 Found 606 hits with Last Name = 'mcallister' and Initial = 'g'
Found 606 hits with Last Name = 'mcallister' and Initial = 'g' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM50571467
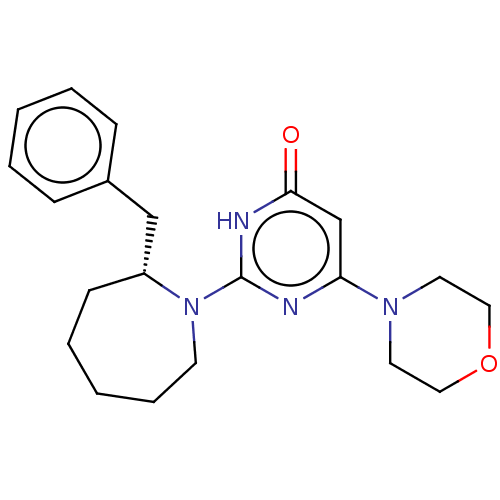
(CHEMBL4850755)Show SMILES O=c1cc(nc([nH]1)N1CCCCC[C@@H]1Cc1ccccc1)N1CCOCC1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant FLAG-tagged human ATM assessed as decrease in p53 S15 phosphorylation using full length myc-tagged p53 as subst... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM82569
(CAS_18426-20-5 | NSC_131212 | R(-)NPA)Show InChI InChI=1S/C19H20FNO2/c1-2-6-21-7-5-12-8-13(20)10-14-17(12)15(21)9-11-3-4-16(22)19(23)18(11)14/h3-4,8,10,15,22-23H,2,5-7,9H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM50020217
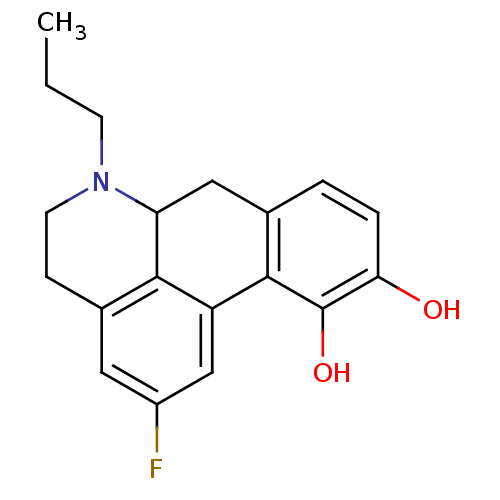
((4aR,10bR)-4-propyl-3,4,4a,5,6,10b-hexahydro-2H-na...)Show InChI InChI=1S/C15H21NO2/c1-2-7-16-8-9-18-15-13-10-12(17)5-3-11(13)4-6-14(15)16/h3,5,10,14-15,17H,2,4,6-9H2,1H3/t14-,15-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM50571471
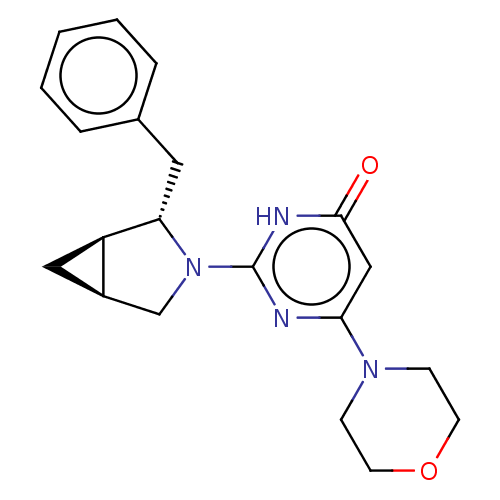
(CHEMBL4858310 | US11685734, Example 213)Show SMILES [H][C@]12C[C@@]1([H])[C@H](Cc1ccccc1)N(C2)c1nc(cc(=O)[nH]1)N1CCOCC1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant FLAG-tagged human ATM assessed as decrease in p53 S15 phosphorylation using full length myc-tagged p53 as subst... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM82569

(CAS_18426-20-5 | NSC_131212 | R(-)NPA)Show InChI InChI=1S/C19H20FNO2/c1-2-6-21-7-5-12-8-13(20)10-14-17(12)15(21)9-11-3-4-16(22)19(23)18(11)14/h3-4,8,10,15,22-23H,2,5-7,9H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM50010289
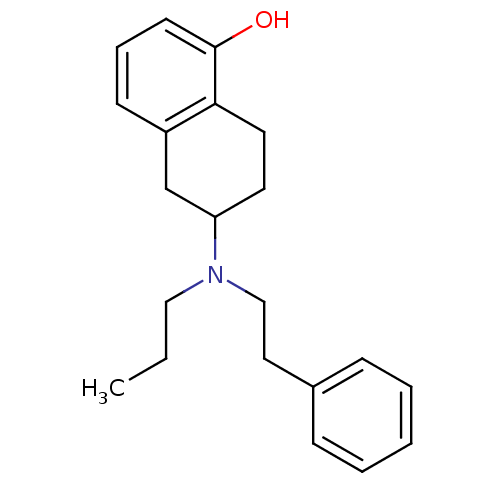
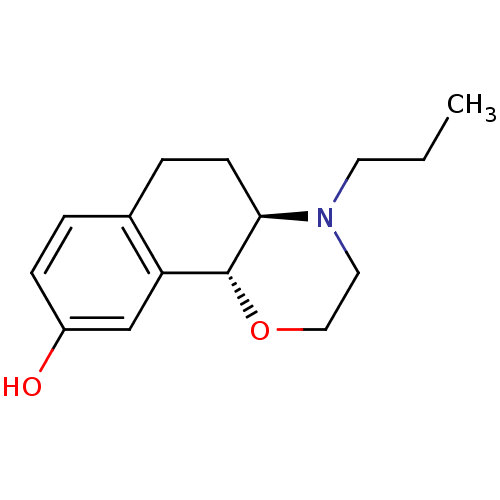
((R)6-(Phenethyl-propyl-amino)-5,6,7,8-tetrahydro-n...)Show InChI InChI=1S/C21H27NO/c1-2-14-22(15-13-17-7-4-3-5-8-17)19-11-12-20-18(16-19)9-6-10-21(20)23/h3-10,19,23H,2,11-16H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM50571463

