 Found 10719 hits with Last Name = 'wei' and Initial = 'a'
Found 10719 hits with Last Name = 'wei' and Initial = 'a' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450717

(CHEMBL317087)Show InChI InChI=1S/C9H10BrClN2O/c10-8-3-7(4-13-9(8)11)14-5-6-1-2-12-6/h3-4,6,12H,1-2,5H2/t6-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450726
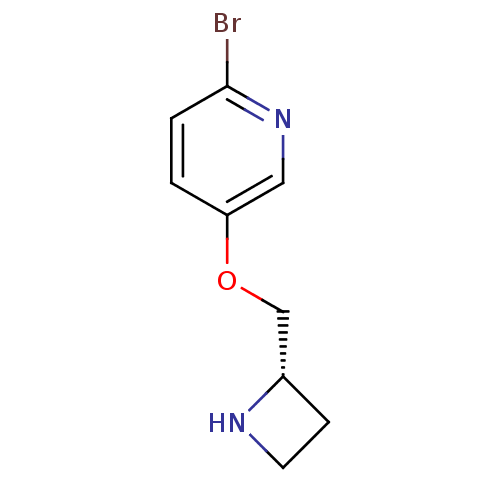
(CHEMBL407258)Show InChI InChI=1S/C9H11BrN2O/c10-9-2-1-8(5-12-9)13-6-7-3-4-11-7/h1-2,5,7,11H,3-4,6H2/t7-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450734
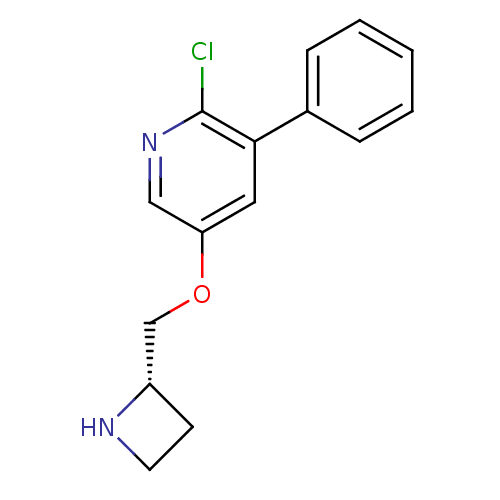
(CHEMBL318869)Show InChI InChI=1S/C15H15ClN2O/c16-15-14(11-4-2-1-3-5-11)8-13(9-18-15)19-10-12-6-7-17-12/h1-5,8-9,12,17H,6-7,10H2/t12-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450710
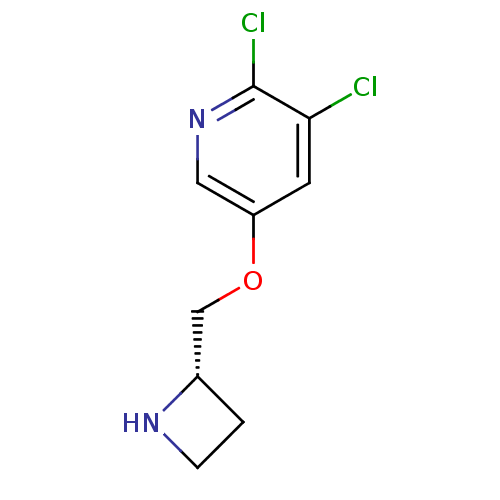
(CHEMBL97555)Show InChI InChI=1S/C9H10Cl2N2O/c10-8-3-7(4-13-9(8)11)14-5-6-1-2-12-6/h3-4,6,12H,1-2,5H2/t6-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450720

(CHEMBL94683)Show InChI InChI=1S/C10H11N3O/c11-4-8-3-10(6-12-5-8)14-7-9-1-2-13-9/h3,5-6,9,13H,1-2,7H2/t9-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50211565
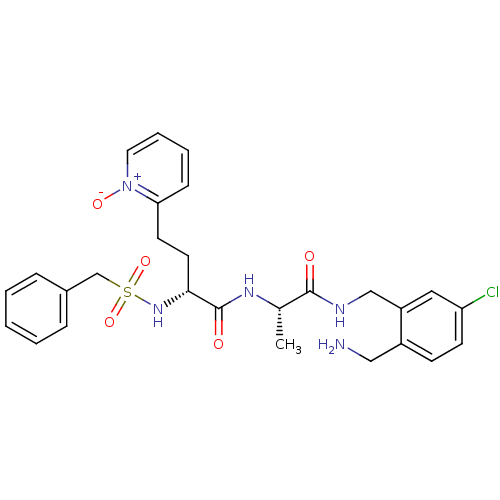
((R)-N-[(S)-1-(2-aminomethyl-5-chloro-benzylcarbamo...)Show SMILES C[C@H](NC(=O)[C@@H](CCc1cccc[n+]1[O-])NS(=O)(=O)Cc1ccccc1)C(=O)NCc1cc(Cl)ccc1CN Show InChI InChI=1S/C27H32ClN5O5S/c1-19(26(34)30-17-22-15-23(28)11-10-21(22)16-29)31-27(35)25(13-12-24-9-5-6-14-33(24)36)32-39(37,38)18-20-7-3-2-4-8-20/h2-11,14-15,19,25,32H,12-13,16-18,29H2,1H3,(H,30,34)(H,31,35)/t19-,25+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Curacyte Discovery GmbH
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
Bioorg Med Chem Lett 17: 3322-9 (2007)
Article DOI: 10.1016/j.bmcl.2007.03.105
BindingDB Entry DOI: 10.7270/Q2NG4Q9K |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450712

(CHEMBL96604)Show InChI InChI=1S/C11H16N2O2/c1-2-14-10-5-11(7-12-6-10)15-8-9-3-4-13-9/h5-7,9,13H,2-4,8H2,1H3/t9-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450713

(CHEMBL96837)Show InChI InChI=1S/C12H18N2O/c1-2-3-10-6-12(8-13-7-10)15-9-11-4-5-14-11/h6-8,11,14H,2-5,9H2,1H3/t11-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50062641
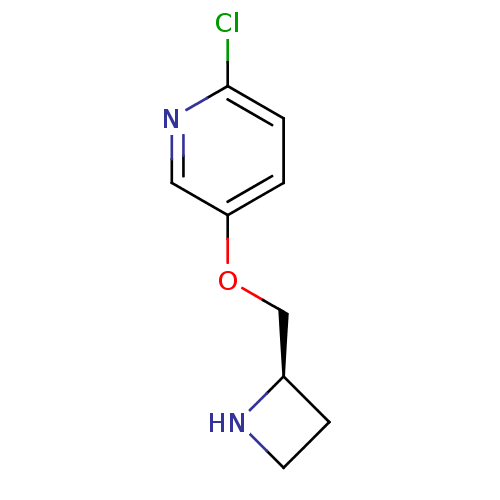
(5-((R)-1-Azetidin-2-ylmethoxy)-2-chloro-pyridine |...)Show InChI InChI=1S/C9H11ClN2O/c10-9-2-1-8(5-12-9)13-6-7-3-4-11-7/h1-2,5,7,11H,3-4,6H2/t7-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50062639
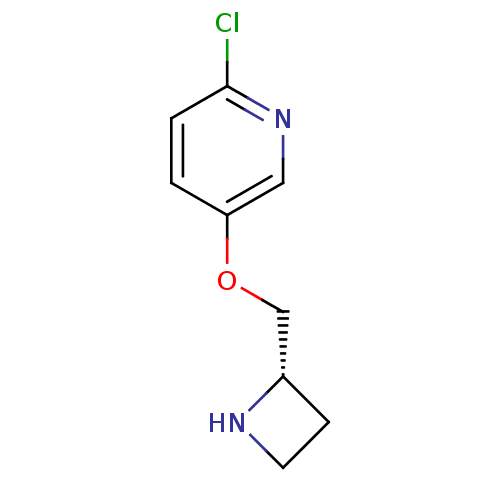
(5-((S)-1-Azetidin-2-ylmethoxy)-2-chloro-pyridine |...)Show InChI InChI=1S/C9H11ClN2O/c10-9-2-1-8(5-12-9)13-6-7-3-4-11-7/h1-2,5,7,11H,3-4,6H2/t7-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Coagulation factor XI
(Homo sapiens (Human)) | BDBM50096792
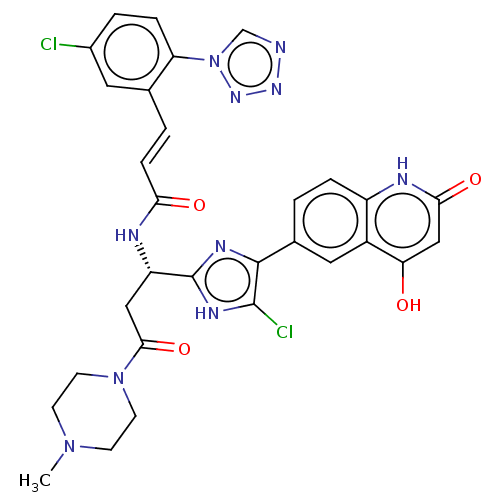
