| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM50391897 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_849757 (CHEMBL2150539) |
|---|
| IC50 | 2400±n/a nM |
|---|
| Citation |  van Eis, MJ; Evenou, JP; Floersheim, P; Gaul, C; Cowan-Jacob, SW; Monovich, L; Rummel, G; Schuler, W; Stark, W; Strauss, A; von Matt, A; Vangrevelinghe, E; Wagner, J; Soldermann, N 2,6-Naphthyridines as potent and selective inhibitors of the novel protein kinase C isozymes. Bioorg Med Chem Lett21:7367-72 (2011) [PubMed] Article van Eis, MJ; Evenou, JP; Floersheim, P; Gaul, C; Cowan-Jacob, SW; Monovich, L; Rummel, G; Schuler, W; Stark, W; Strauss, A; von Matt, A; Vangrevelinghe, E; Wagner, J; Soldermann, N 2,6-Naphthyridines as potent and selective inhibitors of the novel protein kinase C isozymes. Bioorg Med Chem Lett21:7367-72 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM50391897 |
|---|
| n/a |
|---|
| Name | BDBM50391897 |
|---|
| Synonyms: | CHEMBL2147537 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H17N5 |
|---|
| Mol. Mass. | 279.3397 |
|---|
| SMILES | C[C@H](N)CNc1nc(cc2cnccc12)-c1ccncc1 |r| |
|---|
| Structure | 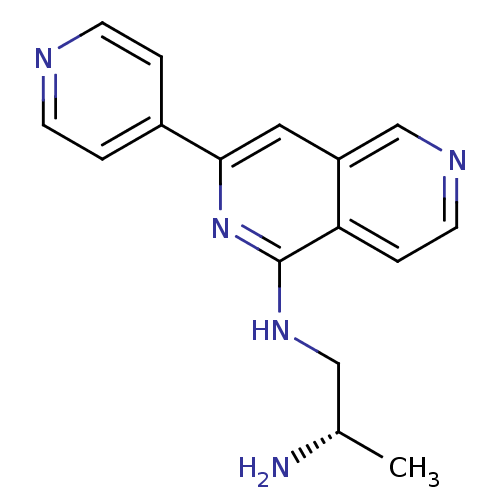
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 van Eis, MJ; Evenou, JP; Floersheim, P; Gaul, C; Cowan-Jacob, SW; Monovich, L; Rummel, G; Schuler, W; Stark, W; Strauss, A; von Matt, A; Vangrevelinghe, E; Wagner, J; Soldermann, N 2,6-Naphthyridines as potent and selective inhibitors of the novel protein kinase C isozymes. Bioorg Med Chem Lett21:7367-72 (2011) [PubMed] Article
van Eis, MJ; Evenou, JP; Floersheim, P; Gaul, C; Cowan-Jacob, SW; Monovich, L; Rummel, G; Schuler, W; Stark, W; Strauss, A; von Matt, A; Vangrevelinghe, E; Wagner, J; Soldermann, N 2,6-Naphthyridines as potent and selective inhibitors of the novel protein kinase C isozymes. Bioorg Med Chem Lett21:7367-72 (2011) [PubMed] Article