(CHEMBL4862803)Show SMILES [H][C@@]12C[C@]1([H])[C@H](Cc1ccccc1)N(C2)c1cc(cc(=O)[nH]1)N1CCOCC1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant FLAG-tagged human ATM assessed as decrease in p53 S15 phosphorylation using full length myc-tagged p53 as subst... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM50334150

(1-(1-(4,4-bis(4-fluorophenyl)butyl)piperidin-4-yl)...)Show SMILES Fc1ccc(cc1)C(CCCN1CCC(CC1)n1c2ccccc2[nH]c1=O)c1ccc(F)cc1 Show InChI InChI=1S/C28H29F2N3O/c29-22-11-7-20(8-12-22)25(21-9-13-23(30)14-10-21)4-3-17-32-18-15-24(16-19-32)33-27-6-2-1-5-26(27)31-28(33)34/h1-2,5-14,24-25H,3-4,15-19H2,(H,31,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50334150

(1-(1-(4,4-bis(4-fluorophenyl)butyl)piperidin-4-yl)...)Show SMILES Fc1ccc(cc1)C(CCCN1CCC(CC1)n1c2ccccc2[nH]c1=O)c1ccc(F)cc1 Show InChI InChI=1S/C28H29F2N3O/c29-22-11-7-20(8-12-22)25(21-9-13-23(30)14-10-21)4-3-17-32-18-15-24(16-19-32)33-27-6-2-1-5-26(27)31-28(33)34/h1-2,5-14,24-25H,3-4,15-19H2,(H,31,34) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PubMed
| 0.390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM50571476
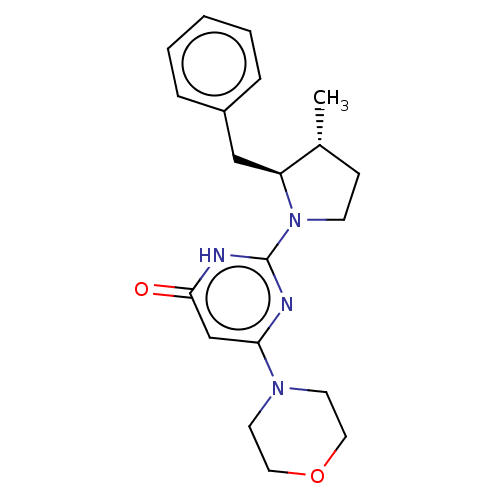
(CHEMBL4852400 | US11685734, Example 215)Show SMILES C[C@@H]1CCN([C@H]1Cc1ccccc1)c1nc(cc(=O)[nH]1)N1CCOCC1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant FLAG-tagged human ATM assessed as decrease in p53 S15 phosphorylation using full length myc-tagged p53 as subst... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50008735
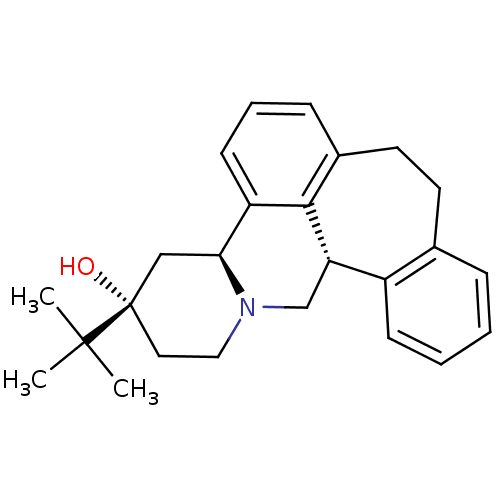
((+)-3-(tert-butyl)-(3S,4aS,13bS)-2,3,4,4a,8,9,13b,...)Show SMILES CC(C)(C)[C@]1(O)CCN2C[C@H]3c4ccccc4CCc4cccc([C@@H]2C1)c34 Show InChI InChI=1S/C25H31NO/c1-24(2,3)25(27)13-14-26-16-21-19-9-5-4-7-17(19)11-12-18-8-6-10-20(23(18)21)22(26)15-25/h4-10,21-22,27H,11-16H2,1-3H3/t21-,22-,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.440 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM50550657
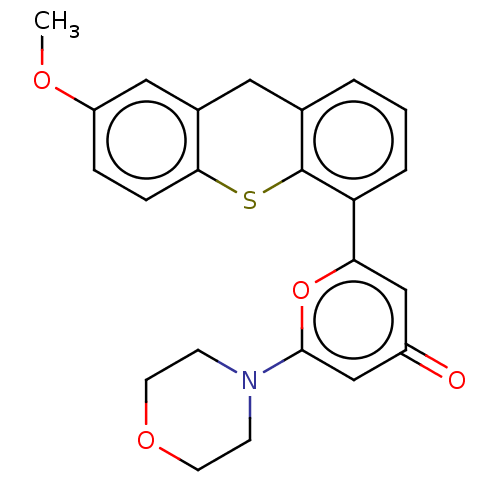
(CHEMBL4792737)Show SMILES COc1ccc2Sc3c(Cc2c1)cccc3-c1cc(=O)cc(o1)N1CCOCC1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant FLAG-tagged human ATM assessed as decrease in p53 S15 phosphorylation using full length myc-tagged p53 as subst... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50241107
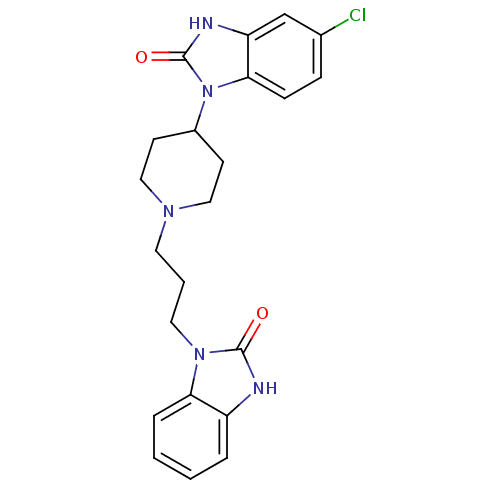
(1-(3-(4-(5-chloro-2-oxo-2,3-dihydrobenzo[d]imidazo...)Show SMILES Clc1ccc2n(C3CCN(CCCn4c5ccccc5[nH]c4=O)CC3)c(=O)[nH]c2c1 Show InChI InChI=1S/C22H24ClN5O2/c23-15-6-7-20-18(14-15)25-22(30)28(20)16-8-12-26(13-9-16)10-3-11-27-19-5-2-1-4-17(19)24-21(27)29/h1-2,4-7,14,16H,3,8-13H2,(H,24,29)(H,25,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
PubMed
| 0.580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM50571474
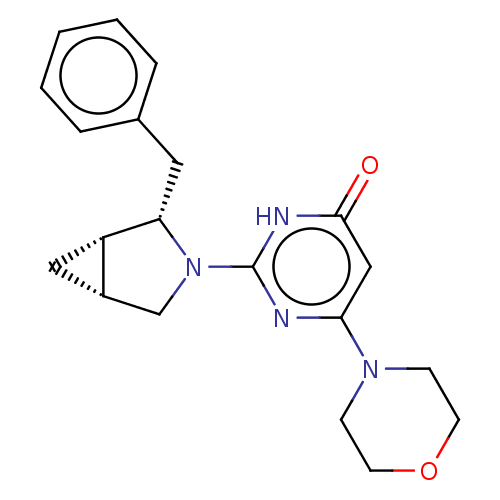
(CHEMBL4857803 | US11685734, Example 211)Show SMILES [H][C@@]12C[C@]1([H])[C@H](Cc1ccccc1)N(C2)c1nc(cc(=O)[nH]1)N1CCOCC1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant FLAG-tagged human ATM assessed as decrease in p53 S15 phosphorylation using full length myc-tagged p53 as subst... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM81993
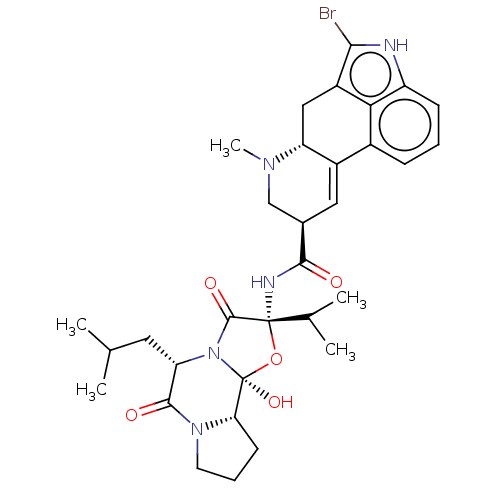
(BROMOCRIPTINE | Bromocriptine+ (GTP+) | Bromocript...)Show SMILES CC(C)C[C@@H]1N2C(=O)[C@](NC(=O)[C@H]3CN(C)[C@@H]4Cc5c(Br)[nH]c6cccc(C4=C3)c56)(O[C@@]2(O)[C@@H]2CCCN2C1=O)C(C)C |r,c:28| Show InChI InChI=1S/C32H40BrN5O5/c1-16(2)12-24-29(40)37-11-7-10-25(37)32(42)38(24)30(41)31(43-32,17(3)4)35-28(39)18-13-20-19-8-6-9-22-26(19)21(27(33)34-22)14-23(20)36(5)15-18/h6,8-9,13,16-18,23-25,34,42H,7,10-12,14-15H2,1-5H3,(H,35,39)/t18-,23-,24+,25+,31-,32+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| DrugBank
PDB
PubMed
| 0.620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM85091