(CHEMBL3580759)Show SMILES CN1CCN(CC1)C(=O)C[C@H](NC(=O)\C=C\c1cc(Cl)ccc1-n1cnnn1)c1nc(c(Cl)[nH]1)-c1ccc2[nH]c(=O)cc(O)c2c1 |r| Show InChI InChI=1S/C30H26Cl2N10O4/c1-40-8-10-41(11-9-40)27(46)14-22(35-25(44)7-3-17-12-19(31)4-6-23(17)42-16-33-38-39-42)30-36-28(29(32)37-30)18-2-5-21-20(13-18)24(43)15-26(45)34-21/h2-7,12-13,15-16,22,43H,8-11,14H2,1H3,(H,35,44)/b7-3+,28-18-/t22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Inhibition of human coagulation factor 11a using p-nitroaniline as substrate assessed as substrate hydrolysis by spectrophotometry |
ACS Med Chem Lett 6: 590-5 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00066
BindingDB Entry DOI: 10.7270/Q2B27X24 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Homo sapiens (Human)) | BDBM50049750
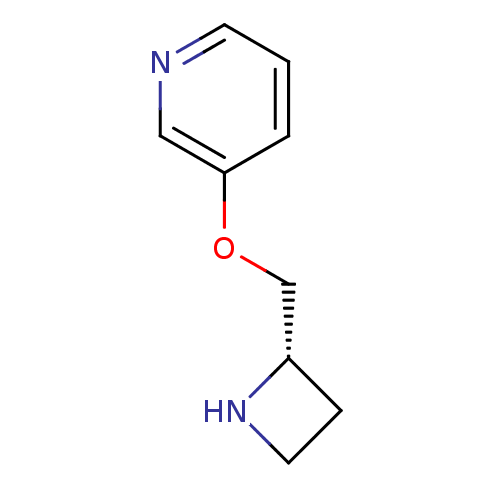
((S)-3-(azetidin-2-ylmethoxy)pyridine | 3-((S)-1-Az...)Show InChI InChI=1S/C9H12N2O/c1-2-9(6-10-4-1)12-7-8-3-5-11-8/h1-2,4,6,8,11H,3,5,7H2/t8-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratory
Curated by ChEMBL
| Assay Description
Compound was evaluated for its ability to displace [3H]-cytisine binding from high-affinity Nicotinic acetylcholine receptor in rat brain (principall... |
J Med Chem 41: 407-12 (1998)
Article DOI: 10.1021/jm9706224
BindingDB Entry DOI: 10.7270/Q2CJ8F56 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450723
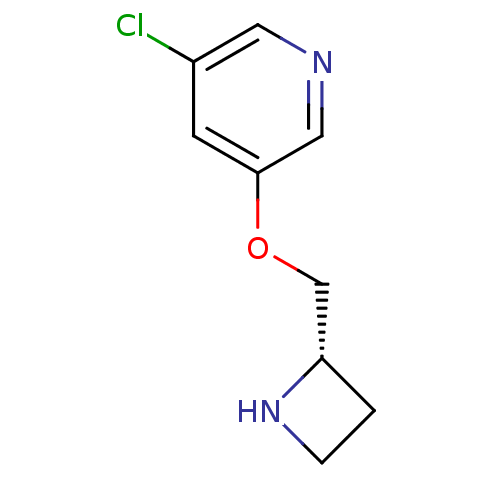
(CHEMBL95113)Show InChI InChI=1S/C9H11ClN2O/c10-7-3-9(5-11-4-7)13-6-8-1-2-12-8/h3-5,8,12H,1-2,6H2/t8-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0420 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50072110
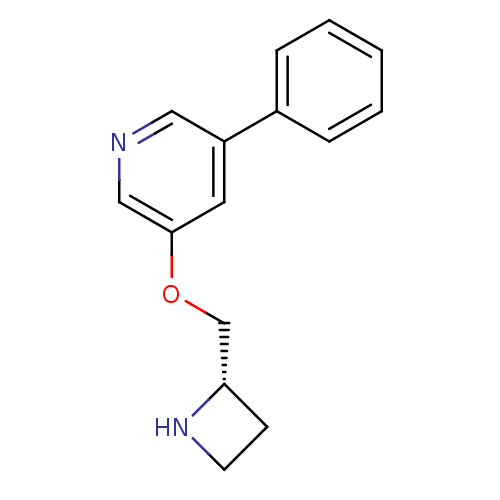
(3-((S)-1-Azetidin-2-ylmethoxy)-5-phenyl-pyridine |...)Show InChI InChI=1S/C15H16N2O/c1-2-4-12(5-3-1)13-8-15(10-16-9-13)18-11-14-6-7-17-14/h1-5,8-10,14,17H,6-7,11H2/t14-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
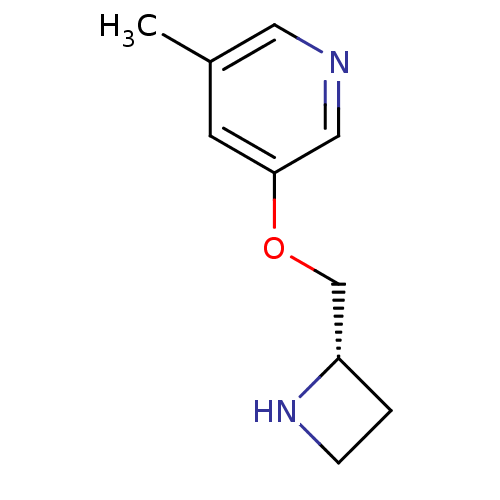
(Rattus norvegicus (Rat)) | BDBM50450728
(CHEMBL96431)Show InChI InChI=1S/C10H14N2O/c1-8-4-10(6-11-5-8)13-7-9-2-3-12-9/h4-6,9,12H,2-3,7H2,1H3/t9-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50066788
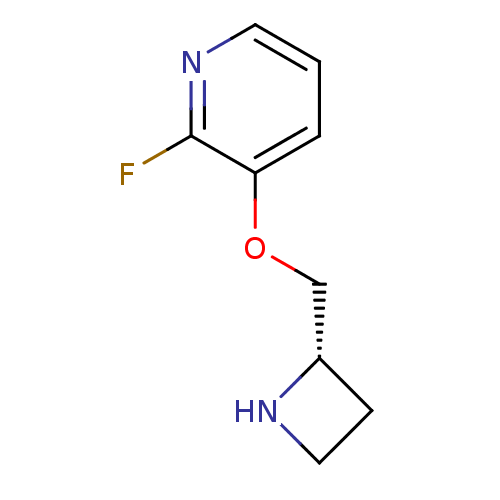
((S)-3-(azetidin-2-ylmethoxy)-2-fluoropyridine | 3-...)Show InChI InChI=1S/C9H11FN2O/c10-9-8(2-1-4-12-9)13-6-7-3-5-11-7/h1-2,4,7,11H,3,5-6H2/t7-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50211578
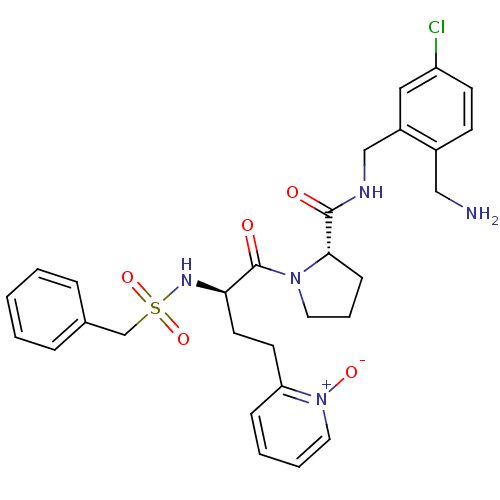
((S)-1-[(R)-4-(1-oxy-pyridin-2-yl)-2-phenylmethanes...)Show SMILES NCc1ccc(Cl)cc1CNC(=O)[C@@H]1CCCN1C(=O)[C@@H](CCc1cccc[n+]1[O-])NS(=O)(=O)Cc1ccccc1 Show InChI InChI=1S/C29H34ClN5O5S/c30-24-12-11-22(18-31)23(17-24)19-32-28(36)27-10-6-15-34(27)29(37)26(14-13-25-9-4-5-16-35(25)38)33-41(39,40)20-21-7-2-1-3-8-21/h1-5,7-9,11-12,16-17,26-27,33H,6,10,13-15,18-20,31H2,(H,32,36)/t26-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Curacyte Discovery GmbH
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
Bioorg Med Chem Lett 17: 3322-9 (2007)
Article DOI: 10.1016/j.bmcl.2007.03.105
BindingDB Entry DOI: 10.7270/Q2NG4Q9K |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50049750

((S)-3-(azetidin-2-ylmethoxy)pyridine | 3-((S)-1-Az...)Show InChI InChI=1S/C9H12N2O/c1-2-9(6-10-4-1)12-7-8-3-5-11-8/h1-2,4,6,8,11H,3,5,7H2/t8-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.0520 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450718
