 Found 288 hits with Last Name = 'trapp' and Initial = 's'
Found 288 hits with Last Name = 'trapp' and Initial = 's' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50102780

(2-(2-Hydroxy-3-phenyl-phenyl)-1H-indole-5-carboxam...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cccc(-c2ccccc2)c1O Show InChI InChI=1S/C21H17N3O/c22-21(23)14-9-10-18-15(11-14)12-19(24-18)17-8-4-7-16(20(17)25)13-5-2-1-3-6-13/h1-12,24-25H,(H3,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibitory concentration against Human Serine Protease Urokinase Plasminogen Activator |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase mTOR
(Homo sapiens (Human)) | BDBM50398399
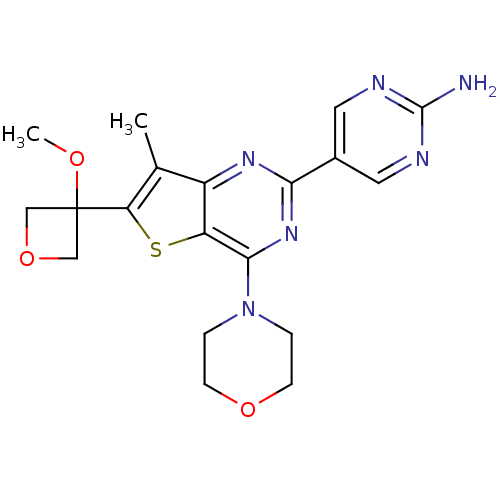
(CHEMBL2178608)Show SMILES COC1(COC1)c1sc2c(nc(nc2c1C)-c1cnc(N)nc1)N1CCOCC1 Show InChI InChI=1S/C19H22N6O3S/c1-11-13-14(29-15(11)19(26-2)9-28-10-19)17(25-3-5-27-6-4-25)24-16(23-13)12-7-21-18(20)22-8-12/h7-8H,3-6,9-10H2,1-2H3,(H2,20,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant mTOR (1360 to 2549 amino acids) assessed as reduction in phosphorylation of (GFP)-4-EBP1 after 30 mins by FRET assay |
J Med Chem 55: 8007-20 (2012)
Article DOI: 10.1021/jm300867c
BindingDB Entry DOI: 10.7270/Q2MC9152 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase mTOR
(Homo sapiens (Human)) | BDBM50398400
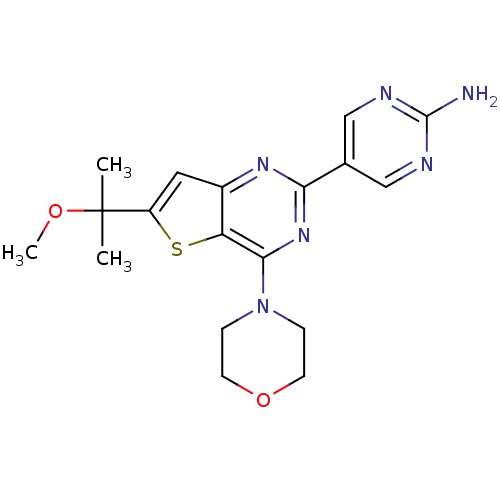
(CHEMBL2178606)Show SMILES COC(C)(C)c1cc2nc(nc(N3CCOCC3)c2s1)-c1cnc(N)nc1 Show InChI InChI=1S/C18H22N6O2S/c1-18(2,25-3)13-8-12-14(27-13)16(24-4-6-26-7-5-24)23-15(22-12)11-9-20-17(19)21-10-11/h8-10H,4-7H2,1-3H3,(H2,19,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant mTOR (1360 to 2549 amino acids) assessed as reduction in phosphorylation of (GFP)-4-EBP1 after 30 mins by FRET assay |
J Med Chem 55: 8007-20 (2012)
Article DOI: 10.1021/jm300867c
BindingDB Entry DOI: 10.7270/Q2MC9152 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase mTOR
(Homo sapiens (Human)) | BDBM50398398

(CHEMBL2178613)Show SMILES CC1(C)OCCc2c1sc1c(nc(nc21)-c1cnc(N)nc1)N1CCOCC1 Show InChI InChI=1S/C19H22N6O2S/c1-19(2)15-12(3-6-27-19)13-14(28-15)17(25-4-7-26-8-5-25)24-16(23-13)11-9-21-18(20)22-10-11/h9-10H,3-8H2,1-2H3,(H2,20,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant mTOR (1360 to 2549 amino acids) assessed as reduction in phosphorylation of (GFP)-4-EBP1 after 30 mins by FRET assay |
J Med Chem 55: 8007-20 (2012)
Article DOI: 10.1021/jm300867c
BindingDB Entry DOI: 10.7270/Q2MC9152 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50102790

