

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50386178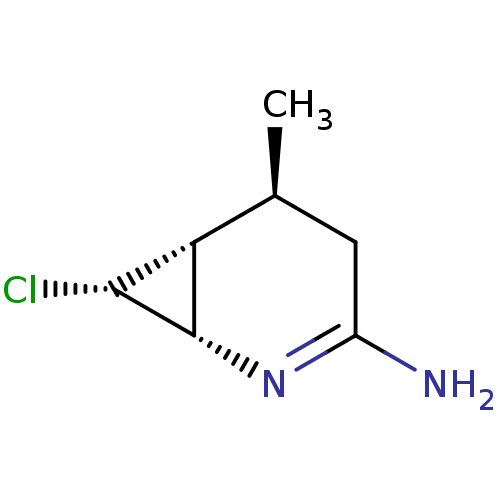 (CHEMBL1800346 | ONO-1714) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 18.6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nycomed GmbH Curated by ChEMBL | Assay Description Inhibition of human eNOS assessed as inhibition of [3H]L-arginine to [3H]L-citrulline conversion by scintillation counting | Bioorg Med Chem Lett 21: 4228-32 (2011) Article DOI: 10.1016/j.bmcl.2011.05.073 BindingDB Entry DOI: 10.7270/Q2H996GJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50225106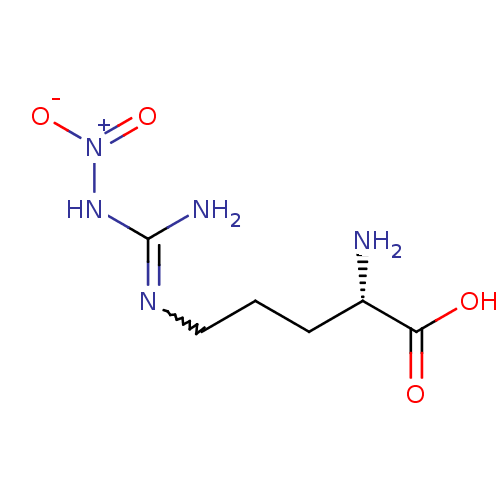 ((2S)-2-amino-5-{[(E)-amino(nitroimino)methyl]amino...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human endothelial nitric oxide synthase (eNOS) isoenzyme. | J Med Chem 41: 2858-71 (1998) Article DOI: 10.1021/jm980072p BindingDB Entry DOI: 10.7270/Q2862H4X | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50289680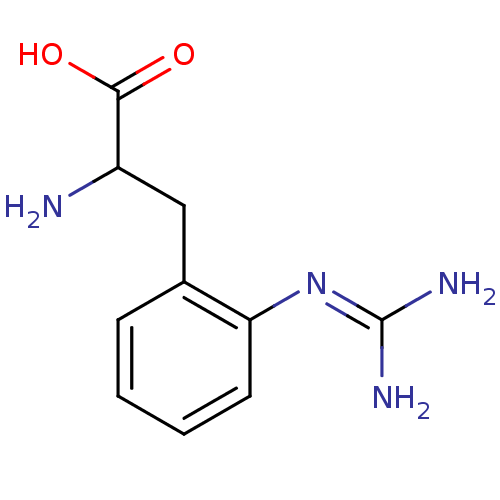 (2-Amino-3-(2-guanidino-phenyl)-propionic acid | CH...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article | 250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory constant for the inhibition of human Endothelial nitric oxide synthase | Bioorg Med Chem Lett 7: 1763-1768 (1997) Article DOI: 10.1016/S0960-894X(97)00309-0 BindingDB Entry DOI: 10.7270/Q2TQ61J5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50289681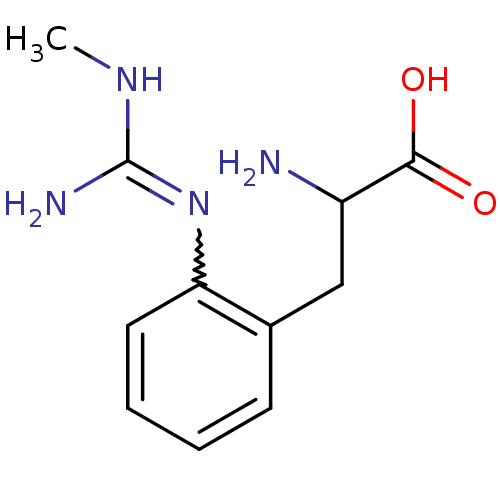 (2-Amino-3-[2-(N'-methyl-guanidino)-phenyl]-propion...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article | 270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory constant for the inhibition of human Endothelial nitric oxide synthase | Bioorg Med Chem Lett 7: 1763-1768 (1997) Article DOI: 10.1016/S0960-894X(97)00309-0 BindingDB Entry DOI: 10.7270/Q2TQ61J5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50065823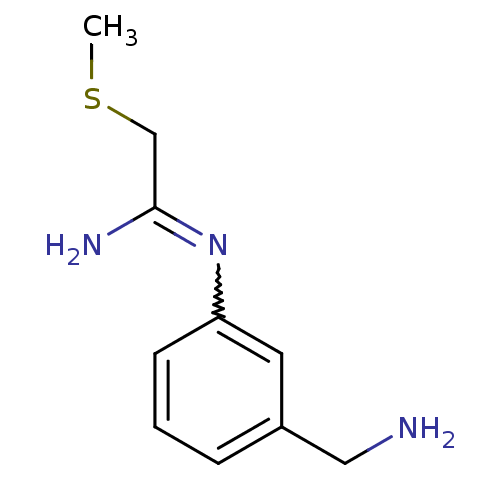 (CHEMBL555794 | N-(3-Aminomethyl-phenyl)-2-methylsu...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human endothelial nitric oxide synthase (eNOS) isoenzyme. | J Med Chem 41: 2858-71 (1998) Article DOI: 10.1021/jm980072p BindingDB Entry DOI: 10.7270/Q2862H4X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50065807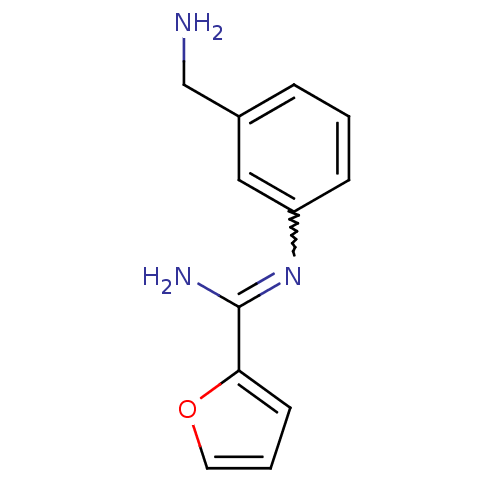 (CHEMBL552825 | N-(3-Aminomethyl-phenyl)-furan-2-ca...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human endothelial nitric oxide synthase (eNOS) isoenzyme. | J Med Chem 41: 2858-71 (1998) Article DOI: 10.1021/jm980072p BindingDB Entry DOI: 10.7270/Q2862H4X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50065843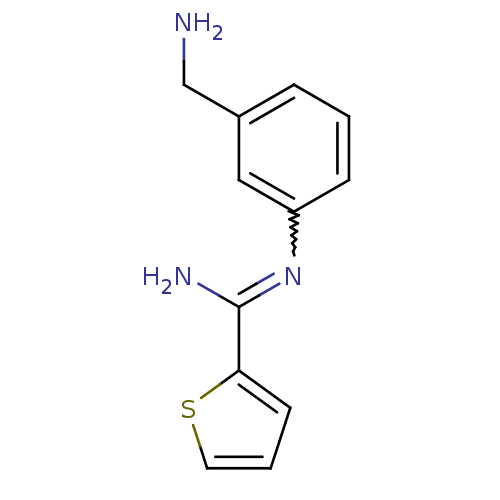 (CHEMBL553081 | CHEMBL555715 | N-(3-Aminomethyl-phe...