

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123658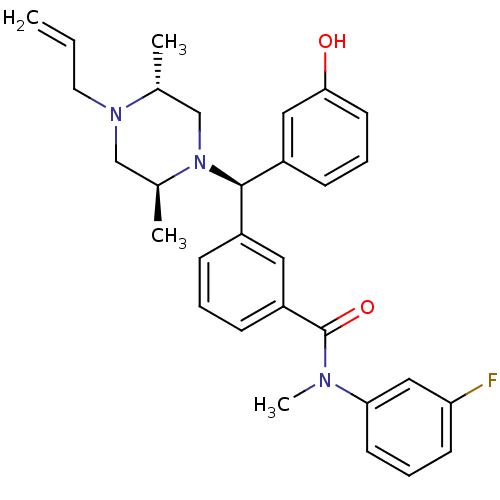 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor delta 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123658 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor delta 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123658 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 0.460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor mu 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123658 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 0.460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor mu 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471981 (CHEMBL446629) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.692 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471983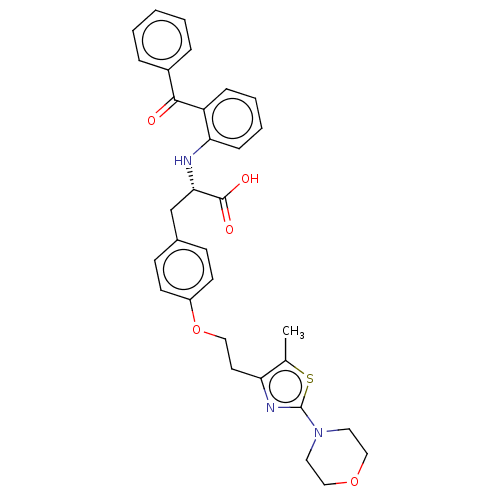 (CHEMBL149876) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.776 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471980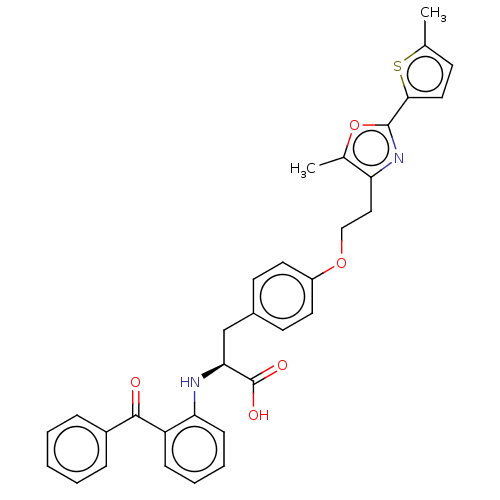 (CHEMBL147095) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.851 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471978 (CHEMBL147090) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.871 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471951 (CHEMBL343210) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.891 