(2-(2-Hydroxy-5-nitro-biphenyl-3-yl)-1H-indole-5-ca...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(cc(c1O)-c1ccccc1)[N+]([O-])=O Show InChI InChI=1S/C21H16N4O3/c22-21(23)13-6-7-18-14(8-13)9-19(24-18)17-11-15(25(27)28)10-16(20(17)26)12-4-2-1-3-5-12/h1-11,24,26H,(H3,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Tissue-type plasminogen activator
(Homo sapiens (Human)) | BDBM50102790

(2-(2-Hydroxy-5-nitro-biphenyl-3-yl)-1H-indole-5-ca...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(cc(c1O)-c1ccccc1)[N+]([O-])=O Show InChI InChI=1S/C21H16N4O3/c22-21(23)13-6-7-18-14(8-13)9-19(24-18)17-11-15(25(27)28)10-16(20(17)26)12-4-2-1-3-5-12/h1-11,24,26H,(H3,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Human Serine Protease tissue type Plasminogen Activator (t-PA). |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase mTOR
(Homo sapiens (Human)) | BDBM50398397
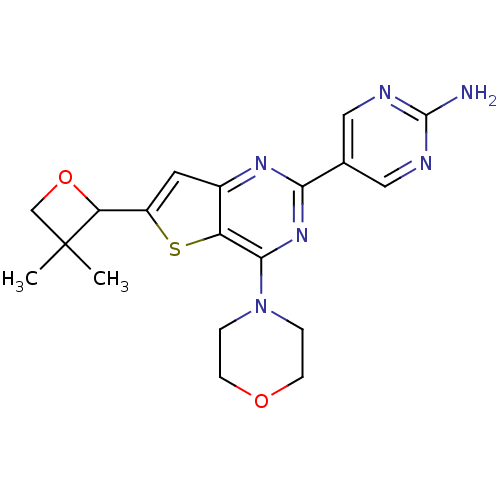
(CHEMBL2178614)Show SMILES CC1(C)COC1c1cc2nc(nc(N3CCOCC3)c2s1)-c1cnc(N)nc1 Show InChI InChI=1S/C19H22N6O2S/c1-19(2)10-27-15(19)13-7-12-14(28-13)17(25-3-5-26-6-4-25)24-16(23-12)11-8-21-18(20)22-9-11/h7-9,15H,3-6,10H2,1-2H3,(H2,20,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant mTOR (1360 to 2549 amino acids) assessed as reduction in phosphorylation of (GFP)-4-EBP1 after 30 mins by FRET assay |
J Med Chem 55: 8007-20 (2012)
Article DOI: 10.1021/jm300867c
BindingDB Entry DOI: 10.7270/Q2MC9152 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50102790

(2-(2-Hydroxy-5-nitro-biphenyl-3-yl)-1H-indole-5-ca...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(cc(c1O)-c1ccccc1)[N+]([O-])=O Show InChI InChI=1S/C21H16N4O3/c22-21(23)13-6-7-18-14(8-13)9-19(24-18)17-11-15(25(27)28)10-16(20(17)26)12-4-2-1-3-5-12/h1-11,24,26H,(H3,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
ComInhibition of Human Serine Protease Urokinase Plasminogen Activator (u-PA). |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase mTOR
(Homo sapiens (Human)) | BDBM50312613
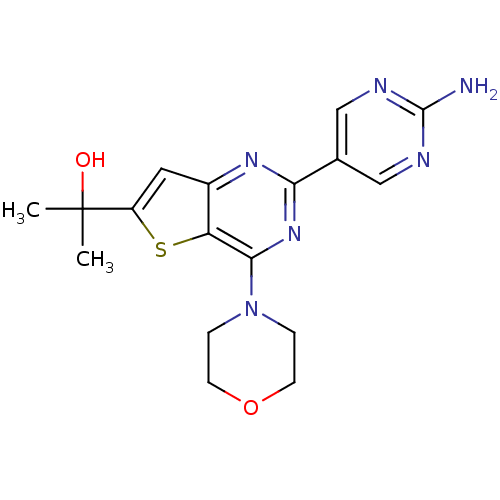
(2-(2-(2-Aminopyrimidin-5-yl)-4-morpholinothieno[3,...)Show SMILES CC(C)(O)c1cc2nc(nc(N3CCOCC3)c2s1)-c1cnc(N)nc1 Show InChI InChI=1S/C17H20N6O2S/c1-17(2,24)12-7-11-13(26-12)15(23-3-5-25-6-4-23)22-14(21-11)10-8-19-16(18)20-9-10/h7-9,24H,3-6H2,1-2H3,(H2,18,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant mTOR (1360 to 2549 amino acids) assessed as reduction in phosphorylation of (GFP)-4-EBP1 after 30 mins by FRET assay |
J Med Chem 55: 8007-20 (2012)
Article DOI: 10.1021/jm300867c
BindingDB Entry DOI: 10.7270/Q2MC9152 |
More data for this
Ligand-Target Pair | |
Tissue-type plasminogen activator
(Homo sapiens (Human)) | BDBM50102780