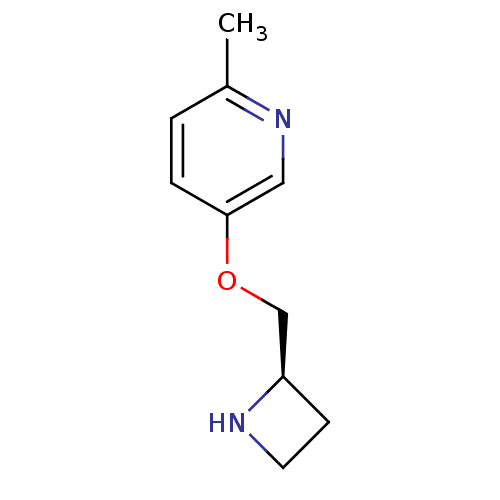
(CHEMBL94670)Show InChI InChI=1S/C10H14N2O/c1-8-2-3-10(6-12-8)13-7-9-4-5-11-9/h2-3,6,9,11H,4-5,7H2,1H3/t9-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Homo sapiens (Human)) | BDBM50062641

(5-((R)-1-Azetidin-2-ylmethoxy)-2-chloro-pyridine |...)Show InChI InChI=1S/C9H11ClN2O/c10-9-2-1-8(5-12-9)13-6-7-3-4-11-7/h1-2,5,7,11H,3-4,6H2/t7-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratory
Curated by ChEMBL
| Assay Description
Compound was evaluated for its ability to displace [125I]alpha-bungarotoxin (alpha-BgT) from K28 cells expressing human Nicotinic acetylcholine recep... |
J Med Chem 41: 407-12 (1998)
Article DOI: 10.1021/jm9706224
BindingDB Entry DOI: 10.7270/Q2CJ8F56 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Homo sapiens (Human)) | BDBM50062641

(5-((R)-1-Azetidin-2-ylmethoxy)-2-chloro-pyridine |...)Show InChI InChI=1S/C9H11ClN2O/c10-9-2-1-8(5-12-9)13-6-7-3-4-11-7/h1-2,5,7,11H,3-4,6H2/t7-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratory
Curated by ChEMBL
| Assay Description
Compound was evaluated for its ability to displace [3H]cytisine from K177 cells expressing human Nicotinic acetylcholine receptor alpha4-beta2 |
J Med Chem 41: 407-12 (1998)
Article DOI: 10.1021/jm9706224
BindingDB Entry DOI: 10.7270/Q2CJ8F56 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450719
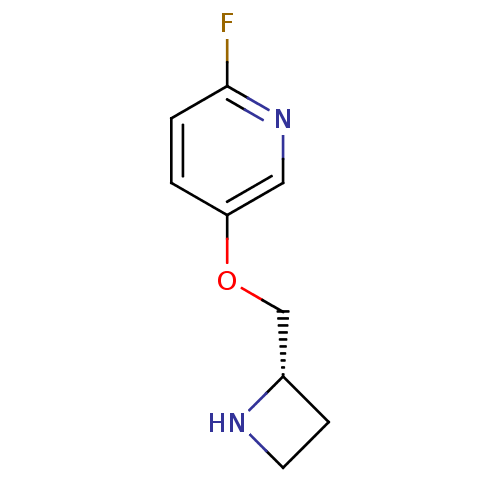
(CHEMBL318121)Show InChI InChI=1S/C9H11FN2O/c10-9-2-1-8(5-12-9)13-6-7-3-4-11-7/h1-2,5,7,11H,3-4,6H2/t7-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0570 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450729
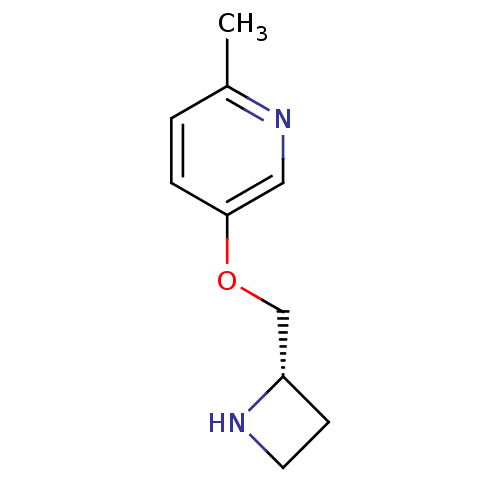
(CHEMBL94877)Show InChI InChI=1S/C10H14N2O/c1-8-2-3-10(6-12-8)13-7-9-4-5-11-9/h2-3,6,9,11H,4-5,7H2,1H3/t9-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0570 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50211567
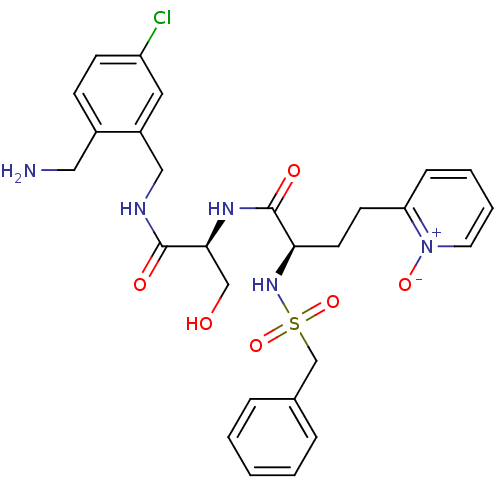
((R)-N-[(S)-1-(2-aminomethyl-5-chloro-benzylcarbamo...)Show SMILES NCc1ccc(Cl)cc1CNC(=O)[C@H](CO)NC(=O)[C@@H](CCc1cccc[n+]1[O-])NS(=O)(=O)Cc1ccccc1 Show InChI InChI=1S/C27H32ClN5O6S/c28-22-10-9-20(15-29)21(14-22)16-30-26(35)25(17-34)31-27(36)24(12-11-23-8-4-5-13-33(23)37)32-40(38,39)18-19-6-2-1-3-7-19/h1-10,13-14,24-25,32,34H,11-12,15-18,29H2,(H,30,35)(H,31,36)/t24-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0590 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Curacyte Discovery GmbH
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
Bioorg Med Chem Lett 17: 3322-9 (2007)
Article DOI: 10.1016/j.bmcl.2007.03.105
BindingDB Entry DOI: 10.7270/Q2NG4Q9K |
More data for this
Ligand-Target Pair | |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50192770
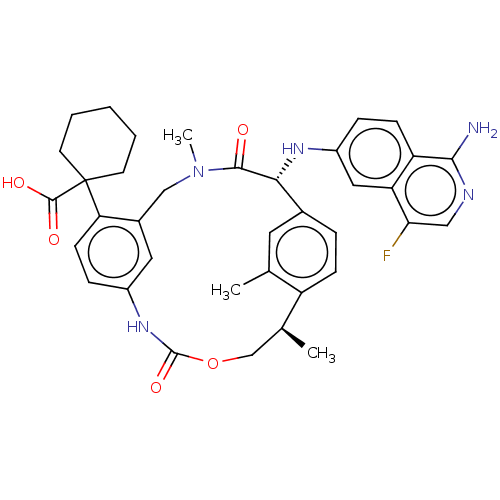
(CHEMBL3956096)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCCCC1)C(O)=O |r| Show InChI InChI=1S/C37H40FN5O5/c1-21-15-23-7-10-27(21)22(2)20-48-36(47)42-25-9-12-30(37(35(45)46)13-5-4-6-14-37)24(16-25)19-43(3)34(44)32(23)41-26-8-11-28-29(17-26)31(38)18-40-33(28)39/h7-12,15-18,22,32,41H,4-6,13-14,19-20H2,1-3H3,(H2,39,40)(H,42,47)(H,45,46)/t22-,32+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
