| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Dimer of Protein farnesyltransferase subunit beta |
|---|
| Ligand | BDBM50048980 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_70728 (CHEMBL680521) |
|---|
| IC50 | 160±n/a nM |
|---|
| Citation |  Leftheris, K; Kline, T; Vite, GD; Cho, YH; Bhide, RS; Patel, DV; Patel, MM; Schmidt, RJ; Weller, HN; Andahazy, ML; Carboni, JM; Gullo-Brown, JL; Lee, FY; Ricca, C; Rose, WC; Yan, N; Barbacid, M; Hunt, JT; Meyers, CA; Seizinger, BR; Zahler, R; Manne, V Development of highly potent inhibitors of Ras farnesyltransferase possessing cellular and in vivo activity. J Med Chem39:224-36 (1996) [PubMed] Article Leftheris, K; Kline, T; Vite, GD; Cho, YH; Bhide, RS; Patel, DV; Patel, MM; Schmidt, RJ; Weller, HN; Andahazy, ML; Carboni, JM; Gullo-Brown, JL; Lee, FY; Ricca, C; Rose, WC; Yan, N; Barbacid, M; Hunt, JT; Meyers, CA; Seizinger, BR; Zahler, R; Manne, V Development of highly potent inhibitors of Ras farnesyltransferase possessing cellular and in vivo activity. J Med Chem39:224-36 (1996) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Dimer of Protein farnesyltransferase subunit beta |
|---|
| Name: | Dimer of Protein farnesyltransferase subunit beta |
|---|
| Synonyms: | Protein Farnesyltransferase (PFT) Chain B |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | n/a |
|---|
| Description: | ASSAY_ID of EBI is 51428 |
|---|
| Components: | This complex has 3 components. |
|---|
| Component 1 |
| Name: | Protein farnesyltransferase subunit beta |
|---|
| Synonyms: | CAAX farnesyltransferase subunit alpha | CAAX farnesyltransferase subunit beta | FNTB | FNTB_HUMAN | FTase-alpha | FTase-beta | GGTase-I-alpha | Protein Farnesyltransferase (PFT) Chain B | Protein farnesyl/geranylgeranyl transferase | Protein farnesyltransferase beta subunit | Protein farnesyltransferase subunit beta | Protein farnesyltransferase/geranylgeranyltransferase type I alpha subunit | Protein farnesyltransferase/geranylgeranyltransferase type-1 subunit alpha | Ras proteins prenyltransferase subunit alpha | Ras proteins prenyltransferase subunit beta | Type I protein geranyl-geranyltransferase subunit alpha |
|---|
| Type: | Enzyme Subunit |
|---|
| Mol. Mass.: | 48766.02 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Protein farnesyltransferase subunit beta |
|---|
| Residue: | 437 |
|---|
| Sequence: | MASPSSFTYYCPPSSSPVWSEPLYSLRPEHARERLQDDSVETVTSIEQAKVEEKIQEVFS
SYKFNHLVPRLVLQREKHFHYLKRGLRQLTDAYECLDASRPWLCYWILHSLELLDEPIPQ
IVATDVCQFLELCQSPEGGFGGGPGQYPHLAPTYAAVNALCIIGTEEAYDIINREKLLQY
LYSLKQPDGSFLMHVGGEVDVRSAYCAASVASLTNIITPDLFEGTAEWIARCQNWEGGIG
GVPGMEAHGGYTFCGLAALVILKRERSLNLKSLLQWVTSRQMRFEGGFQGRCNKLVDGCY
SFWQAGLLPLLHRALHAQGDPALSMSHWMFHQQALQEYILMCCQCPAGGLLDKPGKSRDF
YHTCYCLSGLSIAQHFGSGAMLHDVVLGVPENALQPTHPVYNIGPDKVIQATTYFLQKPV