((+/-)-2-DIPROPYLAMINO- 6,7-DIHYDROXY-1,2,3,4-TETRA...)Show InChI InChI=1S/C16H25NO2/c1-3-7-17(8-4-2)14-6-5-12-10-15(18)16(19)11-13(12)9-14/h10-11,14,18-19H,3-9H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.680 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM50571475
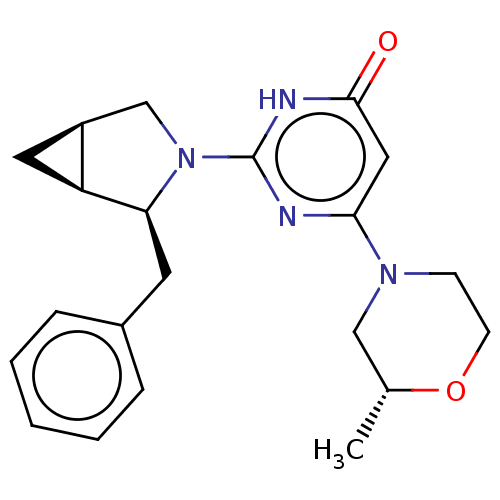
(CHEMBL4853948 | US11685734, Example 292)Show SMILES [H][C@@]12C[C@]1([H])[C@H](Cc1ccccc1)N(C2)c1nc(cc(=O)[nH]1)N1CCO[C@H](C)C1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant FLAG-tagged human ATM assessed as decrease in p53 S15 phosphorylation using full length myc-tagged p53 as subst... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM50571466
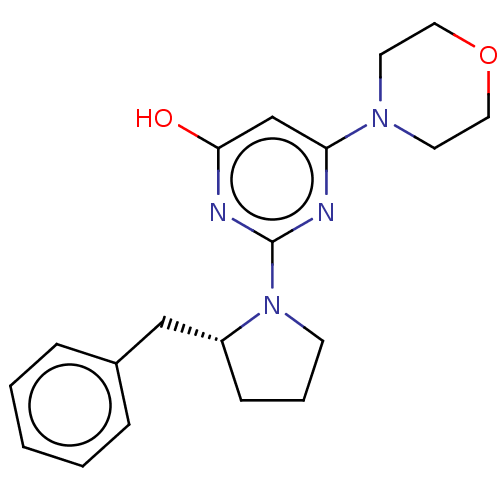
(CHEMBL4878312)Show SMILES Oc1cc(nc(n1)N1CCC[C@@H]1Cc1ccccc1)N1CCOCC1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant FLAG-tagged human ATM assessed as decrease in p53 S15 phosphorylation using full length myc-tagged p53 as subst... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM50571459
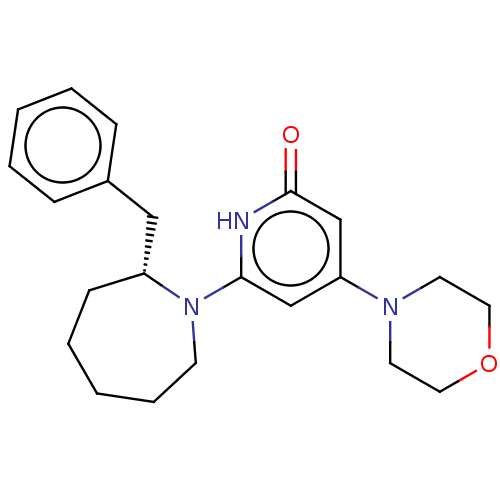
(CHEMBL4872830)Show SMILES O=c1cc(cc([nH]1)N1CCCCC[C@@H]1Cc1ccccc1)N1CCOCC1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant FLAG-tagged human ATM assessed as decrease in p53 S15 phosphorylation using full length myc-tagged p53 as subst... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM50571468
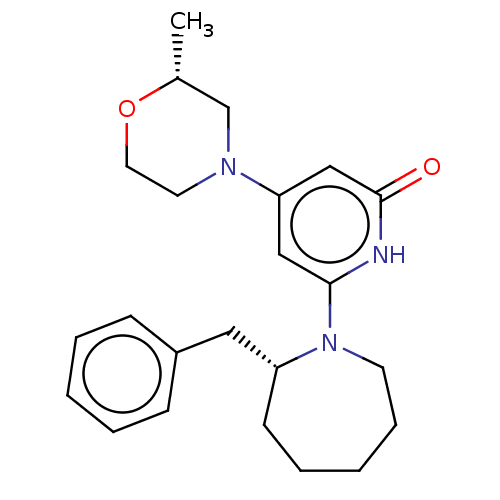
(CHEMBL4845919)Show SMILES C[C@@H]1CN(CCO1)c1cc([nH]c(=O)c1)N1CCCCC[C@@H]1Cc1ccccc1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant FLAG-tagged human ATM assessed as decrease in p53 S15 phosphorylation using full length myc-tagged p53 as subst... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM50571469
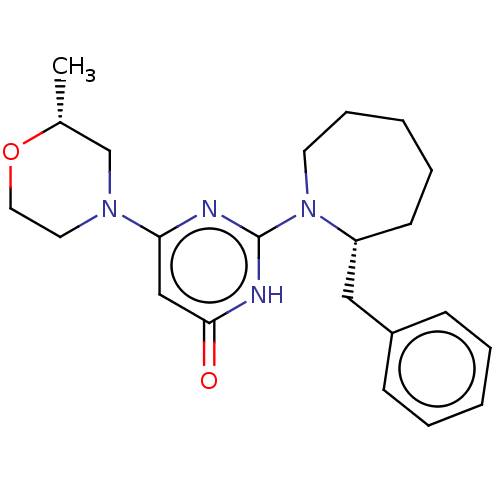