(2-(2-Hydroxy-3-phenyl-phenyl)-1H-indole-5-carboxam...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cccc(-c2ccccc2)c1O Show InChI InChI=1S/C21H17N3O/c22-21(23)14-9-10-18-15(11-14)12-19(24-18)17-8-4-7-16(20(17)25)13-5-2-1-3-6-13/h1-12,24-25H,(H3,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 35 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inihibtion of Human Serine Protease tissue type Plasminogen Activator |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50102767

(2-(3-Bromo-2-hydroxy-phenyl)-1H-indole-5-carboxami...)Show InChI InChI=1S/C15H12BrN3O/c16-11-3-1-2-10(14(11)20)13-7-9-6-8(15(17)18)4-5-12(9)19-13/h1-7,19-20H,(H3,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 35 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibitory concentration against Human Serine Protease Urokinase Plasminogen Activator |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50101866
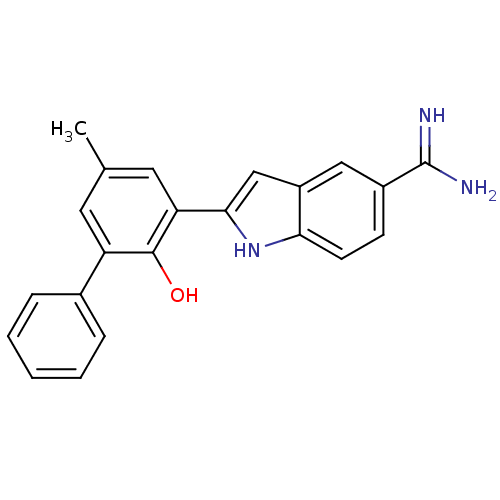
(2-(2-Hydroxy-5-methyl-biphenyl-3-yl)-1H-indole-5-c...)Show SMILES Cc1cc(-c2cc3cc(ccc3[nH]2)C(N)=N)c(O)c(c1)-c1ccccc1 Show InChI InChI=1S/C22H19N3O/c1-13-9-17(14-5-3-2-4-6-14)21(26)18(10-13)20-12-16-11-15(22(23)24)7-8-19(16)25-20/h2-12,25-26H,1H3,(H3,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 38 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibitory concentration against Human Serine Protease Urokinase Plasminogen Activator |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50102767