| PDB
Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human TF-factor 7a using factor 10 as substrate at 37 degC |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450748
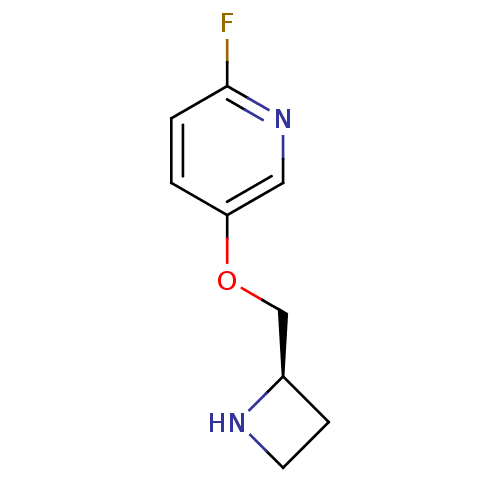
(CHEMBL446410)Show InChI InChI=1S/C9H11FN2O/c10-9-2-1-8(5-12-9)13-6-7-3-4-11-7/h1-2,5,7,11H,3-4,6H2/t7-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0660 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Suppressor of tumorigenicity 14 protein
(Homo sapiens (Human)) | BDBM104907

(US8569313, Inhibitor 15)Show SMILES NCCC1CCN(CC1)C(=O)C(Cc1cccc(c1)C(N)=N)NS(=O)(=O)c1cccc(c1)C1CCC(N)=NC1 |c:39| Show InChI InChI=1S/C28H39N7O3S/c29-12-9-19-10-13-35(14-11-19)28(36)25(16-20-3-1-5-22(15-20)27(31)32)34-39(37,38)24-6-2-4-21(17-24)23-7-8-26(30)33-18-23/h1-6,15,17,19,23,25,34H,7-14,16,18,29H2,(H2,30,33)(H3,31,32) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0690 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Medicines Company (Leipzig) GmbH
US Patent
| Assay Description
Inhibition assay using matriptase enzyme |
US Patent US8569313 (2013)
BindingDB Entry DOI: 10.7270/Q2639ND5 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50558721

(CHEMBL4784791)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(N)=O |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]U69,593 from human KOR expressed in mouse HN9.10 cell membranes incubated for 2 hrs by liquid scintillation counting based radiol... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2ZG6WWT |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450743

(CHEMBL96527)Show InChI InChI=1S/C9H11FN2O/c10-7-3-9(5-11-4-7)13-6-8-1-2-12-8/h3-5,8,12H,1-2,6H2/t8-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0760 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Suppressor of tumorigenicity 14 protein
(Homo sapiens (Human)) | BDBM50253952
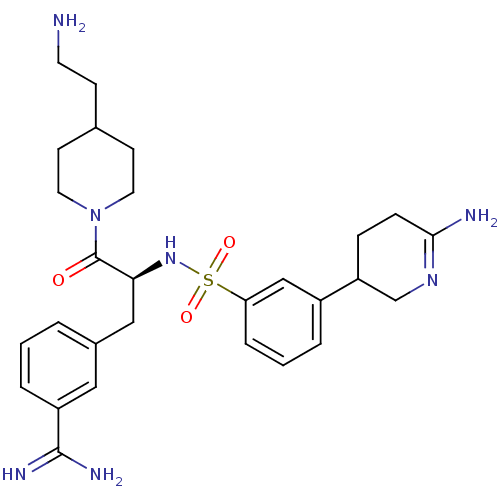
(3-{(S)-3-[4-(2-Amino-ethyl)-piperidin-1-yl]-2-[3-(...)Show SMILES NCCC1CCN(CC1)C(=O)[C@H](Cc1cccc(c1)C(N)=N)NS(=O)(=O)c1cccc(c1)C1CCC(N)=NC1 |r,c:39| Show InChI InChI=1S/C28H39N7O3S/c29-12-9-19-10-13-35(14-11-19)28(36)25(16-20-3-1-5-22(15-20)27(31)32)34-39(37,38)24-6-2-4-21(17-24)23-7-8-26(30)33-18-23/h1-6,15,17,19,23,25,34H,7-14,16,18,29H2,(H2,30,33)(H3,31,32)/t23?,25-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Curacyte Discovery GmbH
Curated by ChEMBL
| Assay Description
Inhibition of matriptase (unknown origin) |
Bioorg Med Chem Lett 19: 67-73 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.019
BindingDB Entry DOI: 10.7270/Q2HH6JXF |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50211585
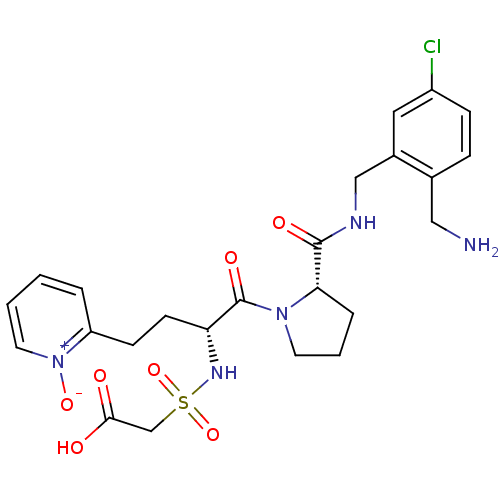
(CHEMBL389960 | [(R)-1-[(S)-2-(2-aminomethyl-5-chlo...)Show SMILES NCc1ccc(Cl)cc1CNC(=O)[C@@H]1CCCN1C(=O)[C@@H](CCc1cccc[n+]1[O-])NS(=O)(=O)CC(O)=O Show InChI InChI=1S/C24H30ClN5O7S/c25-18-7-6-16(13-26)17(12-18)14-27-23(33)21-5-3-10-29(21)24(34)20(28-38(36,37)15-22(31)32)9-8-19-4-1-2-11-30(19)35/h1-2,4,6-7,11-12,20-21,28H,3,5,8-10,13-15,26H2,(H,27,33)(H,31,32)/t20-,21+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Curacyte Discovery GmbH