(CHEMBL4852763 | US11685734, Example 270)Show SMILES C[C@@H]1CN(CCO1)c1cc(=O)[nH]c(n1)N1CCCCC[C@@H]1Cc1ccccc1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant FLAG-tagged human ATM assessed as decrease in p53 S15 phosphorylation using full length myc-tagged p53 as subst... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM50026957
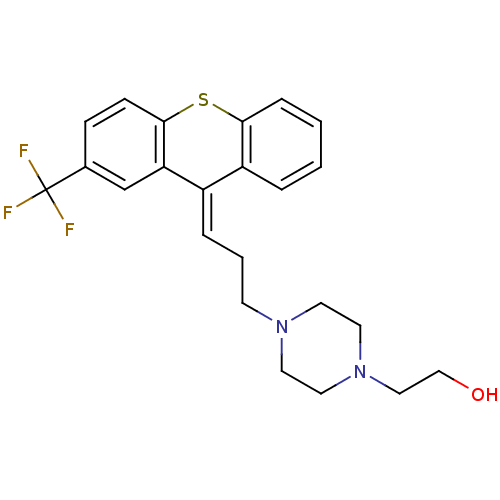
((cis) 2-{4-[3-(2-Trifluoromethyl-thioxanthen-9-yli...)Show SMILES OCCN1CCN(CC\C=C2/c3ccccc3Sc3ccc(cc23)C(F)(F)F)CC1 Show InChI InChI=1S/C23H25F3N2OS/c24-23(25,26)17-7-8-22-20(16-17)18(19-4-1-2-6-21(19)30-22)5-3-9-27-10-12-28(13-11-27)14-15-29/h1-2,4-8,16,29H,3,9-15H2/b18-5+ | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM50106269
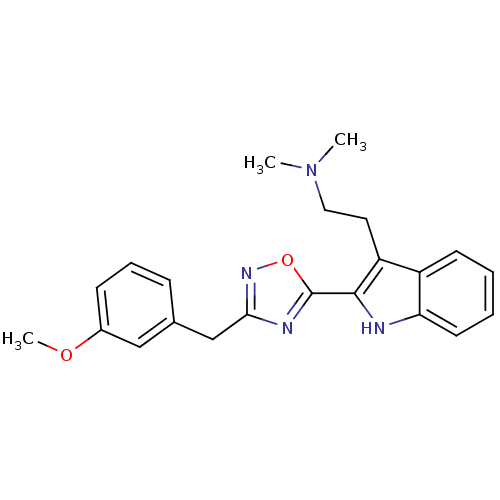
((2-{2-[3-(3-Methoxy-benzyl)-[1,2,4]oxadiazol-5-yl]...)Show SMILES COc1cccc(Cc2noc(n2)-c2[nH]c3ccccc3c2CCN(C)C)c1 Show InChI InChI=1S/C22H24N4O2/c1-26(2)12-11-18-17-9-4-5-10-19(17)23-21(18)22-24-20(25-28-22)14-15-7-6-8-16(13-15)27-3/h4-10,13,23H,11-12,14H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity as displacement of [3H]-5-HT binding to 5-hydroxytryptamine 6 receptor in HeLa cells. |
J Med Chem 44: 3881-95 (2001)
BindingDB Entry DOI: 10.7270/Q2T72GRW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM50106249
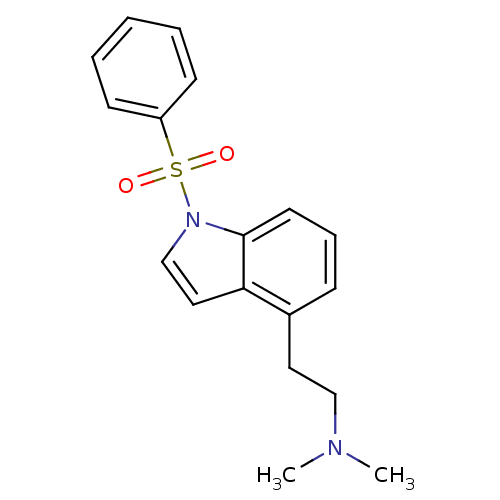
(CHEMBL92139 | [2-(1-Benzenesulfonyl-1H-indol-4-yl)...)Show InChI InChI=1S/C18H20N2O2S/c1-19(2)13-11-15-7-6-10-18-17(15)12-14-20(18)23(21,22)16-8-4-3-5-9-16/h3-10,12,14H,11,13H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity as displacement of [3H]-5-HT binding to 5-hydroxytryptamine 6 receptor in HeLa cells. |
J Med Chem 44: 3881-95 (2001)
BindingDB Entry DOI: 10.7270/Q2T72GRW |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM21398

