| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM50149566 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1558262 (CHEMBL3772396) |
|---|
| IC50 | >50000±n/a nM |
|---|
| Citation |  Demont, EH; Bailey, JM; Bit, RA; Brown, JA; Campbell, CA; Deeks, N; Dowell, SJ; Eldred, C; Gaskin, P; Gray, JR; Haynes, A; Hirst, DJ; Holmes, DS; Kumar, U; Morse, MA; Osborne, GJ; Renaux, JF; Seal, GA; Smethurst, CA; Taylor, S; Watson, R; Willis, R; Witherington, J Discovery of Tetrahydropyrazolopyridine as Sphingosine 1-Phosphate Receptor 3 (S1P3)-Sparing S1P1 Agonists Active at Low Oral Doses. J Med Chem59:1003-20 (2016) [PubMed] Article Demont, EH; Bailey, JM; Bit, RA; Brown, JA; Campbell, CA; Deeks, N; Dowell, SJ; Eldred, C; Gaskin, P; Gray, JR; Haynes, A; Hirst, DJ; Holmes, DS; Kumar, U; Morse, MA; Osborne, GJ; Renaux, JF; Seal, GA; Smethurst, CA; Taylor, S; Watson, R; Willis, R; Witherington, J Discovery of Tetrahydropyrazolopyridine as Sphingosine 1-Phosphate Receptor 3 (S1P3)-Sparing S1P1 Agonists Active at Low Oral Doses. J Med Chem59:1003-20 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM50149566 |
|---|
| n/a |
|---|
| Name | BDBM50149566 |
|---|
| Synonyms: | CHEMBL3769933 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H24ClN5O3S |
|---|
| Mol. Mass. | 461.965 |
|---|
| SMILES | CC(C)Oc1ccc(cc1Cl)-c1nnc(s1)-n1nc2CCN(CCC(O)=O)Cc2c1C |
|---|
| Structure | 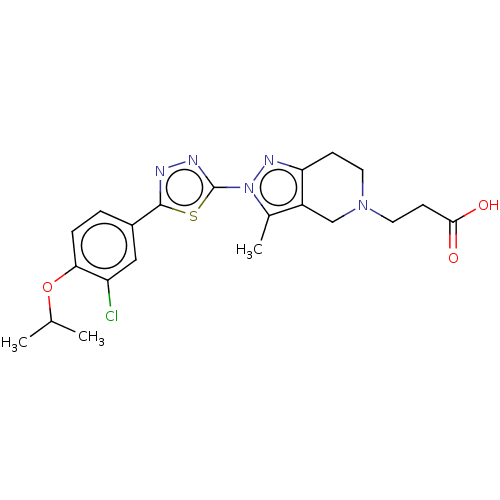
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Demont, EH; Bailey, JM; Bit, RA; Brown, JA; Campbell, CA; Deeks, N; Dowell, SJ; Eldred, C; Gaskin, P; Gray, JR; Haynes, A; Hirst, DJ; Holmes, DS; Kumar, U; Morse, MA; Osborne, GJ; Renaux, JF; Seal, GA; Smethurst, CA; Taylor, S; Watson, R; Willis, R; Witherington, J Discovery of Tetrahydropyrazolopyridine as Sphingosine 1-Phosphate Receptor 3 (S1P3)-Sparing S1P1 Agonists Active at Low Oral Doses. J Med Chem59:1003-20 (2016) [PubMed] Article
Demont, EH; Bailey, JM; Bit, RA; Brown, JA; Campbell, CA; Deeks, N; Dowell, SJ; Eldred, C; Gaskin, P; Gray, JR; Haynes, A; Hirst, DJ; Holmes, DS; Kumar, U; Morse, MA; Osborne, GJ; Renaux, JF; Seal, GA; Smethurst, CA; Taylor, S; Watson, R; Willis, R; Witherington, J Discovery of Tetrahydropyrazolopyridine as Sphingosine 1-Phosphate Receptor 3 (S1P3)-Sparing S1P1 Agonists Active at Low Oral Doses. J Med Chem59:1003-20 (2016) [PubMed] Article