(2-(3-Bromo-2-hydroxy-phenyl)-1H-indole-5-carboxami...)Show InChI InChI=1S/C15H12BrN3O/c16-11-3-1-2-10(14(11)20)13-7-9-6-8(15(17)18)4-5-12(9)19-13/h1-7,19-20H,(H3,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 43 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50102786
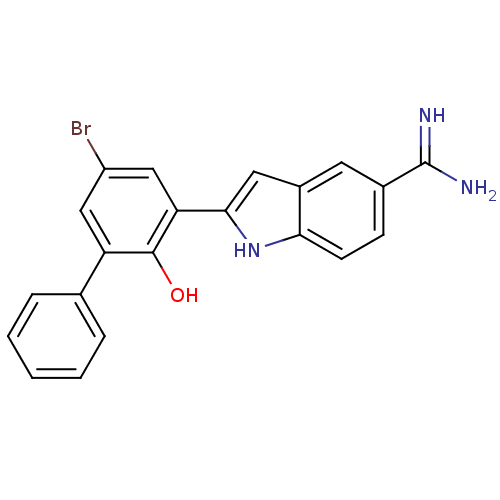
(2-(5-Bromo-2-hydroxy-biphenyl-3-yl)-1H-indole-5-ca...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(Br)cc(-c2ccccc2)c1O Show InChI InChI=1S/C21H16BrN3O/c22-15-10-16(12-4-2-1-3-5-12)20(26)17(11-15)19-9-14-8-13(21(23)24)6-7-18(14)25-19/h1-11,25-26H,(H3,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 43 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibitory concentration against Human Serine Protease Urokinase Plasminogen Activator |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50101873
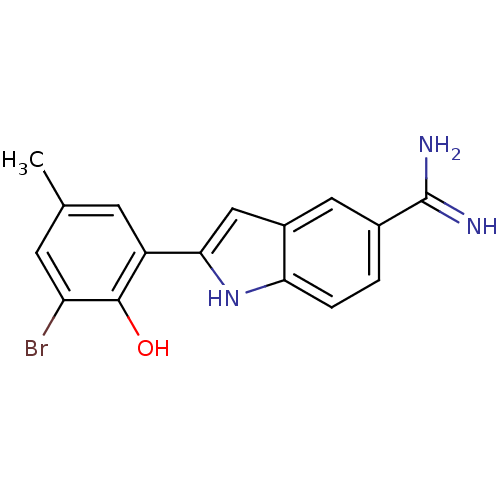
(2-(3-Bromo-2-hydroxy-5-methyl-phenyl)-1H-indole-5-...)Show SMILES Cc1cc(Br)c(O)c(c1)-c1cc2cc(ccc2[nH]1)C(N)=N Show InChI InChI=1S/C16H14BrN3O/c1-8-4-11(15(21)12(17)5-8)14-7-10-6-9(16(18)19)2-3-13(10)20-14/h2-7,20-21H,1H3,(H3,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 52 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50102778
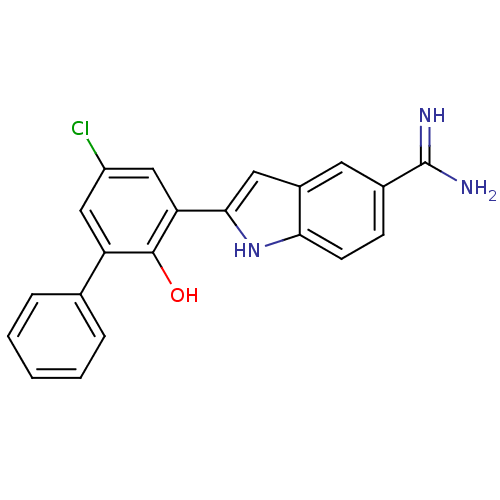
(2-(5-Chloro-2-hydroxy-biphenyl-3-yl)-1H-indole-5-c...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(Cl)cc(-c2ccccc2)c1O Show InChI InChI=1S/C21H16ClN3O/c22-15-10-16(12-4-2-1-3-5-12)20(26)17(11-15)19-9-14-8-13(21(23)24)6-7-18(14)25-19/h1-11,25-26H,(H3,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibitory concentration against Human Serine Protease Urokinase Plasminogen Activator |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50101873

(2-(3-Bromo-2-hydroxy-5-methyl-phenyl)-1H-indole-5-...)Show SMILES Cc1cc(Br)c(O)c(c1)-c1cc2cc(ccc2[nH]1)C(N)=N Show InChI InChI=1S/C16H14BrN3O/c1-8-4-11(15(21)12(17)5-8)14-7-10-6-9(16(18)19)2-3-13(10)20-14/h2-7,20-21H,1H3,(H3,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
ComInhibition of Human Serine Protease Urokinase Plasminogen Activator (u-PA). |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50102790

(2-(2-Hydroxy-5-nitro-biphenyl-3-yl)-1H-indole-5-ca...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(cc(c1O)-c1ccccc1)[N+]([O-])=O Show InChI InChI=1S/C21H16N4O3/c22-21(23)13-6-7-18-14(8-13)9-19(24-18)17-11-15(25(27)28)10-16(20(17)26)12-4-2-1-3-5-12/h1-11,24,26H,(H3,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Human Serine Protease Trypsin. |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50102780