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471959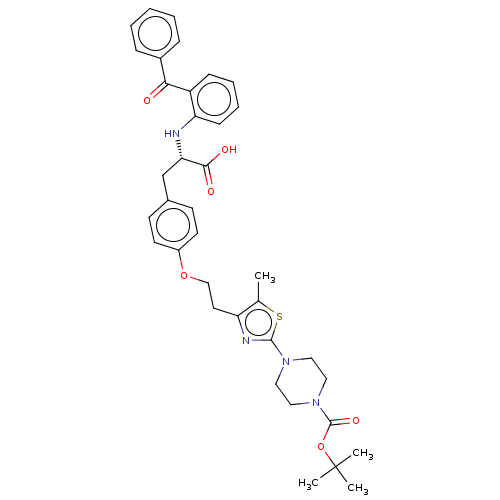 (CHEMBL146301) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.977 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50000092 ((-)-(etorphine) | (-)-morphine | (1S,5R,13R,14S)-1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor mu 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123655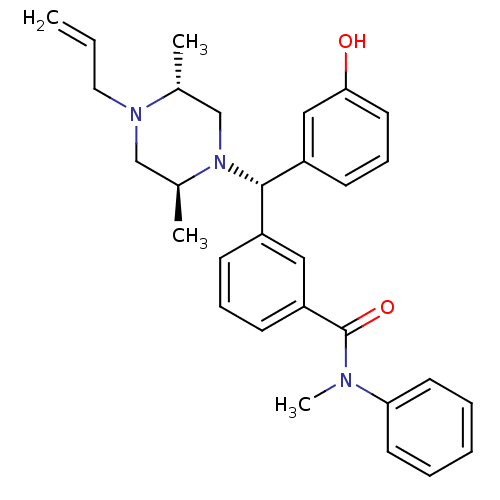 (3-[(S)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.06 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor delta 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123647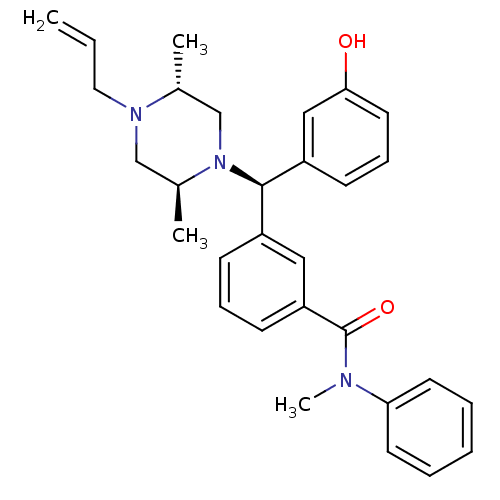 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.06 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor mu 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471953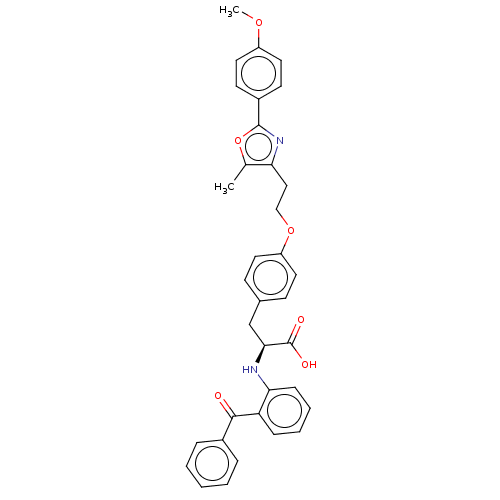 (CHEMBL356382) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471968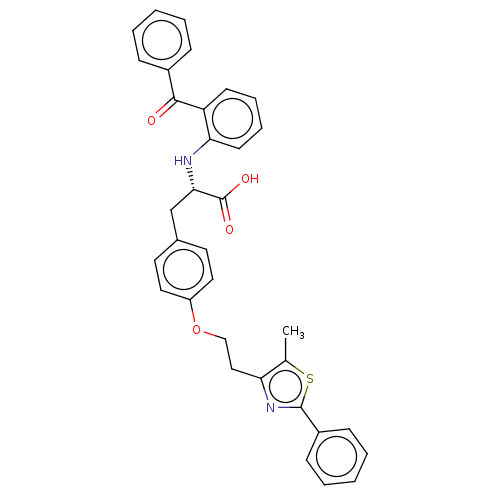 (CHEMBL358379) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50085044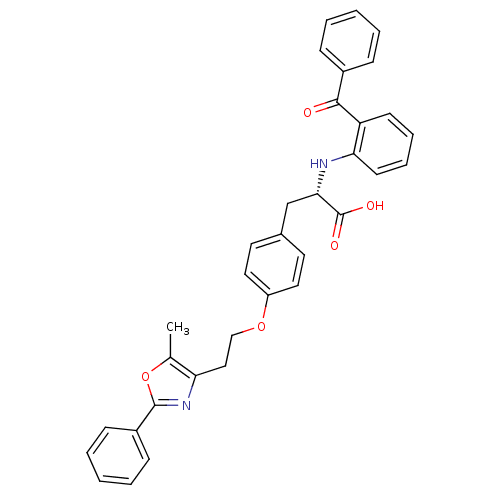 ((S)-2-(2-Benzoyl-phenylamino)-3-{4-[2-(5-methyl-2-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123654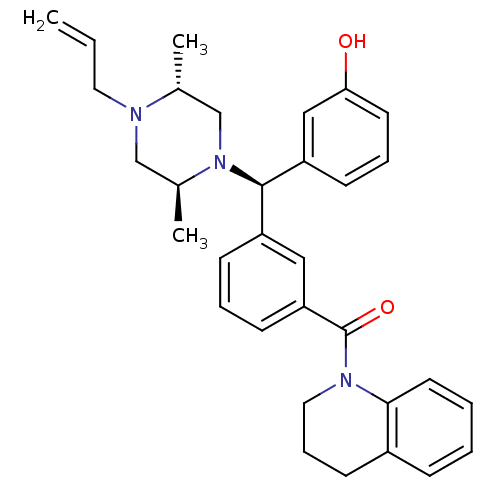 (CHEMBL157660 | {3-[(R)-((2S,5R)-4-Allyl-2,5-dimeth...