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
Bioorg Med Chem Lett 17: 3322-9 (2007)
Article DOI: 10.1016/j.bmcl.2007.03.105
BindingDB Entry DOI: 10.7270/Q2NG4Q9K |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450738

(CHEMBL94207)Show InChI InChI=1S/C9H13N3O/c10-7-3-9(5-11-4-7)13-6-8-1-2-12-8/h3-5,8,12H,1-2,6,10H2/t8-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0820 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50211571
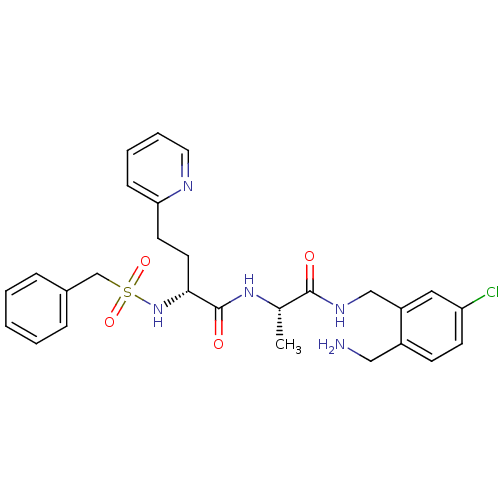
((R)-N-((S)-1-(2-(aminomethyl)-5-chlorobenzylamino)...)Show SMILES C[C@H](NC(=O)[C@@H](CCc1ccccn1)NS(=O)(=O)Cc1ccccc1)C(=O)NCc1cc(Cl)ccc1CN Show InChI InChI=1S/C27H32ClN5O4S/c1-19(26(34)31-17-22-15-23(28)11-10-21(22)16-29)32-27(35)25(13-12-24-9-5-6-14-30-24)33-38(36,37)18-20-7-3-2-4-8-20/h2-11,14-15,19,25,33H,12-13,16-18,29H2,1H3,(H,31,34)(H,32,35)/t19-,25+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0840 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Curacyte Discovery GmbH
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
Bioorg Med Chem Lett 17: 3322-9 (2007)
Article DOI: 10.1016/j.bmcl.2007.03.105
BindingDB Entry DOI: 10.7270/Q2NG4Q9K |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 8
(Homo sapiens (Human)) | BDBM50324510
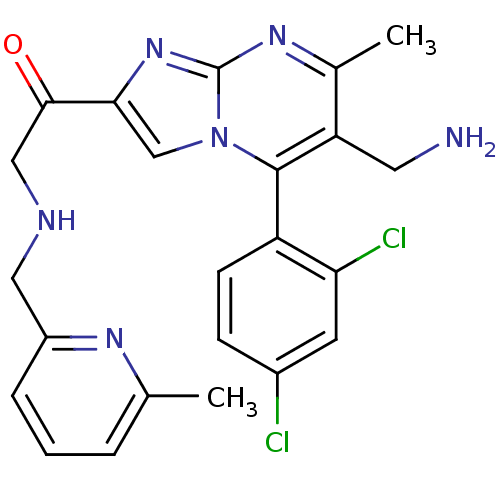
(1-(6-(aminomethyl)-5-(2,4-dichlorophenyl)-7-methyl...)Show SMILES Cc1cccc(CNCC(=O)c2cn3c(c(CN)c(C)nc3n2)-c2ccc(Cl)cc2Cl)n1 |(-4.59,-8.35,;-3.87,-6.99,;-4.69,-5.68,;-3.96,-4.31,;-2.42,-4.28,;-1.62,-5.59,;-.08,-5.54,;.65,-4.18,;2.19,-4.14,;2.92,-2.78,;2.11,-1.48,;4.46,-2.74,;5.4,-3.96,;6.85,-3.44,;8.2,-4.17,;9.51,-3.36,;10.87,-4.08,;12.17,-3.27,;9.45,-1.81,;10.76,-1,;8.1,-1.09,;6.8,-1.9,;5.33,-1.46,;8.17,-5.7,;6.82,-6.44,;6.79,-7.98,;8.11,-8.78,;8.08,-10.32,;9.46,-8.02,;9.48,-6.48,;10.83,-5.74,;-2.34,-6.93,)| Show InChI InChI=1S/C23H22Cl2N6O/c1-13-4-3-5-16(28-13)10-27-11-21(32)20-12-31-22(17-7-6-15(24)8-19(17)25)18(9-26)14(2)29-23(31)30-20/h3-8,12,27H,9-11,26H2,1-2H3 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0860 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Inhibition of human DPP8 |
J Med Chem 53: 5620-8 (2010)
Article DOI: 10.1021/jm100634a
BindingDB Entry DOI: 10.7270/Q2S182PX |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450724
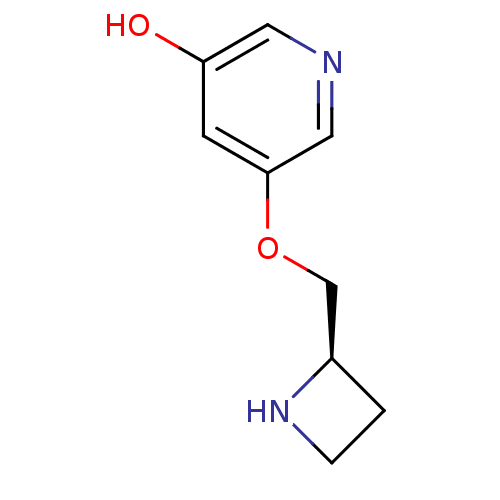
(CHEMBL95128)Show InChI InChI=1S/C9H12N2O2/c12-8-3-9(5-10-4-8)13-6-7-1-2-11-7/h3-5,7,11-12H,1-2,6H2/t7-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0880 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM214798
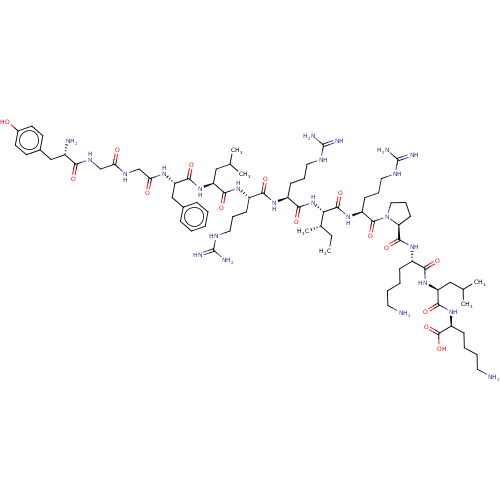