(2-(2-Hydroxy-3-phenyl-phenyl)-1H-indole-5-carboxam...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cccc(-c2ccccc2)c1O Show InChI InChI=1S/C21H17N3O/c22-21(23)14-9-10-18-15(11-14)12-19(24-18)17-8-4-7-16(20(17)25)13-5-2-1-3-6-13/h1-12,24-25H,(H3,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 78 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50102792
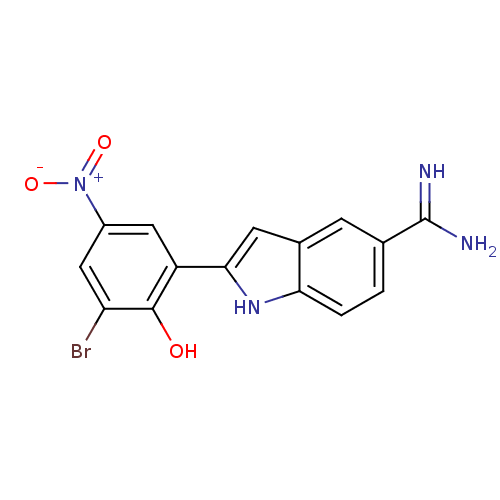
(2-(2-Hydroxy-3-bromo-5-nitro-phenyl)-1H-indole-5-c...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(cc(Br)c1O)[N+]([O-])=O Show InChI InChI=1S/C15H11BrN4O3/c16-11-6-9(20(22)23)5-10(14(11)21)13-4-8-3-7(15(17)18)1-2-12(8)19-13/h1-6,19,21H,(H3,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 78 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50102784
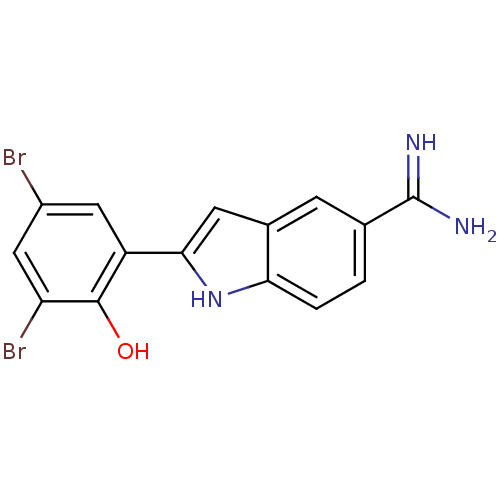
(2-(3,5-Dibromo-2-hydroxy-phenyl)-1H-indole-5-carbo...)Show InChI InChI=1S/C15H11Br2N3O/c16-9-5-10(14(21)11(17)6-9)13-4-8-3-7(15(18)19)1-2-12(8)20-13/h1-6,20-21H,(H3,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 89 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Tissue-type plasminogen activator
(Homo sapiens (Human)) | BDBM50102786

(2-(5-Bromo-2-hydroxy-biphenyl-3-yl)-1H-indole-5-ca...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(Br)cc(-c2ccccc2)c1O Show InChI InChI=1S/C21H16BrN3O/c22-15-10-16(12-4-2-1-3-5-12)20(26)17(11-15)19-9-14-8-13(21(23)24)6-7-18(14)25-19/h1-11,25-26H,(H3,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 94 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inihibtion of Human Serine Protease tissue type Plasminogen Activator |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Plasminogen
(Homo sapiens (Human)) | BDBM50102780

(2-(2-Hydroxy-3-phenyl-phenyl)-1H-indole-5-carboxam...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cccc(-c2ccccc2)c1O Show InChI InChI=1S/C21H17N3O/c22-21(23)14-9-10-18-15(11-14)12-19(24-18)17-8-4-7-16(20(17)25)13-5-2-1-3-6-13/h1-12,24-25H,(H3,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Human Serine Protease Plasmin |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50102784

(2-(3,5-Dibromo-2-hydroxy-phenyl)-1H-indole-5-carbo...)Show InChI InChI=1S/C15H11Br2N3O/c16-9-5-10(14(21)11(17)6-9)13-4-8-3-7(15(18)19)1-2-12(8)20-13/h1-6,20-21H,(H3,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibitory concentration against Human Serine Protease Urokinase Plasminogen Activator |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50102790

(2-(2-Hydroxy-5-nitro-biphenyl-3-yl)-1H-indole-5-ca...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(cc(c1O)-c1ccccc1)[N+]([O-])=O Show InChI InChI=1S/C21H16N4O3/c22-21(23)13-6-7-18-14(8-13)9-19(24-18)17-11-15(25(27)28)10-16(20(17)26)12-4-2-1-3-5-12/h1-11,24,26H,(H3,22,23) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Human Serine Protease Thrombin. |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Tissue-type plasminogen activator
(Homo sapiens (Human)) | BDBM50101866

