 Found 82 hits of Enzyme Inhibition Constant Data
Found 82 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50097579
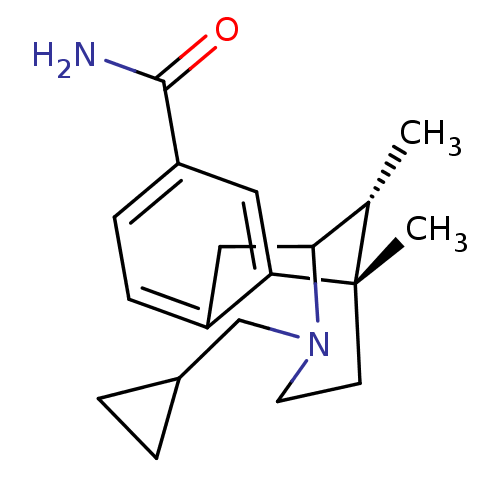
((6S,11R)-3-Cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(N)=O |r,TLB:8:9:1:14.12.13,15:14:1:4.9.3| Show InChI InChI=1S/C19H26N2O/c1-12-17-10-14-5-6-15(18(20)22)9-16(14)19(12,2)7-8-21(17)11-13-3-4-13/h5-6,9,12-13,17H,3-4,7-8,10-11H2,1-2H3,(H2,20,22)/t12-,17?,19-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224870
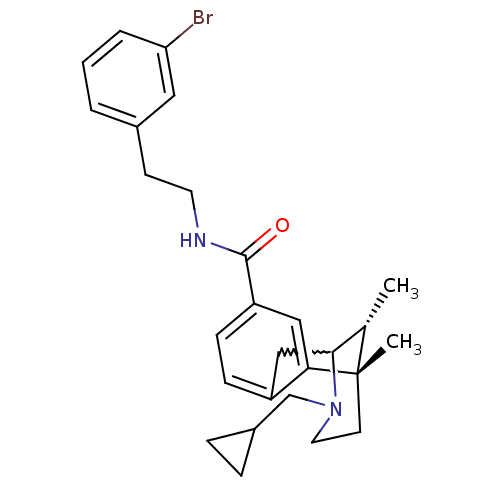
((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1cccc(Br)c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C27H33BrN2O/c1-18-25-16-21-8-9-22(26(31)29-12-10-19-4-3-5-23(28)14-19)15-24(21)27(18,2)11-13-30(25)17-20-6-7-20/h3-5,8-9,14-15,18,20,25H,6-7,10-13,16-17H2,1-2H3,(H,29,31)/t18-,25?,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0630 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224873
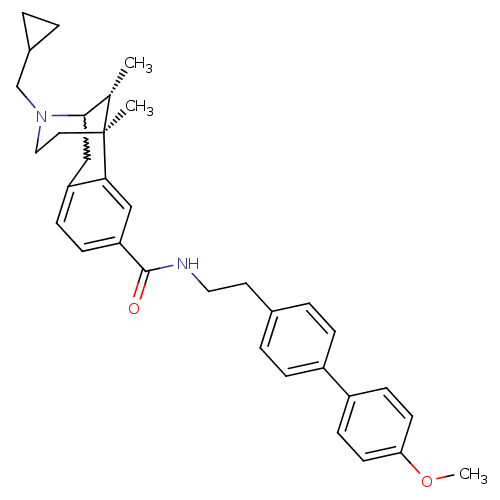
((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES COc1ccc(cc1)-c1ccc(CCNC(=O)c2ccc3CC4[C@H](C)[C@](C)(CCN4CC4CC4)c3c2)cc1 |w:22.22,TLB:35:34:23:29.28.27,30:29:23:20.34.21| Show InChI InChI=1S/C34H40N2O2/c1-23-32-21-28-10-11-29(20-31(28)34(23,2)17-19-36(32)22-25-4-5-25)33(37)35-18-16-24-6-8-26(9-7-24)27-12-14-30(38-3)15-13-27/h6-15,20,23,25,32H,4-5,16-19,21-22H2,1-3H3,(H,35,37)/t23-,32?,34-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0840 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224873

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES COc1ccc(cc1)-c1ccc(CCNC(=O)c2ccc3CC4[C@H](C)[C@](C)(CCN4CC4CC4)c3c2)cc1 |w:22.22,TLB:35:34:23:29.28.27,30:29:23:20.34.21| Show InChI InChI=1S/C34H40N2O2/c1-23-32-21-28-10-11-29(20-31(28)34(23,2)17-19-36(32)22-25-4-5-25)33(37)35-18-16-24-6-8-26(9-7-24)27-12-14-30(38-3)15-13-27/h6-15,20,23,25,32H,4-5,16-19,21-22H2,1-3H3,(H,35,37)/t23-,32?,34-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224871
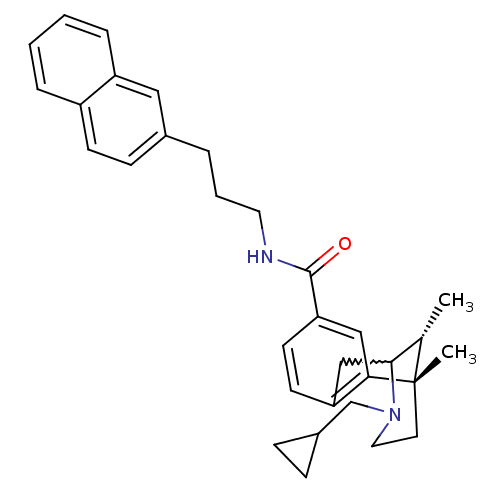
((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCCc1ccc2ccccc2c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C32H38N2O/c1-22-30-20-27-13-14-28(19-29(27)32(22,2)15-17-34(30)21-24-9-10-24)31(35)33-16-5-6-23-11-12-25-7-3-4-8-26(25)18-23/h3-4,7-8,11-14,18-19,22,24,30H,5-6,9-10,15-17,20-21H2,1-2H3,(H,33,35)/t22-,30?,32-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224879
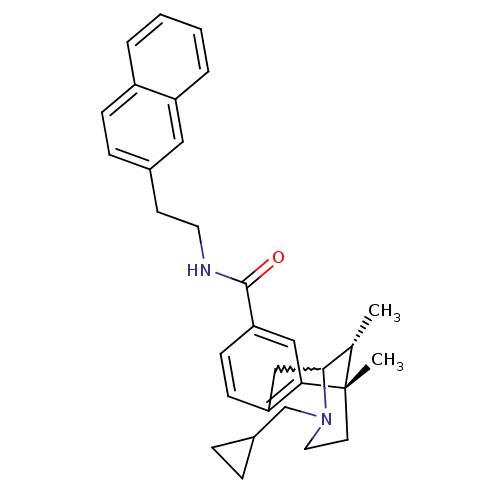
((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc2ccccc2c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C31H36N2O/c1-21-29-19-26-11-12-27(18-28(26)31(21,2)14-16-33(29)20-23-7-8-23)30(34)32-15-13-22-9-10-24-5-3-4-6-25(24)17-22/h3-6,9-12,17-18,21,23,29H,7-8,13-16,19-20H2,1-2H3,(H,32,34)/t21-,29?,31-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224879

