-29(16-24)41-14-4-5-15-42-32-20-26(33(39)37-30)19-28(36-32)22-40-3/h6-11,16,18-20,23,30-31,35,38H,4-5,12-15,17,21-22H2,1-3H3,(H,37,39)/t30-,31+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin E |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus (HCV)) | BDBM50300505
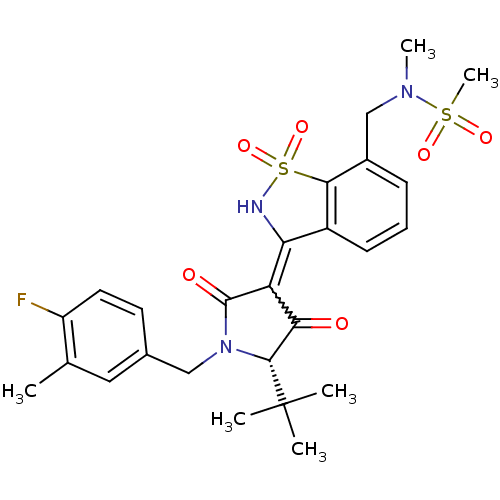
(CHEMBL572683 | N-{3-[(S)-5-tert-Butyl-1-(4-fluoro-...)Show SMILES CN(Cc1cccc2C(NS(=O)(=O)c12)=C1C(=O)[C@@H](N(Cc2ccc(F)c(C)c2)C1=O)C(C)(C)C)S(C)(=O)=O |r,w:14.16| Show InChI InChI=1S/C26H30FN3O6S2/c1-15-12-16(10-11-19(15)27)13-30-24(26(2,3)4)22(31)20(25(30)32)21-18-9-7-8-17(14-29(5)37(6,33)34)23(18)38(35,36)28-21/h7-12,24,28H,13-14H2,1-6H3/t24-/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Palo Alto LLC
Curated by ChEMBL
| Assay Description
Inhibition of HSV1b con1 NS5B polymerase assessed as [3H]UTP incorporation into acid insoluble RNA product |
Bioorg Med Chem Lett 19: 5652-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.08.022
BindingDB Entry DOI: 10.7270/Q2N016K0 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus (HCV)) | BDBM50300501
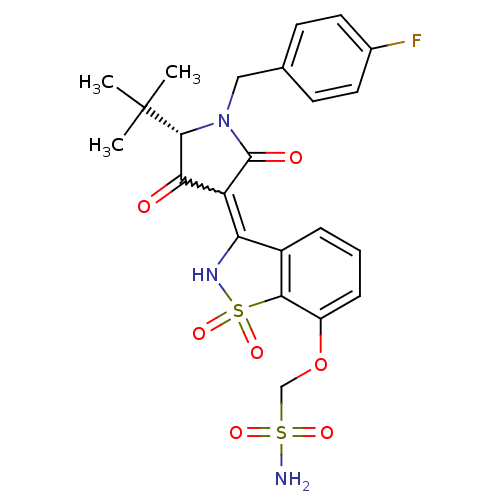
(CHEMBL573175 | {3-[(S)-5-tert-Butyl-1-(4-fluoro-be...)Show SMILES CC(C)(C)[C@@H]1N(Cc2ccc(F)cc2)C(=O)C(C1=O)=C1NS(=O)(=O)c2c1cccc2OCS(N)(=O)=O |r,w:16.17| Show InChI InChI=1S/C23H24FN3O7S2/c1-23(2,3)21-19(28)17(22(29)27(21)11-13-7-9-14(24)10-8-13)18-15-5-4-6-16(34-12-35(25,30)31)20(15)36(32,33)26-18/h4-10,21,26H,11-12H2,1-3H3,(H2,25,30,31)/t21-/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Palo Alto LLC
Curated by ChEMBL
| Assay Description
Inhibition of HSV1b con1 NS5B polymerase assessed as [3H]UTP incorporation into acid insoluble RNA product |
Bioorg Med Chem Lett 19: 5652-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.08.022
BindingDB Entry DOI: 10.7270/Q2N016K0 |
More data for this
Ligand-Target Pair | |
Cathepsin E
(Homo sapiens (Human)) | BDBM50305533

((S)-4-[(R)-1-Hydroxy-2-(3-isopropyl-benzylamino)-e...)Show SMILES CC(C)c1cccc(CNC[C@@H](O)[C@@H]2Cc3cccc(OCCCCOc4cc(cc(C)n4)C(=O)N2)c3)c1 |r| Show InChI InChI=1S/C31H39N3O4/c1-21(2)25-10-6-9-24(15-25)19-32-20-29(35)28-17-23-8-7-11-27(16-23)37-12-4-5-13-38-30-18-26(31(36)34-28)14-22(3)33-30/h6-11,14-16,18,21,28-29,32,35H,4-5,12-13,17,19-20H2,1-3H3,(H,34,36)/t28-,29+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin E |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305536

