| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prostaglandin G/H synthase 1 |
|---|
| Ligand | BDBM50297707 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_583915 (CHEMBL1059967) |
|---|
| IC50 | >100000±n/a nM |
|---|
| Citation |  Beswick, PJ; Blackaby, AP; Bountra, C; Brown, T; Browning, K; Campbell, IB; Corfield, J; Gleave, RJ; Guntrip, SB; Hall, RM; Hindley, S; Lambeth, PF; Lucas, F; Mathews, N; Naylor, A; Player, H; Price, HS; Sidebottom, PJ; Taylor, NL; Webb, G; Wiseman, J Identification and optimisation of a novel series of pyrimidine based cyclooxygenase-2 (COX-2) inhibitors. Utilisation of a biotransformation approach. Bioorg Med Chem Lett19:4509-14 (2009) [PubMed] Article Beswick, PJ; Blackaby, AP; Bountra, C; Brown, T; Browning, K; Campbell, IB; Corfield, J; Gleave, RJ; Guntrip, SB; Hall, RM; Hindley, S; Lambeth, PF; Lucas, F; Mathews, N; Naylor, A; Player, H; Price, HS; Sidebottom, PJ; Taylor, NL; Webb, G; Wiseman, J Identification and optimisation of a novel series of pyrimidine based cyclooxygenase-2 (COX-2) inhibitors. Utilisation of a biotransformation approach. Bioorg Med Chem Lett19:4509-14 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prostaglandin G/H synthase 1 |
|---|
| Name: | Prostaglandin G/H synthase 1 |
|---|
| Synonyms: | COX-1 | COX1 | Cyclooxygenase | Cyclooxygenase-1 | Cyclooxygenase-1 (COX-1) | PGH1_HUMAN | PTGS1 | Prostaglandin G/H synthase (cyclooxygenase) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 68692.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P23219 |
|---|
| Residue: | 599 |
|---|
| Sequence: | MSRSLLLWFLLFLLLLPPLPVLLADPGAPTPVNPCCYYPCQHQGICVRFGLDRYQCDCTR
TGYSGPNCTIPGLWTWLRNSLRPSPSFTHFLLTHGRWFWEFVNATFIREMLMRLVLTVRS
NLIPSPPTYNSAHDYISWESFSNVSYYTRILPSVPKDCPTPMGTKGKKQLPDAQLLARRF
LLRRKFIPDPQGTNLMFAFFAQHFTHQFFKTSGKMGPGFTKALGHGVDLGHIYGDNLERQ
YQLRLFKDGKLKYQVLDGEMYPPSVEEAPVLMHYPRGIPPQSQMAVGQEVFGLLPGLMLY
ATLWLREHNRVCDLLKAEHPTWGDEQLFQTTRLILIGETIKIVIEEYVQQLSGYFLQLKF
DPELLFGVQFQYRNRIAMEFNHLYHWHPLMPDSFKVGSQEYSYEQFLFNTSMLVDYGVEA
LVDAFSRQIAGRIGGGRNMDHHILHVAVDVIRESREMRLQPFNEYRKRFGMKPYTSFQEL
VGEKEMAAELEELYGDIDALEFYPGLLLEKCHPNSIFGESMIEIGAPFSLKGLLGNPICS
PEYWKPSTFGGEVGFNIVKTATLKKLVCLNTKTCPYVSFRVPDASQDDGPAVERPSTEL
|
|
|
|---|
| BDBM50297707 |
|---|
| n/a |
|---|
| Name | BDBM50297707 |
|---|
| Synonyms: | 4-(4-fluorophenyl)-5-(4-(methylsulfonyl)phenyl)-2-(phenylthio)pyrimidine | CHEMBL559422 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H17FN2O2S2 |
|---|
| Mol. Mass. | 436.522 |
|---|
| SMILES | CS(=O)(=O)c1ccc(cc1)-c1cnc(Sc2ccccc2)nc1-c1ccc(F)cc1 |
|---|
| Structure | 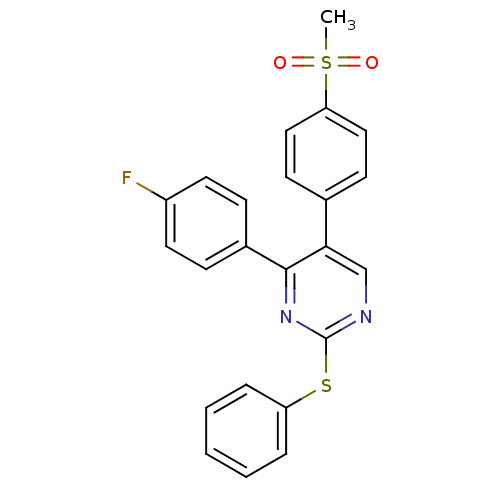
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Beswick, PJ; Blackaby, AP; Bountra, C; Brown, T; Browning, K; Campbell, IB; Corfield, J; Gleave, RJ; Guntrip, SB; Hall, RM; Hindley, S; Lambeth, PF; Lucas, F; Mathews, N; Naylor, A; Player, H; Price, HS; Sidebottom, PJ; Taylor, NL; Webb, G; Wiseman, J Identification and optimisation of a novel series of pyrimidine based cyclooxygenase-2 (COX-2) inhibitors. Utilisation of a biotransformation approach. Bioorg Med Chem Lett19:4509-14 (2009) [PubMed] Article
Beswick, PJ; Blackaby, AP; Bountra, C; Brown, T; Browning, K; Campbell, IB; Corfield, J; Gleave, RJ; Guntrip, SB; Hall, RM; Hindley, S; Lambeth, PF; Lucas, F; Mathews, N; Naylor, A; Player, H; Price, HS; Sidebottom, PJ; Taylor, NL; Webb, G; Wiseman, J Identification and optimisation of a novel series of pyrimidine based cyclooxygenase-2 (COX-2) inhibitors. Utilisation of a biotransformation approach. Bioorg Med Chem Lett19:4509-14 (2009) [PubMed] Article