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc2ccccc2c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C31H36N2O/c1-21-29-19-26-11-12-27(18-28(26)31(21,2)14-16-33(29)20-23-7-8-23)30(34)32-15-13-22-9-10-24-5-3-4-6-25(24)17-22/h3-6,9-12,17-18,21,23,29H,7-8,13-16,19-20H2,1-2H3,(H,32,34)/t21-,29?,31-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224869
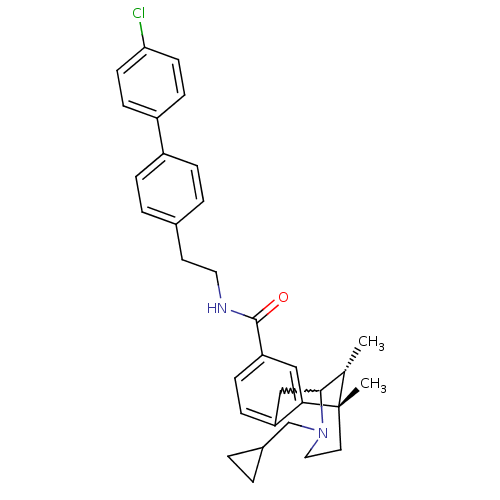
((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccc(Cl)cc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H37ClN2O/c1-22-31-20-27-9-10-28(19-30(27)33(22,2)16-18-36(31)21-24-3-4-24)32(37)35-17-15-23-5-7-25(8-6-23)26-11-13-29(34)14-12-26/h5-14,19,22,24,31H,3-4,15-18,20-21H2,1-2H3,(H,35,37)/t22-,31?,33-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224881

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccc(C)cc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C34H40N2O/c1-23-4-10-27(11-5-23)28-12-8-25(9-13-28)16-18-35-33(37)30-15-14-29-21-32-24(2)34(3,31(29)20-30)17-19-36(32)22-26-6-7-26/h4-5,8-15,20,24,26,32H,6-7,16-19,21-22H2,1-3H3,(H,35,37)/t24-,32?,34-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50194264
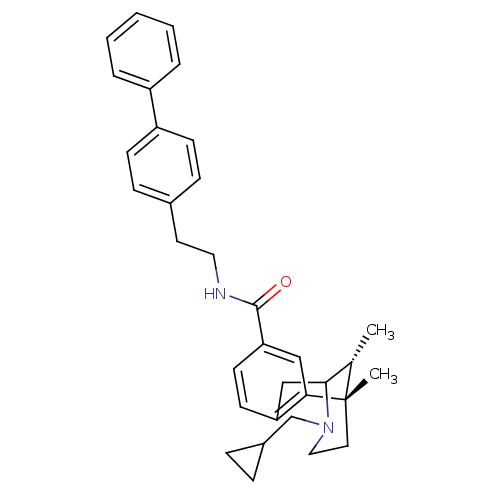
((+/-)-3-(cyclopropylmethyl)-1,2,3,4,5,6-hexahydro-...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccccc1 |r,TLB:8:9:1:14.12.13,15:14:1:4.9.3| Show InChI InChI=1S/C33H38N2O/c1-23-31-21-28-14-15-29(20-30(28)33(23,2)17-19-35(31)22-25-8-9-25)32(36)34-18-16-24-10-12-27(13-11-24)26-6-4-3-5-7-26/h3-7,10-15,20,23,25,31H,8-9,16-19,21-22H2,1-2H3,(H,34,36)/t23-,31?,33-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50097579

((6S,11R)-3-Cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(N)=O |r,TLB:8:9:1:14.12.13,15:14:1:4.9.3| Show InChI InChI=1S/C19H26N2O/c1-12-17-10-14-5-6-15(18(20)22)9-16(14)19(12,2)7-8-21(17)11-13-3-4-13/h5-6,9,12-13,17H,3-4,7-8,10-11H2,1-2H3,(H2,20,22)/t12-,17?,19-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224870

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1cccc(Br)c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C27H33BrN2O/c1-18-25-16-21-8-9-22(26(31)29-12-10-19-4-3-5-23(28)14-19)15-24(21)27(18,2)11-13-30(25)17-20-6-7-20/h3-5,8-9,14-15,18,20,25H,6-7,10-13,16-17H2,1-2H3,(H,29,31)/t18-,25?,27-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224877
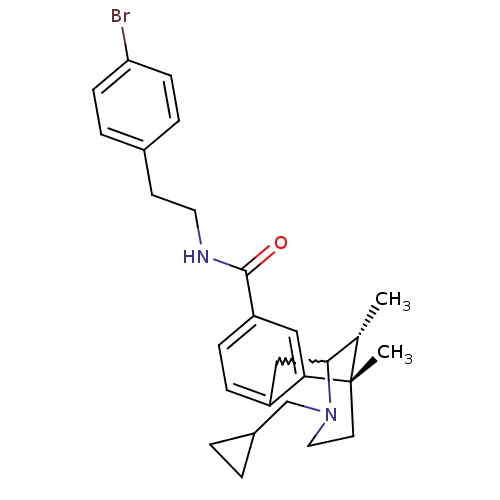
((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(Br)cc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C27H33BrN2O/c1-18-25-16-21-7-8-22(26(31)29-13-11-19-5-9-23(28)10-6-19)15-24(21)27(18,2)12-14-30(25)17-20-3-4-20/h5-10,15,18,20,25H,3-4,11-14,16-17H2,1-2H3,(H,29,31)/t18-,25?,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224869

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccc(Cl)cc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H37ClN2O/c1-22-31-20-27-9-10-28(19-30(27)33(22,2)16-18-36(31)21-24-3-4-24)32(37)35-17-15-23-5-7-25(8-6-23)26-11-13-29(34)14-12-26/h5-14,19,22,24,31H,3-4,15-18,20-21H2,1-2H3,(H,35,37)/t22-,31?,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.710 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224881

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccc(C)cc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C34H40N2O/c1-23-4-10-27(11-5-23)28-12-8-25(9-13-28)16-18-35-33(37)30-15-14-29-21-32-24(2)34(3,31(29)20-30)17-19-36(32)22-26-6-7-26/h4-5,8-15,20,24,26,32H,6-7,16-19,21-22H2,1-3H3,(H,35,37)/t24-,32?,34-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.720 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50194264