(2-(2-Hydroxy-5-methyl-biphenyl-3-yl)-1H-indole-5-c...)Show SMILES Cc1cc(-c2cc3cc(ccc3[nH]2)C(N)=N)c(O)c(c1)-c1ccccc1 Show InChI InChI=1S/C22H19N3O/c1-13-9-17(14-5-3-2-4-6-14)21(26)18(10-13)20-12-16-11-15(22(23)24)7-8-19(16)25-20/h2-12,25-26H,1H3,(H3,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inihibtion of Human Serine Protease tissue type Plasminogen Activator |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50102767

(2-(3-Bromo-2-hydroxy-phenyl)-1H-indole-5-carboxami...)Show InChI InChI=1S/C15H12BrN3O/c16-11-3-1-2-10(14(11)20)13-7-9-6-8(15(17)18)4-5-12(9)19-13/h1-7,19-20H,(H3,17,18) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Activity against Human Serine Protease Thrombin |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50101866

(2-(2-Hydroxy-5-methyl-biphenyl-3-yl)-1H-indole-5-c...)Show SMILES Cc1cc(-c2cc3cc(ccc3[nH]2)C(N)=N)c(O)c(c1)-c1ccccc1 Show InChI InChI=1S/C22H19N3O/c1-13-9-17(14-5-3-2-4-6-14)21(26)18(10-13)20-12-16-11-15(22(23)24)7-8-19(16)25-20/h2-12,25-26H,1H3,(H3,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Activity against Human Serine Protease Trypsin |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50102780

(2-(2-Hydroxy-3-phenyl-phenyl)-1H-indole-5-carboxam...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cccc(-c2ccccc2)c1O Show InChI InChI=1S/C21H17N3O/c22-21(23)14-9-10-18-15(11-14)12-19(24-18)17-8-4-7-16(20(17)25)13-5-2-1-3-6-13/h1-12,24-25H,(H3,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Activity against Human Serine Protease Trypsin |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50102778

(2-(5-Chloro-2-hydroxy-biphenyl-3-yl)-1H-indole-5-c...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(Cl)cc(-c2ccccc2)c1O Show InChI InChI=1S/C21H16ClN3O/c22-15-10-16(12-4-2-1-3-5-12)20(26)17(11-15)19-9-14-8-13(21(23)24)6-7-18(14)25-19/h1-11,25-26H,(H3,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Activity against Human Serine Protease Trypsin |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Tissue-type plasminogen activator
(Homo sapiens (Human)) | BDBM50102778

(2-(5-Chloro-2-hydroxy-biphenyl-3-yl)-1H-indole-5-c...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(Cl)cc(-c2ccccc2)c1O Show InChI InChI=1S/C21H16ClN3O/c22-15-10-16(12-4-2-1-3-5-12)20(26)17(11-15)19-9-14-8-13(21(23)24)6-7-18(14)25-19/h1-11,25-26H,(H3,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inihibtion of Human Serine Protease tissue type Plasminogen Activator |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50101873

(2-(3-Bromo-2-hydroxy-5-methyl-phenyl)-1H-indole-5-...)Show SMILES Cc1cc(Br)c(O)c(c1)-c1cc2cc(ccc2[nH]1)C(N)=N Show InChI InChI=1S/C16H14BrN3O/c1-8-4-11(15(21)12(17)5-8)14-7-10-6-9(16(18)19)2-3-13(10)20-14/h2-7,20-21H,1H3,(H3,18,19) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Human Serine Protease Thrombin. |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Plasminogen
(Homo sapiens (Human)) | BDBM50102790

(2-(2-Hydroxy-5-nitro-biphenyl-3-yl)-1H-indole-5-ca...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(cc(c1O)-c1ccccc1)[N+]([O-])=O Show InChI InChI=1S/C21H16N4O3/c22-21(23)13-6-7-18-14(8-13)9-19(24-18)17-11-15(25(27)28)10-16(20(17)26)12-4-2-1-3-5-12/h1-11,24,26H,(H3,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Human Serine Protease Plasmin. |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50101866

(2-(2-Hydroxy-5-methyl-biphenyl-3-yl)-1H-indole-5-c...)Show SMILES Cc1cc(-c2cc3cc(ccc3[nH]2)C(N)=N)c(O)c(c1)-c1ccccc1 Show InChI InChI=1S/C22H19N3O/c1-13-9-17(14-5-3-2-4-6-14)21(26)18(10-13)20-12-16-11-15(22(23)24)7-8-19(16)25-20/h2-12,25-26H,1H3,(H3,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50102786