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor delta 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123664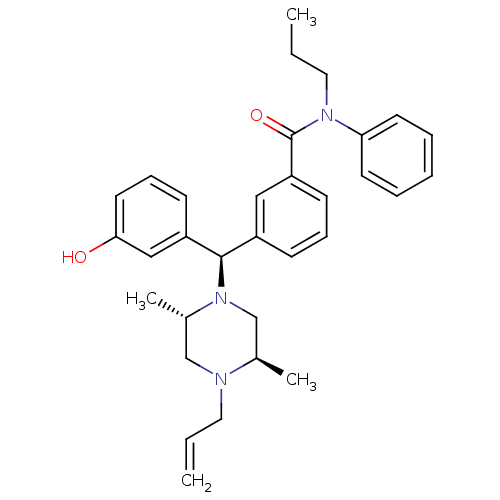 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.26 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor delta 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471966 (CHEMBL147935) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123647 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor delta 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471974 (CHEMBL148950) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123649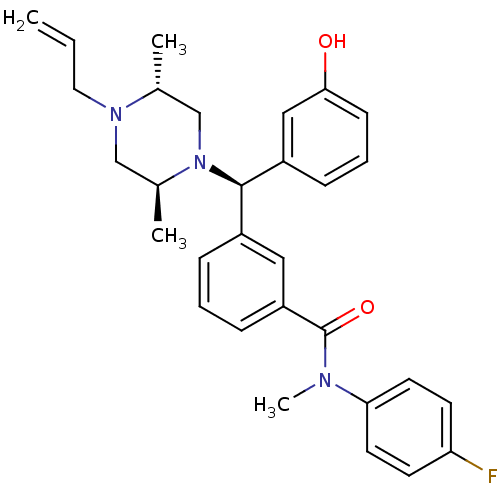 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor delta 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123649 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.49 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor mu 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50008984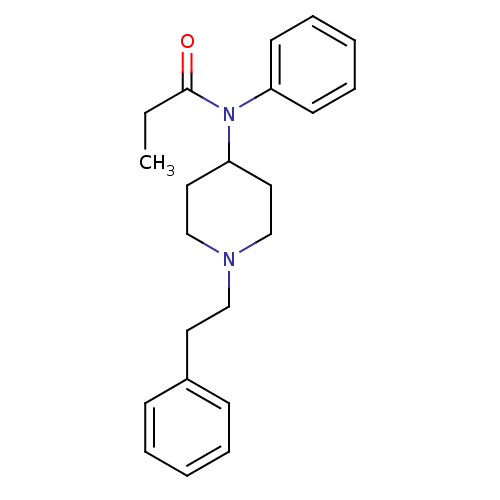 (4-(4-Chloro-benzyl)-2-(1-methyl-azepan-4-yl)-2H-ph...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor mu 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123655 (3-[(S)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor mu 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123665 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.58 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor mu 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471982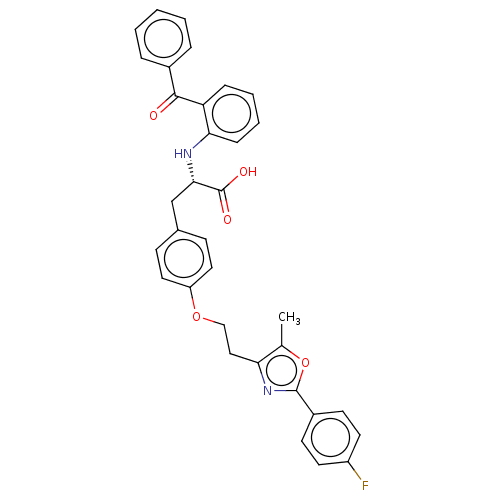 (CHEMBL147384) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123665 