 Found 3044 hits with Last Name = 'qu' and Initial = 'b'
Found 3044 hits with Last Name = 'qu' and Initial = 'b' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Glutamyl aminopeptidase
(Sus scrofa) | BDBM50078120
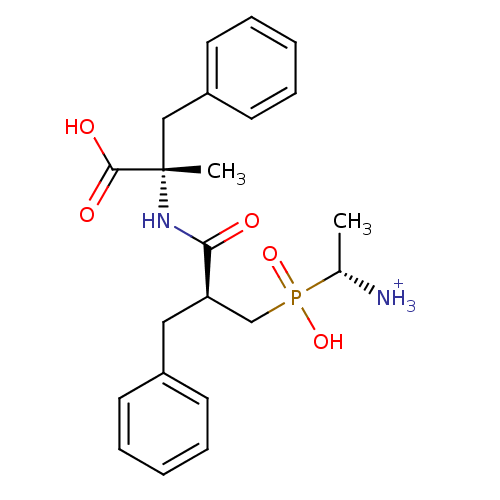
((R)-1-{[(S)-2-((S)-1-Carboxy-1-methyl-2-phenyl-eth...)Show SMILES C[C@H]([NH3+])P(O)(=O)C[C@@H](Cc1ccccc1)C(=O)N[C@@](C)(Cc1ccccc1)C(O)=O Show InChI InChI=1S/C22H29N2O5P/c1-16(23)30(28,29)15-19(13-17-9-5-3-6-10-17)20(25)24-22(2,21(26)27)14-18-11-7-4-8-12-18/h3-12,16,19H,13-15,23H2,1-2H3,(H,24,25)(H,26,27)(H,28,29)/p+1/t16-,19-,22+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibitory activity was measured on pig kidney Aminopeptidase N (activity for A+B stereoisomer) |
Bioorg Med Chem Lett 9: 1511-6 (1999)
BindingDB Entry DOI: 10.7270/Q2H132JR |
More data for this
Ligand-Target Pair | |
Aminopeptidase N
(Sus scrofa (Pig)) | BDBM50078120

((R)-1-{[(S)-2-((S)-1-Carboxy-1-methyl-2-phenyl-eth...)Show SMILES C[C@H]([NH3+])P(O)(=O)C[C@@H](Cc1ccccc1)C(=O)N[C@@](C)(Cc1ccccc1)C(O)=O Show InChI InChI=1S/C22H29N2O5P/c1-16(23)30(28,29)15-19(13-17-9-5-3-6-10-17)20(25)24-22(2,21(26)27)14-18-11-7-4-8-12-18/h3-12,16,19H,13-15,23H2,1-2H3,(H,24,25)(H,26,27)(H,28,29)/p+1/t16-,19-,22+/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibitory activity was measured on pig kidney Aminopeptidase N (activity for C+D stereoisomer) |
Bioorg Med Chem Lett 9: 1511-6 (1999)
BindingDB Entry DOI: 10.7270/Q2H132JR |
More data for this
Ligand-Target Pair | |
Aminopeptidase B
(Mus musculus) | BDBM50078122
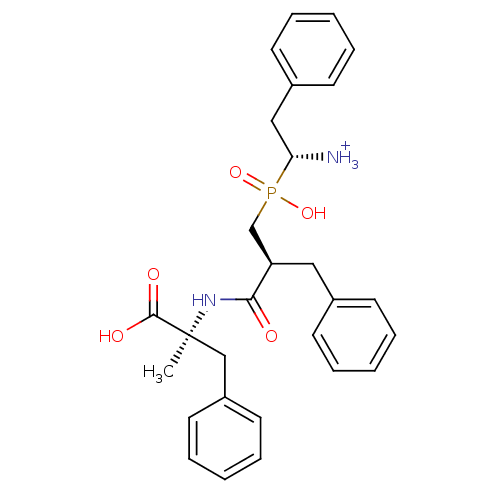
((R)-1-{[(S)-2-((S)-1-Carboxy-1-methyl-2-phenyl-eth...)Show SMILES C[C@@](Cc1ccccc1)(NC(=O)[C@H](Cc1ccccc1)CP(O)(=O)[C@@H]([NH3+])Cc1ccccc1)C(O)=O Show InChI InChI=1S/C28H33N2O5P/c1-28(27(32)33,19-23-15-9-4-10-16-23)30-26(31)24(17-21-11-5-2-6-12-21)20-36(34,35)25(29)18-22-13-7-3-8-14-22/h2-16,24-25H,17-20,29H2,1H3,(H,30,31)(H,32,33)(H,34,35)/p+1/t24-,25-,28+/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| >0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibitory activity was measured on Aminopeptidase B using Arg p.NA as substrate |
Bioorg Med Chem Lett 9: 1511-6 (1999)
BindingDB Entry DOI: 10.7270/Q2H132JR |
More data for this
Ligand-Target Pair | |
Glutamyl aminopeptidase
(Sus scrofa) | BDBM50078122

((R)-1-{[(S)-2-((S)-1-Carboxy-1-methyl-2-phenyl-eth...)Show SMILES C[C@@](Cc1ccccc1)(NC(=O)[C@H](Cc1ccccc1)CP(O)(=O)[C@@H]([NH3+])Cc1ccccc1)C(O)=O Show InChI InChI=1S/C28H33N2O5P/c1-28(27(32)33,19-23-15-9-4-10-16-23)30-26(31)24(17-21-11-5-2-6-12-21)20-36(34,35)25(29)18-22-13-7-3-8-14-22/h2-16,24-25H,17-20,29H2,1H3,(H,30,31)(H,32,33)(H,34,35)/p+1/t24-,25-,28+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibitory activity was measured on Aminopeptidase using GluNA as substrate |
Bioorg Med Chem Lett 9: 1511-6 (1999)
BindingDB Entry DOI: 10.7270/Q2H132JR |
More data for this
Ligand-Target Pair | |
Aminopeptidase B
(Mus musculus) | BDBM50078120

((R)-1-{[(S)-2-((S)-1-Carboxy-1-methyl-2-phenyl-eth...)Show SMILES C[C@H]([NH3+])P(O)(=O)C[C@@H](Cc1ccccc1)C(=O)N[C@@](C)(Cc1ccccc1)C(O)=O Show InChI InChI=1S/C22H29N2O5P/c1-16(23)30(28,29)15-19(13-17-9-5-3-6-10-17)20(25)24-22(2,21(26)27)14-18-11-7-4-8-12-18/h3-12,16,19H,13-15,23H2,1-2H3,(H,24,25)(H,26,27)(H,28,29)/p+1/t16-,19-,22+/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| >0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibitory activity was measured on Aminopeptidase B using Arg p.NA as substrate |
Bioorg Med Chem Lett 9: 1511-6 (1999)
BindingDB Entry DOI: 10.7270/Q2H132JR |
More data for this
Ligand-Target Pair | |
Aminopeptidase N
(Sus scrofa (Pig)) | BDBM50078122

((R)-1-{[(S)-2-((S)-1-Carboxy-1-methyl-2-phenyl-eth...)Show SMILES C[C@@](Cc1ccccc1)(NC(=O)[C@H](Cc1ccccc1)CP(O)(=O)[C@@H]([NH3+])Cc1ccccc1)C(O)=O Show InChI InChI=1S/C28H33N2O5P/c1-28(27(32)33,19-23-15-9-4-10-16-23)30-26(31)24(17-21-11-5-2-6-12-21)20-36(34,35)25(29)18-22-13-7-3-8-14-22/h2-16,24-25H,17-20,29H2,1H3,(H,30,31)(H,32,33)(H,34,35)/p+1/t24-,25-,28+/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibitory activity was measured on pig kidney Aminopeptidase N (activity for C+D stereoisomer) |
Bioorg Med Chem Lett 9: 1511-6 (1999)
BindingDB Entry DOI: 10.7270/Q2H132JR |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50045797
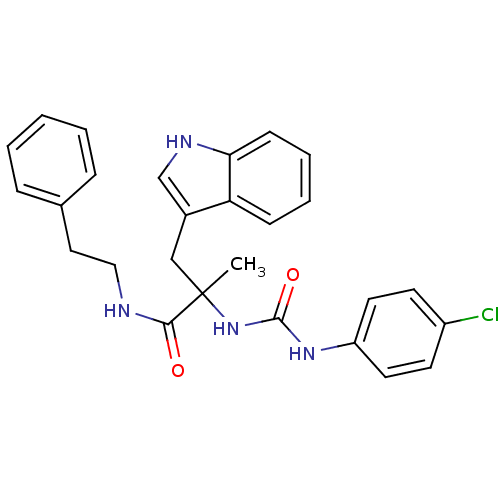
(2-[3-(4-Chloro-phenyl)-ureido]-3-(1H-indol-3-yl)-2...)Show SMILES CC(Cc1c[nH]c2ccccc12)(NC(=O)Nc1ccc(Cl)cc1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C27H27ClN4O2/c1-27(17-20-18-30-24-10-6-5-9-23(20)24,25(33)29-16-15-19-7-3-2-4-8-19)32-26(34)31-22-13-11-21(28)12-14-22/h2-14,18,30H,15-17H2,1H3,(H,29,33)(H2,31,32,34) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| >0.000100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Tested for inhibition of [3H]-pCCK-8 specific binding to cholecystokinin type B receptor in guinea pig brain cortex |
J Med Chem 36: 2868-77 (1993)
BindingDB Entry DOI: 10.7270/Q2MG7NK5 |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50281664