(4-[4-(4-Chloro-phenyl)-4-hydroxy-piperidin-1-yl]-1...)Show SMILES OC1(CCN(CCCC(=O)c2ccc(F)cc2)CC1)c1ccc(Cl)cc1 Show InChI InChI=1S/C21H23ClFNO2/c22-18-7-5-17(6-8-18)21(26)11-14-24(15-12-21)13-1-2-20(25)16-3-9-19(23)10-4-16/h3-10,26H,1-2,11-15H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM82567
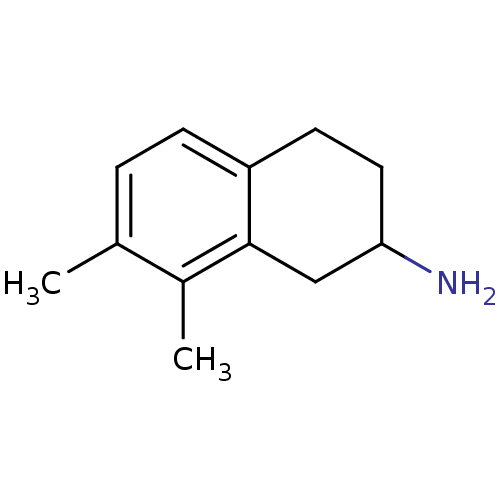
(ADTN DIMETHYL 6,7)Show InChI InChI=1S/C12H17N/c1-8-3-4-10-5-6-11(13)7-12(10)9(8)2/h3-4,11H,5-7,13H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50026957