((+/-)-3-(cyclopropylmethyl)-1,2,3,4,5,6-hexahydro-...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccccc1 |r,TLB:8:9:1:14.12.13,15:14:1:4.9.3| Show InChI InChI=1S/C33H38N2O/c1-23-31-21-28-14-15-29(20-30(28)33(23,2)17-19-35(31)22-25-8-9-25)32(36)34-18-16-24-10-12-27(13-11-24)26-6-4-3-5-7-26/h3-7,10-15,20,23,25,31H,8-9,16-19,21-22H2,1-2H3,(H,34,36)/t23-,31?,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.740 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224879

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc2ccccc2c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C31H36N2O/c1-21-29-19-26-11-12-27(18-28(26)31(21,2)14-16-33(29)20-23-7-8-23)30(34)32-15-13-22-9-10-24-5-3-4-6-25(24)17-22/h3-6,9-12,17-18,21,23,29H,7-8,13-16,19-20H2,1-2H3,(H,32,34)/t21-,29?,31-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224872
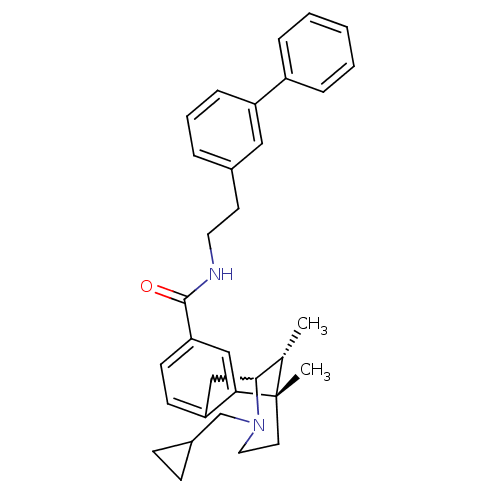
((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1cccc(c1)-c1ccccc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H38N2O/c1-23-31-21-28-13-14-29(20-30(28)33(23,2)16-18-35(31)22-25-11-12-25)32(36)34-17-15-24-7-6-10-27(19-24)26-8-4-3-5-9-26/h3-10,13-14,19-20,23,25,31H,11-12,15-18,21-22H2,1-2H3,(H,34,36)/t23-,31?,33-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.950 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224868
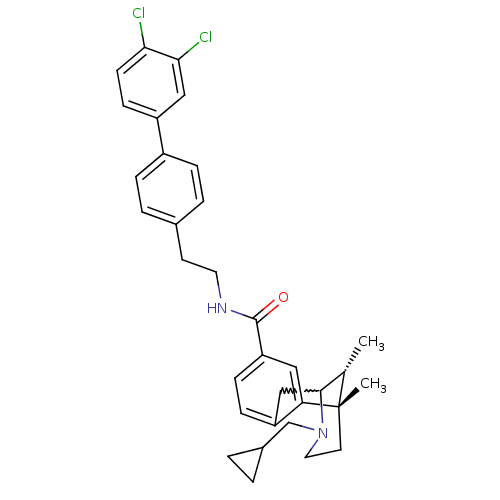
((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccc(Cl)c(Cl)c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H36Cl2N2O/c1-21-31-19-26-9-10-27(17-28(26)33(21,2)14-16-37(31)20-23-3-4-23)32(38)36-15-13-22-5-7-24(8-6-22)25-11-12-29(34)30(35)18-25/h5-12,17-18,21,23,31H,3-4,13-16,19-20H2,1-2H3,(H,36,38)/t21-,31?,33-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.980 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224868

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccc(Cl)c(Cl)c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H36Cl2N2O/c1-21-31-19-26-9-10-27(17-28(26)33(21,2)14-16-37(31)20-23-3-4-23)32(38)36-15-13-22-5-7-24(8-6-22)25-11-12-29(34)30(35)18-25/h5-12,17-18,21,23,31H,3-4,13-16,19-20H2,1-2H3,(H,36,38)/t21-,31?,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224874
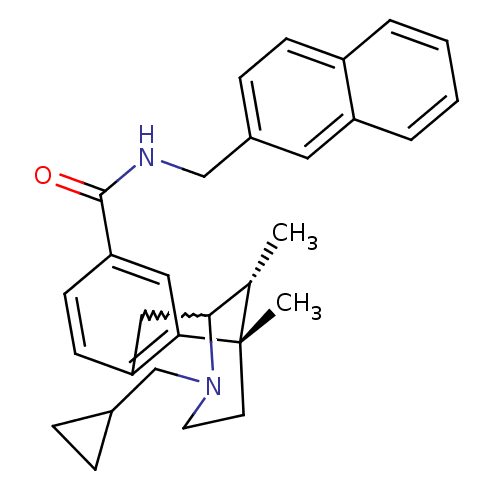
((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCc1ccc2ccccc2c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C30H34N2O/c1-20-28-17-25-11-12-26(16-27(25)30(20,2)13-14-32(28)19-21-7-8-21)29(33)31-18-22-9-10-23-5-3-4-6-24(23)15-22/h3-6,9-12,15-16,20-21,28H,7-8,13-14,17-19H2,1-2H3,(H,31,33)/t20-,28?,30-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224873

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES COc1ccc(cc1)-c1ccc(CCNC(=O)c2ccc3CC4[C@H](C)[C@](C)(CCN4CC4CC4)c3c2)cc1 |w:22.22,TLB:35:34:23:29.28.27,30:29:23:20.34.21| Show InChI InChI=1S/C34H40N2O2/c1-23-32-21-28-10-11-29(20-31(28)34(23,2)17-19-36(32)22-25-4-5-25)33(37)35-18-16-24-6-8-26(9-7-24)27-12-14-30(38-3)15-13-27/h6-15,20,23,25,32H,4-5,16-19,21-22H2,1-3H3,(H,35,37)/t23-,32?,34-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50194264

((+/-)-3-(cyclopropylmethyl)-1,2,3,4,5,6-hexahydro-...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccccc1 |r,TLB:8:9:1:14.12.13,15:14:1:4.9.3| Show InChI InChI=1S/C33H38N2O/c1-23-31-21-28-14-15-29(20-30(28)33(23,2)17-19-35(31)22-25-8-9-25)32(36)34-18-16-24-10-12-27(13-11-24)26-6-4-3-5-7-26/h3-7,10-15,20,23,25,31H,8-9,16-19,21-22H2,1-2H3,(H,34,36)/t23-,31?,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224871