(CHEMBL262894 | p-Tyr(SO3Na)-gNle-mGly-Trp-(N-Me)Nl...)Show SMILES CCCC[C@H](NC(=O)[C@@H](N)Cc1ccc(OS([O-])(=O)=O)cc1)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCCC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C47H61N9O13S/c1-3-5-15-35(53-43(61)33(48)22-29-18-20-31(21-19-29)69-70(66,67)68)44(62)51-27-40(57)52-38(24-30-26-50-34-17-11-10-14-32(30)34)46(64)54-36(16-6-4-2)45(63)56-39(25-41(58)59)47(65)55-37(42(49)60)23-28-12-8-7-9-13-28/h7-14,17-21,26,33,35-39,50H,3-6,15-16,22-25,27,48H2,1-2H3,(H2,49,60)(H,51,62)(H,52,57)(H,53,61)(H,54,64)(H,55,65)(H,56,63)(H,58,59)(H,66,67,68)/p-1/t33-,35-,36-,37-,38-,39-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Tested for the inhibition of [3H]-pCCK-8 to Cholecystokinin type B receptor in guinea pig brain |
Bioorg Med Chem Lett 3: 847-850 (1993)
Article DOI: 10.1016/S0960-894X(00)80678-2
BindingDB Entry DOI: 10.7270/Q2P26Z28 |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50046128
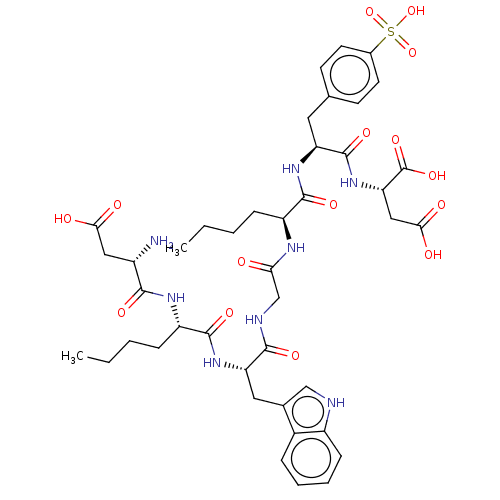
(2-[2-(2-{2-[2-{2-[3-Carboxy-2-(2-methylamino-3-phe...)Show SMILES CCCC[C@H](NC(=O)CNC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CCCC)NC(=O)[C@@H](N)CC(O)=O)C(=O)N[C@@H](Cc1ccc(cc1)S(O)(=O)=O)C(=O)N[C@@H](CC(O)=O)C(O)=O Show InChI InChI=1S/C42H56N8O15S/c1-3-5-10-29(39(58)48-31(41(60)50-33(42(61)62)20-36(54)55)17-23-13-15-25(16-14-23)66(63,64)65)46-34(51)22-45-38(57)32(18-24-21-44-28-12-8-7-9-26(24)28)49-40(59)30(11-6-4-2)47-37(56)27(43)19-35(52)53/h7-9,12-16,21,27,29-33,44H,3-6,10-11,17-20,22,43H2,1-2H3,(H,45,57)(H,46,51)(H,47,56)(H,48,58)(H,49,59)(H,50,60)(H,52,53)(H,54,55)(H,61,62)(H,63,64,65)/t27-,29-,30-,31-,32-,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]pCCK-8 from cholecystokinin type B receptor in guinea pig brain membrane |
J Med Chem 36: 166-72 (1993)
BindingDB Entry DOI: 10.7270/Q26M37FJ |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50016504

((S)-3-(2-{[(S)-2-(2-{2-[2-tert-Butoxycarbonylamino...)Show SMILES CCCC[C@H](NC(=O)[C@H](Cc1ccc(OS([O-])(=O)=O)cc1)NC(=O)OC(C)(C)C)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCCC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C52H69N9O15S/c1-6-8-18-37(57-48(68)40(61-51(71)75-52(3,4)5)26-32-21-23-34(24-22-32)76-77(72,73)74)46(66)55-30-43(62)56-41(27-33-29-54-36-20-14-13-17-35(33)36)49(69)58-38(19-9-7-2)47(67)60-42(28-44(63)64)50(70)59-39(45(53)65)25-31-15-11-10-12-16-31/h10-17,20-24,29,37-42,54H,6-9,18-19,25-28,30H2,1-5H3,(H2,53,65)(H,55,66)(H,56,62)(H,57,68)(H,58,69)(H,59,70)(H,60,67)(H,61,71)(H,63,64)(H,72,73,74)/p-1/t37-,38-,39-,40-,41-,42-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Tested for the inhibition of [3H]-pCCK-8 binding to Cholecystokinin type B receptor in guinea pig brain |
Bioorg Med Chem Lett 3: 847-850 (1993)
Article DOI: 10.1016/S0960-894X(00)80678-2
BindingDB Entry DOI: 10.7270/Q2P26Z28 |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A/Gastrin/cholecystokinin type B receptor
(Mus musculus-MOUSE) | BDBM50016425
((S)-3-{(S)-2-[(S)-2-(2-{(S)-2-[(S)-2-tert-Butoxyca...)Show SMILES CCCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)OC(C)(C)C)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCCC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C52H69N9O15S/c1-6-8-18-37(57-48(68)40(61-51(71)75-52(3,4)5)26-32-21-23-34(24-22-32)76-77(72,73)74)46(66)55-30-43(62)56-41(27-33-29-54-36-20-14-13-17-35(33)36)49(69)58-38(19-9-7-2)47(67)60-42(28-44(63)64)50(70)59-39(45(53)65)25-31-15-11-10-12-16-31/h10-17,20-24,29,37-42,54H,6-9,18-19,25-28,30H2,1-5H3,(H2,53,65)(H,55,66)(H,56,62)(H,57,68)(H,58,69)(H,59,70)(H,60,67)(H,61,71)(H,63,64)(H,72,73,74)/t37-,38-,39-,40-,41-,42-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.191 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Binding affinity measured by inhibiting [3H]Boc[Nle28,31]CCK27-33 specific binding to Cholecystokinin receptor in mouse brain membranes at a KD conce... |
J Med Chem 30: 962-8 (1987)
BindingDB Entry DOI: 10.7270/Q21J98R1 |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50016425

((S)-3-{(S)-2-[(S)-2-(2-{(S)-2-[(S)-2-tert-Butoxyca...)Show SMILES CCCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)OC(C)(C)C)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCCC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C52H69N9O15S/c1-6-8-18-37(57-48(68)40(61-51(71)75-52(3,4)5)26-32-21-23-34(24-22-32)76-77(72,73)74)46(66)55-30-43(62)56-41(27-33-29-54-36-20-14-13-17-35(33)36)49(69)58-38(19-9-7-2)47(67)60-42(28-44(63)64)50(70)59-39(45(53)65)25-31-15-11-10-12-16-31/h10-17,20-24,29,37-42,54H,6-9,18-19,25-28,30H2,1-5H3,(H2,53,65)(H,55,66)(H,56,62)(H,57,68)(H,58,69)(H,59,70)(H,60,67)(H,61,71)(H,63,64)(H,72,73,74)/t37-,38-,39-,40-,41-,42-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of 0.2 nM [3H]-pCCK-8 from guinea pig brain membranes |
J Med Chem 32: 1184-90 (1989)
BindingDB Entry DOI: 10.7270/Q2ZG6R7N |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Cavia porcellus) | BDBM50016504

((S)-3-(2-{[(S)-2-(2-{2-[2-tert-Butoxycarbonylamino...)Show SMILES CCCC[C@H](NC(=O)[C@H](Cc1ccc(OS([O-])(=O)=O)cc1)NC(=O)OC(C)(C)C)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCCC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C52H69N9O15S/c1-6-8-18-37(57-48(68)40(61-51(71)75-52(3,4)5)26-32-21-23-34(24-22-32)76-77(72,73)74)46(66)55-30-43(62)56-41(27-33-29-54-36-20-14-13-17-35(33)36)49(69)58-38(19-9-7-2)47(67)60-42(28-44(63)64)50(70)59-39(45(53)65)25-31-15-11-10-12-16-31/h10-17,20-24,29,37-42,54H,6-9,18-19,25-28,30H2,1-5H3,(H2,53,65)(H,55,66)(H,56,62)(H,57,68)(H,58,69)(H,59,70)(H,60,67)(H,61,71)(H,63,64)(H,72,73,74)/p-1/t37-,38-,39-,40-,41-,42-/m0/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
The compound was tested for the inhibition of [3H]-Propionyl specific binding to Cholecystokinin 8 receptor of guinea pig brain |
J Med Chem 32: 445-9 (1989)
BindingDB Entry DOI: 10.7270/Q2S181GT |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM21147

((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C49H62N10O16S3/c1-76-18-16-34(55-47(69)37(58-44(66)32(50)23-41(61)62)21-28-12-14-30(15-13-28)75-78(72,73)74)45(67)53-26-40(60)54-38(22-29-25-52-33-11-7-6-10-31(29)33)48(70)56-35(17-19-77-2)46(68)59-39(24-42(63)64)49(71)57-36(43(51)65)20-27-8-4-3-5-9-27/h3-15,25,32,34-39,52H,16-24,26,50H2,1-2H3,(H2,51,65)(H,53,67)(H,54,60)(H,55,69)(H,56,70)(H,57,71)(H,58,66)(H,59,68)(H,61,62)(H,63,64)(H,72,73,74)/t32-,34-,35-,36-,37-,38-,39-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of 0.2 nM [3H]-pCCK-8 from guinea pig brain membranes |
J Med Chem 32: 1184-90 (1989)
BindingDB Entry DOI: 10.7270/Q2ZG6R7N |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Cavia porcellus) | BDBM21147

((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C49H62N10O16S3/c1-76-18-16-34(55-47(69)37(58-44(66)32(50)23-41(61)62)21-28-12-14-30(15-13-28)75-78(72,73)74)45(67)53-26-40(60)54-38(22-29-25-52-33-11-7-6-10-31(29)33)48(70)56-35(17-19-77-2)46(68)59-39(24-42(63)64)49(71)57-36(43(51)65)20-27-8-4-3-5-9-27/h3-15,25,32,34-39,52H,16-24,26,50H2,1-2H3,(H2,51,65)(H,53,67)(H,54,60)(H,55,69)(H,56,70)(H,57,71)(H,58,66)(H,59,68)(H,61,62)(H,63,64)(H,72,73,74)/t32-,34-,35-,36-,37-,38-,39-/m0/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
The compound was tested for the inhibition of [3H]-Propionyl specific binding to CCK-8 receptor of guinea pig brain |
J Med Chem 32: 445-9 (1989)
BindingDB Entry DOI: 10.7270/Q2S181GT |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Cavia porcellus) | BDBM21147

((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C49H62N10O16S3/c1-76-18-16-34(55-47(69)37(58-44(66)32(50)23-41(61)62)21-28-12-14-30(15-13-28)75-78(72,73)74)45(67)53-26-40(60)54-38(22-29-25-52-33-11-7-6-10-31(29)33)48(70)56-35(17-19-77-2)46(68)59-39(24-42(63)64)49(71)57-36(43(51)65)20-27-8-4-3-5-9-27/h3-15,25,32,34-39,52H,16-24,26,50H2,1-2H3,(H2,51,65)(H,53,67)(H,54,60)(H,55,69)(H,56,70)(H,57,71)(H,58,66)(H,59,68)(H,61,62)(H,63,64)(H,72,73,74)/t32-,34-,35-,36-,37-,38-,39-/m0/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of 0.1 nM [3H]-pCCK-8 from guinea pig pancreatic membranes |
J Med Chem 32: 1184-90 (1989)
BindingDB Entry DOI: 10.7270/Q2ZG6R7N |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Cavia porcellus) | BDBM21147