((S)-4-{(R)-1-Hydroxy-2-[1-(3-isopropyl-phenyl)-cyc...)Show SMILES COCc1cc2cc(OCCCCOc3cccc(C[C@H](NC2=O)[C@H](O)CNC2(CC2)c2cccc(c2)C(C)C)c3)n1 |r| Show InChI InChI=1S/C34H43N3O5/c1-23(2)25-9-7-10-27(18-25)34(12-13-34)35-21-31(38)30-17-24-8-6-11-29(16-24)41-14-4-5-15-42-32-20-26(33(39)37-30)19-28(36-32)22-40-3/h6-11,16,18-20,23,30-31,35,38H,4-5,12-15,17,21-22H2,1-3H3,(H,37,39)/t30-,31+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 expressed in Escherichia coli |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus (HCV)) | BDBM50300500
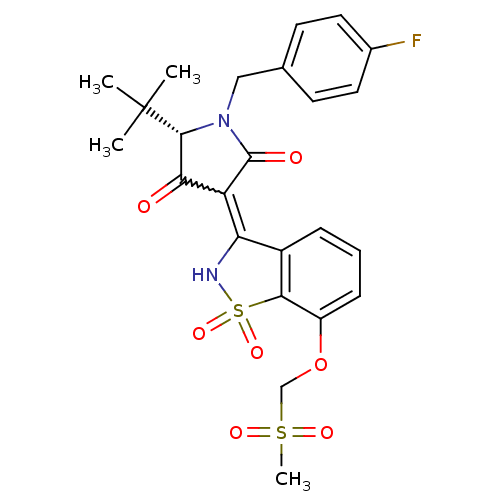
((S)-5-tert-Butyl-1-(4-fluoro-benzyl)-4-hydroxy-3-(...)Show SMILES CC(C)(C)[C@@H]1N(Cc2ccc(F)cc2)C(=O)C(C1=O)=C1NS(=O)(=O)c2c1cccc2OCS(C)(=O)=O |r,w:16.17| Show InChI InChI=1S/C24H25FN2O7S2/c1-24(2,3)22-20(28)18(23(29)27(22)12-14-8-10-15(25)11-9-14)19-16-6-5-7-17(34-13-35(4,30)31)21(16)36(32,33)26-19/h5-11,22,26H,12-13H2,1-4H3/t22-/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Palo Alto LLC
Curated by ChEMBL
| Assay Description
Inhibition of HSV1b con1 NS5B polymerase assessed as [3H]UTP incorporation into acid insoluble RNA product |
Bioorg Med Chem Lett 19: 5652-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.08.022
BindingDB Entry DOI: 10.7270/Q2N016K0 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus (HCV)) | BDBM30491
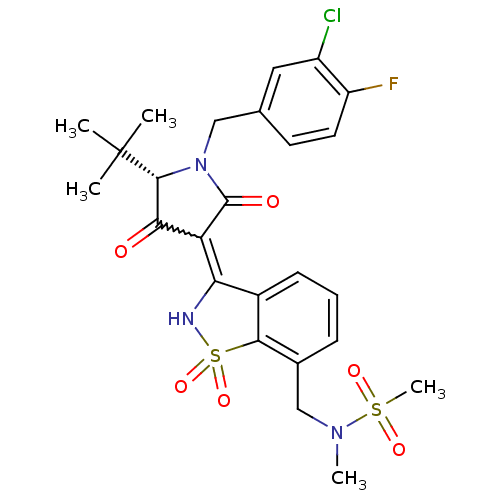
(1,1-dioxoisothiazole analog., 35)Show SMILES CN(Cc1cccc2C(NS(=O)(=O)c12)=C1C(=O)[C@@H](N(Cc2ccc(F)c(Cl)c2)C1=O)C(C)(C)C)S(C)(=O)=O |r,w:14.16| Show InChI InChI=1S/C25H27ClFN3O6S2/c1-25(2,3)23-21(31)19(24(32)30(23)12-14-9-10-18(27)17(26)11-14)20-16-8-6-7-15(13-29(4)37(5,33)34)22(16)38(35,36)28-20/h6-11,23,28H,12-13H2,1-5H3/t23-/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Palo Alto LLC
Curated by ChEMBL
| Assay Description
Inhibition of HSV1b con1 NS5B polymerase assessed as [3H]UTP incorporation into acid insoluble RNA product |
Bioorg Med Chem Lett 19: 5652-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.08.022
BindingDB Entry DOI: 10.7270/Q2N016K0 |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50305537

((S)-4-{(R)-1-Hydroxy-2-[1-(3-isopropyl-phenyl)-cyc...)Show SMILES CC(C)c1cccc(c1)C1(CC1)NC[C@@H](O)[C@@H]1Cc2cccc(OCCCCOc3cc(cc(C)n3)C(=O)N1)c2 |r| Show InChI InChI=1S/C33H41N3O4/c1-22(2)25-9-7-10-27(19-25)33(12-13-33)34-21-30(37)29-18-24-8-6-11-28(17-24)39-14-4-5-15-40-31-20-26(32(38)36-29)16-23(3)35-31/h6-11,16-17,19-20,22,29-30,34,37H,4-5,12-15,18,21H2,1-3H3,(H,36,38)/t29-,30+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin D |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus (HCV)) | BDBM50300506
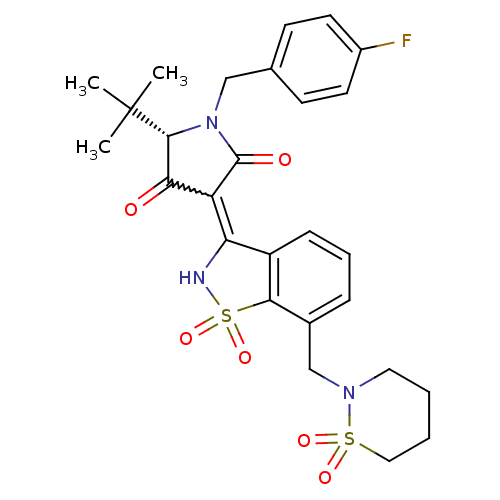
((S)-5-tert-Butyl-3-[7-(1,1-dioxo-1lambda*6*-[1,2]t...)Show SMILES CC(C)(C)[C@@H]1N(Cc2ccc(F)cc2)C(=O)C(C1=O)=C1NS(=O)(=O)c2c1cccc2CN1CCCCS1(=O)=O |r,w:16.17| Show InChI InChI=1S/C27H30FN3O6S2/c1-27(2,3)25-23(32)21(26(33)31(25)15-17-9-11-19(28)12-10-17)22-20-8-6-7-18(24(20)39(36,37)29-22)16-30-13-4-5-14-38(30,34)35/h6-12,25,29H,4-5,13-16H2,1-3H3/t25-/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Palo Alto LLC
Curated by ChEMBL
| Assay Description
Inhibition of HSV1b con1 NS5B polymerase assessed as [3H]UTP incorporation into acid insoluble RNA product |
Bioorg Med Chem Lett 19: 5652-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.08.022
BindingDB Entry DOI: 10.7270/Q2N016K0 |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305527