PGFEELKDETSAEPATD
|
|
|
|---|
| Component 2 |
| Name: | Protein farnesyltransferase subunit beta |
|---|
| Synonyms: | CAAX farnesyltransferase subunit alpha | CAAX farnesyltransferase subunit beta | FNTB | FNTB_HUMAN | FTase-alpha | FTase-beta | GGTase-I-alpha | Protein Farnesyltransferase (PFT) Chain B | Protein farnesyl/geranylgeranyl transferase | Protein farnesyltransferase beta subunit | Protein farnesyltransferase subunit beta | Protein farnesyltransferase/geranylgeranyltransferase type I alpha subunit | Protein farnesyltransferase/geranylgeranyltransferase type-1 subunit alpha | Ras proteins prenyltransferase subunit alpha | Ras proteins prenyltransferase subunit beta | Type I protein geranyl-geranyltransferase subunit alpha |
|---|
| Type: | Enzyme Subunit |
|---|
| Mol. Mass.: | 48766.02 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Protein farnesyltransferase subunit beta |
|---|
| Residue: | 437 |
|---|
| Sequence: | MASPSSFTYYCPPSSSPVWSEPLYSLRPEHARERLQDDSVETVTSIEQAKVEEKIQEVFS
SYKFNHLVPRLVLQREKHFHYLKRGLRQLTDAYECLDASRPWLCYWILHSLELLDEPIPQ
IVATDVCQFLELCQSPEGGFGGGPGQYPHLAPTYAAVNALCIIGTEEAYDIINREKLLQY
LYSLKQPDGSFLMHVGGEVDVRSAYCAASVASLTNIITPDLFEGTAEWIARCQNWEGGIG
GVPGMEAHGGYTFCGLAALVILKRERSLNLKSLLQWVTSRQMRFEGGFQGRCNKLVDGCY
SFWQAGLLPLLHRALHAQGDPALSMSHWMFHQQALQEYILMCCQCPAGGLLDKPGKSRDF
YHTCYCLSGLSIAQHFGSGAMLHDVVLGVPENALQPTHPVYNIGPDKVIQATTYFLQKPV
PGFEELKDETSAEPATD
|
|
|
|---|
| BDBM50048980 |
|---|
| n/a |
|---|
| Name | BDBM50048980 |
|---|
| Synonyms: | (S)-2-{(E)-2-[(S)-2-((R)-2-Amino-3-mercapto-propionylamino)-3-methyl-butyrylamino]-3-phenyl-acryloylamino}-4-methylsulfanyl-butyric acid | CHEMBL419091 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H32N4O5S2 |
|---|
| Mol. Mass. | 496.643 |
|---|
| SMILES | CSCC[C@H](NC(=O)C(\NC(=O)[C@@H](NC(=O)[C@@H](N)CS)C(C)C)=C/c1ccccc1)C(O)=O |
|---|
| Structure | 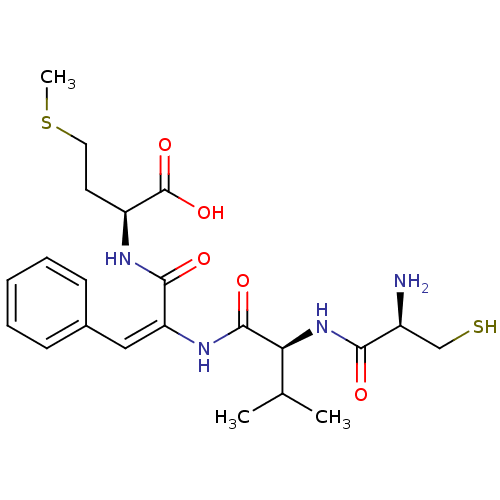
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Leftheris, K; Kline, T; Vite, GD; Cho, YH; Bhide, RS; Patel, DV; Patel, MM; Schmidt, RJ; Weller, HN; Andahazy, ML; Carboni, JM; Gullo-Brown, JL; Lee, FY; Ricca, C; Rose, WC; Yan, N; Barbacid, M; Hunt, JT; Meyers, CA; Seizinger, BR; Zahler, R; Manne, V Development of highly potent inhibitors of Ras farnesyltransferase possessing cellular and in vivo activity. J Med Chem39:224-36 (1996) [PubMed] Article
Leftheris, K; Kline, T; Vite, GD; Cho, YH; Bhide, RS; Patel, DV; Patel, MM; Schmidt, RJ; Weller, HN; Andahazy, ML; Carboni, JM; Gullo-Brown, JL; Lee, FY; Ricca, C; Rose, WC; Yan, N; Barbacid, M; Hunt, JT; Meyers, CA; Seizinger, BR; Zahler, R; Manne, V Development of highly potent inhibitors of Ras farnesyltransferase possessing cellular and in vivo activity. J Med Chem39:224-36 (1996) [PubMed] Article