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human endothelial nitric oxide synthase (eNOS) isoenzyme. | J Med Chem 41: 2858-71 (1998) Article DOI: 10.1021/jm980072p BindingDB Entry DOI: 10.7270/Q2862H4X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50058459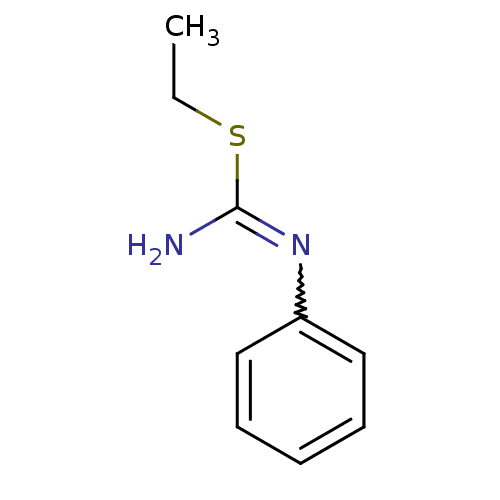 (2-Ethyl-1-phenyl-isothiourea; hydriodide | CHEMBL4...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human vascular endothelial nitric oxide synthase. | J Med Chem 40: 1901-5 (1997) Article DOI: 10.1021/jm960785c BindingDB Entry DOI: 10.7270/Q2SJ1M94 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50230993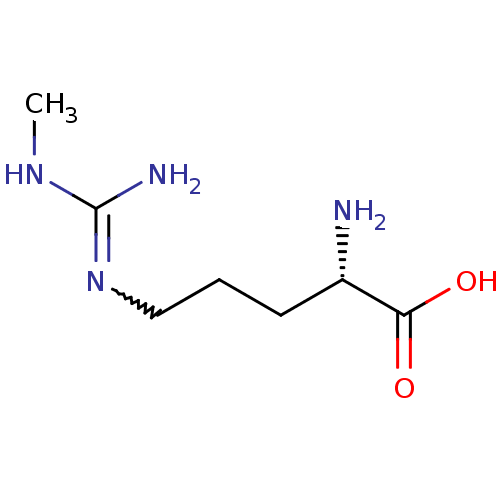 ((2S)-2-amino-5-[(N-methylcarbamimidoyl)amino]penta...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article | 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory constant for the inhibition of human Endothelial nitric oxide synthase | Bioorg Med Chem Lett 7: 1763-1768 (1997) Article DOI: 10.1016/S0960-894X(97)00309-0 BindingDB Entry DOI: 10.7270/Q2TQ61J5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50230993 ((2S)-2-amino-5-[(N-methylcarbamimidoyl)amino]penta...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human endothelial nitric oxide synthase (eNOS) isoenzyme. | J Med Chem 41: 2858-71 (1998) Article DOI: 10.1021/jm980072p BindingDB Entry DOI: 10.7270/Q2862H4X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50072297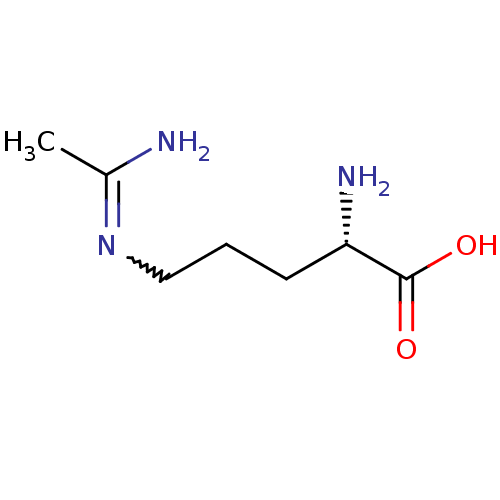 ((S)-5-Acetimidoylamino-2-amino-pent | (S)-5-Acetim...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid UniChem Patents Similars | Article | 810 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory constant for the inhibition of human Endothelial nitric oxide synthase | Bioorg Med Chem Lett 7: 1763-1768 (1997) Article DOI: 10.1016/S0960-894X(97)00309-0 BindingDB Entry DOI: 10.7270/Q2TQ61J5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50058469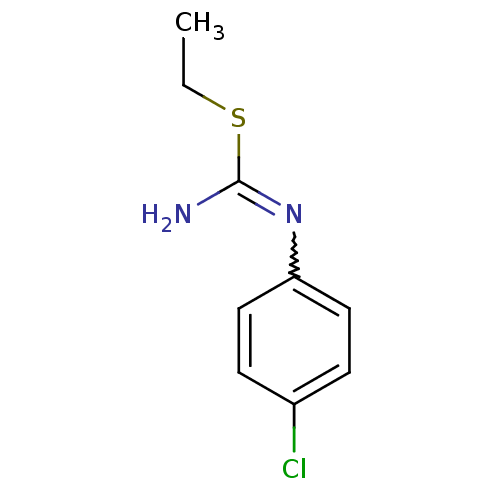 (1-(4-Chloro-phenyl)-2-ethyl-isothiourea; hydrochlo...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human vascular endothelial nitric oxide synthase. | J Med Chem 40: 1901-5 (1997) Article DOI: 10.1021/jm960785c BindingDB Entry DOI: 10.7270/Q2SJ1M94 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50058443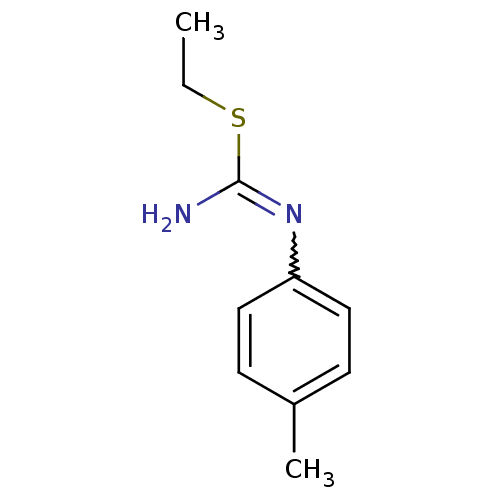 (2-Ethyl-1-p-tolyl-isothiourea; hydrochloride | CHE...