| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Leukotriene A-4 hydrolase |
|---|
| Ligand | BDBM24201 |
|---|
| Substrate/Competitor | BDBM24200 |
|---|
| Meas. Tech. | Epoxide Hydrolase Assay |
|---|
| pH | 7.4±n/a |
|---|
| Temperature | 295.15±n/a K |
|---|
| IC50 | 14±9 nM |
|---|
| Citation |  Grice, CA; Tays, KL; Savall, BM; Wei, J; Butler, CR; Axe, FU; Bembenek, SD; Fourie, AM; Dunford, PJ; Lundeen, K; Coles, F; Xue, X; Riley, JP; Williams, KN; Karlsson, L; Edwards, JP Identification of a potent, selective, and orally active leukotriene a4 hydrolase inhibitor with anti-inflammatory activity. J Med Chem51:4150-69 (2008) [PubMed] Article Grice, CA; Tays, KL; Savall, BM; Wei, J; Butler, CR; Axe, FU; Bembenek, SD; Fourie, AM; Dunford, PJ; Lundeen, K; Coles, F; Xue, X; Riley, JP; Williams, KN; Karlsson, L; Edwards, JP Identification of a potent, selective, and orally active leukotriene a4 hydrolase inhibitor with anti-inflammatory activity. J Med Chem51:4150-69 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Inhibition_Run data, Solution Info, Assay Method |
|---|
| |
| Leukotriene A-4 hydrolase |
|---|
| Name: | Leukotriene A-4 hydrolase |
|---|
| Synonyms: | LKHA4_HUMAN | LTA-4 hydrolase | LTA4 | LTA4H | Leukotriene A(4) hydrolase | Leukotriene A-4 hydrolase (LTA4H) | Leukotriene A4 hydrolase |
|---|
| Type: | Hydrolase; metalloprotease |
|---|
| Mol. Mass.: | 69280.41 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Human recombinant LTA4H. |
|---|
| Residue: | 611 |
|---|
| Sequence: | MPEIVDTCSLASPASVCRTKHLHLRCSVDFTRRTLTGTAALTVQSQEDNLRSLVLDTKDL
TIEKVVINGQEVKYALGERQSYKGSPMEISLPIALSKNQEIVIEISFETSPKSSALQWLT
PEQTSGKEHPYLFSQCQAIHCRAILPCQDTPSVKLTYTAEVSVPKELVALMSAIRDGETP
DPEDPSRKIYKFIQKVPIPCYLIALVVGALESRQIGPRTLVWSEKEQVEKSAYEFSETES
MLKIAEDLGGPYVWGQYDLLVLPPSFPYGGMENPCLTFVTPTLLAGDKSLSNVIAHEISH
SWTGNLVTNKTWDHFWLNEGHTVYLERHICGRLFGEKFRHFNALGGWGELQNSVKTFGET
HPFTKLVVDLTDIDPDVAYSSVPYEKGFALLFYLEQLLGGPEIFLGFLKAYVEKFSYKSI
TTDDWKDFLYSYFKDKVDVLNQVDWNAWLYSPGLPPIKPNYDMTLTNACIALSQRWITAK
EDDLNSFNATDLKDLSSHQLNEFLAQTLQRAPLPLGHIKRMQEVYNFNAINNSEIRFRWL
RLCIQSKWEDAIPLALKMATEQGRMKFTRPLFKDLAAFDKSHDQAVRTYQEHKASMHPVT
AMLVGKDLKVD
|
|
|
|---|
| BDBM24201 |
|---|
| BDBM24200 |
|---|
| Name | BDBM24201 |
|---|
| Synonyms: | 2-{4-[2-(pyrrolidin-1-yl)ethoxy]phenoxy}-1,3-benzothiazole | Benzthiazole compound, 11a |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H20N2O2S |
|---|
| Mol. Mass. | 340.439 |
|---|
| SMILES | C(CN1CCCC1)Oc1ccc(Oc2nc3ccccc3s2)cc1 |
|---|
| Structure | 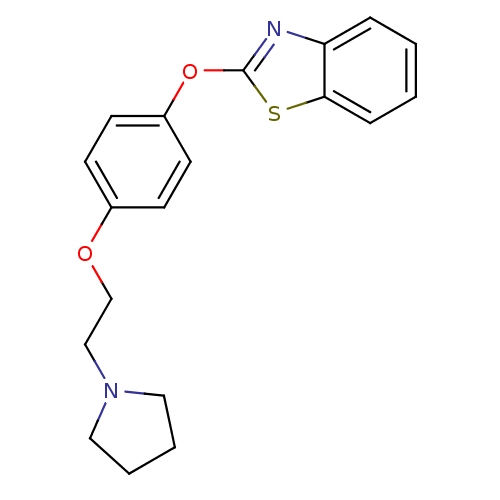
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Grice, CA; Tays, KL; Savall, BM; Wei, J; Butler, CR; Axe, FU; Bembenek, SD; Fourie, AM; Dunford, PJ; Lundeen, K; Coles, F; Xue, X; Riley, JP; Williams, KN; Karlsson, L; Edwards, JP Identification of a potent, selective, and orally active leukotriene a4 hydrolase inhibitor with anti-inflammatory activity. J Med Chem51:4150-69 (2008) [PubMed] Article
Grice, CA; Tays, KL; Savall, BM; Wei, J; Butler, CR; Axe, FU; Bembenek, SD; Fourie, AM; Dunford, PJ; Lundeen, K; Coles, F; Xue, X; Riley, JP; Williams, KN; Karlsson, L; Edwards, JP Identification of a potent, selective, and orally active leukotriene a4 hydrolase inhibitor with anti-inflammatory activity. J Med Chem51:4150-69 (2008) [PubMed] Article