| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 11-beta-hydroxysteroid dehydrogenase 1 |
|---|
| Ligand | BDBM50273458 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_561241 (CHEMBL1013596) |
|---|
| Ki | 300±n/a nM |
|---|
| Citation |  Fotsch, C; Bartberger, MD; Bercot, EA; Chen, M; Cupples, R; Emery, M; Fretland, J; Guram, A; Hale, C; Han, N; Hickman, D; Hungate, RW; Hayashi, M; Komorowski, R; Liu, Q; Matsumoto, G; St Jean, DJ; Ursu, S; Véniant, M; Xu, G; Ye, Q; Yuan, C; Zhang, J; Zhang, X; Tu, H; Wang, M Further studies with the 2-amino-1,3-thiazol-4(5H)-one class of 11beta-hydroxysteroid dehydrogenase type 1 inhibitors: reducing pregnane X receptor activity and exploring activity in a monkey pharmacodynamic model. J Med Chem51:7953-67 (2008) [PubMed] Article Fotsch, C; Bartberger, MD; Bercot, EA; Chen, M; Cupples, R; Emery, M; Fretland, J; Guram, A; Hale, C; Han, N; Hickman, D; Hungate, RW; Hayashi, M; Komorowski, R; Liu, Q; Matsumoto, G; St Jean, DJ; Ursu, S; Véniant, M; Xu, G; Ye, Q; Yuan, C; Zhang, J; Zhang, X; Tu, H; Wang, M Further studies with the 2-amino-1,3-thiazol-4(5H)-one class of 11beta-hydroxysteroid dehydrogenase type 1 inhibitors: reducing pregnane X receptor activity and exploring activity in a monkey pharmacodynamic model. J Med Chem51:7953-67 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 11-beta-hydroxysteroid dehydrogenase 1 |
|---|
| Name: | 11-beta-hydroxysteroid dehydrogenase 1 |
|---|
| Synonyms: | 11-DH | 11-beta-HSD1 | 11-beta-Hydroxysteroid Dehydrogenase 1 (11-beta-HSD1) | 11-beta-hydroxysteroid dehydrogenase 1 | Corticosteroid 11-beta-dehydrogenase isozyme 1 | Corticosteroid 11-beta-dehydrogenase isozyme 1 (11-beta-HSD-1) | DHI1_RAT | Hsd11 | Hsd11b1 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 31889.48 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | P16232 |
|---|
| Residue: | 288 |
|---|
| Sequence: | MKKYLLPVLVLCLGYYYSTNEEFRPEMLQGKKVIVTGASKGIGREMAYHLSKMGAHVVLT
ARSEEGLQKVVSRCLELGAASAHYIAGTMEDMAFAERFVVEAGKLLGGLDMLILNHITQT
TMSLFHDDIHSVRRSMEVNFLSYVVLSTAALPMLKQSNGSIAIISSMAGKMTQPLIASYS
ASKFALDGFFSTIRKEHLMTKVNVSITLCVLGFIDTETALKETSGIILSQAAPKEECALE
IIKGTVLRKDEVYYDKSSWTPLLLGNPGRRIMEFLSLRSYNRDLFVSN
|
|
|
|---|
| BDBM50273458 |
|---|
| n/a |
|---|
| Name | BDBM50273458 |
|---|
| Synonyms: | (5S)-2-{[(1S)-1-(4-fluorophenyl)ethyl]amino}-5-(1-hydroxy-1-methylethyl)-5-methyl-1,3-thiazol-4(5H)-one | (S)-2-[(S)-1-(4-Fluorophenyl)ethylamino]-5-(2-hydroxypropan-2-yl)-5-methylthiazol-4(5H)-one | CHEMBL455907 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H19FN2O2S |
|---|
| Mol. Mass. | 310.387 |
|---|
| SMILES | C[C@H](NC1=NC(=O)[C@@](C)(S1)C(C)(C)O)c1ccc(F)cc1 |r,t:3| |
|---|
| Structure | 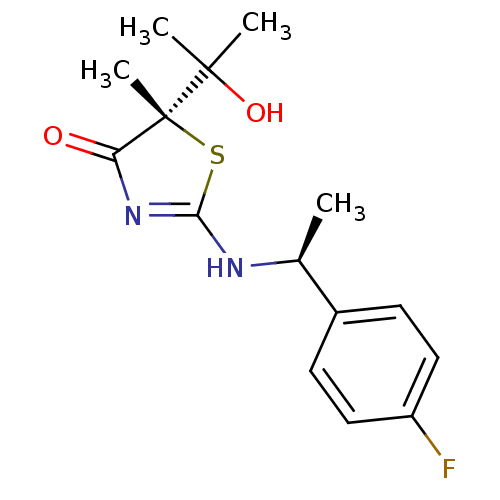
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Fotsch, C; Bartberger, MD; Bercot, EA; Chen, M; Cupples, R; Emery, M; Fretland, J; Guram, A; Hale, C; Han, N; Hickman, D; Hungate, RW; Hayashi, M; Komorowski, R; Liu, Q; Matsumoto, G; St Jean, DJ; Ursu, S; Véniant, M; Xu, G; Ye, Q; Yuan, C; Zhang, J; Zhang, X; Tu, H; Wang, M Further studies with the 2-amino-1,3-thiazol-4(5H)-one class of 11beta-hydroxysteroid dehydrogenase type 1 inhibitors: reducing pregnane X receptor activity and exploring activity in a monkey pharmacodynamic model. J Med Chem51:7953-67 (2008) [PubMed] Article
Fotsch, C; Bartberger, MD; Bercot, EA; Chen, M; Cupples, R; Emery, M; Fretland, J; Guram, A; Hale, C; Han, N; Hickman, D; Hungate, RW; Hayashi, M; Komorowski, R; Liu, Q; Matsumoto, G; St Jean, DJ; Ursu, S; Véniant, M; Xu, G; Ye, Q; Yuan, C; Zhang, J; Zhang, X; Tu, H; Wang, M Further studies with the 2-amino-1,3-thiazol-4(5H)-one class of 11beta-hydroxysteroid dehydrogenase type 1 inhibitors: reducing pregnane X receptor activity and exploring activity in a monkey pharmacodynamic model. J Med Chem51:7953-67 (2008) [PubMed] Article