((4S)-4-[(1R)-1-hydroxy-2-({1-[3-(1-methylethyl)phe...)Show SMILES COCc1cc2NCCCCOc3cccc(C[C@H](NC(=O)c(c1)c2)[C@H](O)CNC1(CC1)c1cccc(c1)C(C)C)c3 |r| Show InChI InChI=1S/C35H45N3O4/c1-24(2)27-9-7-10-29(20-27)35(12-13-35)37-22-33(39)32-19-25-8-6-11-31(18-25)42-15-5-4-14-36-30-17-26(23-41-3)16-28(21-30)34(40)38-32/h6-11,16-18,20-21,24,32-33,36-37,39H,4-5,12-15,19,22-23H2,1-3H3,(H,38,40)/t32-,33+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 expressed in Escherichia coli |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305537

((S)-4-{(R)-1-Hydroxy-2-[1-(3-isopropyl-phenyl)-cyc...)Show SMILES CC(C)c1cccc(c1)C1(CC1)NC[C@@H](O)[C@@H]1Cc2cccc(OCCCCOc3cc(cc(C)n3)C(=O)N1)c2 |r| Show InChI InChI=1S/C33H41N3O4/c1-22(2)25-9-7-10-27(19-25)33(12-13-33)34-21-30(37)29-18-24-8-6-11-28(17-24)39-14-4-5-15-40-31-20-26(32(38)36-29)16-23(3)35-31/h6-11,16-17,19-20,22,29-30,34,37H,4-5,12-15,18,21H2,1-3H3,(H,36,38)/t29-,30+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 expressed in Escherichia coli |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305543
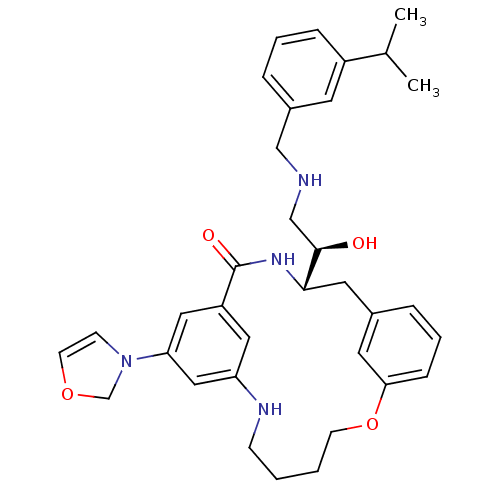
((S)-4-[(R)-1-Hydroxy-2-(3-isopropyl-benzylamino)-e...)Show SMILES CC(C)c1cccc(CNC[C@@H](O)[C@@H]2Cc3cccc(OCCCCNc4cc(cc(c4)C(=O)N2)N2COC=C2)c3)c1 |r,c:40| Show InChI InChI=1S/C34H42N4O4/c1-24(2)27-9-5-8-26(15-27)21-35-22-33(39)32-17-25-7-6-10-31(16-25)42-13-4-3-11-36-29-18-28(34(40)37-32)19-30(20-29)38-12-14-41-23-38/h5-10,12,14-16,18-20,24,32-33,35-36,39H,3-4,11,13,17,21-23H2,1-2H3,(H,37,40)/t32-,33+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 expressed in Escherichia coli |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305527