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human vascular endothelial nitric oxide synthase. | J Med Chem 40: 1901-5 (1997) Article DOI: 10.1021/jm960785c BindingDB Entry DOI: 10.7270/Q2SJ1M94 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50258688 (CHEMBL4062164) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 1.03E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Chemistry, Department of Molecular Biosciences, Chemistry of Life Processes Institute, Center for Molecular Innovation and Drug Discovery, Northwestern University , Evanston, Illinois 6 Curated by ChEMBL | Assay Description Inhibition of recombinant human eNOS expressed in Escherichia coli using L-arginine as substrate after 30 secs by hemoglobin capture assay | J Med Chem 60: 7146-7165 (2017) Article DOI: 10.1021/acs.jmedchem.7b00835 BindingDB Entry DOI: 10.7270/Q2639S64 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50065813 (CHEMBL555584 | N-(3-Aminomethyl-phenyl)-2-fluoro-a...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human endothelial nitric oxide synthase (eNOS) isoenzyme. | J Med Chem 41: 2858-71 (1998) Article DOI: 10.1021/jm980072p BindingDB Entry DOI: 10.7270/Q2862H4X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50341684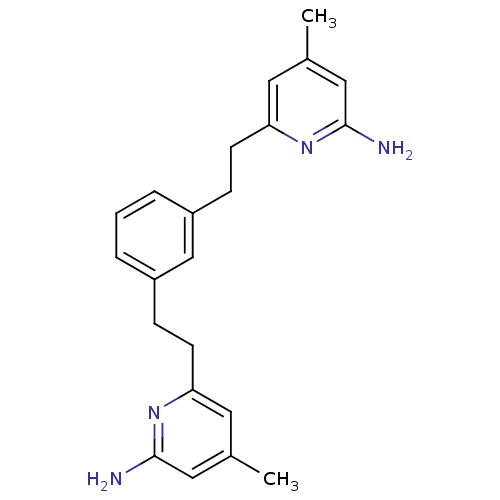 (6,6'-(2,2'-(1,3-Phenylene)bis(ethane-2,1-diyl))bis...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | US Patent | 1.41E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astex | Assay Description The NOSs isoform assays involved subjecting 3-8 to an oxyhemoglobin NO capture assay using a Biotek Gen5� microplate reader. IC50 values for each com... | J Med Chem 52: 379-88 (2009) BindingDB Entry DOI: 10.7270/Q21N83G1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50592473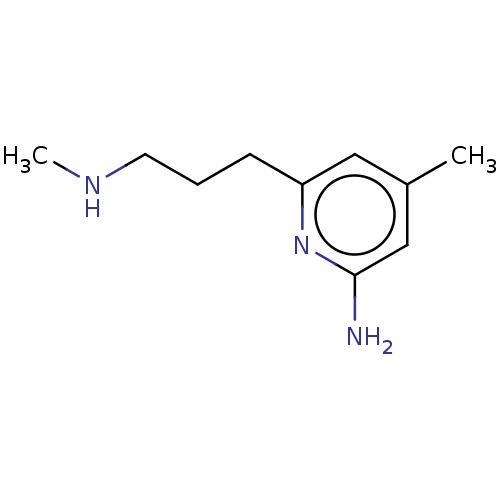 (CHEMBL5191295) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem | PDB Article PubMed | 1.47E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Citation and Details Article DOI: 10.1016/j.bmc.2022.116878 BindingDB Entry DOI: 10.7270/Q2VQ36P6 | ||||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50258622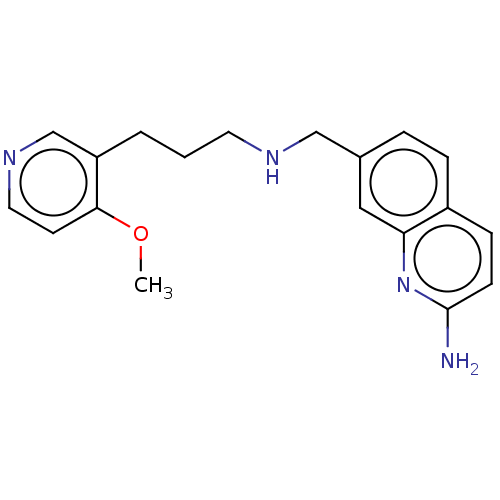 (CHEMBL4097898) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | Article PubMed | 1.58E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Chemistry, Department of Molecular Biosciences, Chemistry of Life Processes Institute, Center for Molecular Innovation and Drug Discovery, Northwestern University , Evanston, Illinois 6 Curated by ChEMBL | Assay Description Inhibition of recombinant human eNOS expressed in Escherichia coli using L-arginine as substrate after 30 secs by hemoglobin capture assay | J Med Chem 60: 7146-7165 (2017) Article DOI: 10.1021/acs.jmedchem.7b00835 BindingDB Entry DOI: 10.7270/Q2639S64 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50289677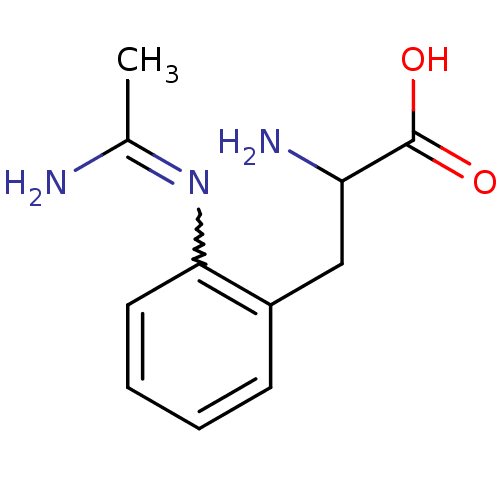 (3-(2-Acetimidoylamino-phenyl)-2-amino-propionic ac...