(Dynorphin A (1-17) | YGGFLRRIRPKLK)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(O)=O |r| Show InChI InChI=1S/C75H126N24O15/c1-7-45(6)61(70(111)94-53(25-17-35-87-75(83)84)71(112)99-36-18-26-58(99)69(110)93-50(21-11-13-31-76)64(105)96-56(38-44(4)5)67(108)95-54(72(113)114)22-12-14-32-77)98-65(106)52(24-16-34-86-74(81)82)91-63(104)51(23-15-33-85-73(79)80)92-66(107)55(37-43(2)3)97-68(109)57(40-46-19-9-8-10-20-46)90-60(102)42-88-59(101)41-89-62(103)49(78)39-47-27-29-48(100)30-28-47/h8-10,19-20,27-30,43-45,49-58,61,100H,7,11-18,21-26,31-42,76-78H2,1-6H3,(H,88,101)(H,89,103)(H,90,102)(H,91,104)(H,92,107)(H,93,110)(H,94,111)(H,95,108)(H,96,105)(H,97,109)(H,98,106)(H,113,114)(H4,79,80,85)(H4,81,82,86)(H4,83,84,87)/t45-,49-,50-,51-,52-,53-,54-,55-,56-,57-,58-,61-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]U69,593 from human KOR expressed in mouse HN9.10 cell membranes incubated for 2 hrs by liquid scintillation counting based radiol... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2ZG6WWT |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50211564
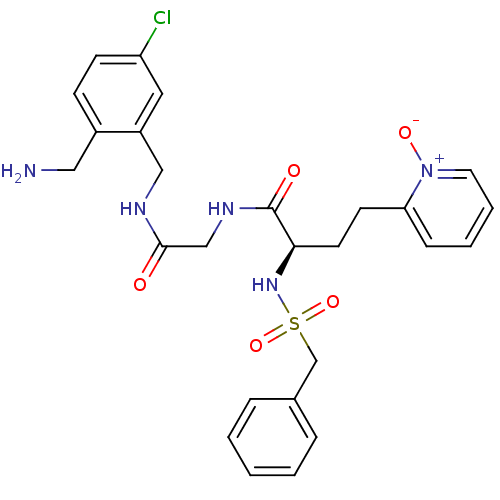
((R)-N-[(5-chloro-2-ethyl-benzylcarbamoyl)-methyl]-...)Show SMILES NCc1ccc(Cl)cc1CNC(=O)CNC(=O)[C@@H](CCc1cccc[n+]1[O-])NS(=O)(=O)Cc1ccccc1 Show InChI InChI=1S/C26H30ClN5O5S/c27-22-10-9-20(15-28)21(14-22)16-29-25(33)17-30-26(34)24(12-11-23-8-4-5-13-32(23)35)31-38(36,37)18-19-6-2-1-3-7-19/h1-10,13-14,24,31H,11-12,15-18,28H2,(H,29,33)(H,30,34)/t24-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0950 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Curacyte Discovery GmbH
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a |
Bioorg Med Chem Lett 17: 3322-9 (2007)
Article DOI: 10.1016/j.bmcl.2007.03.105
BindingDB Entry DOI: 10.7270/Q2NG4Q9K |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50010704
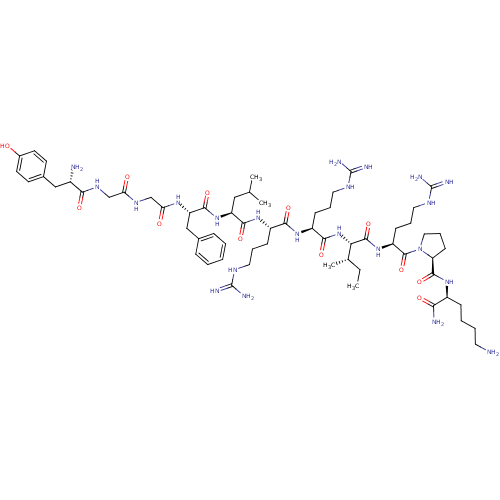
(CHEMBL216640 | Dyn A(1-11)-NH2 | Dynorphin A analo...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(N)=O Show InChI InChI=1S/C63H104N22O12/c1-5-37(4)51(59(96)82-45(20-13-29-75-63(71)72)60(97)85-30-14-21-48(85)58(95)79-42(52(66)89)17-9-10-26-64)84-55(92)44(19-12-28-74-62(69)70)80-54(91)43(18-11-27-73-61(67)68)81-56(93)46(31-36(2)3)83-57(94)47(33-38-15-7-6-8-16-38)78-50(88)35-76-49(87)34-77-53(90)41(65)32-39-22-24-40(86)25-23-39/h6-8,15-16,22-25,36-37,41-48,51,86H,5,9-14,17-21,26-35,64-65H2,1-4H3,(H2,66,89)(H,76,87)(H,77,90)(H,78,88)(H,79,95)(H,80,91)(H,81,93)(H,82,96)(H,83,94)(H,84,92)(H4,67,68,73)(H4,69,70,74)(H4,71,72,75)/t37-,41-,42-,43-,44-,45-,46-,47-,48-,51-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]U69,593 from human KOR expressed in mouse HN9.10 cell membranes incubated for 2 hrs by liquid scintillation counting based radiol... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2ZG6WWT |
More data for this
Ligand-Target Pair | |
Coagulation factor XI
(Homo sapiens (Human)) | BDBM50096792

(CHEMBL3580759)Show SMILES CN1CCN(CC1)C(=O)C[C@H](NC(=O)\C=C\c1cc(Cl)ccc1-n1cnnn1)c1nc(c(Cl)[nH]1)-c1ccc2[nH]c(=O)cc(O)c2c1 |r| Show InChI InChI=1S/C30H26Cl2N10O4/c1-40-8-10-41(11-9-40)27(46)14-22(35-25(44)7-3-17-12-19(31)4-6-23(17)42-16-33-38-39-42)30-36-28(29(32)37-30)18-2-5-21-20(13-18)24(43)15-26(45)34-21/h2-7,12-13,15-16,22,43H,8-11,14H2,1H3,(H,35,44)/b7-3+,28-18-/t22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Inhibition of human coagulation factor 11a using p-nitroaniline as substrate assessed as substrate hydrolysis at 37 degC by spectrophotometry |