((4S)-4-[(1R)-1-hydroxy-2-({1-[3-(1-methylethyl)phe...)Show SMILES COCc1cc2NCCCCOc3cccc(C[C@H](NC(=O)c(c1)c2)[C@H](O)CNC1(CC1)c1cccc(c1)C(C)C)c3 |r| Show InChI InChI=1S/C35H45N3O4/c1-24(2)27-9-7-10-29(20-27)35(12-13-35)37-22-33(39)32-19-25-8-6-11-31(18-25)42-15-5-4-14-36-30-17-26(23-41-3)16-28(21-30)34(40)38-32/h6-11,16-18,20-21,24,32-33,36-37,39H,4-5,12-15,19,22-23H2,1-3H3,(H,38,40)/t32-,33+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 expressed in APP-overexpressing CHO cells |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Histone deacetylase 1
(Homo sapiens (Human)) | BDBM50292317
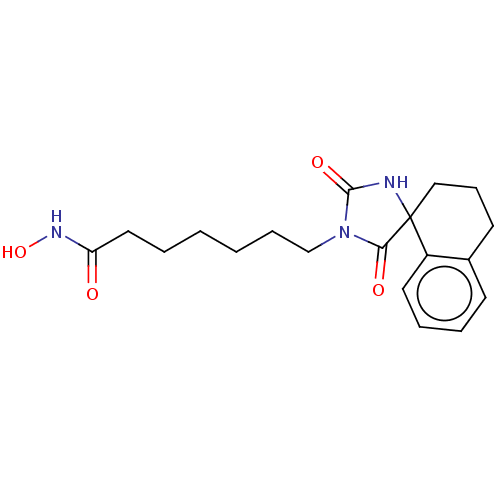
(CHEMBL4172557)Show InChI InChI=1S/C19H25N3O4/c23-16(21-26)11-3-1-2-6-13-22-17(24)19(20-18(22)25)12-7-9-14-8-4-5-10-15(14)19/h4-5,8,10,26H,1-3,6-7,9,11-13H2,(H,20,25)(H,21,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
Minia University
Curated by ChEMBL
| Assay Description
Inhibition of HDAC1 in human HeLa cells after 72 hrs by ELISA |
Eur J Med Chem 146: 79-92 (2018)
Article DOI: 10.1016/j.ejmech.2018.01.021
BindingDB Entry DOI: 10.7270/Q2HD7Z6B |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50294219
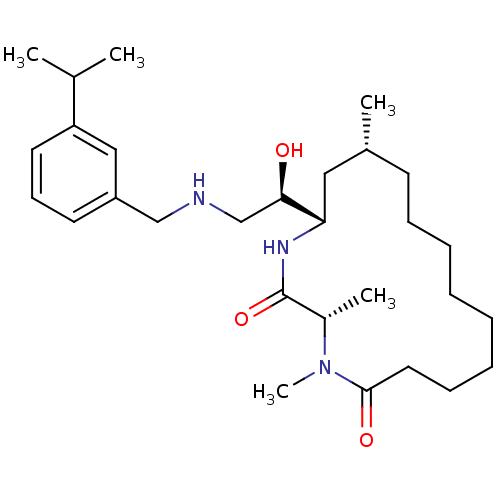
((3S,14R,16S)-16-((R)-1-hydroxy-2-(3-isopropylbenzy...)Show SMILES CC(C)c1cccc(CNC[C@@H](O)[C@@H]2C[C@H](C)CCCCCCCCC(=O)N(C)[C@@H](C)C(=O)N2)c1 |r| Show InChI InChI=1S/C29H49N3O3/c1-21(2)25-15-12-14-24(18-25)19-30-20-27(33)26-17-22(3)13-10-8-6-7-9-11-16-28(34)32(5)23(4)29(35)31-26/h12,14-15,18,21-23,26-27,30,33H,6-11,13,16-17,19-20H2,1-5H3,(H,31,35)/t22-,23+,26+,27-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
| MMDB
PDB
Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 |
Bioorg Med Chem Lett 19: 1366-70 (2009)
Article DOI: 10.1016/j.bmcl.2009.01.055
BindingDB Entry DOI: 10.7270/Q2SB45S3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305534
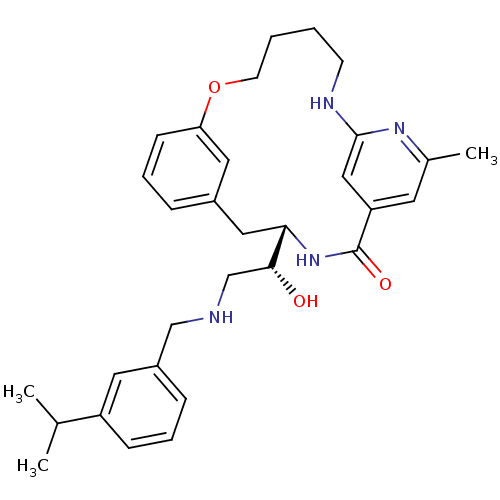
((S)-4-[(R)-1-Hydroxy-2-(3-isopropyl-benzylamino)-e...)Show SMILES CC(C)c1cccc(CNC[C@@H](O)[C@@H]2Cc3cccc(OCCCCNc4cc(cc(C)n4)C(=O)N2)c3)c1 |r| Show InChI InChI=1S/C31H40N4O3/c1-21(2)25-10-6-9-24(15-25)19-32-20-29(36)28-17-23-8-7-11-27(16-23)38-13-5-4-12-33-30-18-26(31(37)35-28)14-22(3)34-30/h6-11,14-16,18,21,28-29,32,36H,4-5,12-13,17,19-20H2,1-3H3,(H,33,34)(H,35,37)/t28-,29+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 expressed in Escherichia coli |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Integrin alpha-IIb/beta-3
(Homo sapiens (Human)) | BDBM50075566
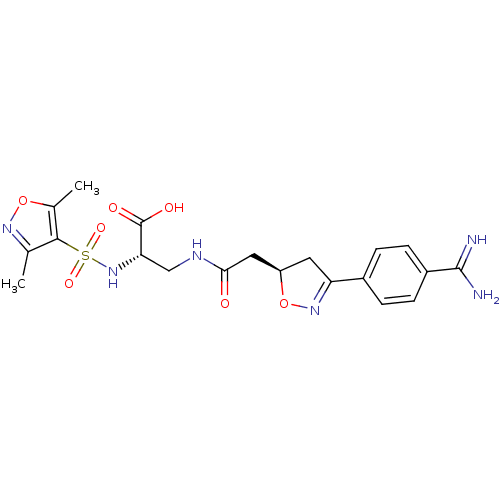
((S)-3-{2-[(R)-3-(4-Carbamimidoyl-phenyl)-4,5-dihyd...)Show SMILES Cc1noc(C)c1S(=O)(=O)N[C@@H](CNC(=O)C[C@H]1CC(=NO1)c1ccc(cc1)C(N)=N)C(O)=O |c:20| Show InChI InChI=1S/C20H24N6O7S/c1-10-18(11(2)32-24-10)34(30,31)26-16(20(28)29)9-23-17(27)8-14-7-15(25-33-14)12-3-5-13(6-4-12)19(21)22/h3-6,14,16,26H,7-9H2,1-2H3,(H3,21,22)(H,23,27)(H,28,29)/t14-,16+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 29 | n/a | n/a | n/a | n/a | n/a | n/a |
The DuPont Merck Pharmaceutical Company
Curated by ChEMBL
| Assay Description
Inhibitory activity against Fibrinogen Receptor |
J Med Chem 42: 1178-92 (1999)
Article DOI: 10.1021/jm980348t
BindingDB Entry DOI: 10.7270/Q2WD3ZQZ |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM13531
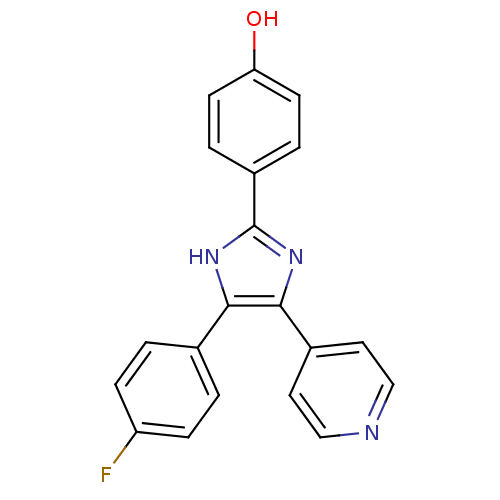
(4-(4-Fluorophenyl)-2-(4-hydroxyphenyl)-5-(4-pyridy...)Show SMILES Oc1ccc(cc1)-c1nc(c([nH]1)-c1ccc(F)cc1)-c1ccncc1 Show InChI InChI=1S/C20H14FN3O/c21-16-5-1-13(2-6-16)18-19(14-9-11-22-12-10-14)24-20(23-18)15-3-7-17(25)8-4-15/h1-12,25H,(H,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Palo Alto LLC
| Assay Description
Inhibition of human recombinant active p38alpha MAP kinase was tested by measuring the incorporation of 33P from gamma-[33P] ATP into myelin basic pr... |
J Med Chem 49: 1562-75 (2006)
Article DOI: 10.1021/jm050736c
BindingDB Entry DOI: 10.7270/Q23776ZN |
More data for this
Ligand-Target Pair | |
Histone deacetylase 1
(Homo sapiens (Human)) | BDBM19149

(CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...)Show InChI InChI=1S/C14H20N2O3/c17-13(15-12-8-4-3-5-9-12)10-6-1-2-7-11-14(18)16-19/h3-5,8-9,19H,1-2,6-7,10-11H2,(H,15,17)(H,16,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
Minia University
Curated by ChEMBL
| Assay Description
Inhibition of HDAC1 in human HeLa cells after 72 hrs by ELISA |
Eur J Med Chem 146: 79-92 (2018)
Article DOI: 10.1016/j.ejmech.2018.01.021
BindingDB Entry DOI: 10.7270/Q2HD7Z6B |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Histone deacetylase 6
(Homo sapiens (Human)) | BDBM19149

(CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...)Show InChI InChI=1S/C14H20N2O3/c17-13(15-12-8-4-3-5-9-12)10-6-1-2-7-11-14(18)16-19/h3-5,8-9,19H,1-2,6-7,10-11H2,(H,15,17)(H,16,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
Minia University
Curated by ChEMBL
| Assay Description
Inhibition of HDAC6 in human HeLa cells after 72 hrs by ELISA |
Eur J Med Chem 146: 79-92 (2018)
Article DOI: 10.1016/j.ejmech.2018.01.021
BindingDB Entry DOI: 10.7270/Q2HD7Z6B |
More data for this
Ligand-Target Pair | |
Histone deacetylase 1
(Homo sapiens (Human)) | BDBM50292331
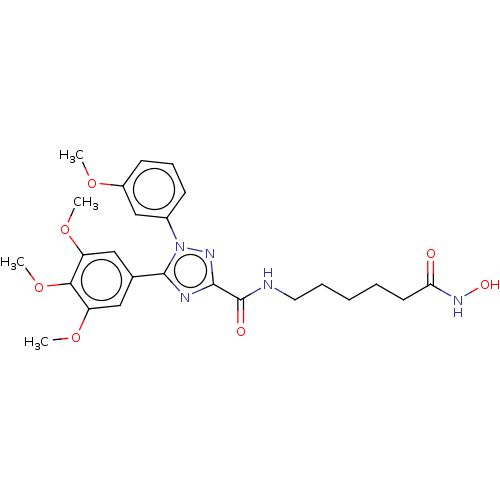
(CHEMBL4168150)Show SMILES COc1cccc(c1)-n1nc(nc1-c1cc(OC)c(OC)c(OC)c1)C(=O)NCCCCCC(=O)NO Show InChI InChI=1S/C25H31N5O7/c1-34-18-10-8-9-17(15-18)30-24(16-13-19(35-2)22(37-4)20(14-16)36-3)27-23(28-30)25(32)26-12-7-5-6-11-21(31)29-33/h8-10,13-15,33H,5-7,11-12H2,1-4H3,(H,26,32)(H,29,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 38 | n/a | n/a | n/a | n/a | n/a | n/a |
Minia University
Curated by ChEMBL
| Assay Description
Inhibition of HDAC1 in human HeLa cells after 72 hrs by ELISA |
Eur J Med Chem 146: 79-92 (2018)
Article DOI: 10.1016/j.ejmech.2018.01.021
BindingDB Entry DOI: 10.7270/Q2HD7Z6B |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305533