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory constant for the inhibition of human Endothelial nitric oxide synthase | Bioorg Med Chem Lett 7: 1763-1768 (1997) Article DOI: 10.1016/S0960-894X(97)00309-0 BindingDB Entry DOI: 10.7270/Q2TQ61J5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50058448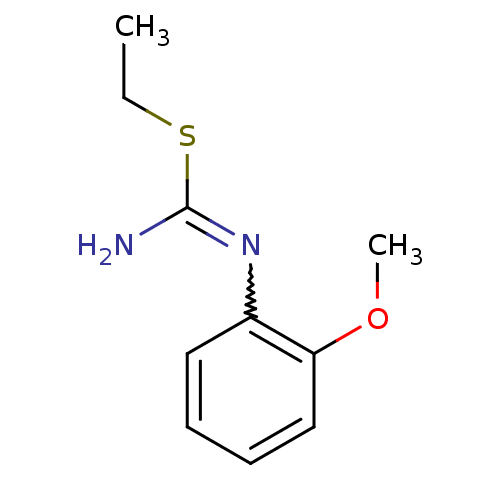 (2-Ethyl-1-(2-methoxy-phenyl)-isothiourea; hydrochl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human vascular endothelial nitric oxide synthase. | J Med Chem 40: 1901-5 (1997) Article DOI: 10.1021/jm960785c BindingDB Entry DOI: 10.7270/Q2SJ1M94 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50058455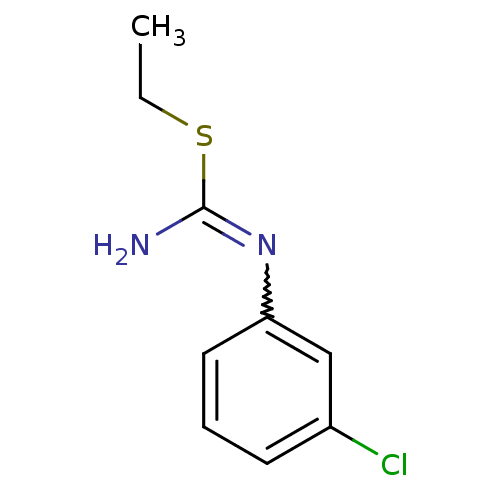 (1-(3-Chloro-phenyl)-2-ethyl-isothiourea; hydrochlo...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human vascular endothelial nitric oxide synthase. | J Med Chem 40: 1901-5 (1997) Article DOI: 10.1021/jm960785c BindingDB Entry DOI: 10.7270/Q2SJ1M94 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50058441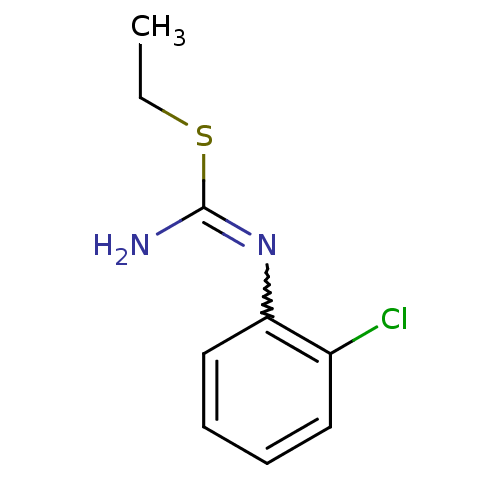 (1-(2-Chloro-phenyl)-2-ethyl-isothiourea; hydrochlo...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human vascular endothelial nitric oxide synthase. | J Med Chem 40: 1901-5 (1997) Article DOI: 10.1021/jm960785c BindingDB Entry DOI: 10.7270/Q2SJ1M94 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50065848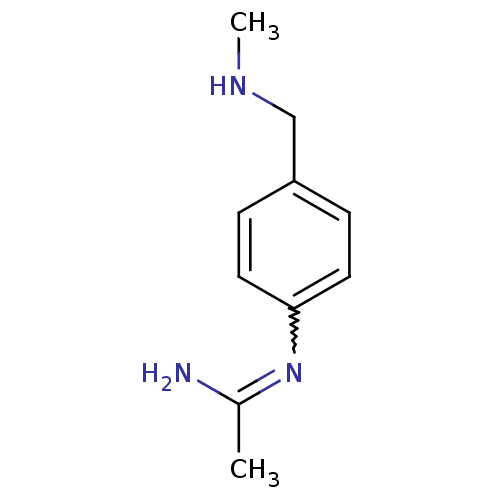 (CHEMBL539793 | N-(4-Methylaminomethyl-phenyl)-acet...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human endothelial nitric oxide synthase (eNOS) isoenzyme. | J Med Chem 41: 2858-71 (1998) Article DOI: 10.1021/jm980072p BindingDB Entry DOI: 10.7270/Q2862H4X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50592472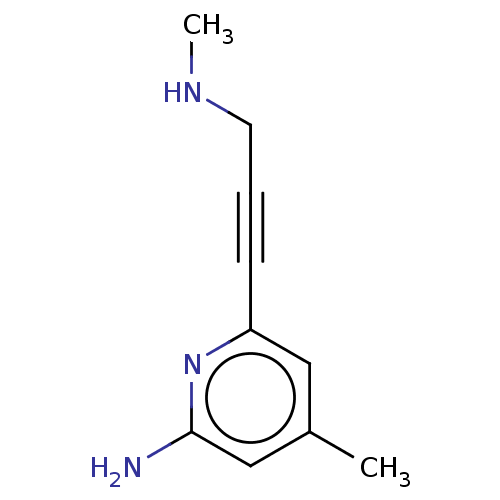 (CHEMBL5176637) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem | PDB Article PubMed | 1.92E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Citation and Details Article DOI: 10.1016/j.bmc.2022.116878 BindingDB Entry DOI: 10.7270/Q2VQ36P6 | ||||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50058440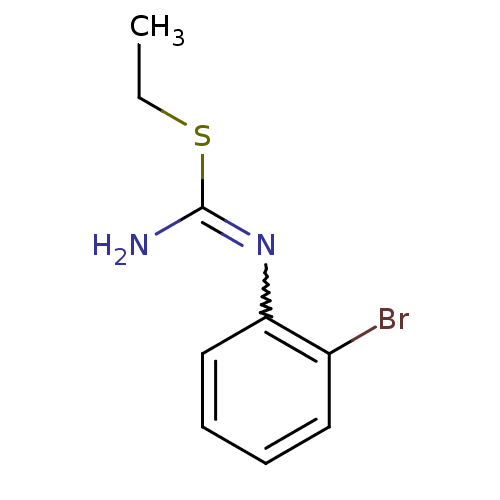 (1-(2-Bromo-phenyl)-2-ethyl-isothiourea; hydriodide...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human vascular endothelial nitric oxide synthase. | J Med Chem 40: 1901-5 (1997) Article DOI: 10.1021/jm960785c BindingDB Entry DOI: 10.7270/Q2SJ1M94 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50258602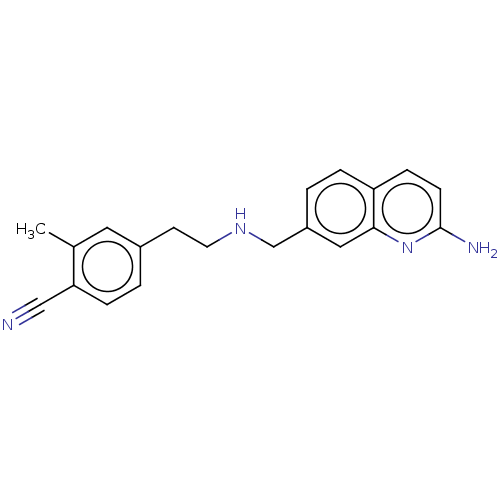 (CHEMBL4068062) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 2.08E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Chemistry, Department