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCCc1ccc2ccccc2c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C32H38N2O/c1-22-30-20-27-13-14-28(19-29(27)32(22,2)15-17-34(30)21-24-9-10-24)31(35)33-16-5-6-23-11-12-25-7-3-4-8-26(25)18-23/h3-4,7-8,11-14,18-19,22,24,30H,5-6,9-10,15-17,20-21H2,1-2H3,(H,33,35)/t22-,30?,32-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224878
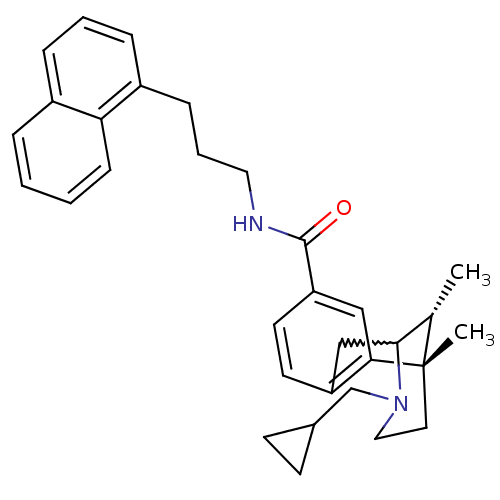
((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCCc1cccc2ccccc12 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C32H38N2O/c1-22-30-20-26-14-15-27(19-29(26)32(22,2)16-18-34(30)21-23-12-13-23)31(35)33-17-6-10-25-9-5-8-24-7-3-4-11-28(24)25/h3-5,7-9,11,14-15,19,22-23,30H,6,10,12-13,16-18,20-21H2,1-2H3,(H,33,35)/t22-,30?,32-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50083926

((6S,11R)-3-Cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1[C@H]2Cc3ccc(O)cc3[C@@]1(C)CCN2CC1CC1 |r,TLB:16:15:1:4.10.3| Show InChI InChI=1S/C18H25NO/c1-12-17-9-14-5-6-15(20)10-16(14)18(12,2)7-8-19(17)11-13-3-4-13/h5-6,10,12-13,17,20H,3-4,7-9,11H2,1-2H3/t12-,17?,18-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50083926

((6S,11R)-3-Cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1[C@H]2Cc3ccc(O)cc3[C@@]1(C)CCN2CC1CC1 |r,TLB:16:15:1:4.10.3| Show InChI InChI=1S/C18H25NO/c1-12-17-9-14-5-6-15(20)10-16(14)18(12,2)7-8-19(17)11-13-3-4-13/h5-6,10,12-13,17,20H,3-4,7-9,11H2,1-2H3/t12-,17?,18-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50083926

((6S,11R)-3-Cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1[C@H]2Cc3ccc(O)cc3[C@@]1(C)CCN2CC1CC1 |r,TLB:16:15:1:4.10.3| Show InChI InChI=1S/C18H25NO/c1-12-17-9-14-5-6-15(20)10-16(14)18(12,2)7-8-19(17)11-13-3-4-13/h5-6,10,12-13,17,20H,3-4,7-9,11H2,1-2H3/t12-,17?,18-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224872

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1cccc(c1)-c1ccccc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H38N2O/c1-23-31-21-28-13-14-29(20-30(28)33(23,2)16-18-35(31)22-25-11-12-25)32(36)34-17-15-24-7-6-10-27(19-24)26-8-4-3-5-9-26/h3-10,13-14,19-20,23,25,31H,11-12,15-18,21-22H2,1-2H3,(H,34,36)/t23-,31?,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224878

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCCc1cccc2ccccc12 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C32H38N2O/c1-22-30-20-26-14-15-27(19-29(26)32(22,2)16-18-34(30)21-23-12-13-23)31(35)33-17-6-10-25-9-5-8-24-7-3-4-11-28(24)25/h3-5,7-9,11,14-15,19,22-23,30H,6,10,12-13,16-18,20-21H2,1-2H3,(H,33,35)/t22-,30?,32-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224867
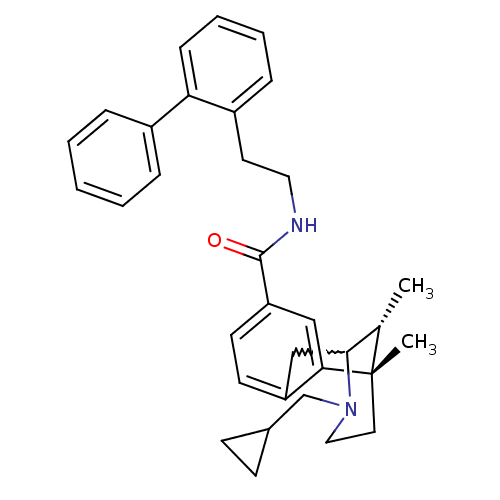
((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccccc1-c1ccccc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H38N2O/c1-23-31-21-27-14-15-28(20-30(27)33(23,2)17-19-35(31)22-24-12-13-24)32(36)34-18-16-26-10-6-7-11-29(26)25-8-4-3-5-9-25/h3-11,14-15,20,23-24,31H,12-13,16-19,21-22H2,1-2H3,(H,34,36)/t23-,31?,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224877

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(Br)cc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C27H33BrN2O/c1-18-25-16-21-7-8-22(26(31)29-13-11-19-5-9-23(28)10-6-19)15-24(21)27(18,2)12-14-30(25)17-20-3-4-20/h5-10,15,18,20,25H,3-4,11-14,16-17H2,1-2H3,(H,29,31)/t18-,25?,27-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224875
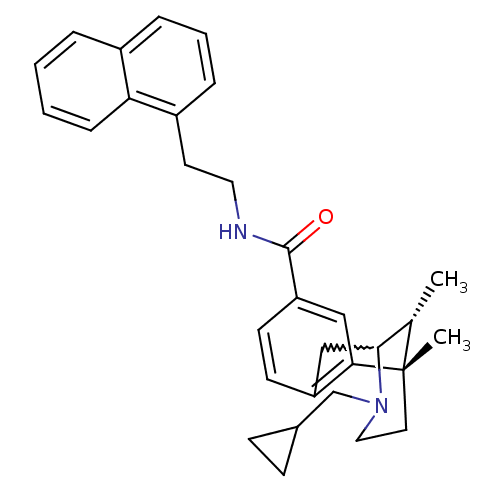
((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1cccc2ccccc12 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C31H36N2O/c1-21-29-19-25-12-13-26(18-28(25)31(21,2)15-17-33(29)20-22-10-11-22)30(34)32-16-14-24-8-5-7-23-6-3-4-9-27(23)24/h3-9,12-13,18,21-22,29H,10-11,14-17,19-20H2,1-2H3,(H,32,34)/t21-,29?,31-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224880
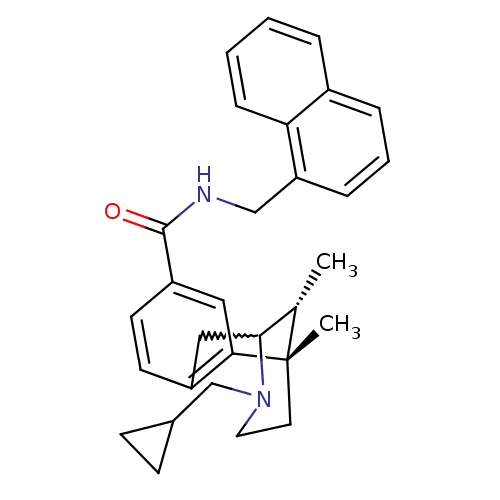
((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCc1cccc2ccccc12 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C30H34N2O/c1-20-28-17-23-12-13-24(16-27(23)30(20,2)14-15-32(28)19-21-10-11-21)29(33)31-18-25-8-5-7-22-6-3-4-9-26(22)25/h3-9,12-13,16,20-21,28H,10-11,14-15,17-19H2,1-2H3,(H,31,33)/t20-,28?,30-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224877