((S)-4-[(R)-1-Hydroxy-2-(3-isopropyl-benzylamino)-e...)Show SMILES CC(C)c1cccc(CNC[C@@H](O)[C@@H]2Cc3cccc(OCCCCOc4cc(cc(C)n4)C(=O)N2)c3)c1 |r| Show InChI InChI=1S/C31H39N3O4/c1-21(2)25-10-6-9-24(15-25)19-32-20-29(35)28-17-23-8-7-11-27(16-23)37-12-4-5-13-38-30-18-26(31(36)34-28)14-22(3)33-30/h6-11,14-16,18,21,28-29,32,35H,4-5,12-13,17,19-20H2,1-3H3,(H,34,36)/t28-,29+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 42 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 expressed in Escherichia coli |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus (HCV)) | BDBM50300495

(CHEMBL572718 | N-{3-[(S)-5-tert-Butyl-1-(4-fluoro-...)Show SMILES CC(C)(C)[C@@H]1N(Cc2ccc(F)cc2)C(=O)C(C1=O)=C1NS(=O)(=O)c2cc(CNS(C)(=O)=O)ccc12 |r,w:16.17| Show InChI InChI=1S/C24H26FN3O6S2/c1-24(2,3)22-21(29)19(23(30)28(22)13-14-5-8-16(25)9-6-14)20-17-10-7-15(12-26-35(4,31)32)11-18(17)36(33,34)27-20/h5-11,22,26-27H,12-13H2,1-4H3/t22-/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 43 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Palo Alto LLC
Curated by ChEMBL
| Assay Description
Inhibition of HSV1b con1 NS5B polymerase assessed as [3H]UTP incorporation into acid insoluble RNA product |
Bioorg Med Chem Lett 19: 5652-6 (2009)
Article DOI: 10.1016/j.bmcl.2009.08.022
BindingDB Entry DOI: 10.7270/Q2N016K0 |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50294219

((3S,14R,16S)-16-((R)-1-hydroxy-2-(3-isopropylbenzy...)Show SMILES CC(C)c1cccc(CNC[C@@H](O)[C@@H]2C[C@H](C)CCCCCCCCC(=O)N(C)[C@@H](C)C(=O)N2)c1 |r| Show InChI InChI=1S/C29H49N3O3/c1-21(2)25-15-12-14-24(18-25)19-30-20-27(33)26-17-22(3)13-10-8-6-7-9-11-16-28(34)32(5)23(4)29(35)31-26/h12,14-15,18,21-23,26-27,30,33H,6-11,13,16-17,19-20H2,1-5H3,(H,31,35)/t22-,23+,26+,27-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
| MMDB
PDB
Article
PubMed
| n/a | n/a | 45 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 expressed in CHO cells expressing human recombinant APP assessed as amyloid beta40 aggregation |
Bioorg Med Chem Lett 19: 1366-70 (2009)
Article DOI: 10.1016/j.bmcl.2009.01.055
BindingDB Entry DOI: 10.7270/Q2SB45S3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305536

((S)-4-{(R)-1-Hydroxy-2-[1-(3-isopropyl-phenyl)-cyc...)Show SMILES COCc1cc2cc(OCCCCOc3cccc(C[C@H](NC2=O)[C@H](O)CNC2(CC2)c2cccc(c2)C(C)C)c3)n1 |r| Show InChI InChI=1S/C34H43N3O5/c1-23(2)25-9-7-10-27(18-25)34(12-13-34)35-21-31(38)30-17-24-8-6-11-29(16-24)41-14-4-5-15-42-32-20-26(33(39)37-30)19-28(36-32)22-40-3/h6-11,16,18-20,23,30-31,35,38H,4-5,12-15,17,21-22H2,1-3H3,(H,37,39)/t30-,31+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 47 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 expressed in APP-overexpressing CHO cells |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305528
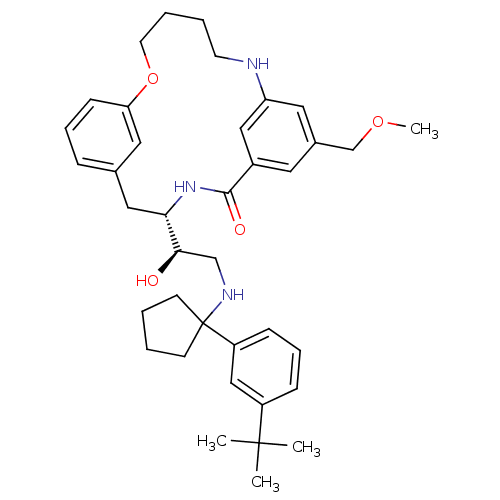
((S)-4-{(R)-2-[1-(3-tert-Butyl-phenyl)-cyclopentyla...)Show SMILES COCc1cc2NCCCCOc3cccc(C[C@H](NC(=O)c(c1)c2)[C@H](O)CNC1(CCCC1)c1cccc(c1)C(C)(C)C)c3 |r| Show InChI InChI=1S/C38H51N3O4/c1-37(2,3)30-12-10-13-31(24-30)38(15-5-6-16-38)40-25-35(42)34-22-27-11-9-14-33(21-27)45-18-8-7-17-39-32-20-28(26-44-4)19-29(23-32)36(43)41-34/h9-14,19-21,23-24,34-35,39-40,42H,5-8,15-18,22,25-26H2,1-4H3,(H,41,43)/t34-,35+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 expressed in Escherichia coli |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305530