of Molecular Biosciences, Chemistry of Life Processes Institute, Center for Molecular Innovation and Drug Discovery, Northwestern University , Evanston, Illinois 6 Curated by ChEMBL | Assay Description Inhibition of recombinant human eNOS expressed in Escherichia coli using L-arginine as substrate after 30 secs by hemoglobin capture assay | J Med Chem 60: 7146-7165 (2017) Article DOI: 10.1021/acs.jmedchem.7b00835 BindingDB Entry DOI: 10.7270/Q2639S64 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50446252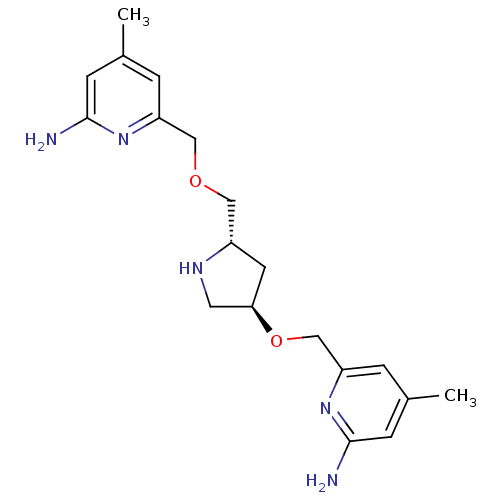 (CHEMBL3109186 | US9732037, Compound 6) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB US Patent | 2.18E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description NOS inhibition assays of representative compounds 1-21 were undertaken, and the results are summarized in Table 1, below. All NOS isoforms were expre... | US Patent US9732037 (2017) BindingDB Entry DOI: 10.7270/Q26W9D6H | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50446249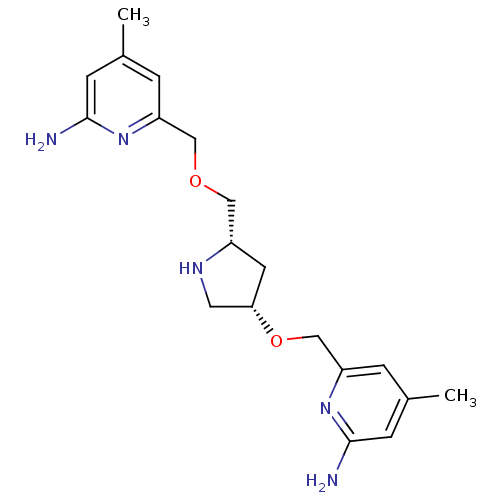 (CHEMBL3109189 | US9732037, Compound 8) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | 2.36E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description NOS inhibition assays of representative compounds 1-21 were undertaken, and the results are summarized in Table 1, below. All NOS isoforms were expre... | US Patent US9732037 (2017) BindingDB Entry DOI: 10.7270/Q26W9D6H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50058452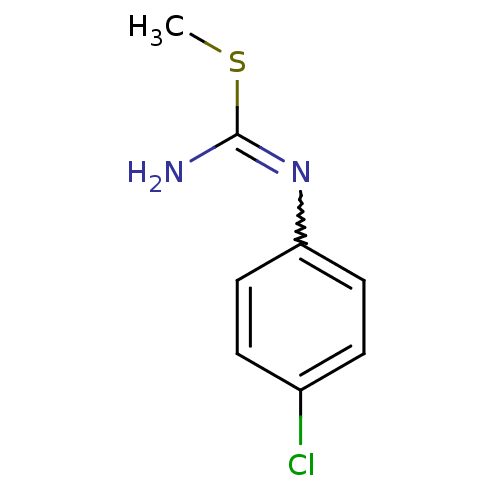 (1-(4-Chloro-phenyl)-2-methyl-isothiourea; hydriodi...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human vascular endothelial nitric oxide synthase. | J Med Chem 40: 1901-5 (1997) Article DOI: 10.1021/jm960785c BindingDB Entry DOI: 10.7270/Q2SJ1M94 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50341682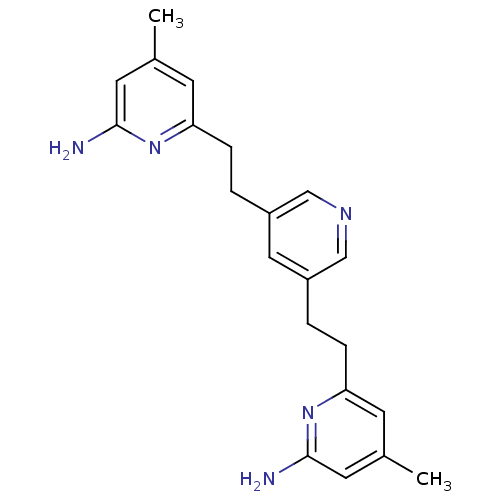 (6,6'-(2,2'-(Pyridine-3,5-diyl)bis(ethane-2,1-diyl)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | PDB US Patent | 2.68E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astex | Assay Description The NOSs isoform assays involved subjecting 3-8 to an oxyhemoglobin NO capture assay using a Biotek Gen5� microplate reader. IC50 values for each com... | J Med Chem 52: 379-88 (2009) BindingDB Entry DOI: 10.7270/Q21N83G1 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50341682 (6,6'-(2,2'-(Pyridine-3,5-diyl)bis(ethane-2,1-diyl)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 2.68E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of California, Irvine | Assay Description Inhibition assay using nitric oxide synthases. | Biochemistry 49: 10803-10 (2010) Article