((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C49H62N10O16S3/c1-76-18-16-34(55-47(69)37(58-44(66)32(50)23-41(61)62)21-28-12-14-30(15-13-28)75-78(72,73)74)45(67)53-26-40(60)54-38(22-29-25-52-33-11-7-6-10-31(29)33)48(70)56-35(17-19-77-2)46(68)59-39(24-42(63)64)49(71)57-36(43(51)65)20-27-8-4-3-5-9-27/h3-15,25,32,34-39,52H,16-24,26,50H2,1-2H3,(H2,51,65)(H,53,67)(H,54,60)(H,55,69)(H,56,70)(H,57,71)(H,58,66)(H,59,68)(H,61,62)(H,63,64)(H,72,73,74)/t32-,34-,35-,36-,37-,38-,39-/m0/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibition of [3H]pCCK-8 binding to Cholecystokinin type A receptor of Guinea pig pancreatic membranes |
J Med Chem 40: 647-58 (1997)
Article DOI: 10.1021/jm9603072
BindingDB Entry DOI: 10.7270/Q2PG1SDX |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM21147

((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C49H62N10O16S3/c1-76-18-16-34(55-47(69)37(58-44(66)32(50)23-41(61)62)21-28-12-14-30(15-13-28)75-78(72,73)74)45(67)53-26-40(60)54-38(22-29-25-52-33-11-7-6-10-31(29)33)48(70)56-35(17-19-77-2)46(68)59-39(24-42(63)64)49(71)57-36(43(51)65)20-27-8-4-3-5-9-27/h3-15,25,32,34-39,52H,16-24,26,50H2,1-2H3,(H2,51,65)(H,53,67)(H,54,60)(H,55,69)(H,56,70)(H,57,71)(H,58,66)(H,59,68)(H,61,62)(H,63,64)(H,72,73,74)/t32-,34-,35-,36-,37-,38-,39-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]pCCK-8 from cholecystokinin type B receptor in guinea pig brain membrane |
J Med Chem 36: 166-72 (1993)
BindingDB Entry DOI: 10.7270/Q26M37FJ |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A/Gastrin/cholecystokinin type B receptor
(Mus musculus-MOUSE) | BDBM21147

((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C49H62N10O16S3/c1-76-18-16-34(55-47(69)37(58-44(66)32(50)23-41(61)62)21-28-12-14-30(15-13-28)75-78(72,73)74)45(67)53-26-40(60)54-38(22-29-25-52-33-11-7-6-10-31(29)33)48(70)56-35(17-19-77-2)46(68)59-39(24-42(63)64)49(71)57-36(43(51)65)20-27-8-4-3-5-9-27/h3-15,25,32,34-39,52H,16-24,26,50H2,1-2H3,(H2,51,65)(H,53,67)(H,54,60)(H,55,69)(H,56,70)(H,57,71)(H,58,66)(H,59,68)(H,61,62)(H,63,64)(H,72,73,74)/t32-,34-,35-,36-,37-,38-,39-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Potency in displacing [3H]propionyl-Cholecystokinin from mouse brain membranes. |
J Med Chem 31: 966-70 (1988)
BindingDB Entry DOI: 10.7270/Q2XP775P |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50449787

(CHEMBL2062154 | PD-134308)Show SMILES [H][C@@]12C[C@@]3([H])C[C@@]([H])(C1)C(OC(=O)N[C@](C)(Cc1c[nH]c4ccccc14)C(=O)NC[C@H](NC(=O)CCC(O)=O)c1ccccc1)[C@@]([H])(C2)C3 |wU:30.41,14.15,45.49,3.3,wD:6.6,1.0,TLB:5:3:47:9.6.8,10:9:47:3.48.2,THB:5:6:47:3.48.2,10:9:1.47.8:3.5.48,2:3:9:1.47.8,2:1:9:3.5.48,(-14.99,-2.1,;-13.56,-2.66,;-14.77,-3.94,;-13.26,-3.53,;-13.35,-5.06,;-11.86,-4.09,;-10.83,-2.82,;-9.38,-3.33,;-12.24,-3.16,;-10.83,-1.28,;-9.29,-1.31,;-8.5,.01,;-9.25,1.36,;-6.96,-.01,;-6.19,1.3,;-5.42,-.02,;-7.44,2.2,;-7.28,3.74,;-8.44,4.76,;-7.81,6.18,;-6.29,6.03,;-5.14,7.07,;-3.69,6.6,;-3.34,5.08,;-4.49,4.06,;-5.94,4.52,;-4.66,1.42,;-3.99,2.8,;-3.79,.15,;-2.27,.27,;-1.4,-1.02,;-2.08,-2.4,;-1.22,-3.69,;.32,-3.58,;-1.9,-5.07,;-3.43,-5.16,;-4.1,-6.57,;-5.64,-6.67,;-3.25,-7.83,;.14,-.91,;.99,-2.2,;2.51,-2.08,;3.19,-.7,;2.32,.59,;.8,.47,;-12.23,-.7,;-12.2,.82,;-13.58,-1.18,;-13.27,-1.94,)| Show InChI InChI=1S/C35H42N4O6/c1-35(18-26-19-36-28-10-6-5-9-27(26)28,39-34(44)45-32-24-14-21-13-22(16-24)17-25(32)15-21)33(43)37-20-29(23-7-3-2-4-8-23)38-30(40)11-12-31(41)42/h2-10,19,21-22,24-25,29,32,36H,11-18,20H2,1H3,(H,37,43)(H,38,40)(H,39,44)(H,41,42)/t21-,22+,24-,25+,29-,32?,35+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Compound was tested for the affinity against Cholecystokinin type B receptor on guinea pig cortex. |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50021325

(4-methyl-(1S,5R,13R,14S)-12-oxa-4-azapentacyclo[9....)Show SMILES CN1CC[C@@]23[C@H]4Oc5c2c(C[C@@H]1C3CC[C@@H]4O)ccc5O |TLB:13:12:8.9.10:1.3.2| Show InChI InChI=1S/C17H21NO3/c1-18-7-6-17-10-3-5-13(20)16(17)21-15-12(19)4-2-9(14(15)17)8-11(10)18/h2,4,10-11,13,16,19-20H,3,5-8H2,1H3/t10?,11-,13+,16+,17+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Eur J Pharmacol 383: 209-14 (1999)
Article DOI: 10.1016/s0014-2999(99)00610-x
BindingDB Entry DOI: 10.7270/Q2TT4PJ9 |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50281665
(Boc-Phe(CH2-SO3Na)-gNle-mCIy-Trp-(N-Me)Nle-Asp-Phe...)Show SMILES CCCC[C@H](NC(=O)[C@H](Cc1ccc(CS([O-])(=O)=O)cc1)NC(=O)OC(C)(C)C)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCCC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C53H71N9O14S/c1-6-8-18-38(58-49(69)41(62-52(72)76-53(3,4)5)26-33-21-23-34(24-22-33)31-77(73,74)75)47(67)56-30-44(63)57-42(27-35-29-55-37-20-14-13-17-36(35)37)50(70)59-39(19-9-7-2)48(68)61-43(28-45(64)65)51(71)60-40(46(54)66)25-32-15-11-10-12-16-32/h10-17,20-24,29,38-43,55H,6-9,18-19,25-28,30-31H2,1-5H3,(H2,54,66)(H,56,67)(H,57,63)(H,58,69)(H,59,70)(H,60,71)(H,61,68)(H,62,72)(H,64,65)(H,73,74,75)/p-1/t38-,39-,40-,41-,42-,43-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
| 0.460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Tested for the inhibition of [3H]-pCCK-8 to Cholecystokinin type B receptor in guinea pig brain |
Bioorg Med Chem Lett 3: 847-850 (1993)
Article DOI: 10.1016/S0960-894X(00)80678-2
BindingDB Entry DOI: 10.7270/Q2P26Z28 |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Rat 6B) | BDBM82276

(L31,P34-NPY,human | NPY Leu31, Pro34, human, rat |...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1c[nH]cn1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](C)NC(=O)[C@H](CCSC)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C188H282N54O56S/c1-15-94(8)148(178(292)231-127(81-139(191)251)165(279)223-122(74-92(4)5)168(282)235-147(93(6)7)177(291)237-149(99(13)245)179(293)220-119(31-21-66-206-188(200)201)183(297)241-70-25-35-137(241)174(288)218-114(30-20-65-205-187(198)199)155(269)221-120(150(193)264)76-101-39-49-107(247)50-40-101)236-169(283)125(79-104-45-55-110(250)56-46-104)226-164(278)126(80-105-86-202-90-208-105)227-157(271)113(29-19-64-204-186(196)197)216-161(275)121(73-91(2)3)222-153(267)96(10)210-170(284)132(88-243)233-163(277)124(78-103-43-53-109(249)54-44-103)225-162(276)123(77-102-41-51-108(248)52-42-102)224-156(270)112(28-18-63-203-185(194)195)214-151(265)95(9)209-154(268)117(61-72-299-14)217-166(280)129(84-145(260)261)229-159(273)116(58-60-143(256)257)215-152(266)97(11)211-173(287)135-33-23-67-238(135)180(294)98(12)212-160(274)128(83-144(258)259)228-158(272)115(57-59-142(254)255)213-141(253)87-207-172(286)134-32-22-69-240(134)184(298)131(82-140(192)252)232-167(281)130(85-146(262)263)230-175(289)138-36-26-71-242(138)182(296)118(27-16-17-62-189)219-171(285)133(89-244)234-176(290)136-34-24-68-239(136)181(295)111(190)75-100-37-47-106(246)48-38-100/h37-56,86,90-99,111-138,147-149,243-250H,15-36,57-85,87-89,189-190H2,1-14H3,(H2,191,251)(H2,192,252)(H2,193,264)(H,202,208)(H,207,286)(H,209,268)(H,210,284)(H,211,287)(H,212,274)(H,213,253)(H,214,265)(H,215,266)(H,216,275)(H,217,280)(H,218,288)(H,219,285)(H,220,293)(H,221,269)(H,222,267)(H,223,279)(H,224,270)(H,225,276)(H,226,278)(H,227,271)(H,228,272)(H,229,273)(H,230,289)(H,231,292)(H,232,281)(H,233,277)(H,234,290)(H,235,282)(H,236,283)(H,237,291)(H,254,255)(H,256,257)(H,258,259)(H,260,261)(H,262,263)(H4,194,195,203)(H4,196,197,204)(H4,198,199,205)(H4,200,201,206)/t94-,95-,96-,97-,98-,99+,111-,112-,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,147-,148-,149-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer Corporation
Curated by PDSP Ki Database
| |
J Biol Chem 271: 26315-9 (1996)
Article DOI: 10.1074/jbc.271.42.26315
BindingDB Entry DOI: 10.7270/Q20G3HPR |
More data for this
Ligand-Target Pair | |
Growth hormone secretagogue receptor type 1
(Ovis aries) | BDBM50222885
(6-(4-fluorophenoxy)-3-[(1-isopropylpiperidin-3-yl)...)Show SMILES CC(C)N1CCCC(Cn2c(nc3ccc(Oc4ccc(F)cc4)cc3c2=O)-c2ccccc2C)C1 |w:7.7| Show InChI InChI=1S/C30H32FN3O2/c1-20(2)33-16-6-8-22(18-33)19-34-29(26-9-5-4-7-21(26)3)32-28-15-14-25(17-27(28)30(34)35)36-24-12-10-23(31)11-13-24/h4-5,7,9-15,17,20,22H,6,8,16,18-19H2,1-3H3 | PDB
KEGG
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer Pharmaceuticals Corporation
Curated by ChEMBL
| Assay Description
Displacement of [125I]ghrelin from ovine recombinant GHSR1a expressed in HEK293F cells after 6 hrs by scintillation proximity assay |
J Med Chem 50: 5202-16 (2007)
Article DOI: 10.1021/jm070071+
BindingDB Entry DOI: 10.7270/Q2WH2QT2 |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Rat 6B) | BDBM50091652