((S)-4-{(R)-1-Hydroxy-2-[1-(3-isopropyl-phenyl)-1-m...)Show SMILES COCc1cc2NCCCCOc3cccc(C[C@H](NC(=O)c(c1)c2)[C@H](O)CNC(C)(C)c1cccc(c1)C(C)C)c3 |r| Show InChI InChI=1S/C35H47N3O4/c1-24(2)27-11-9-12-29(20-27)35(3,4)37-22-33(39)32-19-25-10-8-13-31(18-25)42-15-7-6-14-36-30-17-26(23-41-5)16-28(21-30)34(40)38-32/h8-13,16-18,20-21,24,32-33,36-37,39H,6-7,14-15,19,22-23H2,1-5H3,(H,38,40)/t32-,33+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 expressed in Escherichia coli |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305533

((S)-4-[(R)-1-Hydroxy-2-(3-isopropyl-benzylamino)-e...)Show SMILES CC(C)c1cccc(CNC[C@@H](O)[C@@H]2Cc3cccc(OCCCCOc4cc(cc(C)n4)C(=O)N2)c3)c1 |r| Show InChI InChI=1S/C31H39N3O4/c1-21(2)25-10-6-9-24(15-25)19-32-20-29(35)28-17-23-8-7-11-27(16-23)37-12-4-5-13-38-30-18-26(31(36)34-28)14-22(3)33-30/h6-11,14-16,18,21,28-29,32,35H,4-5,12-13,17,19-20H2,1-3H3,(H,34,36)/t28-,29+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 55 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 expressed in APP-overexpressing CHO cells |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Histone deacetylase 1
(Homo sapiens (Human)) | BDBM50292318
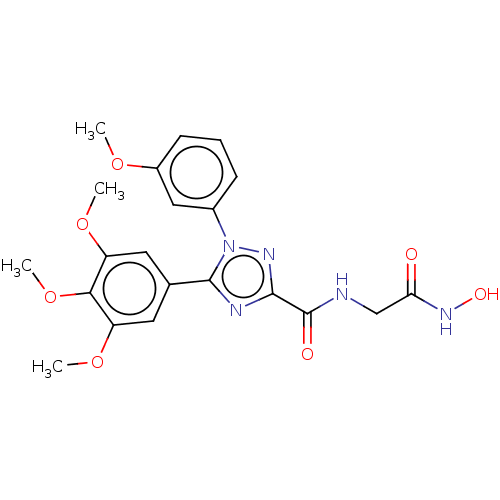
(CHEMBL4174925)Show SMILES COc1cccc(c1)-n1nc(nc1-c1cc(OC)c(OC)c(OC)c1)C(=O)NCC(=O)NO Show InChI InChI=1S/C21H23N5O7/c1-30-14-7-5-6-13(10-14)26-20(23-19(24-26)21(28)22-11-17(27)25-29)12-8-15(31-2)18(33-4)16(9-12)32-3/h5-10,29H,11H2,1-4H3,(H,22,28)(H,25,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 58 | n/a | n/a | n/a | n/a | n/a | n/a |
Minia University
Curated by ChEMBL
| Assay Description
Inhibition of HDAC1 in human HeLa cells after 72 hrs by ELISA |
Eur J Med Chem 146: 79-92 (2018)
Article DOI: 10.1016/j.ejmech.2018.01.021
BindingDB Entry DOI: 10.7270/Q2HD7Z6B |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305535
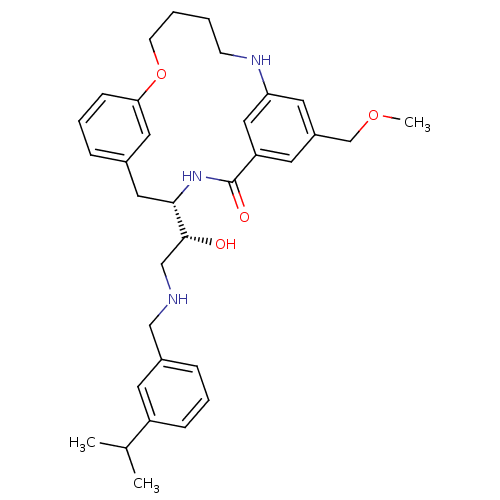
((S)-4-[(R)-1-Hydroxy-2-(3-isopropyl-benzylamino)-e...)Show SMILES COCc1cc2NCCCCOc3cccc(C[C@H](NC(=O)c(c1)c2)[C@H](O)CNCc1cccc(c1)C(C)C)c3 |r| Show InChI InChI=1S/C33H43N3O4/c1-23(2)27-10-6-9-25(14-27)20-34-21-32(37)31-18-24-8-7-11-30(17-24)40-13-5-4-12-35-29-16-26(22-39-3)15-28(19-29)33(38)36-31/h6-11,14-17,19,23,31-32,34-35,37H,4-5,12-13,18,20-22H2,1-3H3,(H,36,38)/t31-,32+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 expressed in Escherichia coli |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305529
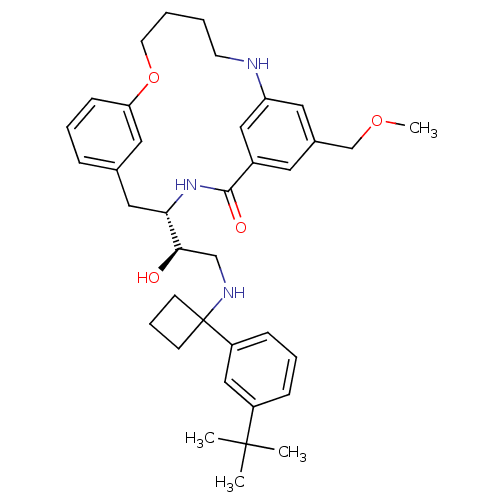
((S)-4-{(R)-2-[1-(3-tert-Butyl-phenyl)-cyclobutylam...)Show SMILES COCc1cc2NCCCCOc3cccc(C[C@H](NC(=O)c(c1)c2)[C@H](O)CNC1(CCC1)c1cccc(c1)C(C)(C)C)c3 |r| Show InChI InChI=1S/C37H49N3O4/c1-36(2,3)29-11-8-12-30(23-29)37(14-9-15-37)39-24-34(41)33-21-26-10-7-13-32(20-26)44-17-6-5-16-38-31-19-27(25-43-4)18-28(22-31)35(42)40-33/h7-8,10-13,18-20,22-23,33-34,38-39,41H,5-6,9,14-17,21,24-25H2,1-4H3,(H,40,42)/t33-,34+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 67 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 expressed in Escherichia coli |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305537