ACS Med Chem Lett 6: 590-5 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00066
BindingDB Entry DOI: 10.7270/Q2B27X24 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450715

(CHEMBL96370)Show InChI InChI=1S/C11H16N2O/c1-2-9-5-11(7-12-6-9)14-8-10-3-4-13-10/h5-7,10,13H,2-4,8H2,1H3/t10-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50040123
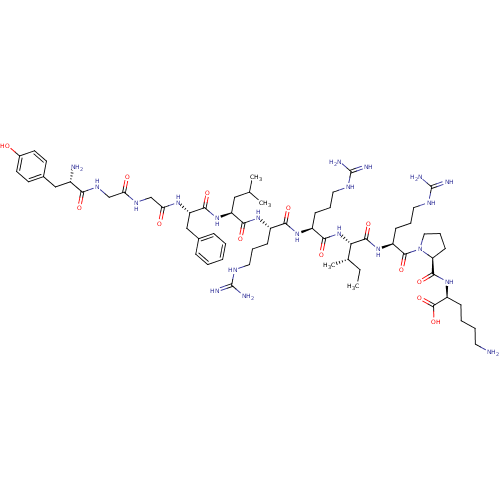
(CHEMBL438223 | HTry-Gly-Gly-Phe-Leu-Arg-Arg-lle-Ar...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(O)=O Show InChI InChI=1S/C63H103N21O13/c1-5-37(4)51(58(94)80-44(20-13-29-74-63(70)71)59(95)84-30-14-21-48(84)57(93)81-45(60(96)97)17-9-10-26-64)83-54(90)43(19-12-28-73-62(68)69)78-53(89)42(18-11-27-72-61(66)67)79-55(91)46(31-36(2)3)82-56(92)47(33-38-15-7-6-8-16-38)77-50(87)35-75-49(86)34-76-52(88)41(65)32-39-22-24-40(85)25-23-39/h6-8,15-16,22-25,36-37,41-48,51,85H,5,9-14,17-21,26-35,64-65H2,1-4H3,(H,75,86)(H,76,88)(H,77,87)(H,78,89)(H,79,91)(H,80,94)(H,81,93)(H,82,92)(H,83,90)(H,96,97)(H4,66,67,72)(H4,68,69,73)(H4,70,71,74)/t37-,41-,42-,43-,44-,45-,46-,47-,48-,51-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]U69,593 from human KOR expressed in mouse HN9.10 cell membranes incubated for 2 hrs by liquid scintillation counting based radiol... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2ZG6WWT |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450747
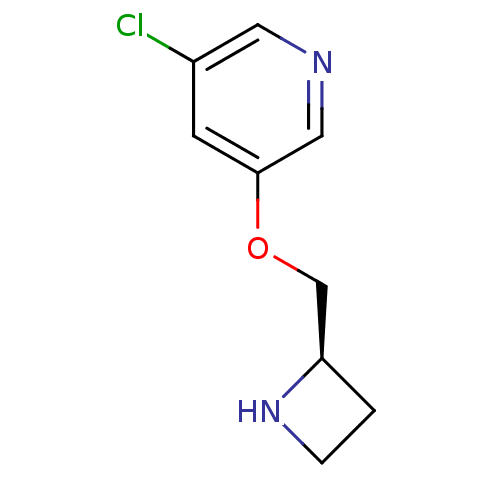
(CHEMBL95132)Show InChI InChI=1S/C9H11ClN2O/c10-7-3-9(5-11-4-7)13-6-8-1-2-12-8/h3-5,8,12H,1-2,6H2/t8-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Adenosine receptor A1
(Rattus norvegicus (rat)) | BDBM50494549
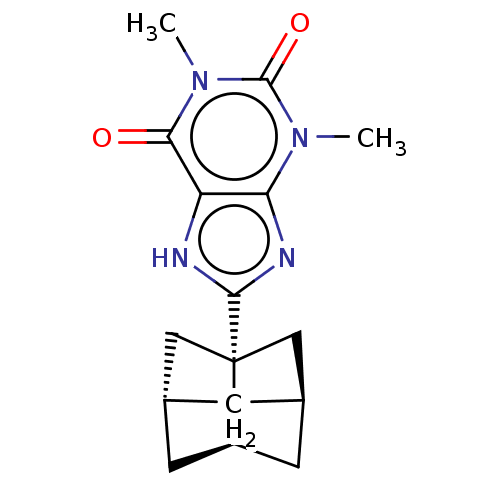
(CHEMBL3093327)Show SMILES [H][C@@]12C[C@@]3([H])C[C@]([H])(C[C@]3(C1)c1nc3n(C)c(=O)n(C)c(=O)c3[nH]1)C2 |r,wD:9.12,6.6,3.3,1.0,TLB:5:6:10:2.3,THB:5:3:10:24.6.8,(14.09,-14.42,;12.36,-15.15,;12.34,-13.62,;10.87,-13.16,;12.72,-13.48,;13.43,-13.25,;13.03,-14.75,;14.92,-14.74,;11.3,-15.18,;9.98,-14.41,;10.9,-15.65,;8.44,-14.44,;7.53,-15.7,;6.07,-15.24,;4.75,-16.03,;4.75,-17.57,;3.4,-15.27,;2.07,-16.06,;3.39,-13.73,;2.04,-12.98,;4.71,-12.95,;4.68,-11.41,;6.05,-13.7,;7.5,-13.21,;13.44,-16.24,)| Show InChI InChI=1S/C16H20N4O2/c1-19-12-11(13(21)20(2)15(19)22)17-14(18-12)16-6-8-3-9(7-16)5-10(16)4-8/h8-10H,3-7H2,1-2H3,(H,17,18)/t8-,9-,10-,16+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.124 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Binding affinity to rat adenosine A1 receptor |
Bioorg Med Chem 21: 7435-52 (2013)
Article DOI: 10.1016/j.bmc.2013.09.044
BindingDB Entry DOI: 10.7270/Q2TT4TWF |
More data for this
Ligand-Target Pair | |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50192768
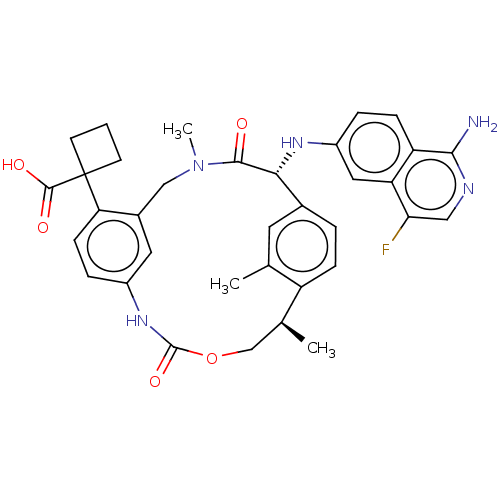
(CHEMBL3900166)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCC1)C(O)=O |r| Show InChI InChI=1S/C35H36FN5O5/c1-19-13-21-5-8-25(19)20(2)18-46-34(45)40-23-7-10-28(35(33(43)44)11-4-12-35)22(14-23)17-41(3)32(42)30(21)39-24-6-9-26-27(15-24)29(36)16-38-31(26)37/h5-10,13-16,20,30,39H,4,11-12,17-18H2,1-3H3,(H2,37,38)(H,40,45)(H,43,44)/t20-,30+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human TF-factor 7a using factor 10 as substrate at 37 degC |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450737