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(Br)cc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C27H33BrN2O/c1-18-25-16-21-7-8-22(26(31)29-13-11-19-5-9-23(28)10-6-19)15-24(21)27(18,2)12-14-30(25)17-20-3-4-20/h5-10,15,18,20,25H,3-4,11-14,16-17H2,1-2H3,(H,29,31)/t18-,25?,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224868

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccc(Cl)c(Cl)c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H36Cl2N2O/c1-21-31-19-26-9-10-27(17-28(26)33(21,2)14-16-37(31)20-23-3-4-23)32(38)36-15-13-22-5-7-24(8-6-22)25-11-12-29(34)30(35)18-25/h5-12,17-18,21,23,31H,3-4,13-16,19-20H2,1-2H3,(H,36,38)/t21-,31?,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224869

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccc(Cl)cc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H37ClN2O/c1-22-31-20-27-9-10-28(19-30(27)33(22,2)16-18-36(31)21-24-3-4-24)32(37)35-17-15-23-5-7-25(8-6-23)26-11-13-29(34)14-12-26/h5-14,19,22,24,31H,3-4,15-18,20-21H2,1-2H3,(H,35,37)/t22-,31?,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224881

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccc(C)cc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C34H40N2O/c1-23-4-10-27(11-5-23)28-12-8-25(9-13-28)16-18-35-33(37)30-15-14-29-21-32-24(2)34(3,31(29)20-30)17-19-36(32)22-26-6-7-26/h4-5,8-15,20,24,26,32H,6-7,16-19,21-22H2,1-3H3,(H,35,37)/t24-,32?,34-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224870

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1cccc(Br)c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C27H33BrN2O/c1-18-25-16-21-8-9-22(26(31)29-12-10-19-4-3-5-23(28)14-19)15-24(21)27(18,2)11-13-30(25)17-20-6-7-20/h3-5,8-9,14-15,18,20,25H,6-7,10-13,16-17H2,1-2H3,(H,29,31)/t18-,25?,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224876

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccccc1Br |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C27H33BrN2O/c1-18-25-16-21-9-10-22(26(31)29-13-11-20-5-3-4-6-24(20)28)15-23(21)27(18,2)12-14-30(25)17-19-7-8-19/h3-6,9-10,15,18-19,25H,7-8,11-14,16-17H2,1-2H3,(H,29,31)/t18-,25?,27-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224875

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1cccc2ccccc12 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C31H36N2O/c1-21-29-19-25-12-13-26(18-28(25)31(21,2)15-17-33(29)20-22-10-11-22)30(34)32-16-14-24-8-5-7-23-6-3-4-9-27(23)24/h3-9,12-13,18,21-22,29H,10-11,14-17,19-20H2,1-2H3,(H,32,34)/t21-,29?,31-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50097579

((6S,11R)-3-Cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(N)=O |r,TLB:8:9:1:14.12.13,15:14:1:4.9.3| Show InChI InChI=1S/C19H26N2O/c1-12-17-10-14-5-6-15(18(20)22)9-16(14)19(12,2)7-8-21(17)11-13-3-4-13/h5-6,9,12-13,17H,3-4,7-8,10-11H2,1-2H3,(H2,20,22)/t12-,17?,19-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 5.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224872

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1cccc(c1)-c1ccccc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H38N2O/c1-23-31-21-28-13-14-29(20-30(28)33(23,2)16-18-35(31)22-25-11-12-25)32(36)34-17-15-24-7-6-10-27(19-24)26-8-4-3-5-9-26/h3-10,13-14,19-20,23,25,31H,11-12,15-18,21-22H2,1-2H3,(H,34,36)/t23-,31?,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 5.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224867

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccccc1-c1ccccc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H38N2O/c1-23-31-21-27-14-15-28(20-30(27)33(23,2)17-19-35(31)22-24-12-13-24)32(36)34-18-16-26-10-6-7-11-29(26)25-8-4-3-5-9-25/h3-11,14-15,20,23-24,31H,12-13,16-19,21-22H2,1-2H3,(H,34,36)/t23-,31?,33-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 6.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224871

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCCc1ccc2ccccc2c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C32H38N2O/c1-22-30-20-27-13-14-28(19-29(27)32(22,2)15-17-34(30)21-24-9-10-24)31(35)33-16-5-6-23-11-12-25-7-3-4-8-26(25)18-23/h3-4,7-8,11-14,18-19,22,24,30H,5-6,9-10,15-17,20-21H2,1-2H3,(H,33,35)/t22-,30?,32-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224878

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCCc1cccc2ccccc12 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C32H38N2O/c1-22-30-20-26-14-15-27(19-29(26)32(22,2)16-18-34(30)21-23-12-13-23)31(35)33-17-6-10-25-9-5-8-24-7-3-4-11-28(24)25/h3-5,7-9,11,14-15,19,22-23,30H,6,10,12-13,16-18,20-21H2,1-2H3,(H,33,35)/t22-,30?,32-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224880

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCc1cccc2ccccc12 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C30H34N2O/c1-20-28-17-23-12-13-24(16-27(23)30(20,2)14-15-32(28)19-21-10-11-21)29(33)31-18-25-8-5-7-22-6-3-4-9-26(22)25/h3-9,12-13,16,20-21,28H,10-11,14-15,17-19H2,1-2H3,(H,31,33)/t20-,28?,30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224874

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCc1ccc2ccccc2c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C30H34N2O/c1-20-28-17-25-11-12-26(16-27(25)30(20,2)13-14-32(28)19-21-7-8-21)29(33)31-18-22-9-10-23-5-3-4-6-24(23)15-22/h3-6,9-12,15-16,20-21,28H,7-8,13-14,17-19H2,1-2H3,(H,31,33)/t20-,28?,30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224876

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccccc1Br |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C27H33BrN2O/c1-18-25-16-21-9-10-22(26(31)29-13-11-20-5-3-4-6-24(20)28)15-23(21)27(18,2)12-14-30(25)17-19-7-8-19/h3-6,9-10,15,18-19,25H,7-8,11-14,16-17H2,1-2H3,(H,29,31)/t18-,25?,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]U-69593 from kappa opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224867