((cis) 2-{4-[3-(2-Trifluoromethyl-thioxanthen-9-yli...)Show SMILES OCCN1CCN(CC\C=C2/c3ccccc3Sc3ccc(cc23)C(F)(F)F)CC1 Show InChI InChI=1S/C23H25F3N2OS/c24-23(25,26)17-7-8-22-20(16-17)18(19-4-1-2-6-21(19)30-22)5-3-9-27-10-12-28(13-11-27)14-15-29/h1-2,4-8,16,29H,3,9-15H2/b18-5+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM50020222
((+/-)-7-(dipropylamino)-5,6,7,8-tetrahydronaphthal...)Show InChI InChI=1S/C16H25NO/c1-3-9-17(10-4-2)15-7-5-13-6-8-16(18)12-14(13)11-15/h6,8,12,15,18H,3-5,7,9-11H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM82566
(ADTN 6,7)Show SMILES NC1CCc2ccc(OC(=O)c3ccccc3)c(OC(=O)c3ccccc3)c2C1 Show InChI InChI=1S/C24H21NO4/c25-19-13-11-16-12-14-21(28-23(26)17-7-3-1-4-8-17)22(20(16)15-19)29-24(27)18-9-5-2-6-10-18/h1-10,12,14,19H,11,13,15,25H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM50571464
(CHEMBL4874640 | US11685734, Example 43)Show SMILES [H][C@]12C[C@@]1([H])[C@H](Cc1ccccc1)N(C2)c1cc(cc(=O)[nH]1)N1CCOCC1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant FLAG-tagged human ATM assessed as decrease in p53 S15 phosphorylation using full length myc-tagged p53 as subst... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM50571472

(CHEMBL4867495 | US11685734, Example 291)Show SMILES [H][C@]12C[C@@]1([H])[C@H](Cc1ccccc1)N(C2)c1nc(cc(=O)[nH]1)N1CCO[C@H](C)C1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant FLAG-tagged human ATM assessed as decrease in p53 S15 phosphorylation using full length myc-tagged p53 as subst... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM81993

(BROMOCRIPTINE | Bromocriptine+ (GTP+) | Bromocript...)Show SMILES CC(C)C[C@@H]1N2C(=O)[C@](NC(=O)[C@H]3CN(C)[C@@H]4Cc5c(Br)[nH]c6cccc(C4=C3)c56)(O[C@@]2(O)[C@@H]2CCCN2C1=O)C(C)C |r,c:28| Show InChI InChI=1S/C32H40BrN5O5/c1-16(2)12-24-29(40)37-11-7-10-25(37)32(42)38(24)30(41)31(43-32,17(3)4)35-28(39)18-13-20-19-8-6-9-22-26(19)21(27(33)34-22)14-23(20)36(5)15-18/h6,8-9,13,16-18,23-25,34,42H,7,10-12,14-15H2,1-5H3,(H,35,39)/t18-,23-,24+,25+,31-,32+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM50008735

((+)-3-(tert-butyl)-(3S,4aS,13bS)-2,3,4,4a,8,9,13b,...)Show SMILES CC(C)(C)[C@]1(O)CCN2C[C@H]3c4ccccc4CCc4cccc([C@@H]2C1)c34 Show InChI InChI=1S/C25H31NO/c1-24(2,3)25(27)13-14-26-16-21-19-9-5-4-7-17(19)11-12-18-8-6-10-20(23(18)21)22(26)15-25/h4-10,21-22,27H,11-16H2,1-3H3/t21-,22-,25-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM34141
(CHEMBL76237 | MS-245)Show SMILES COc1ccc2n(cc(CCN(C)C)c2c1)S(=O)(=O)c1ccccc1 Show InChI InChI=1S/C19H22N2O3S/c1-20(2)12-11-15-14-21(19-10-9-16(24-3)13-18(15)19)25(22,23)17-7-5-4-6-8-17/h4-10,13-14H,11-12H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity as displacement of [3H]-5-HT binding to 5-hydroxytryptamine 6 receptor in HeLa cells. |
J Med Chem 44: 3881-95 (2001)
BindingDB Entry DOI: 10.7270/Q2T72GRW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM50106251
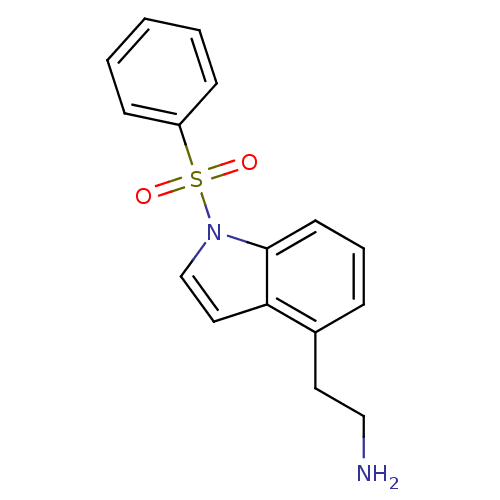
(2-(1-Benzenesulfonyl-1H-indol-4-yl)-ethylamine | C...)Show InChI InChI=1S/C16H16N2O2S/c17-11-9-13-5-4-8-16-15(13)10-12-18(16)21(19,20)14-6-2-1-3-7-14/h1-8,10,12H,9,11,17H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity as displacement of [3H]-5-HT binding to 5-hydroxytryptamine 6 receptor in HeLa cells. |
J Med Chem 44: 3881-95 (2001)
BindingDB Entry DOI: 10.7270/Q2T72GRW |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM21398

