| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Serine/threonine-protein kinase B-raf |
|---|
| Ligand | BDBM50264718 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1699092 (CHEMBL4050074) |
|---|
| IC50 | 40±n/a nM |
|---|
| Citation |  Wang, L; Zhang, Q; Zhu, G; Zhang, Z; Zhi, Y; Zhang, L; Mao, T; Zhou, X; Chen, Y; Lu, T; Tang, W Design, synthesis and evaluation of derivatives based on pyrimidine scaffold as potent Pan-Raf inhibitors to overcome resistance. Eur J Med Chem130:86-106 (2017) [PubMed] Article Wang, L; Zhang, Q; Zhu, G; Zhang, Z; Zhi, Y; Zhang, L; Mao, T; Zhou, X; Chen, Y; Lu, T; Tang, W Design, synthesis and evaluation of derivatives based on pyrimidine scaffold as potent Pan-Raf inhibitors to overcome resistance. Eur J Med Chem130:86-106 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Serine/threonine-protein kinase B-raf |
|---|
| Name: | Serine/threonine-protein kinase B-raf |
|---|
| Synonyms: | B-RAF | B-Raf Protein Kinase | B-Raf proto-oncogene serine/threonine-protein kinase | BRAF | BRAF1 | BRAF_HUMAN | RAFB1 | p94 | v-Raf murine sarcoma viral oncogene homolog B1 |
|---|
| Type: | Serine/threonine-protein kinase |
|---|
| Mol. Mass.: | 84446.00 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P15056 |
|---|
| Residue: | 766 |
|---|
| Sequence: | MAALSGGGGGGAEPGQALFNGDMEPEAGAGAGAAASSAADPAIPEEVWNIKQMIKLTQEH
IEALLDKFGGEHNPPSIYLEAYEEYTSKLDALQQREQQLLESLGNGTDFSVSSSASMDTV
TSSSSSSLSVLPSSLSVFQNPTDVARSNPKSPQKPIVRVFLPNKQRTVVPARCGVTVRDS
LKKALMMRGLIPECCAVYRIQDGEKKPIGWDTDISWLTGEELHVEVLENVPLTTHNFVRK
TFFTLAFCDFCRKLLFQGFRCQTCGYKFHQRCSTEVPLMCVNYDQLDLLFVSKFFEHHPI
PQEEASLAETALTSGSSPSAPASDSIGPQILTSPSPSKSIPIPQPFRPADEDHRNQFGQR
DRSSSAPNVHINTIEPVNIDDLIRDQGFRGDGGSTTGLSATPPASLPGSLTNVKALQKSP
GPQRERKSSSSSEDRNRMKTLGRRDSSDDWEIPDGQITVGQRIGSGSFGTVYKGKWHGDV
AVKMLNVTAPTPQQLQAFKNEVGVLRKTRHVNILLFMGYSTKPQLAIVTQWCEGSSLYHH
LHIIETKFEMIKLIDIARQTAQGMDYLHAKSIIHRDLKSNNIFLHEDLTVKIGDFGLATV
KSRWSGSHQFEQLSGSILWMAPEVIRMQDKNPYSFQSDVYAFGIVLYELMTGQLPYSNIN
NRDQIIFMVGRGYLSPDLSKVRSNCPKAMKRLMAECLKKKRDERPLFPQILASIELLARS
LPKIHRSASEPSLNRAGFQTEDFSLYACASPKTPIQAGGYGAFPVH
|
|
|
|---|
| BDBM50264718 |
|---|
| n/a |
|---|
| Name | BDBM50264718 |
|---|
| Synonyms: | CHEMBL4085294 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C30H32ClF3N6O2 |
|---|
| Mol. Mass. | 601.062 |
|---|
| SMILES | CN1CCC(CNc2cc(ncn2)C2(CC2)C(=O)Nc2cc(ccc2C)C(=O)Nc2ccc(Cl)c(c2)C(F)(F)F)CC1 |
|---|
| Structure | 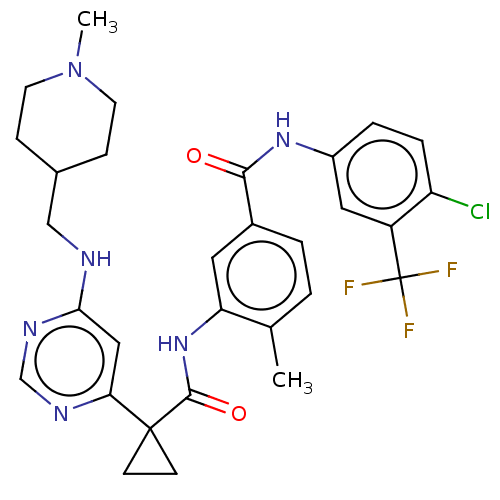
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wang, L; Zhang, Q; Zhu, G; Zhang, Z; Zhi, Y; Zhang, L; Mao, T; Zhou, X; Chen, Y; Lu, T; Tang, W Design, synthesis and evaluation of derivatives based on pyrimidine scaffold as potent Pan-Raf inhibitors to overcome resistance. Eur J Med Chem130:86-106 (2017) [PubMed] Article
Wang, L; Zhang, Q; Zhu, G; Zhang, Z; Zhi, Y; Zhang, L; Mao, T; Zhou, X; Chen, Y; Lu, T; Tang, W Design, synthesis and evaluation of derivatives based on pyrimidine scaffold as potent Pan-Raf inhibitors to overcome resistance. Eur J Med Chem130:86-106 (2017) [PubMed] Article