(CHEMBL269503 | PYY | PYY, rat | Peptide YY(PYY)(YP...)Show SMILES CC(C)C[C@H](NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CO)NC(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1c[nH]cn1)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(O)=O Show InChI InChI=1S/C190H287N53O58/c1-92(2)74-124(166(280)216-114(27-18-66-204-187(195)196)158(272)231-131(83-107-86-203-91-209-107)171(285)230-130(81-105-41-51-111(251)52-42-105)169(283)225-125(75-93(3)4)167(281)232-132(84-143(194)254)172(286)226-127(77-95(7)8)173(287)238-150(96(9)10)180(294)239-151(101(15)247)181(295)222-117(30-21-69-207-190(201)202)156(270)218-119(55-60-142(193)253)161(275)215-116(29-20-68-206-189(199)200)159(273)234-134(186(300)301)82-106-43-53-112(252)54-44-106)227-175(289)135(88-244)235-153(267)97(11)210-164(278)128(79-103-37-47-109(249)48-38-103)229-170(284)129(80-104-39-49-110(250)50-40-104)228-157(271)115(28-19-67-205-188(197)198)217-174(288)136(89-245)236-168(282)126(76-94(5)6)224-163(277)121(58-63-147(260)261)219-162(276)122(59-64-148(262)263)221-179(293)141-34-25-73-243(141)185(299)137(90-246)237-154(268)98(12)211-165(279)133(85-149(264)265)233-160(274)118(56-61-145(256)257)214-144(255)87-208-176(290)138-31-22-70-240(138)182(296)100(14)213-155(269)120(57-62-146(258)259)220-178(292)140-33-24-72-242(140)184(298)123(26-16-17-65-191)223-152(266)99(13)212-177(291)139-32-23-71-241(139)183(297)113(192)78-102-35-45-108(248)46-36-102/h35-54,86,91-101,113-141,150-151,244-252H,16-34,55-85,87-90,191-192H2,1-15H3,(H2,193,253)(H2,194,254)(H,203,209)(H,208,290)(H,210,278)(H,211,279)(H,212,291)(H,213,269)(H,214,255)(H,215,275)(H,216,280)(H,217,288)(H,218,270)(H,219,276)(H,220,292)(H,221,293)(H,222,295)(H,223,266)(H,224,277)(H,225,283)(H,226,286)(H,227,289)(H,228,271)(H,229,284)(H,230,285)(H,231,272)(H,232,281)(H,233,274)(H,234,273)(H,235,267)(H,236,282)(H,237,268)(H,238,287)(H,239,294)(H,256,257)(H,258,259)(H,260,261)(H,262,263)(H,264,265)(H,300,301)(H4,195,196,204)(H4,197,198,205)(H4,199,200,206)(H4,201,202,207)/t97-,98-,99-,100-,101+,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,139-,140-,141-,150-,151-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer Corporation
Curated by PDSP Ki Database
| |
J Biol Chem 271: 26315-9 (1996)
Article DOI: 10.1074/jbc.271.42.26315
BindingDB Entry DOI: 10.7270/Q20G3HPR |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Rat 6B) | BDBM50015490
(CHEMBL438945 | H-YPSKPDNPGEDAPAEDMARYYSALRHYINLITR...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1c[nH]cn1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](C)NC(=O)[C@H](CCSC)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C189H285N55O57S/c1-15-93(7)148(179(295)234-128(81-140(193)254)168(284)226-123(74-92(5)6)171(287)239-149(94(8)16-2)180(296)240-150(99(13)247)181(297)222-115(31-22-67-208-189(202)203)156(272)220-117(56-59-139(192)253)161(277)218-113(29-20-65-206-187(198)199)157(273)224-121(151(195)267)76-101-38-48-107(249)49-39-101)238-172(288)126(79-104-44-54-110(252)55-45-104)229-167(283)127(80-105-86-204-90-210-105)230-159(275)114(30-21-66-207-188(200)201)219-164(280)122(73-91(3)4)225-154(270)96(10)212-173(289)133(88-245)236-166(282)125(78-103-42-52-109(251)53-43-103)228-165(281)124(77-102-40-50-108(250)51-41-102)227-158(274)112(28-19-64-205-186(196)197)216-152(268)95(9)211-155(271)119(62-72-302-14)221-169(285)130(84-146(263)264)232-162(278)118(58-61-144(259)260)217-153(269)97(11)213-176(292)136-33-24-68-241(136)182(298)98(12)214-163(279)129(83-145(261)262)231-160(276)116(57-60-143(257)258)215-142(256)87-209-175(291)135-32-23-70-243(135)185(301)132(82-141(194)255)235-170(286)131(85-147(265)266)233-177(293)138-35-26-71-244(138)184(300)120(27-17-18-63-190)223-174(290)134(89-246)237-178(294)137-34-25-69-242(137)183(299)111(191)75-100-36-46-106(248)47-37-100/h36-55,86,90-99,111-138,148-150,245-252H,15-35,56-85,87-89,190-191H2,1-14H3,(H2,192,253)(H2,193,254)(H2,194,255)(H2,195,267)(H,204,210)(H,209,291)(H,211,271)(H,212,289)(H,213,292)(H,214,279)(H,215,256)(H,216,268)(H,217,269)(H,218,277)(H,219,280)(H,220,272)(H,221,285)(H,222,297)(H,223,290)(H,224,273)(H,225,270)(H,226,284)(H,227,274)(H,228,281)(H,229,283)(H,230,275)(H,231,276)(H,232,278)(H,233,293)(H,234,295)(H,235,286)(H,236,282)(H,237,294)(H,238,288)(H,239,287)(H,240,296)(H,257,258)(H,259,260)(H,261,262)(H,263,264)(H,265,266)(H4,196,197,205)(H4,198,199,206)(H4,200,201,207)(H4,202,203,208)/t93-,94-,95-,96-,97-,98-,99+,111-,112-,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,148-,149-,150-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer Corporation
Curated by PDSP Ki Database
| |
J Biol Chem 271: 26315-9 (1996)
Article DOI: 10.1074/jbc.271.42.26315
BindingDB Entry DOI: 10.7270/Q20G3HPR |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A/Gastrin/cholecystokinin type B receptor
(Mus musculus-MOUSE) | BDBM21147

((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C49H62N10O16S3/c1-76-18-16-34(55-47(69)37(58-44(66)32(50)23-41(61)62)21-28-12-14-30(15-13-28)75-78(72,73)74)45(67)53-26-40(60)54-38(22-29-25-52-33-11-7-6-10-31(29)33)48(70)56-35(17-19-77-2)46(68)59-39(24-42(63)64)49(71)57-36(43(51)65)20-27-8-4-3-5-9-27/h3-15,25,32,34-39,52H,16-24,26,50H2,1-2H3,(H2,51,65)(H,53,67)(H,54,60)(H,55,69)(H,56,70)(H,57,71)(H,58,66)(H,59,68)(H,61,62)(H,63,64)(H,72,73,74)/t32-,34-,35-,36-,37-,38-,39-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.491 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Binding affinity measured by inhibiting [3H]Boc[Nle28,31]CCK27-33 specific binding to Cholecystokinin receptor in mouse brain membranes at a KD conce... |
J Med Chem 30: 962-8 (1987)
BindingDB Entry DOI: 10.7270/Q21J98R1 |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Rat 6B) | BDBM50015490