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccccc1-c1ccccc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H38N2O/c1-23-31-21-27-14-15-28(20-30(27)33(23,2)17-19-35(31)22-24-12-13-24)32(36)34-18-16-26-10-6-7-11-29(26)25-8-4-3-5-9-25/h3-11,14-15,20,23-24,31H,12-13,16-19,21-22H2,1-2H3,(H,34,36)/t23-,31?,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224875

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1cccc2ccccc12 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C31H36N2O/c1-21-29-19-25-12-13-26(18-28(25)31(21,2)15-17-33(29)20-22-10-11-22)30(34)32-16-14-24-8-5-7-23-6-3-4-9-27(23)24/h3-9,12-13,18,21-22,29H,10-11,14-17,19-20H2,1-2H3,(H,32,34)/t21-,29?,31-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 24 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224874

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCc1ccc2ccccc2c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C30H34N2O/c1-20-28-17-25-11-12-26(16-27(25)30(20,2)13-14-32(28)19-21-7-8-21)29(33)31-18-22-9-10-23-5-3-4-6-24(23)15-22/h3-6,9-12,15-16,20-21,28H,7-8,13-14,17-19H2,1-2H3,(H,31,33)/t20-,28?,30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 31 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224880

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCc1cccc2ccccc12 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C30H34N2O/c1-20-28-17-23-12-13-24(16-27(23)30(20,2)14-15-32(28)19-21-10-11-21)29(33)31-18-25-8-5-7-22-6-3-4-9-26(22)25/h3-9,12-13,16,20-21,28H,10-11,14-15,17-19H2,1-2H3,(H,31,33)/t20-,28?,30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 52 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224876

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccccc1Br |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C27H33BrN2O/c1-18-25-16-21-9-10-22(26(31)29-13-11-20-5-3-4-6-24(20)28)15-23(21)27(18,2)12-14-30(25)17-19-7-8-19/h3-6,9-10,15,18-19,25H,7-8,11-14,16-17H2,1-2H3,(H,29,31)/t18-,25?,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Displacement of [3H]naltrindole from delta opioid receptor expressed in CHO cells |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224873

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES COc1ccc(cc1)-c1ccc(CCNC(=O)c2ccc3CC4[C@H](C)[C@](C)(CCN4CC4CC4)c3c2)cc1 |w:22.22,TLB:35:34:23:29.28.27,30:29:23:20.34.21| Show InChI InChI=1S/C34H40N2O2/c1-23-32-21-28-10-11-29(20-31(28)34(23,2)17-19-36(32)22-25-4-5-25)33(37)35-18-16-24-6-8-26(9-7-24)27-12-14-30(38-3)15-13-27/h6-15,20,23,25,32H,4-5,16-19,21-22H2,1-3H3,(H,35,37)/t23-,32?,34-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Antagonist activity at human mu opioid receptor expressed in CHO cell membrane assessed as inhibition of DAMGO induced [35S]GTP-gamma-S binding |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50194264

((+/-)-3-(cyclopropylmethyl)-1,2,3,4,5,6-hexahydro-...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccccc1 |r,TLB:8:9:1:14.12.13,15:14:1:4.9.3| Show InChI InChI=1S/C33H38N2O/c1-23-31-21-28-14-15-29(20-30(28)33(23,2)17-19-35(31)22-25-8-9-25)32(36)34-18-16-24-10-12-27(13-11-24)26-6-4-3-5-7-26/h3-7,10-15,20,23,25,31H,8-9,16-19,21-22H2,1-2H3,(H,34,36)/t23-,31?,33-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Antagonist activity at human mu opioid receptor expressed in CHO cell membrane assessed as inhibition of DAMGO induced [35S]GTP-gamma-S binding |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224870

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1cccc(Br)c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C27H33BrN2O/c1-18-25-16-21-8-9-22(26(31)29-12-10-19-4-3-5-23(28)14-19)15-24(21)27(18,2)11-13-30(25)17-20-6-7-20/h3-5,8-9,14-15,18,20,25H,6-7,10-13,16-17H2,1-2H3,(H,29,31)/t18-,25?,27-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 230 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Antagonist activity at human mu opioid receptor expressed in CHO cell membrane assessed as inhibition of DAMGO induced [35S]GTP-gamma-S binding |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224871

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCCc1ccc2ccccc2c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C32H38N2O/c1-22-30-20-27-13-14-28(19-29(27)32(22,2)15-17-34(30)21-24-9-10-24)31(35)33-16-5-6-23-11-12-25-7-3-4-8-26(25)18-23/h3-4,7-8,11-14,18-19,22,24,30H,5-6,9-10,15-17,20-21H2,1-2H3,(H,33,35)/t22-,30?,32-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 320 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Antagonist activity at human mu opioid receptor expressed in CHO cell membrane assessed as inhibition of DAMGO induced [35S]GTP-gamma-S binding |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224870

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1cccc(Br)c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C27H33BrN2O/c1-18-25-16-21-8-9-22(26(31)29-12-10-19-4-3-5-23(28)14-19)15-24(21)27(18,2)11-13-30(25)17-20-6-7-20/h3-5,8-9,14-15,18,20,25H,6-7,10-13,16-17H2,1-2H3,(H,29,31)/t18-,25?,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 4.60 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human kappa opioid receptor expressed in CHO cell membrane by [35S]GTP-gamma-S binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224867

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccccc1-c1ccccc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H38N2O/c1-23-31-21-27-14-15-28(20-30(27)33(23,2)17-19-35(31)22-24-12-13-24)32(36)34-18-16-26-10-6-7-11-29(26)25-8-4-3-5-9-25/h3-11,14-15,20,23-24,31H,12-13,16-19,21-22H2,1-2H3,(H,34,36)/t23-,31?,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 42 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human kappa opioid receptor expressed in CHO cell membrane by [35S]GTP-gamma-S binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224871