DOI: 10.1021/bi1013479 BindingDB Entry DOI: 10.7270/Q2KS6Q51 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50258601 (CHEMBL4089246) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Chemistry, Department of Molecular Biosciences, Chemistry of Life Processes Institute, Center for Molecular Innovation and Drug Discovery, Northwestern University , Evanston, Illinois 6 Curated by ChEMBL | Assay Description Inhibition of recombinant human eNOS expressed in Escherichia coli using L-arginine as substrate after 30 secs by hemoglobin capture assay | J Med Chem 60: 7146-7165 (2017) Article DOI: 10.1021/acs.jmedchem.7b00835 BindingDB Entry DOI: 10.7270/Q2639S64 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50058447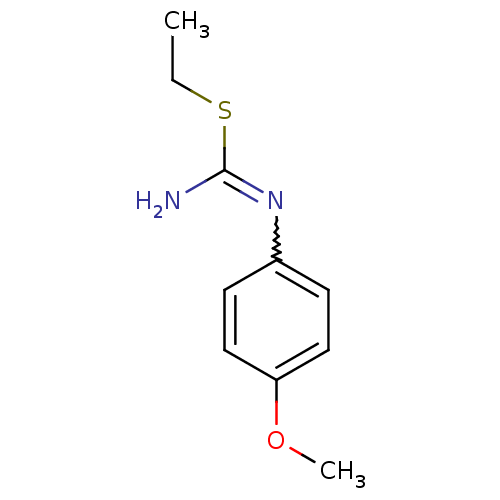 (2-Ethyl-1-(4-methoxy-phenyl)-isothiourea; hydriodi...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human vascular endothelial nitric oxide synthase. | J Med Chem 40: 1901-5 (1997) Article DOI: 10.1021/jm960785c BindingDB Entry DOI: 10.7270/Q2SJ1M94 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50058444 (2-Ethyl-1-(4-hydroxy-phenyl)-isothiourea; hydriodi...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human vascular endothelial nitric oxide synthase. | J Med Chem 40: 1901-5 (1997) Article DOI: 10.1021/jm960785c BindingDB Entry DOI: 10.7270/Q2SJ1M94 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50058468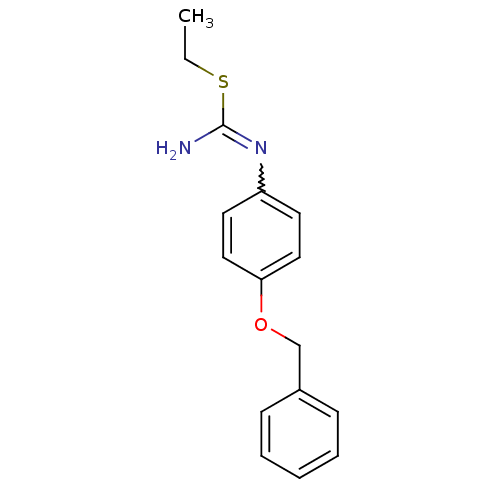 (1-(4-Benzyloxy-phenyl)-2-ethyl-isothiourea; hydrio...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human vascular endothelial nitric oxide synthase. | J Med Chem 40: 1901-5 (1997) Article DOI: 10.1021/jm960785c BindingDB Entry DOI: 10.7270/Q2SJ1M94 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50441007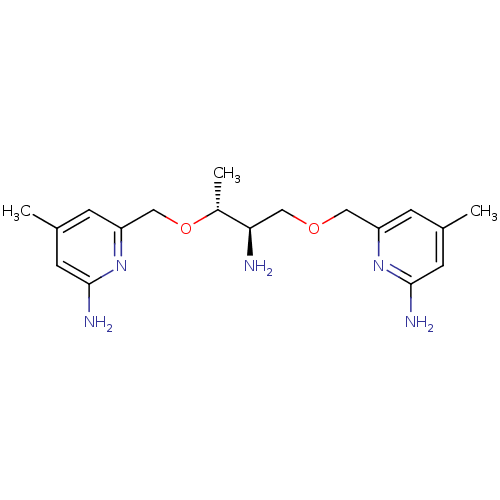 (CHEMBL2430149 | US9732037, Compound 19) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description NOS inhibition assays of representative compounds 1-21 were undertaken, and the results are summarized in Table 1, below. All NOS isoforms were expre... | US Patent US9732037 (2017) BindingDB Entry DOI: 10.7270/Q26W9D6H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50065842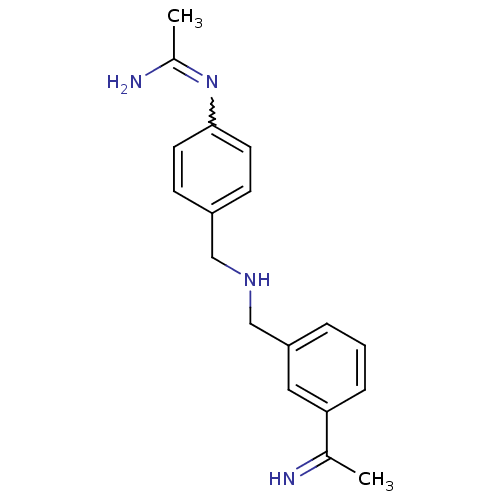 (CHEMBL540048 | N-(4-{[3-(1-Imino-ethyl)-benzylamin...