((S)-4-{(R)-1-Hydroxy-2-[1-(3-isopropyl-phenyl)-cyc...)Show SMILES CC(C)c1cccc(c1)C1(CC1)NC[C@@H](O)[C@@H]1Cc2cccc(OCCCCOc3cc(cc(C)n3)C(=O)N1)c2 |r| Show InChI InChI=1S/C33H41N3O4/c1-22(2)25-9-7-10-27(19-25)33(12-13-33)34-21-30(37)29-18-24-8-6-11-28(17-24)39-14-4-5-15-40-31-20-26(32(38)36-29)16-23(3)35-31/h6-11,16-17,19-20,22,29-30,34,37H,4-5,12-15,18,21H2,1-3H3,(H,36,38)/t29-,30+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 expressed in APP-overexpressing CHO cells |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Histone deacetylase 2
(Homo sapiens (Human)) | BDBM19149

(CHEMBL98 | N-hydroxy-N'-phenyloctanediamide | SAHA...)Show InChI InChI=1S/C14H20N2O3/c17-13(15-12-8-4-3-5-9-12)10-6-1-2-7-11-14(18)16-19/h3-5,8-9,19H,1-2,6-7,10-11H2,(H,15,17)(H,16,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 81 | n/a | n/a | n/a | n/a | n/a | n/a |
Minia University
Curated by ChEMBL
| Assay Description
Inhibition of VLA-4 from HL60 lysate in a protein-based ligand binding assay. |
Eur J Med Chem 146: 79-92 (2018)
Article DOI: 10.1016/j.ejmech.2018.01.021
BindingDB Entry DOI: 10.7270/Q2HD7Z6B |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305540
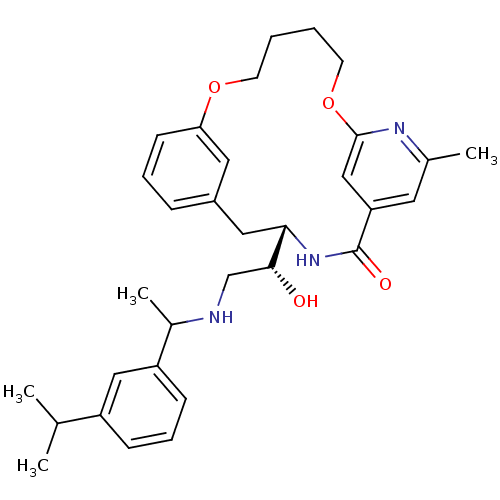
((S)-4-{(R)-1-Hydroxy-2-[1-(3-isopropyl-phenyl)-eth...)Show SMILES CC(C)c1cccc(c1)C(C)NC[C@@H](O)[C@@H]1Cc2cccc(OCCCCOc3cc(cc(C)n3)C(=O)N1)c2 |r| Show InChI InChI=1S/C32H41N3O4/c1-21(2)25-10-8-11-26(18-25)23(4)33-20-30(36)29-17-24-9-7-12-28(16-24)38-13-5-6-14-39-31-19-27(32(37)35-29)15-22(3)34-31/h7-12,15-16,18-19,21,23,29-30,33,36H,5-6,13-14,17,20H2,1-4H3,(H,35,37)/t23?,29-,30+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 83 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 expressed in Escherichia coli |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data