(2-(5-Bromo-2-hydroxy-biphenyl-3-yl)-1H-indole-5-ca...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(Br)cc(-c2ccccc2)c1O Show InChI InChI=1S/C21H16BrN3O/c22-15-10-16(12-4-2-1-3-5-12)20(26)17(11-15)19-9-14-8-13(21(23)24)6-7-18(14)25-19/h1-11,25-26H,(H3,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50102792

(2-(2-Hydroxy-3-bromo-5-nitro-phenyl)-1H-indole-5-c...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(cc(Br)c1O)[N+]([O-])=O Show InChI InChI=1S/C15H11BrN4O3/c16-11-6-9(20(22)23)5-10(14(11)21)13-4-8-3-7(15(17)18)1-2-12(8)19-13/h1-6,19,21H,(H3,17,18) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Human Serine Protease Thrombin. |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Plasminogen
(Homo sapiens (Human)) | BDBM50101866

(2-(2-Hydroxy-5-methyl-biphenyl-3-yl)-1H-indole-5-c...)Show SMILES Cc1cc(-c2cc3cc(ccc3[nH]2)C(N)=N)c(O)c(c1)-c1ccccc1 Show InChI InChI=1S/C22H19N3O/c1-13-9-17(14-5-3-2-4-6-14)21(26)18(10-13)20-12-16-11-15(22(23)24)7-8-19(16)25-20/h2-12,25-26H,1H3,(H3,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Human Serine Protease Plasmin |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50102785
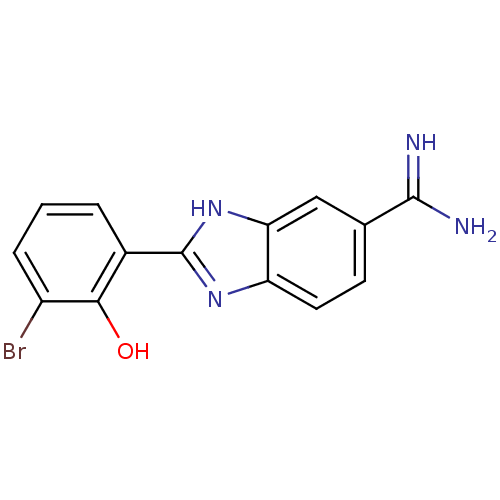
(2-(3-Bromo-2-hydroxy-phenyl)-1H-benzoimidazole-5-c...)Show InChI InChI=1S/C14H11BrN4O/c15-9-3-1-2-8(12(9)20)14-18-10-5-4-7(13(16)17)6-11(10)19-14/h1-6,20H,(H3,16,17)(H,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
ComInhibition of Human Serine Protease Urokinase Plasminogen Activator (u-PA). |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Plasminogen
(Homo sapiens (Human)) | BDBM50102767

(2-(3-Bromo-2-hydroxy-phenyl)-1H-indole-5-carboxami...)Show InChI InChI=1S/C15H12BrN3O/c16-11-3-1-2-10(14(11)20)13-7-9-6-8(15(17)18)4-5-12(9)19-13/h1-7,19-20H,(H3,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Human Serine Protease Plasmin |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50102792

(2-(2-Hydroxy-3-bromo-5-nitro-phenyl)-1H-indole-5-c...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(cc(Br)c1O)[N+]([O-])=O Show InChI InChI=1S/C15H11BrN4O3/c16-11-6-9(20(22)23)5-10(14(11)21)13-4-8-3-7(15(17)18)1-2-12(8)19-13/h1-6,19,21H,(H3,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
ComInhibition of Human Serine Protease Urokinase Plasminogen Activator (u-PA). |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Tissue-type plasminogen activator
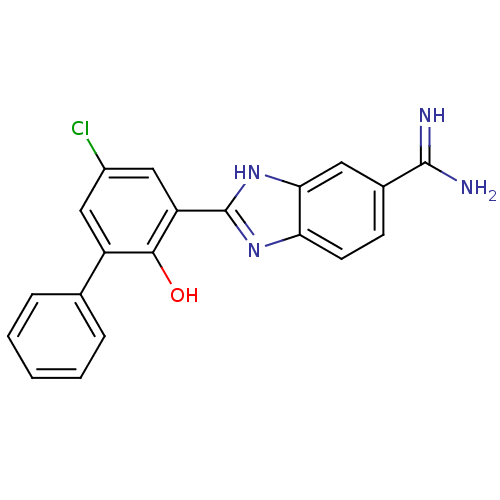
(Homo sapiens (Human)) | BDBM50102791
(2-(5-Chloro-2-hydroxy-biphenyl-3-yl)-1H-benzoimida...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(Cl)cc(-c2ccccc2)c1O Show InChI InChI=1S/C20H15ClN4O/c21-13-9-14(11-4-2-1-3-5-11)18(26)15(10-13)20-24-16-7-6-12(19(22)23)8-17(16)25-20/h1-10,26H,(H3,22,23)(H,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inihibtion of Human Serine Protease tissue type Plasminogen Activator |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50102789