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCCc1ccc2ccccc2c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C32H38N2O/c1-22-30-20-27-13-14-28(19-29(27)32(22,2)15-17-34(30)21-24-9-10-24)31(35)33-16-5-6-23-11-12-25-7-3-4-8-26(25)18-23/h3-4,7-8,11-14,18-19,22,24,30H,5-6,9-10,15-17,20-21H2,1-2H3,(H,33,35)/t22-,30?,32-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 24 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human mu opioid receptor expressed in CHO cell membrane by [35S]GTPgammaS binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50039029
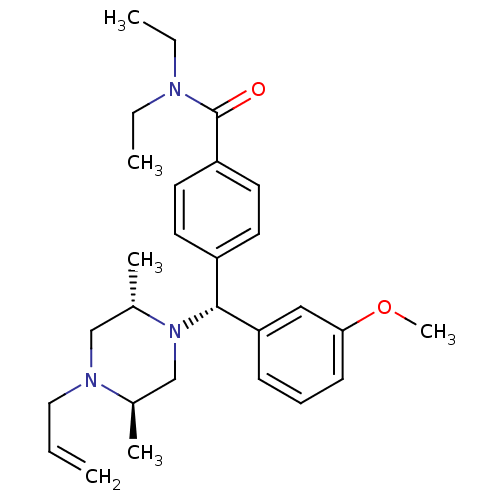
((+)-4-((alpha R)-((2S,5R)-4-allyl-2,5-dimethylpipe...)Show SMILES CCN(CC)C(=O)c1ccc(cc1)[C@@H](N1C[C@@H](C)N(CC=C)C[C@@H]1C)c1cccc(OC)c1 |r| Show InChI InChI=1S/C28H39N3O2/c1-7-17-30-19-22(5)31(20-21(30)4)27(25-11-10-12-26(18-25)33-6)23-13-15-24(16-14-23)28(32)29(8-2)9-3/h7,10-16,18,21-22,27H,1,8-9,17,19-20H2,2-6H3/t21-,22+,27-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 4.80 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human delta opioid receptor expressed in CHO cell membrane by [35S]GTP-gamma-S binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50194264

((+/-)-3-(cyclopropylmethyl)-1,2,3,4,5,6-hexahydro-...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccccc1 |r,TLB:8:9:1:14.12.13,15:14:1:4.9.3| Show InChI InChI=1S/C33H38N2O/c1-23-31-21-28-14-15-29(20-30(28)33(23,2)17-19-35(31)22-25-8-9-25)32(36)34-18-16-24-10-12-27(13-11-24)26-6-4-3-5-7-26/h3-7,10-15,20,23,25,31H,8-9,16-19,21-22H2,1-2H3,(H,34,36)/t23-,31?,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 4.40 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human kappa opioid receptor expressed in CHO cell membrane by [35S]GTP-gamma-S binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224872

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1cccc(c1)-c1ccccc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H38N2O/c1-23-31-21-28-13-14-29(20-30(28)33(23,2)16-18-35(31)22-25-11-12-25)32(36)34-17-15-24-7-6-10-27(19-24)26-8-4-3-5-9-26/h3-10,13-14,19-20,23,25,31H,11-12,15-18,21-22H2,1-2H3,(H,34,36)/t23-,31?,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 12 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human kappa opioid receptor expressed in CHO cell membrane by [35S]GTP-gamma-S binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224867

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccccc1-c1ccccc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H38N2O/c1-23-31-21-27-14-15-28(20-30(27)33(23,2)17-19-35(31)22-24-12-13-24)32(36)34-18-16-26-10-6-7-11-29(26)25-8-4-3-5-9-25/h3-11,14-15,20,23-24,31H,12-13,16-19,21-22H2,1-2H3,(H,34,36)/t23-,31?,33-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 12 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human mu opioid receptor expressed in CHO cell membrane by [35S]GTPgammaS binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224872

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1cccc(c1)-c1ccccc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H38N2O/c1-23-31-21-28-13-14-29(20-30(28)33(23,2)16-18-35(31)22-25-11-12-25)32(36)34-17-15-24-7-6-10-27(19-24)26-8-4-3-5-9-26/h3-10,13-14,19-20,23,25,31H,11-12,15-18,21-22H2,1-2H3,(H,34,36)/t23-,31?,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 18 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human delta opioid receptor expressed in CHO cell membrane by [35S]GTP-gamma-S binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224869

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccc(Cl)cc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H37ClN2O/c1-22-31-20-27-9-10-28(19-30(27)33(22,2)16-18-36(31)21-24-3-4-24)32(37)35-17-15-23-5-7-25(8-6-23)26-11-13-29(34)14-12-26/h5-14,19,22,24,31H,3-4,15-18,20-21H2,1-2H3,(H,35,37)/t22-,31?,33-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.5 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human mu opioid receptor expressed in CHO cell membrane by [35S]GTPgammaS binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224870

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1cccc(Br)c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C27H33BrN2O/c1-18-25-16-21-8-9-22(26(31)29-12-10-19-4-3-5-23(28)14-19)15-24(21)27(18,2)11-13-30(25)17-20-6-7-20/h3-5,8-9,14-15,18,20,25H,6-7,10-13,16-17H2,1-2H3,(H,29,31)/t18-,25?,27-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 5.60 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human mu opioid receptor expressed in CHO cell membrane by [35S]GTPgammaS binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224870

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1cccc(Br)c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C27H33BrN2O/c1-18-25-16-21-8-9-22(26(31)29-12-10-19-4-3-5-23(28)14-19)15-24(21)27(18,2)11-13-30(25)17-20-6-7-20/h3-5,8-9,14-15,18,20,25H,6-7,10-13,16-17H2,1-2H3,(H,29,31)/t18-,25?,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 14 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human delta opioid receptor expressed in CHO cell membrane by [35S]GTP-gamma-S binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224877

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(Br)cc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C27H33BrN2O/c1-18-25-16-21-7-8-22(26(31)29-13-11-19-5-9-23(28)10-6-19)15-24(21)27(18,2)12-14-30(25)17-20-3-4-20/h5-10,15,18,20,25H,3-4,11-14,16-17H2,1-2H3,(H,29,31)/t18-,25?,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 20 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human kappa opioid receptor expressed in CHO cell membrane by [35S]GTP-gamma-S binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224869

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccc(Cl)cc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H37ClN2O/c1-22-31-20-27-9-10-28(19-30(27)33(22,2)16-18-36(31)21-24-3-4-24)32(37)35-17-15-23-5-7-25(8-6-23)26-11-13-29(34)14-12-26/h5-14,19,22,24,31H,3-4,15-18,20-21H2,1-2H3,(H,35,37)/t22-,31?,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human delta opioid receptor expressed in CHO cell membrane by [35S]GTP-gamma-S binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224873

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES COc1ccc(cc1)-c1ccc(CCNC(=O)c2ccc3CC4[C@H](C)[C@](C)(CCN4CC4CC4)c3c2)cc1 |w:22.22,TLB:35:34:23:29.28.27,30:29:23:20.34.21| Show InChI InChI=1S/C34H40N2O2/c1-23-32-21-28-10-11-29(20-31(28)34(23,2)17-19-36(32)22-25-4-5-25)33(37)35-18-16-24-6-8-26(9-7-24)27-12-14-30(38-3)15-13-27/h6-15,20,23,25,32H,4-5,16-19,21-22H2,1-3H3,(H,35,37)/t23-,32?,34-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 0.680 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human delta opioid receptor expressed in CHO cell membrane by [35S]GTP-gamma-S binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224871