(CHEMBL438945 | H-YPSKPDNPGEDAPAEDMARYYSALRHYINLITR...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1c[nH]cn1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](C)NC(=O)[C@H](CCSC)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C189H285N55O57S/c1-15-93(7)148(179(295)234-128(81-140(193)254)168(284)226-123(74-92(5)6)171(287)239-149(94(8)16-2)180(296)240-150(99(13)247)181(297)222-115(31-22-67-208-189(202)203)156(272)220-117(56-59-139(192)253)161(277)218-113(29-20-65-206-187(198)199)157(273)224-121(151(195)267)76-101-38-48-107(249)49-39-101)238-172(288)126(79-104-44-54-110(252)55-45-104)229-167(283)127(80-105-86-204-90-210-105)230-159(275)114(30-21-66-207-188(200)201)219-164(280)122(73-91(3)4)225-154(270)96(10)212-173(289)133(88-245)236-166(282)125(78-103-42-52-109(251)53-43-103)228-165(281)124(77-102-40-50-108(250)51-41-102)227-158(274)112(28-19-64-205-186(196)197)216-152(268)95(9)211-155(271)119(62-72-302-14)221-169(285)130(84-146(263)264)232-162(278)118(58-61-144(259)260)217-153(269)97(11)213-176(292)136-33-24-68-241(136)182(298)98(12)214-163(279)129(83-145(261)262)231-160(276)116(57-60-143(257)258)215-142(256)87-209-175(291)135-32-23-70-243(135)185(301)132(82-141(194)255)235-170(286)131(85-147(265)266)233-177(293)138-35-26-71-244(138)184(300)120(27-17-18-63-190)223-174(290)134(89-246)237-178(294)137-34-25-69-242(137)183(299)111(191)75-100-36-46-106(248)47-37-100/h36-55,86,90-99,111-138,148-150,245-252H,15-35,56-85,87-89,190-191H2,1-14H3,(H2,192,253)(H2,193,254)(H2,194,255)(H2,195,267)(H,204,210)(H,209,291)(H,211,271)(H,212,289)(H,213,292)(H,214,279)(H,215,256)(H,216,268)(H,217,269)(H,218,277)(H,219,280)(H,220,272)(H,221,285)(H,222,297)(H,223,290)(H,224,273)(H,225,270)(H,226,284)(H,227,274)(H,228,281)(H,229,283)(H,230,275)(H,231,276)(H,232,278)(H,233,293)(H,234,295)(H,235,286)(H,236,282)(H,237,294)(H,238,288)(H,239,287)(H,240,296)(H,257,258)(H,259,260)(H,261,262)(H,263,264)(H,265,266)(H4,196,197,205)(H4,198,199,206)(H4,200,201,207)(H4,202,203,208)/t93-,94-,95-,96-,97-,98-,99+,111-,112-,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,148-,149-,150-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer Corporation
Curated by PDSP Ki Database
| |
J Biol Chem 271: 26315-9 (1996)
Article DOI: 10.1074/jbc.271.42.26315
BindingDB Entry DOI: 10.7270/Q20G3HPR |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50061266
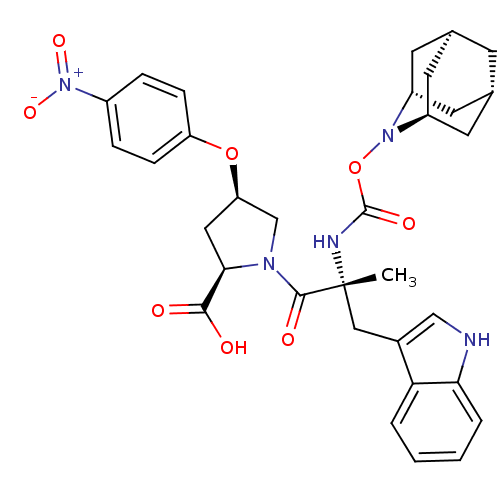
((2R,4R)-1-[(R)-2-(2-Aza-tricyclo[3.3.1.1*3,7*]dec-...)Show SMILES C[C@](Cc1c[nH]c2ccccc12)(NC(=O)ON1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2)C(=O)N1C[C@@H](C[C@@H]1C(O)=O)Oc1ccc(cc1)[N+]([O-])=O |TLB:24:23:25:19.18.20,15:16:18:21.25.20,THB:22:23:18:21.25.20,22:21:18:16.23.24,15:16:25:19.18.20| Show InChI InChI=1S/C33H37N5O8/c1-33(16-21-17-34-28-5-3-2-4-27(21)28,35-32(42)46-37-23-11-19-10-20(13-23)14-24(37)12-19)31(41)36-18-26(15-29(36)30(39)40)45-25-8-6-22(7-9-25)38(43)44/h2-9,17,19-20,23-24,26,29,34H,10-16,18H2,1H3,(H,35,42)(H,39,40)/t19-,20+,23-,24+,26-,29-,33-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Binding affinity (affinity state 1) for Cholecystokinin type B receptor, was determined using CHO cells |
J Med Chem 40: 3947-56 (1998)
Article DOI: 10.1021/jm970439a
BindingDB Entry DOI: 10.7270/Q27H1K8Z |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50018060
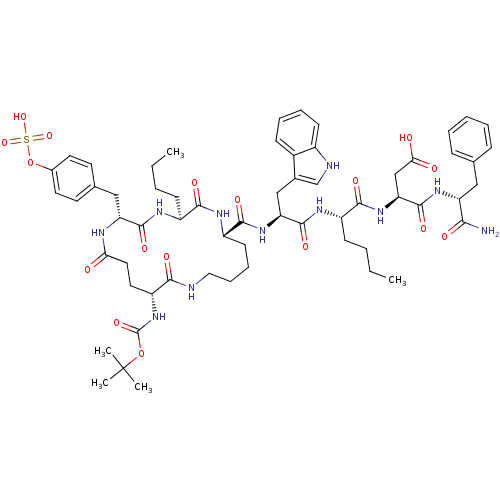
(Boc-cyclo-(gama-D-Glu-Tyr(SO3H)-Nle-D-Lys)-Trp-Nle...)Show SMILES CCCC[C@H](NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@@H]1CCCCNC(=O)[C@@H](CCC(=O)N[C@H](Cc2ccc(OS(O)(=O)=O)cc2)C(=O)N[C@@H](CCCC)C(=O)N1)NC(=O)OC(C)(C)C)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C61H83N11O17S/c1-6-8-20-42-54(78)66-44(23-15-16-30-63-53(77)45(72-60(84)88-61(3,4)5)28-29-50(73)65-47(57(81)67-42)32-37-24-26-39(27-25-37)89-90(85,86)87)56(80)70-48(33-38-35-64-41-22-14-13-19-40(38)41)58(82)68-43(21-9-7-2)55(79)71-49(34-51(74)75)59(83)69-46(52(62)76)31-36-17-11-10-12-18-36/h10-14,17-19,22,24-27,35,42-49,64H,6-9,15-16,20-21,23,28-34H2,1-5H3,(H2,62,76)(H,63,77)(H,65,73)(H,66,78)(H,67,81)(H,68,82)(H,69,83)(H,70,80)(H,71,79)(H,72,84)(H,74,75)(H,85,86,87)/t42-,43-,44-,45+,46+,47+,48-,49-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.560 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of 0.2 nM [3H]-pCCK-8 from guinea pig brain membranes |
J Med Chem 32: 1184-90 (1989)
BindingDB Entry DOI: 10.7270/Q2ZG6R7N |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Rat 6B) | BDBM84999
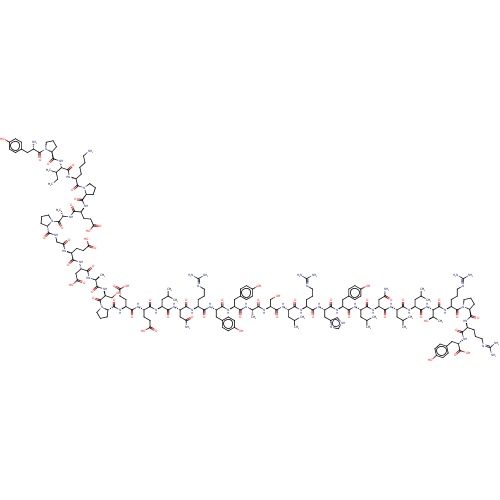
(PYY,[leu31,Pro34], human)Show SMILES CCC(C)[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)NCC(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CO)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1c[nH]cn1)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(O)=O Show InChI InChI=1S/C194H295N53O58/c1-17-101(12)155(241-182(294)144-37-25-73-244(144)187(299)117(196)82-106-39-49-112(251)50-40-106)184(296)224-125(29-18-19-67-195)188(300)245-74-27-36-143(245)181(293)222-122(60-64-151(261)262)159(271)216-104(15)186(298)243-72-24-34-141(243)179(291)212-92-148(258)217-121(59-63-150(259)260)163(275)237-138(90-154(267)268)167(279)215-103(14)158(270)240-149(94-249)305(304)247-76-28-38-145(247)183(295)223-124(62-66-153(265)266)164(276)220-123(61-65-152(263)264)165(277)226-128(78-97(4)5)170(282)235-136(88-146(197)256)175(287)219-118(30-20-68-208-191(199)200)160(272)231-133(84-108-43-53-114(253)54-44-108)173(285)232-132(83-107-41-51-113(252)52-42-107)166(278)214-102(13)157(269)239-140(93-248)178(290)230-127(77-96(2)3)168(280)218-119(31-21-69-209-192(201)202)161(273)234-135(87-111-91-207-95-213-111)174(286)233-134(85-109-45-55-115(254)56-46-109)172(284)227-130(80-99(8)9)171(283)236-137(89-147(198)257)176(288)228-129(79-98(6)7)169(281)229-131(81-100(10)11)177(289)242-156(105(16)250)185(297)225-126(33-23-71-211-194(205)206)189(301)246-75-26-35-142(246)180(292)221-120(32-22-70-210-193(203)204)162(274)238-139(190(302)303)86-110-47-57-116(255)58-48-110/h39-58,91,95-105,117-145,149,155-156,248-255H,17-38,59-90,92-94,195-196H2,1-16H3,(H2,197,256)(H2,198,257)(H,207,213)(H,212,291)(H,214,278)(H,215,279)(H,216,271)(H,217,258)(H,218,280)(H,219,287)(H,220,276)(H,221,292)(H,222,293)(H,223,295)(H,224,296)(H,225,297)(H,226,277)(H,227,284)(H,228,288)(H,229,281)(H,230,290)(H,231,272)(H,232,285)(H,233,286)(H,234,273)(H,235,282)(H,236,283)(H,237,275)(H,238,274)(H,239,269)(H,240,270)(H,241,294)(H,242,289)(H,259,260)(H,261,262)(H,263,264)(H,265,266)(H,267,268)(H,302,303)(H4,199,200,208)(H4,201,202,209)(H4,203,204,210)(H4,205,206,211)/t101?,102-,103-,104-,105+,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,139-,140-,141-,142-,143-,144-,145-,149+,155-,156-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.570 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer Corporation
Curated by PDSP Ki Database
| |
J Biol Chem 271: 26315-9 (1996)
Article DOI: 10.1074/jbc.271.42.26315
BindingDB Entry DOI: 10.7270/Q20G3HPR |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM21147