(4-[4-(4-Chloro-phenyl)-4-hydroxy-piperidin-1-yl]-1...)Show SMILES OC1(CCN(CCCC(=O)c2ccc(F)cc2)CC1)c1ccc(Cl)cc1 Show InChI InChI=1S/C21H23ClFNO2/c22-18-7-5-17(6-8-18)21(26)11-14-24(15-12-21)13-1-2-20(25)16-3-9-19(23)10-4-16/h3-10,26H,1-2,11-15H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PubMed
| 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM50106250
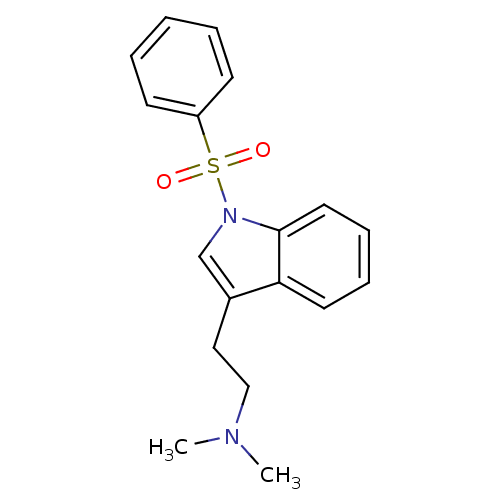
(CHEMBL93868 | N,N-dimethyl-2-(1-(phenylsulfonyl)-1...)Show InChI InChI=1S/C18H20N2O2S/c1-19(2)13-12-15-14-20(18-11-7-6-10-17(15)18)23(21,22)16-8-4-3-5-9-16/h3-11,14H,12-13H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity as displacement of [3H]-5-HT binding to 5-hydroxytryptamine 6 receptor in HeLa cells. |
J Med Chem 44: 3881-95 (2001)
BindingDB Entry DOI: 10.7270/Q2T72GRW |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM50241107

(1-(3-(4-(5-chloro-2-oxo-2,3-dihydrobenzo[d]imidazo...)Show SMILES Clc1ccc2n(C3CCN(CCCn4c5ccccc5[nH]c4=O)CC3)c(=O)[nH]c2c1 Show InChI InChI=1S/C22H24ClN5O2/c23-15-6-7-20-18(14-15)25-22(30)28(20)16-8-12-26(13-9-16)10-3-11-27-19-5-2-1-4-17(19)24-21(27)29/h1-2,4-7,14,16H,3,8-13H2,(H,24,29)(H,25,30) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM50571461
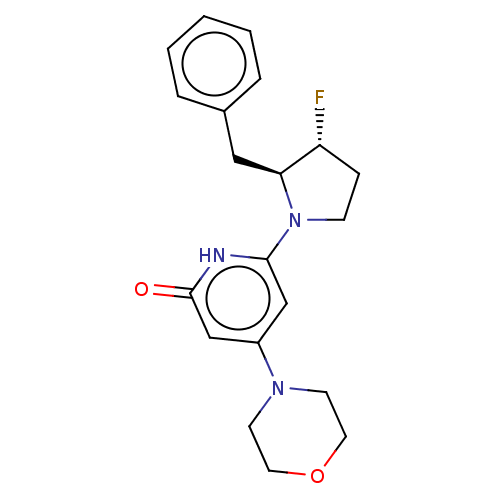
(CHEMBL4866506 | US11685734, Example 96)Show SMILES F[C@@H]1CCN([C@H]1Cc1ccccc1)c1cc(cc(=O)[nH]1)N1CCOCC1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant FLAG-tagged human ATM assessed as decrease in p53 S15 phosphorylation using full length myc-tagged p53 as subst... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM50106246

(CHEMBL126340 | [1-Benzenesulfonyl-4-(2-dimethylami...)Show SMILES CN(C)CCc1cccc2n(c(cc12)C(=O)c1ccccc1)S(=O)(=O)c1ccccc1 Show InChI InChI=1S/C25H24N2O3S/c1-26(2)17-16-19-12-9-15-23-22(19)18-24(25(28)20-10-5-3-6-11-20)27(23)31(29,30)21-13-7-4-8-14-21/h3-15,18H,16-17H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity as displacement of [3H]-5-HT binding to 5-hydroxytryptamine 6 receptor in HeLa cells. |
J Med Chem 44: 3881-95 (2001)
BindingDB Entry DOI: 10.7270/Q2T72GRW |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM50001888