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCCc1ccc2ccccc2c1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C32H38N2O/c1-22-30-20-27-13-14-28(19-29(27)32(22,2)15-17-34(30)21-24-9-10-24)31(35)33-16-5-6-23-11-12-25-7-3-4-8-26(25)18-23/h3-4,7-8,11-14,18-19,22,24,30H,5-6,9-10,15-17,20-21H2,1-2H3,(H,33,35)/t22-,30?,32-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 6.30 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human kappa opioid receptor expressed in CHO cell membrane by [35S]GTP-gamma-S binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224872

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1cccc(c1)-c1ccccc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H38N2O/c1-23-31-21-28-13-14-29(20-30(28)33(23,2)16-18-35(31)22-25-11-12-25)32(36)34-17-15-24-7-6-10-27(19-24)26-8-4-3-5-9-26/h3-10,13-14,19-20,23,25,31H,11-12,15-18,21-22H2,1-2H3,(H,34,36)/t23-,31?,33-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 6.80 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human mu opioid receptor expressed in CHO cell membrane by [35S]GTPgammaS binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM21015

((2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-(4-hydroxyphenyl...)Show SMILES C[C@@H](NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)NCC(=O)N(C)[C@@H](Cc1ccccc1)C(=O)NCCO Show InChI InChI=1S/C26H35N5O6/c1-17(30-25(36)21(27)14-19-8-10-20(33)11-9-19)24(35)29-16-23(34)31(2)22(26(37)28-12-13-32)15-18-6-4-3-5-7-18/h3-11,17,21-22,32-33H,12-16,27H2,1-2H3,(H,28,37)(H,29,35)(H,30,36)/t17-,21+,22+/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 55 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human mu opioid receptor expressed in CHO cell membrane by [35S]GTPgammaS binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224873

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES COc1ccc(cc1)-c1ccc(CCNC(=O)c2ccc3CC4[C@H](C)[C@](C)(CCN4CC4CC4)c3c2)cc1 |w:22.22,TLB:35:34:23:29.28.27,30:29:23:20.34.21| Show InChI InChI=1S/C34H40N2O2/c1-23-32-21-28-10-11-29(20-31(28)34(23,2)17-19-36(32)22-25-4-5-25)33(37)35-18-16-24-6-8-26(9-7-24)27-12-14-30(38-3)15-13-27/h6-15,20,23,25,32H,4-5,16-19,21-22H2,1-3H3,(H,35,37)/t23-,32?,34-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human kappa opioid receptor expressed in CHO cell membrane by [35S]GTP-gamma-S binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224873

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES COc1ccc(cc1)-c1ccc(CCNC(=O)c2ccc3CC4[C@H](C)[C@](C)(CCN4CC4CC4)c3c2)cc1 |w:22.22,TLB:35:34:23:29.28.27,30:29:23:20.34.21| Show InChI InChI=1S/C34H40N2O2/c1-23-32-21-28-10-11-29(20-31(28)34(23,2)17-19-36(32)22-25-4-5-25)33(37)35-18-16-24-6-8-26(9-7-24)27-12-14-30(38-3)15-13-27/h6-15,20,23,25,32H,4-5,16-19,21-22H2,1-3H3,(H,35,37)/t23-,32?,34-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 11 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human mu opioid receptor expressed in CHO cell membrane by [35S]GTPgammaS binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50224869

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccc(Cl)cc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C33H37ClN2O/c1-22-31-20-27-9-10-28(19-30(27)33(22,2)16-18-36(31)21-24-3-4-24)32(37)35-17-15-23-5-7-25(8-6-23)26-11-13-29(34)14-12-26/h5-14,19,22,24,31H,3-4,15-18,20-21H2,1-2H3,(H,35,37)/t22-,31?,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 6.40 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human kappa opioid receptor expressed in CHO cell membrane by [35S]GTP-gamma-S binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50224877

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(Br)cc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C27H33BrN2O/c1-18-25-16-21-7-8-22(26(31)29-13-11-19-5-9-23(28)10-6-19)15-24(21)27(18,2)12-14-30(25)17-20-3-4-20/h5-10,15,18,20,25H,3-4,11-14,16-17H2,1-2H3,(H,29,31)/t18-,25?,27-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 20 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human delta opioid receptor expressed in CHO cell membrane by [35S]GTP-gamma-S binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50000296
(CHEMBL441765 | CHEMBL482811 | U-50488H | US1149237...)Show SMILES CN([C@@H]1CCCC[C@H]1N1CCCC1)C(=O)Cc1ccc(Cl)c(Cl)c1 |r| Show InChI InChI=1S/C19H26Cl2N2O/c1-22(19(24)13-14-8-9-15(20)16(21)12-14)17-6-2-3-7-18(17)23-10-4-5-11-23/h8-9,12,17-18H,2-7,10-11,13H2,1H3/t17-,18-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 36 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human kappa opioid receptor expressed in CHO cell membrane by [35S]GTP-gamma-S binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50224877

((6S,11R)-3-cyclopropylmethyl-6,11-dimethyl-1,2,3,4...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(Br)cc1 |w:2.2,TLB:8:9:1:14.13.12,15:14:1:4.9.3| Show InChI InChI=1S/C27H33BrN2O/c1-18-25-16-21-7-8-22(26(31)29-13-11-19-5-9-23(28)10-6-19)15-24(21)27(18,2)12-14-30(25)17-20-3-4-20/h5-10,15,18,20,25H,3-4,11-14,16-17H2,1-2H3,(H,29,31)/t18-,25?,27-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 16 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human mu opioid receptor expressed in CHO cell membrane by [35S]GTPgammaS binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM50194264

((+/-)-3-(cyclopropylmethyl)-1,2,3,4,5,6-hexahydro-...)Show SMILES C[C@H]1C2Cc3ccc(cc3[C@@]1(C)CCN2CC1CC1)C(=O)NCCc1ccc(cc1)-c1ccccc1 |r,TLB:8:9:1:14.12.13,15:14:1:4.9.3| Show InChI InChI=1S/C33H38N2O/c1-23-31-21-28-14-15-29(20-30(28)33(23,2)17-19-35(31)22-25-8-9-25)32(36)34-18-16-24-10-12-27(13-11-24)26-6-4-3-5-7-26/h3-7,10-15,20,23,25,31H,8-9,16-19,21-22H2,1-2H3,(H,34,36)/t23-,31?,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 3 | n/a | n/a | n/a | n/a |
Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human delta opioid receptor expressed in CHO cell membrane by [35S]GTP-gamma-S binding assay |
Bioorg Med Chem Lett 17: 6516-20 (2007)
Article DOI: 10.1016/j.bmcl.2007.09.082
BindingDB Entry DOI: 10.7270/Q21R6RCT |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data