((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C49H62N10O16S3/c1-76-18-16-34(55-47(69)37(58-44(66)32(50)23-41(61)62)21-28-12-14-30(15-13-28)75-78(72,73)74)45(67)53-26-40(60)54-38(22-29-25-52-33-11-7-6-10-31(29)33)48(70)56-35(17-19-77-2)46(68)59-39(24-42(63)64)49(71)57-36(43(51)65)20-27-8-4-3-5-9-27/h3-15,25,32,34-39,52H,16-24,26,50H2,1-2H3,(H2,51,65)(H,53,67)(H,54,60)(H,55,69)(H,56,70)(H,57,71)(H,58,66)(H,59,68)(H,61,62)(H,63,64)(H,72,73,74)/t32-,34-,35-,36-,37-,38-,39-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Affinity to inhibit [3H]pCCK-8 specific binding on rat brain Cholecystokinin type B receptor expressed in CHO cells |
J Med Chem 40: 647-58 (1997)
Article DOI: 10.1021/jm9603072
BindingDB Entry DOI: 10.7270/Q2PG1SDX |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM21147

((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C49H62N10O16S3/c1-76-18-16-34(55-47(69)37(58-44(66)32(50)23-41(61)62)21-28-12-14-30(15-13-28)75-78(72,73)74)45(67)53-26-40(60)54-38(22-29-25-52-33-11-7-6-10-31(29)33)48(70)56-35(17-19-77-2)46(68)59-39(24-42(63)64)49(71)57-36(43(51)65)20-27-8-4-3-5-9-27/h3-15,25,32,34-39,52H,16-24,26,50H2,1-2H3,(H2,51,65)(H,53,67)(H,54,60)(H,55,69)(H,56,70)(H,57,71)(H,58,66)(H,59,68)(H,61,62)(H,63,64)(H,72,73,74)/t32-,34-,35-,36-,37-,38-,39-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
| 0.640 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Tested for the inhibition of [3H]-pCCK-8 binding to Cholecystokinin type B receptor in guinea pig brain |
Bioorg Med Chem Lett 3: 847-850 (1993)
Article DOI: 10.1016/S0960-894X(00)80678-2
BindingDB Entry DOI: 10.7270/Q2P26Z28 |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Rat 6B) | BDBM82276

(L31,P34-NPY,human | NPY Leu31, Pro34, human, rat |...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1c[nH]cn1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](C)NC(=O)[C@H](CCSC)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C188H282N54O56S/c1-15-94(8)148(178(292)231-127(81-139(191)251)165(279)223-122(74-92(4)5)168(282)235-147(93(6)7)177(291)237-149(99(13)245)179(293)220-119(31-21-66-206-188(200)201)183(297)241-70-25-35-137(241)174(288)218-114(30-20-65-205-187(198)199)155(269)221-120(150(193)264)76-101-39-49-107(247)50-40-101)236-169(283)125(79-104-45-55-110(250)56-46-104)226-164(278)126(80-105-86-202-90-208-105)227-157(271)113(29-19-64-204-186(196)197)216-161(275)121(73-91(2)3)222-153(267)96(10)210-170(284)132(88-243)233-163(277)124(78-103-43-53-109(249)54-44-103)225-162(276)123(77-102-41-51-108(248)52-42-102)224-156(270)112(28-18-63-203-185(194)195)214-151(265)95(9)209-154(268)117(61-72-299-14)217-166(280)129(84-145(260)261)229-159(273)116(58-60-143(256)257)215-152(266)97(11)211-173(287)135-33-23-67-238(135)180(294)98(12)212-160(274)128(83-144(258)259)228-158(272)115(57-59-142(254)255)213-141(253)87-207-172(286)134-32-22-69-240(134)184(298)131(82-140(192)252)232-167(281)130(85-146(262)263)230-175(289)138-36-26-71-242(138)182(296)118(27-16-17-62-189)219-171(285)133(89-244)234-176(290)136-34-24-68-239(136)181(295)111(190)75-100-37-47-106(246)48-38-100/h37-56,86,90-99,111-138,147-149,243-250H,15-36,57-85,87-89,189-190H2,1-14H3,(H2,191,251)(H2,192,252)(H2,193,264)(H,202,208)(H,207,286)(H,209,268)(H,210,284)(H,211,287)(H,212,274)(H,213,253)(H,214,265)(H,215,266)(H,216,275)(H,217,280)(H,218,288)(H,219,285)(H,220,293)(H,221,269)(H,222,267)(H,223,279)(H,224,270)(H,225,276)(H,226,278)(H,227,271)(H,228,272)(H,229,273)(H,230,289)(H,231,292)(H,232,281)(H,233,277)(H,234,290)(H,235,282)(H,236,283)(H,237,291)(H,254,255)(H,256,257)(H,258,259)(H,260,261)(H,262,263)(H4,194,195,203)(H4,196,197,204)(H4,198,199,205)(H4,200,201,206)/t94-,95-,96-,97-,98-,99+,111-,112-,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,147-,148-,149-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.640 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer Corporation
Curated by PDSP Ki Database
| |
J Biol Chem 271: 26315-9 (1996)
Article DOI: 10.1074/jbc.271.42.26315
BindingDB Entry DOI: 10.7270/Q20G3HPR |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Cavia porcellus) | BDBM21147

((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C49H62N10O16S3/c1-76-18-16-34(55-47(69)37(58-44(66)32(50)23-41(61)62)21-28-12-14-30(15-13-28)75-78(72,73)74)45(67)53-26-40(60)54-38(22-29-25-52-33-11-7-6-10-31(29)33)48(70)56-35(17-19-77-2)46(68)59-39(24-42(63)64)49(71)57-36(43(51)65)20-27-8-4-3-5-9-27/h3-15,25,32,34-39,52H,16-24,26,50H2,1-2H3,(H2,51,65)(H,53,67)(H,54,60)(H,55,69)(H,56,70)(H,57,71)(H,58,66)(H,59,68)(H,61,62)(H,63,64)(H,72,73,74)/t32-,34-,35-,36-,37-,38-,39-/m0/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.640 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
The compound was tested for the inhibition of [3H]-Propionyl specific binding to Cholecystokinin 8 receptor of guinea pig brain |
J Med Chem 32: 445-9 (1989)
BindingDB Entry DOI: 10.7270/Q2S181GT |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Cavia porcellus) | BDBM21147

((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C49H62N10O16S3/c1-76-18-16-34(55-47(69)37(58-44(66)32(50)23-41(61)62)21-28-12-14-30(15-13-28)75-78(72,73)74)45(67)53-26-40(60)54-38(22-29-25-52-33-11-7-6-10-31(29)33)48(70)56-35(17-19-77-2)46(68)59-39(24-42(63)64)49(71)57-36(43(51)65)20-27-8-4-3-5-9-27/h3-15,25,32,34-39,52H,16-24,26,50H2,1-2H3,(H2,51,65)(H,53,67)(H,54,60)(H,55,69)(H,56,70)(H,57,71)(H,58,66)(H,59,68)(H,61,62)(H,63,64)(H,72,73,74)/t32-,34-,35-,36-,37-,38-,39-/m0/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
| 0.640 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Tested for the inhibition of [3H]-pCCK-8 binding to Cholecystokinin type A receptor in pancreatic membranes of guinea-pig |
Bioorg Med Chem Lett 3: 847-850 (1993)
Article DOI: 10.1016/S0960-894X(00)80678-2
BindingDB Entry DOI: 10.7270/Q2P26Z28 |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM21147