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human endothelial nitric oxide synthase (eNOS) isoenzyme. | J Med Chem 41: 2858-71 (1998) Article DOI: 10.1021/jm980072p BindingDB Entry DOI: 10.7270/Q2862H4X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50441008 (CHEMBL2430150 | US9732037, Compound 20) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | PDB US Patent | 3.04E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description NOS inhibition assays of representative compounds 1-21 were undertaken, and the results are summarized in Table 1, below. All NOS isoforms were expre... | US Patent US9732037 (2017) BindingDB Entry DOI: 10.7270/Q26W9D6H | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50058442 (2-Ethyl-1-(3-methoxy-phenyl)-isothiourea; hydriodi...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human vascular endothelial nitric oxide synthase. | J Med Chem 40: 1901-5 (1997) Article DOI: 10.1021/jm960785c BindingDB Entry DOI: 10.7270/Q2SJ1M94 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50446251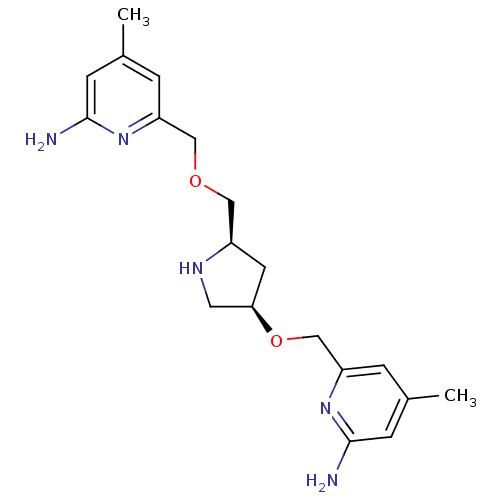 (CHEMBL3109187 | US9732037, Compound 7) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | 3.47E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description NOS inhibition assays of representative compounds 1-21 were undertaken, and the results are summarized in Table 1, below. All NOS isoforms were expre... | US Patent US9732037 (2017) BindingDB Entry DOI: 10.7270/Q26W9D6H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM389162 (US9951014, Name 4) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 3.61E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astex | Assay Description The NOSs isoform assays involved subjecting 3-8 to an oxyhemoglobin NO capture assay using a Biotek Gen5� microplate reader. IC50 values for each com... | J Med Chem 52: 379-88 (2009) BindingDB Entry DOI: 10.7270/Q21N83G1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50592474 (CHEMBL5189279) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem | PDB Article PubMed | 3.66E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Citation and Details Article DOI: 10.1016/j.bmc.2022.116878 BindingDB Entry DOI: 10.7270/Q2VQ36P6 | ||||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50065847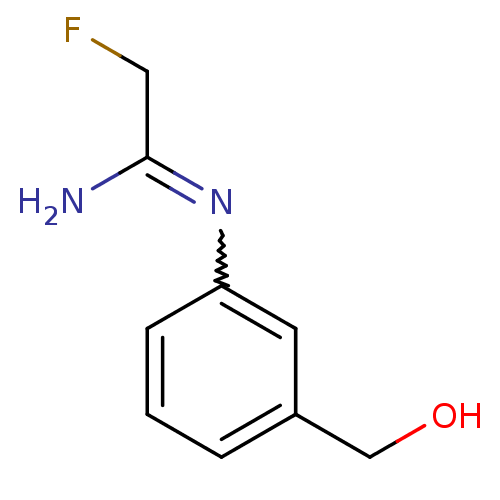 (2-Fluoro-N-(3-hydroxymethyl-phenyl)-acetamidine | ...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 3.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human endothelial nitric oxide synthase (eNOS) isoenzyme. | J Med Chem 41: 2858-71 (1998) Article DOI: 10.1021/jm980072p BindingDB Entry DOI: 10.7270/Q2862H4X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM275050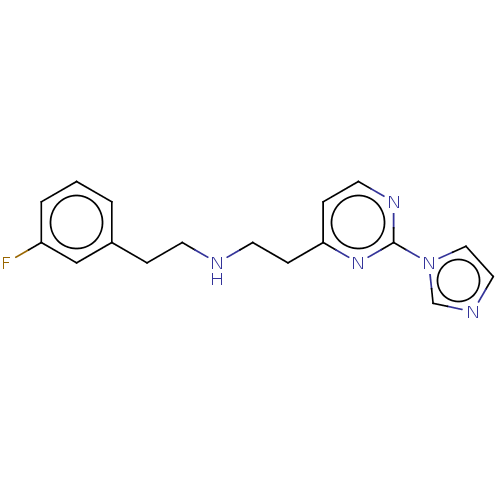 (US9878996, Compound 6) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem | PDB US Patent | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description Assays were performed at the NIMH Psychoactive Drug Screening Program (PDSP) at UNC-Chapel Hill. | US Patent US9878996 (2018) BindingDB Entry DOI: 10.7270/Q2PN97PN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50058445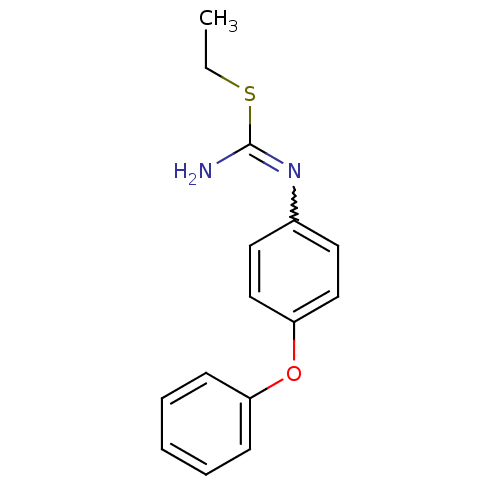 (2-Ethyl-1-(4-phenoxy-phenyl)-isothiourea; hydriodi...