(2-(3-Bromo-2-hydroxy-5-methyl-phenyl)-1H-benzoimid...)Show SMILES Cc1cc(Br)c(O)c(c1)-c1nc2ccc(cc2[nH]1)C(N)=N Show InChI InChI=1S/C15H13BrN4O/c1-7-4-9(13(21)10(16)5-7)15-19-11-3-2-8(14(17)18)6-12(11)20-15/h2-6,21H,1H3,(H3,17,18)(H,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
ComInhibition of Human Serine Protease Urokinase Plasminogen Activator (u-PA). |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50102778

(2-(5-Chloro-2-hydroxy-biphenyl-3-yl)-1H-indole-5-c...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(Cl)cc(-c2ccccc2)c1O Show InChI InChI=1S/C21H16ClN3O/c22-15-10-16(12-4-2-1-3-5-12)20(26)17(11-15)19-9-14-8-13(21(23)24)6-7-18(14)25-19/h1-11,25-26H,(H3,23,24) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Activity against Human Serine Protease Thrombin |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50102767

(2-(3-Bromo-2-hydroxy-phenyl)-1H-indole-5-carboxami...)Show InChI InChI=1S/C15H12BrN3O/c16-11-3-1-2-10(14(11)20)13-7-9-6-8(15(17)18)4-5-12(9)19-13/h1-7,19-20H,(H3,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Activity against Human Serine Protease Trypsin |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Plasminogen
(Homo sapiens (Human)) | BDBM50102778

(2-(5-Chloro-2-hydroxy-biphenyl-3-yl)-1H-indole-5-c...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(Cl)cc(-c2ccccc2)c1O Show InChI InChI=1S/C21H16ClN3O/c22-15-10-16(12-4-2-1-3-5-12)20(26)17(11-15)19-9-14-8-13(21(23)24)6-7-18(14)25-19/h1-11,25-26H,(H3,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Human Serine Protease Plasmin |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50102785

(2-(3-Bromo-2-hydroxy-phenyl)-1H-benzoimidazole-5-c...)Show InChI InChI=1S/C14H11BrN4O/c15-9-3-1-2-8(12(9)20)14-18-10-5-4-7(13(16)17)6-11(10)19-14/h1-6,20H,(H3,16,17)(H,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50102780

(2-(2-Hydroxy-3-phenyl-phenyl)-1H-indole-5-carboxam...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cccc(-c2ccccc2)c1O Show InChI InChI=1S/C21H17N3O/c22-21(23)14-9-10-18-15(11-14)12-19(24-18)17-8-4-7-16(20(17)25)13-5-2-1-3-6-13/h1-12,24-25H,(H3,22,23) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Activity against Human Serine Protease Thrombin |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50102784

(2-(3,5-Dibromo-2-hydroxy-phenyl)-1H-indole-5-carbo...)Show InChI InChI=1S/C15H11Br2N3O/c16-9-5-10(14(21)11(17)6-9)13-4-8-3-7(15(18)19)1-2-12(8)20-13/h1-6,20-21H,(H3,18,19) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Activity against Human Serine Protease Thrombin |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50101873

(2-(3-Bromo-2-hydroxy-5-methyl-phenyl)-1H-indole-5-...)Show SMILES Cc1cc(Br)c(O)c(c1)-c1cc2cc(ccc2[nH]1)C(N)=N Show InChI InChI=1S/C16H14BrN3O/c1-8-4-11(15(21)12(17)5-8)14-7-10-6-9(16(18)19)2-3-13(10)20-14/h2-7,20-21H,1H3,(H3,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Human Serine Protease Trypsin. |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50102778

(2-(5-Chloro-2-hydroxy-biphenyl-3-yl)-1H-indole-5-c...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(Cl)cc(-c2ccccc2)c1O Show InChI InChI=1S/C21H16ClN3O/c22-15-10-16(12-4-2-1-3-5-12)20(26)17(11-15)19-9-14-8-13(21(23)24)6-7-18(14)25-19/h1-11,25-26H,(H3,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Binding affinity against human coagulation factor X |
J Med Chem 44: 2753-71 (2001)
BindingDB Entry DOI: 10.7270/Q2RX9BC7 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data