((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C49H62N10O16S3/c1-76-18-16-34(55-47(69)37(58-44(66)32(50)23-41(61)62)21-28-12-14-30(15-13-28)75-78(72,73)74)45(67)53-26-40(60)54-38(22-29-25-52-33-11-7-6-10-31(29)33)48(70)56-35(17-19-77-2)46(68)59-39(24-42(63)64)49(71)57-36(43(51)65)20-27-8-4-3-5-9-27/h3-15,25,32,34-39,52H,16-24,26,50H2,1-2H3,(H2,51,65)(H,53,67)(H,54,60)(H,55,69)(H,56,70)(H,57,71)(H,58,66)(H,59,68)(H,61,62)(H,63,64)(H,72,73,74)/t32-,34-,35-,36-,37-,38-,39-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.640 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibition of [3H]pCCK-8 binding to Guinea pig cortex membrane Cholecystokinin type B receptor |
J Med Chem 40: 647-58 (1997)
Article DOI: 10.1021/jm9603072
BindingDB Entry DOI: 10.7270/Q2PG1SDX |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Rat 6B) | BDBM82277
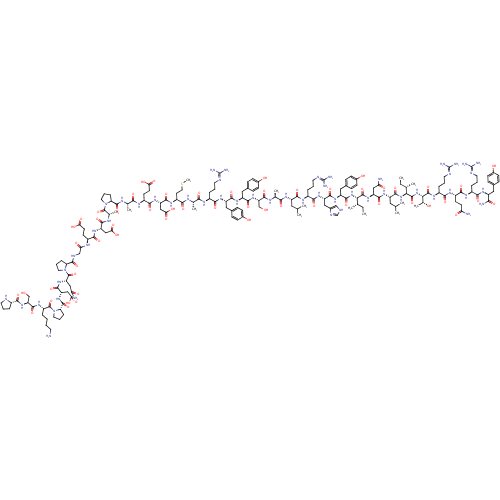
(NPY2-36, human | NPY2-36, rat, human)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1c[nH]cn1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](C)NC(=O)[C@H](CCSC)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C180H276N54O55S/c1-15-88(7)140(171(284)225-121(76-132(183)243)161(274)217-116(70-87(5)6)164(277)230-141(89(8)16-2)172(285)231-142(94(13)237)173(286)213-108(32-23-64-199-180(192)193)149(262)211-110(52-55-131(182)242)154(267)209-106(30-21-62-197-178(188)189)150(263)215-114(143(185)256)71-95-36-44-100(238)45-37-95)229-165(278)119(74-98-42-50-103(241)51-43-98)220-160(273)120(75-99-81-194-85-201-99)221-152(265)107(31-22-63-198-179(190)191)210-157(270)115(69-86(3)4)216-146(259)91(10)203-166(279)126(83-235)228-159(272)118(73-97-40-48-102(240)49-41-97)219-158(271)117(72-96-38-46-101(239)47-39-96)218-151(264)105(29-20-61-196-177(186)187)207-144(257)90(9)202-148(261)112(58-68-290-14)212-162(275)123(79-138(252)253)223-155(268)111(54-57-136(248)249)208-145(258)92(11)204-169(282)129-34-25-65-232(129)174(287)93(12)205-156(269)122(78-137(250)251)222-153(266)109(53-56-135(246)247)206-134(245)82-200-168(281)128-33-24-66-233(128)176(289)125(77-133(184)244)226-163(276)124(80-139(254)255)224-170(283)130-35-26-67-234(130)175(288)113(27-17-18-59-181)214-167(280)127(84-236)227-147(260)104-28-19-60-195-104/h36-51,81,85-94,104-130,140-142,195,235-241H,15-35,52-80,82-84,181H2,1-14H3,(H2,182,242)(H2,183,243)(H2,184,244)(H2,185,256)(H,194,201)(H,200,281)(H,202,261)(H,203,279)(H,204,282)(H,205,269)(H,206,245)(H,207,257)(H,208,258)(H,209,267)(H,210,270)(H,211,262)(H,212,275)(H,213,286)(H,214,280)(H,215,263)(H,216,259)(H,217,274)(H,218,264)(H,219,271)(H,220,273)(H,221,265)(H,222,266)(H,223,268)(H,224,283)(H,225,284)(H,226,276)(H,227,260)(H,228,272)(H,229,278)(H,230,277)(H,231,285)(H,246,247)(H,248,249)(H,250,251)(H,252,253)(H,254,255)(H4,186,187,196)(H4,188,189,197)(H4,190,191,198)(H4,192,193,199)/t88-,89-,90-,91-,92-,93-,94+,104-,105-,106-,107-,108-,109-,110-,111-,112-,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,140-,141-,142-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.650 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer Corporation
Curated by PDSP Ki Database
| |
J Biol Chem 271: 26315-9 (1996)
Article DOI: 10.1074/jbc.271.42.26315
BindingDB Entry DOI: 10.7270/Q20G3HPR |
More data for this
Ligand-Target Pair | |
Neprilysin
(Homo sapiens (Human)) | BDBM50407297
(CHEMBL2052008)Show SMILES C[C@H](NC(=O)[C@H](CS)[C@@H](C)c1ccccc1)C(O)=O |r| Show InChI InChI=1S/C14H19NO3S/c1-9(11-6-4-3-5-7-11)12(8-19)13(16)15-10(2)14(17)18/h3-7,9-10,12,19H,8H2,1-2H3,(H,15,16)(H,17,18)/t9-,10-,12+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
In vivo inhibitory potency against neutral endopeptidase by displacement of [3H]-HACBOGly binding in mouse kidney |
J Med Chem 37: 1070-83 (1994)
BindingDB Entry DOI: 10.7270/Q26W995T |
More data for this
Ligand-Target Pair | |
Neprilysin
(Oryctolagus cuniculus (rabbit)) | BDBM21653
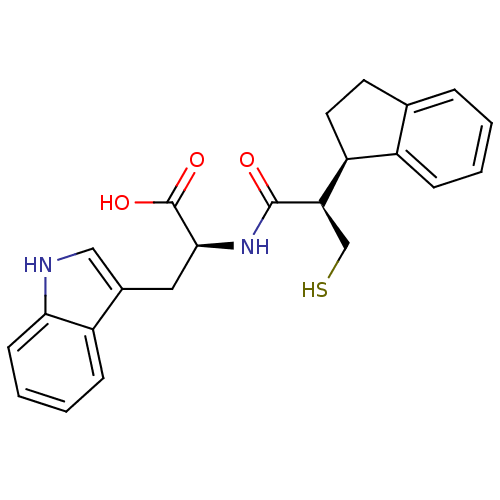
((2S)-2-[(2R)-2-[(1R)-2,3-dihydro-1H-inden-1-yl]-3-...)Show SMILES OC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CS)[C@H]1CCc2ccccc12 |r| Show InChI InChI=1S/C23H24N2O3S/c26-22(19(13-29)18-10-9-14-5-1-2-6-16(14)18)25-21(23(27)28)11-15-12-24-20-8-4-3-7-17(15)20/h1-8,12,18-19,21,24,29H,9-11,13H2,(H,25,26)(H,27,28)/t18-,19+,21-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.700 | -54.4 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
CNRS
| Assay Description
NEP was preincubated in black 96-well microplates with or without increasing concentrations of inhibitors. DGPA was added, and the reaction was stopp... |
J Med Chem 45: 1477-86 (2002)
Article DOI: 10.1021/jm0005454
BindingDB Entry DOI: 10.7270/Q2T72FQ1 |
More data for this
Ligand-Target Pair | |
Cholecystokinin receptor type A
(Cavia porcellus) | BDBM21147

((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C49H62N10O16S3/c1-76-18-16-34(55-47(69)37(58-44(66)32(50)23-41(61)62)21-28-12-14-30(15-13-28)75-78(72,73)74)45(67)53-26-40(60)54-38(22-29-25-52-33-11-7-6-10-31(29)33)48(70)56-35(17-19-77-2)46(68)59-39(24-42(63)64)49(71)57-36(43(51)65)20-27-8-4-3-5-9-27/h3-15,25,32,34-39,52H,16-24,26,50H2,1-2H3,(H2,51,65)(H,53,67)(H,54,60)(H,55,69)(H,56,70)(H,57,71)(H,58,66)(H,59,68)(H,61,62)(H,63,64)(H,72,73,74)/t32-,34-,35-,36-,37-,38-,39-/m0/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]-pCCK-8 from cholecystokinin-A receptor in guinea pig pancreatic membrane |
J Med Chem 36: 166-72 (1993)
BindingDB Entry DOI: 10.7270/Q26M37FJ |
More data for this
Ligand-Target Pair | |
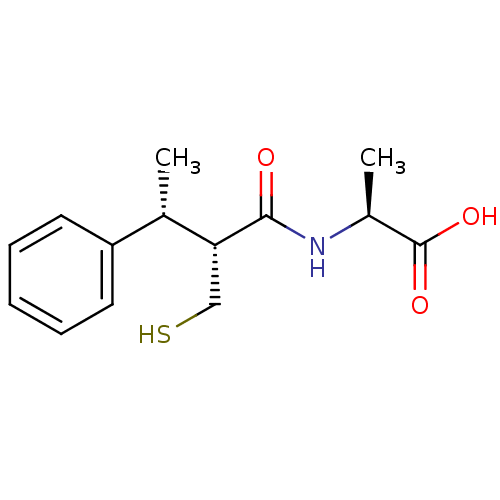
Neprilysin
(Homo sapiens (Human)) | BDBM50407297

(CHEMBL2052008)Show SMILES C[C@H](NC(=O)[C@H](CS)[C@@H](C)c1ccccc1)C(O)=O |r| Show InChI InChI=1S/C14H19NO3S/c1-9(11-6-4-3-5-7-11)12(8-19)13(16)15-10(2)14(17)18/h3-7,9-10,12,19H,8H2,1-2H3,(H,15,16)(H,17,18)/t9-,10-,12+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
In vivo inhibitory potency against neutral endopeptidase by displacement of [3H]-HACBOGly binding in mouse kidney |
J Med Chem 37: 1070-83 (1994)
BindingDB Entry DOI: 10.7270/Q26W995T |
More data for this
Ligand-Target Pair | |
Neprilysin
(Homo sapiens (Human)) | BDBM50024101
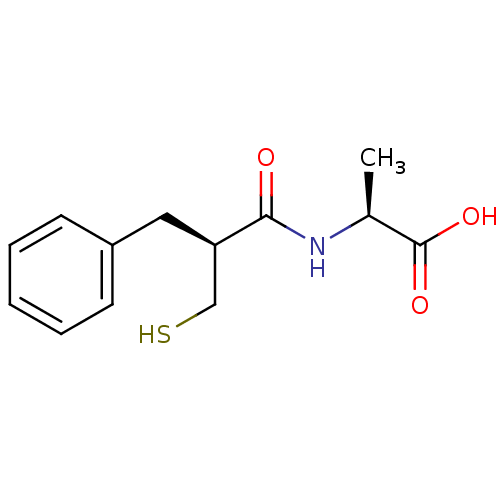
((S)-2-((S)-2-Mercaptomethyl-3-phenyl-propionylamin...)Show InChI InChI=1S/C13H17NO3S/c1-9(13(16)17)14-12(15)11(8-18)7-10-5-3-2-4-6-10/h2-6,9,11,18H,7-8H2,1H3,(H,14,15)(H,16,17)/t9-,11+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaleads , Paris BioPark, 11 Rue Watt, 75013 Paris, France.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant NEP using Suc-Ala-Ala-Phe-AMC as substrate after 30 mins by fluorimetry |
J Med Chem 57: 5748-63 (2014)
Article DOI: 10.1021/jm500602h
BindingDB Entry DOI: 10.7270/Q2Q241TH |
More data for this
Ligand-Target Pair | |
Neprilysin
(Oryctolagus cuniculus (rabbit)) | BDBM21653

((2S)-2-[(2R)-2-[(1R)-2,3-dihydro-1H-inden-1-yl]-3-...)Show SMILES OC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CS)[C@H]1CCc2ccccc12 |r| Show InChI InChI=1S/C23H24N2O3S/c26-22(19(13-29)18-10-9-14-5-1-2-6-16(14)18)25-21(23(27)28)11-15-12-24-20-8-4-3-7-17(15)20/h1-8,12,18-19,21,24,29H,9-11,13H2,(H,25,26)(H,27,28)/t18-,19+,21-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
In vitro inhibition of Neutral endopeptidase. |
Bioorg Med Chem Lett 12: 2001-5 (2002)
BindingDB Entry DOI: 10.7270/Q27080S4 |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50015490