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity against human vascular endothelial nitric oxide synthase. | J Med Chem 40: 1901-5 (1997) Article DOI: 10.1021/jm960785c BindingDB Entry DOI: 10.7270/Q2SJ1M94 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM275053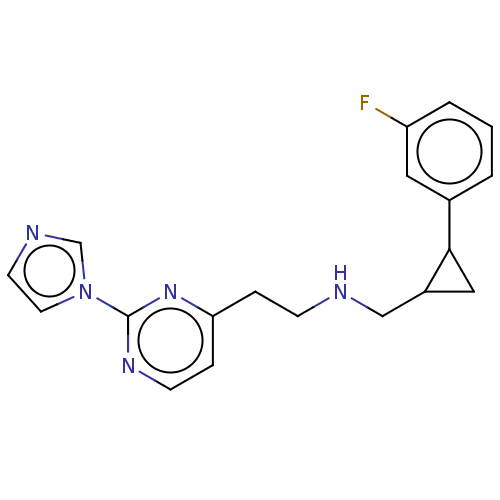 (US9878996, Compound 26) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem | PDB US Patent | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description Assays were performed at the NIMH Psychoactive Drug Screening Program (PDSP) at UNC-Chapel Hill. | US Patent US9878996 (2018) BindingDB Entry DOI: 10.7270/Q2PN97PN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50266590 (CHEMBL4066044) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 4.19E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition of recombinant human full-length eNOS expressed in Escherichia coli BL21(DE3) assessed as reduction in nitric oxide production using L-arg... | J Med Chem 60: 3958-3978 (2017) Article DOI: 10.1021/acs.jmedchem.7b00259 BindingDB Entry DOI: 10.7270/Q2RV0R5D | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50341684 (6,6'-(2,2'-(1,3-Phenylene)bis(ethane-2,1-diyl))bis...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 4.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of California, Irvine | Assay Description Inhibition assay using nitric oxide synthases. | Biochemistry 49: 10803-10 (2010) Article DOI: 10.1021/bi1013479 BindingDB Entry DOI: 10.7270/Q2KS6Q51 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM389152 (US9951014, Name 23d) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 4.29E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astex | Assay Description The NOSs isoform assays involved subjecting 3-8 to an oxyhemoglobin NO capture assay using a Biotek Gen5� microplate reader. IC50 values for each com... | J Med Chem 52: 379-88 (2009) BindingDB Entry DOI: 10.7270/Q21N83G1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50014713 (CHEMBL3262022 | US10759791, Compound 6 | US9951014...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | US Patent | 4.77E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astex | Assay Description The NOSs isoform assays involved subjecting 3-8 to an oxyhemoglobin NO capture assay using a Biotek Gen5� microplate reader. IC50 values for each com... | J Med Chem 52: 379-88 (2009) BindingDB Entry DOI: 10.7270/Q21N83G1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 284 total ) | Next | Last >> |