((chloropromazine) [3-(2-Chloro-phenothiazin-10-yl)...)Show InChI InChI=1S/C17H19ClN2S/c1-19(2)10-5-11-20-14-6-3-4-7-16(14)21-17-9-8-13(18)12-15(17)20/h3-4,6-9,12H,5,10-11H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1F
(Homo sapiens (Human)) | BDBM50106249

(CHEMBL92139 | [2-(1-Benzenesulfonyl-1H-indol-4-yl)...)Show InChI InChI=1S/C18H20N2O2S/c1-19(2)13-11-15-7-6-10-18-17(15)12-14-20(18)23(21,22)16-8-4-3-5-9-16/h3-10,12,14H,11,13H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity against 5-hydroxytryptamine 1F receptor in CHO cells using [3H]-5-HT radioligand |
J Med Chem 44: 3881-95 (2001)
BindingDB Entry DOI: 10.7270/Q2T72GRW |
More data for this
Ligand-Target Pair | |
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM50571473

(CHEMBL4856420 | US11685734, Example 39)Show SMILES [H][C@@]12C[C@]1([H])[C@H](Cc1ccccc1)N(C2)c1cc(cc(=O)[nH]1)N1CCO[C@H](C)C1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant FLAG-tagged human ATM assessed as decrease in p53 S15 phosphorylation using full length myc-tagged p53 as subst... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase catalytic subunit type 3
(Homo sapiens (Human)) | BDBM50571463

(CHEMBL4862803)Show SMILES [H][C@@]12C[C@]1([H])[C@H](Cc1ccccc1)N(C2)c1cc(cc(=O)[nH]1)N1CCOCC1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant GST-tagged human VPS34 expressed in baculovirus expression system using PI/PS as substrate incubated for 60 min... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
Serine-protein kinase ATM
(Homo sapiens (Human)) | BDBM50571446

(CHEMBL4879094)Show SMILES COc1ccc2Sc3c(Cc2c1)cccc3-c1cc(cc(=O)[nH]1)N1CCOCC1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant FLAG-tagged human ATM assessed as decrease in p53 S15 phosphorylation using full length myc-tagged p53 as subst... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1D
(Homo sapiens (Human)) | BDBM50106251

(2-(1-Benzenesulfonyl-1H-indol-4-yl)-ethylamine | C...)Show InChI InChI=1S/C16H16N2O2S/c17-11-9-13-5-4-8-16-15(13)10-12-18(16)21(19,20)14-6-2-1-3-7-14/h1-8,10,12H,9,11,17H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 4.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity against 5-hydroxytryptamine 1D receptor in CHO cells using [3H]-5-HT as radioligand |
J Med Chem 44: 3881-95 (2001)
BindingDB Entry DOI: 10.7270/Q2T72GRW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Rattus norvegicus (rat)) | BDBM50106251

(2-(1-Benzenesulfonyl-1H-indol-4-yl)-ethylamine | C...)Show InChI InChI=1S/C16H16N2O2S/c17-11-9-13-5-4-8-16-15(13)10-12-18(16)21(19,20)14-6-2-1-3-7-14/h1-8,10,12H,9,11,17H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 4.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity for 5-hydroxytryptamine 2 receptor of rat cortical membrane using [3H]-DOB radioligand |
J Med Chem 44: 3881-95 (2001)
BindingDB Entry DOI: 10.7270/Q2T72GRW |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Homo sapiens (Human)) | BDBM81792
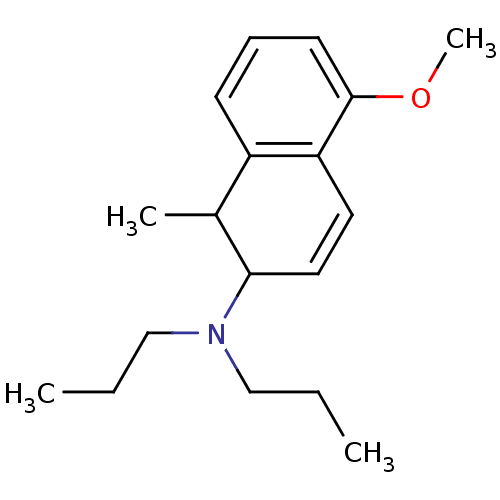
(CAS_5311190 | NSC_5311190 | UH232 | UH232(+))Show InChI InChI=1S/C18H27NO/c1-5-12-19(13-6-2)17-11-10-16-15(14(17)3)8-7-9-18(16)20-4/h7-11,14,17H,5-6,12-13H2,1-4H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| PubMed
| 4.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase catalytic subunit type 3
(Homo sapiens (Human)) | BDBM50571474

(CHEMBL4857803 | US11685734, Example 211)Show SMILES [H][C@@]12C[C@]1([H])[C@H](Cc1ccccc1)N(C2)c1nc(cc(=O)[nH]1)N1CCOCC1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of full length recombinant GST-tagged human VPS34 expressed in baculovirus expression system using PI/PS as substrate incubated for 60 min... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00114
BindingDB Entry DOI: 10.7270/Q2F76HB1 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50010289

((R)6-(Phenethyl-propyl-amino)-5,6,7,8-tetrahydro-n...)Show InChI InChI=1S/C21H27NO/c1-2-14-22(15-13-17-7-4-3-5-8-17)19-11-12-20-18(16-19)9-6-10-21(20)23/h3-10,19,23H,2,11-16H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 5.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neuroscience Research Centre
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 268: 417-26 (1994)
BindingDB Entry DOI: 10.7270/Q2C827SR |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data