(CHEMBL438945 | H-YPSKPDNPGEDAPAEDMARYYSALRHYINLITR...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1c[nH]cn1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](C)NC(=O)[C@H](CCSC)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C189H285N55O57S/c1-15-93(7)148(179(295)234-128(81-140(193)254)168(284)226-123(74-92(5)6)171(287)239-149(94(8)16-2)180(296)240-150(99(13)247)181(297)222-115(31-22-67-208-189(202)203)156(272)220-117(56-59-139(192)253)161(277)218-113(29-20-65-206-187(198)199)157(273)224-121(151(195)267)76-101-38-48-107(249)49-39-101)238-172(288)126(79-104-44-54-110(252)55-45-104)229-167(283)127(80-105-86-204-90-210-105)230-159(275)114(30-21-66-207-188(200)201)219-164(280)122(73-91(3)4)225-154(270)96(10)212-173(289)133(88-245)236-166(282)125(78-103-42-52-109(251)53-43-103)228-165(281)124(77-102-40-50-108(250)51-41-102)227-158(274)112(28-19-64-205-186(196)197)216-152(268)95(9)211-155(271)119(62-72-302-14)221-169(285)130(84-146(263)264)232-162(278)118(58-61-144(259)260)217-153(269)97(11)213-176(292)136-33-24-68-241(136)182(298)98(12)214-163(279)129(83-145(261)262)231-160(276)116(57-60-143(257)258)215-142(256)87-209-175(291)135-32-23-70-243(135)185(301)132(82-141(194)255)235-170(286)131(85-147(265)266)233-177(293)138-35-26-71-244(138)184(300)120(27-17-18-63-190)223-174(290)134(89-246)237-178(294)137-34-25-69-242(137)183(299)111(191)75-100-36-46-106(248)47-37-100/h36-55,86,90-99,111-138,148-150,245-252H,15-35,56-85,87-89,190-191H2,1-14H3,(H2,192,253)(H2,193,254)(H2,194,255)(H2,195,267)(H,204,210)(H,209,291)(H,211,271)(H,212,289)(H,213,292)(H,214,279)(H,215,256)(H,216,268)(H,217,269)(H,218,277)(H,219,280)(H,220,272)(H,221,285)(H,222,297)(H,223,290)(H,224,273)(H,225,270)(H,226,284)(H,227,274)(H,228,281)(H,229,283)(H,230,275)(H,231,276)(H,232,278)(H,233,293)(H,234,295)(H,235,286)(H,236,282)(H,237,294)(H,238,288)(H,239,287)(H,240,296)(H,257,258)(H,259,260)(H,261,262)(H,263,264)(H,265,266)(H4,196,197,205)(H4,198,199,206)(H4,200,201,207)(H4,202,203,208)/t93-,94-,95-,96-,97-,98-,99+,111-,112-,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,148-,149-,150-/m0/s1 | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.730 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer Corporation
Curated by PDSP Ki Database
| |
J Biol Chem 271: 26315-9 (1996)
Article DOI: 10.1074/jbc.271.42.26315
BindingDB Entry DOI: 10.7270/Q20G3HPR |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50046121
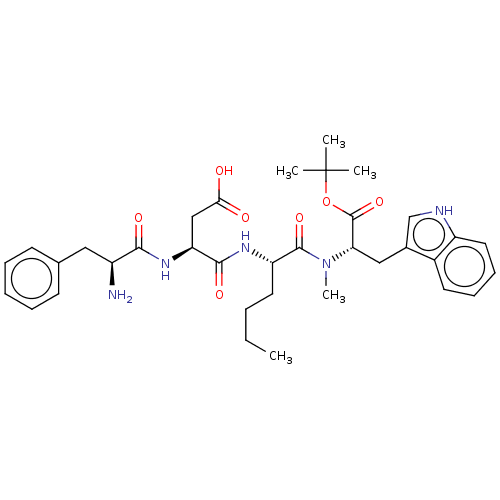
(3-(2-Amino-3-phenyl-propionylamino)-N-(1-{[1-tert-...)Show SMILES CCCC[C@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H](N)Cc1ccccc1)C(=O)N(C)[C@@H](Cc1c[nH]c2ccccc12)C(=O)OC(C)(C)C Show InChI InChI=1S/C35H47N5O7/c1-6-7-16-27(38-32(44)28(20-30(41)42)39-31(43)25(36)18-22-13-9-8-10-14-22)33(45)40(5)29(34(46)47-35(2,3)4)19-23-21-37-26-17-12-11-15-24(23)26/h8-15,17,21,25,27-29,37H,6-7,16,18-20,36H2,1-5H3,(H,38,44)(H,39,43)(H,41,42)/t25-,27-,28-,29-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]pCCK-8 from cholecystokinin type B receptor in guinea pig brain membrane |
J Med Chem 36: 166-72 (1993)
BindingDB Entry DOI: 10.7270/Q26M37FJ |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(Homo sapiens (Human)) | BDBM50056453
(3-(2-{[2-tert-Butoxycarbonylamino-3-(1H-indol-3-yl...)Show SMILES CCCCC(N(C)C(=O)C(Cc1c[nH]c2ccccc12)NC(=O)OC(C)(C)C)C(=O)NC(CC(O)=O)C(=O)NC(Cc1ccccc1)C(N)=O Show InChI InChI=1S/C36H48N6O8/c1-6-7-17-29(33(47)40-27(20-30(43)44)32(46)39-26(31(37)45)18-22-13-9-8-10-14-22)42(5)34(48)28(41-35(49)50-36(2,3)4)19-23-21-38-25-16-12-11-15-24(23)25/h8-16,21,26-29,38H,6-7,17-20H2,1-5H3,(H2,37,45)(H,39,46)(H,40,47)(H,41,49)(H,43,44) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Inhibition of [3H]pCCK-8 binding to Guinea pig cortex membrane Cholecystokinin type B receptor |
J Med Chem 40: 647-58 (1997)
Article DOI: 10.1021/jm9603072
BindingDB Entry DOI: 10.7270/Q2PG1SDX |
More data for this
Ligand-Target Pair | |
Growth hormone secretagogue receptor type 1
(Ovis aries) | BDBM50222867
((S)-6-(4-fluorophenoxy)-3-((1-isopropylpiperidin-3...)Show SMILES CC(C)N1CCC[C@H](Cn2c(nc3ccc(Oc4ccc(F)cc4)cc3c2=O)-c2ccccc2C)C1 Show InChI InChI=1S/C30H32FN3O2/c1-20(2)33-16-6-8-22(18-33)19-34-29(26-9-5-4-7-21(26)3)32-28-15-14-25(17-27(28)30(34)35)36-24-12-10-23(31)11-13-24/h4-5,7,9-15,17,20,22H,6,8,16,18-19H2,1-3H3/t22-/m0/s1 | PDB
KEGG
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.860 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer Pharmaceuticals Corporation
Curated by ChEMBL
| Assay Description
Displacement of [125I]ghrelin from ovine recombinant GHSR1a expressed in HEK293F cells after 6 hrs by scintillation proximity assay |
J Med Chem 50: 5202-16 (2007)
Article DOI: 10.1021/jm070071+
BindingDB Entry DOI: 10.7270/Q2WH2QT2 |
More data for this
Ligand-Target Pair | |
Glutamyl aminopeptidase
(Homo sapiens (Human)) | BDBM50083386

(1-{[1-(2,3-Dicarboxy-pyrrolidine-1-carbonyl)-2-met...)Show SMILES CC[C@@H](C)[C@H](NC(=O)[C@H](S)[C@H]([NH3+])CCS([O-])(=O)=O)C(=O)N1CCC([C@H]1C(O)=O)C(O)=O Show InChI InChI=1S/C17H29N3O9S2/c1-3-8(2)11(19-14(21)13(30)10(18)5-7-31(27,28)29)15(22)20-6-4-9(16(23)24)12(20)17(25)26/h8-13,30H,3-7,18H2,1-2H3,(H,19,21)(H,23,24)(H,25,26)(H,27,28,29)/t8-,9?,10-,11+,12+,13-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.873 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Binding affinity against recombinant Aminopeptidase A |
J Med Chem 42: 5197-211 (2000)
BindingDB Entry DOI: 10.7270/Q2028S8F |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM82276

(L31,P34-NPY,human | NPY Leu31, Pro34, human, rat |...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1c[nH]cn1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](C)NC(=O)[C@H](CCSC)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C188H282N54O56S/c1-15-94(8)148(178(292)231-127(81-139(191)251)165(279)223-122(74-92(4)5)168(282)235-147(93(6)7)177(291)237-149(99(13)245)179(293)220-119(31-21-66-206-188(200)201)183(297)241-70-25-35-137(241)174(288)218-114(30-20-65-205-187(198)199)155(269)221-120(150(193)264)76-101-39-49-107(247)50-40-101)236-169(283)125(79-104-45-55-110(250)56-46-104)226-164(278)126(80-105-86-202-90-208-105)227-157(271)113(29-19-64-204-186(196)197)216-161(275)121(73-91(2)3)222-153(267)96(10)210-170(284)132(88-243)233-163(277)124(78-103-43-53-109(249)54-44-103)225-162(276)123(77-102-41-51-108(248)52-42-102)224-156(270)112(28-18-63-203-185(194)195)214-151(265)95(9)209-154(268)117(61-72-299-14)217-166(280)129(84-145(260)261)229-159(273)116(58-60-143(256)257)215-152(266)97(11)211-173(287)135-33-23-67-238(135)180(294)98(12)212-160(274)128(83-144(258)259)228-158(272)115(57-59-142(254)255)213-141(253)87-207-172(286)134-32-22-69-240(134)184(298)131(82-140(192)252)232-167(281)130(85-146(262)263)230-175(289)138-36-26-71-242(138)182(296)118(27-16-17-62-189)219-171(285)133(89-244)234-176(290)136-34-24-68-239(136)181(295)111(190)75-100-37-47-106(246)48-38-100/h37-56,86,90-99,111-138,147-149,243-250H,15-36,57-85,87-89,189-190H2,1-14H3,(H2,191,251)(H2,192,252)(H2,193,264)(H,202,208)(H,207,286)(H,209,268)(H,210,284)(H,211,287)(H,212,274)(H,213,253)(H,214,265)(H,215,266)(H,216,275)(H,217,280)(H,218,288)(H,219,285)(H,220,293)(H,221,269)(H,222,267)(H,223,279)(H,224,270)(H,225,276)(H,226,278)(H,227,271)(H,228,272)(H,229,273)(H,230,289)(H,231,292)(H,232,281)(H,233,277)(H,234,290)(H,235,282)(H,236,283)(H,237,291)(H,254,255)(H,256,257)(H,258,259)(H,260,261)(H,262,263)(H4,194,195,203)(H4,196,197,204)(H4,198,199,205)(H4,200,201,206)/t94-,95-,96-,97-,98-,99+,111-,112-,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,147-,148-,149-/m0/s1 | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.880 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer Corporation
Curated by PDSP Ki Database
| |
J Biol Chem 271: 26315-9 (1996)
Article DOI: 10.1074/jbc.271.42.26315
BindingDB Entry DOI: 10.7270/Q20G3HPR |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data