(CHEMBL97314)Show InChI InChI=1S/C10H14N2O/c1-8-4-10(6-11-5-8)13-7-9-2-3-12-9/h4-6,9,12H,2-3,7H2,1H3/t9-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM189445
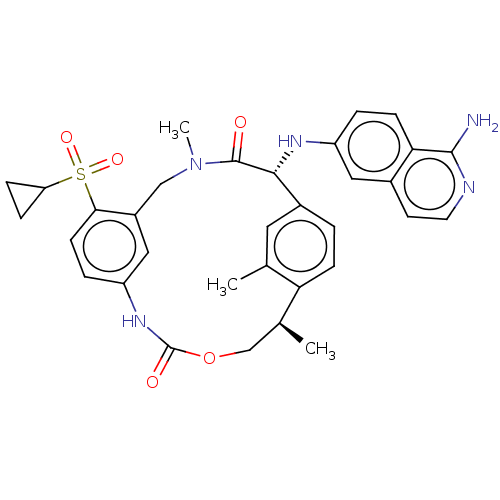
(US9174974, Example 31)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)nccc4c3)c3ccc1c(C)c3)c2)S(=O)(=O)C1CC1 |r| Show InChI InChI=1S/C33H35N5O5S/c1-19-14-22-4-9-27(19)20(2)18-43-33(40)37-25-6-11-29(44(41,42)26-7-8-26)23(16-25)17-38(3)32(39)30(22)36-24-5-10-28-21(15-24)12-13-35-31(28)34/h4-6,9-16,20,26,30,36H,7-8,17-18H2,1-3H3,(H2,34,35)(H,37,40)/t20-,30+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of human TF-factor 7a (366 to 11 residues) using factor 10 as substrate after 60 mins |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450744
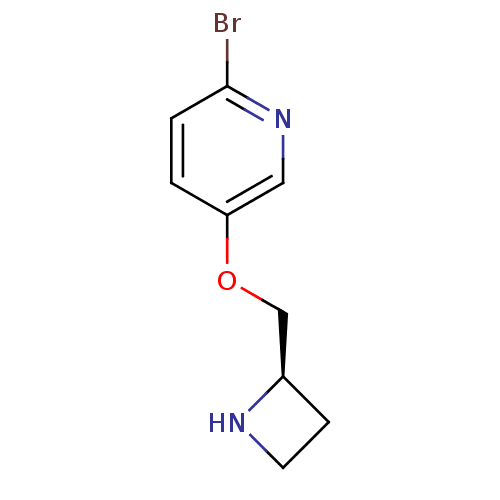
(CHEMBL314303)Show InChI InChI=1S/C9H11BrN2O/c10-9-2-1-8(5-12-9)13-6-7-3-4-11-7/h1-2,5,7,11H,3-4,6H2/t7-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Coagulation factor XI
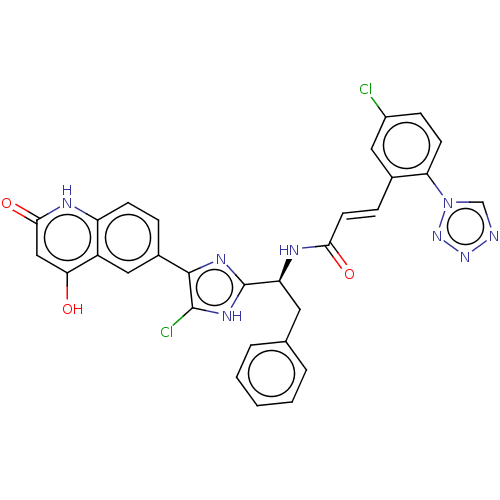
(Homo sapiens (Human)) | BDBM50096843
(CHEMBL3580754)Show SMILES Oc1cc(=O)[nH]c2ccc(cc12)-c1nc([nH]c1Cl)[C@H](Cc1ccccc1)NC(=O)\C=C\c1cc(Cl)ccc1-n1cnnn1 |r| Show InChI InChI=1S/C30H20Cl2N8O3/c31-20-8-10-24(40-16-33-38-39-40)18(13-20)7-11-26(42)35-23(12-17-4-2-1-3-5-17)30-36-28(29(32)37-30)19-6-9-22-21(14-19)25(41)15-27(43)34-22/h1-11,13-16,23,41H,12H2,(H,35,42)/b11-7+,28-19-/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Inhibition of human coagulation factor 11a using p-nitroaniline as substrate assessed as substrate hydrolysis by spectrophotometry |
ACS Med Chem Lett 6: 590-5 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00066
BindingDB Entry DOI: 10.7270/Q2B27X24 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450731
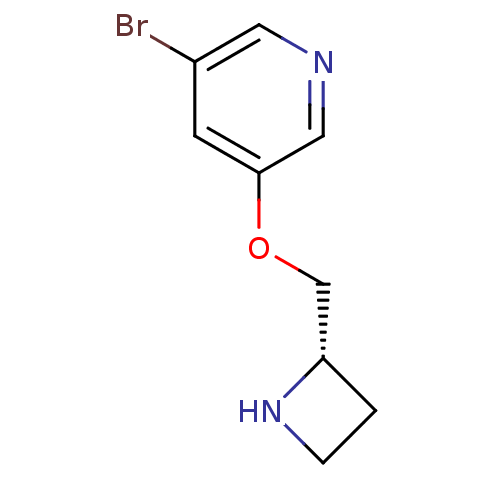
(CHEMBL96733)Show InChI InChI=1S/C9H11BrN2O/c10-7-3-9(5-11-4-7)13-6-8-1-2-12-8/h3-5,8,12H,1-2,6H2/t8-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 9
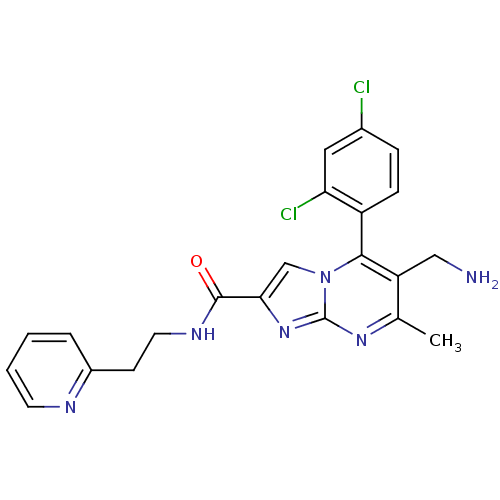
(Homo sapiens (Human)) | BDBM50324512
(6-(aminomethyl)-5-(2,4-dichlorophenyl)-7-methyl-N-...)Show SMILES Cc1nc2nc(cn2c(c1CN)-c1ccc(Cl)cc1Cl)C(=O)NCCc1ccccn1 |(10.76,-1,;9.45,-1.81,;8.1,-1.09,;6.8,-1.9,;5.33,-1.46,;4.46,-2.74,;5.4,-3.96,;6.85,-3.44,;8.2,-4.17,;9.51,-3.36,;10.87,-4.08,;12.17,-3.27,;8.17,-5.7,;6.82,-6.44,;6.79,-7.98,;8.11,-8.78,;8.08,-10.32,;9.46,-8.02,;9.48,-6.48,;10.83,-5.74,;2.92,-2.78,;2.11,-1.48,;2.19,-4.14,;.65,-4.18,;-.08,-5.54,;-1.62,-5.59,;-2.34,-6.93,;-3.87,-6.99,;-4.69,-5.68,;-3.96,-4.31,;-2.42,-4.28,)| Show InChI InChI=1S/C22H20Cl2N6O/c1-13-17(11-25)20(16-6-5-14(23)10-18(16)24)30-12-19(29-22(30)28-13)21(31)27-9-7-15-4-2-3-8-26-15/h2-6,8,10,12H,7,9,11,25H2,1H3,(H,27,31) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Inhibition of human DPP9 |
J Med Chem 53: 5620-8 (2010)
Article DOI: 10.1021/jm100634a
BindingDB Entry DOI: 10.7270/Q2S182PX |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data