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.69 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor delta 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123662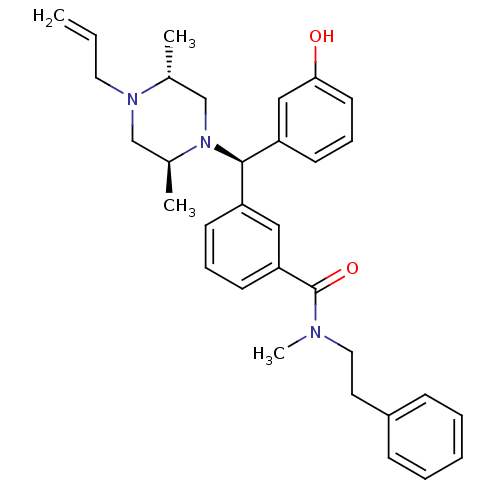 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor delta 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471955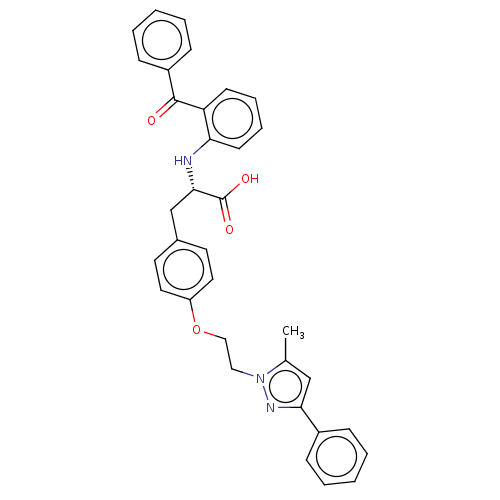 (CHEMBL148797) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50039026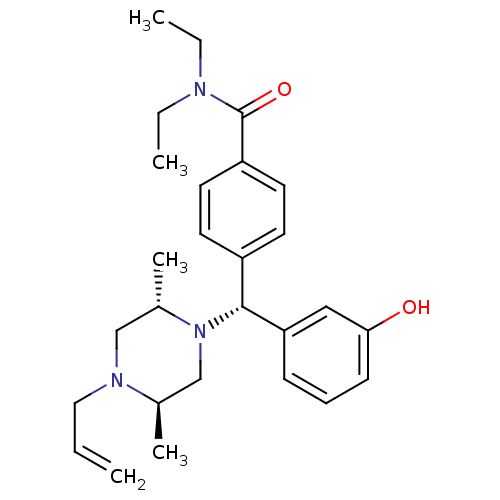 (4-((R)-((2S,5R)-4-allyl-2,5-dimethylpiperazin-1-yl...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor delta 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123664 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.82 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor mu 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123657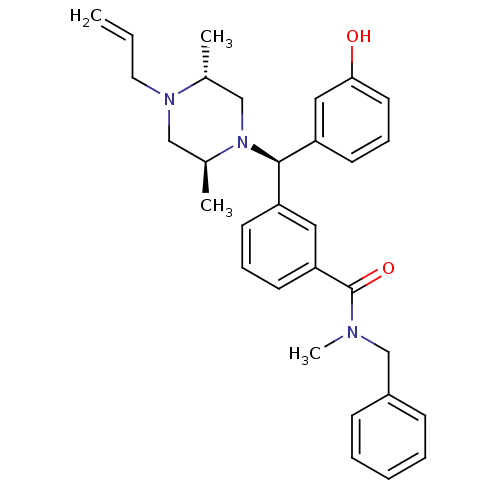 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.88 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor delta 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471970 (CHEMBL148459) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123660 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.92 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor delta 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471948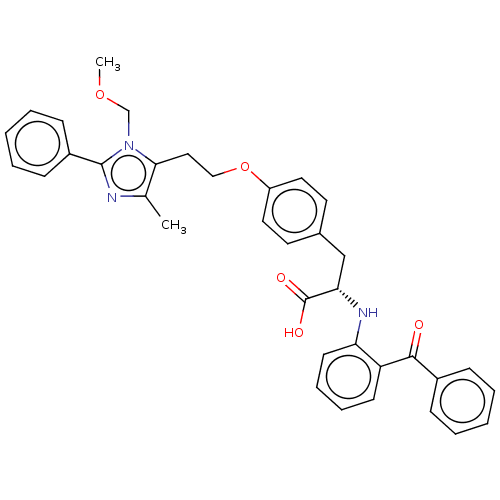 (CHEMBL149647) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123656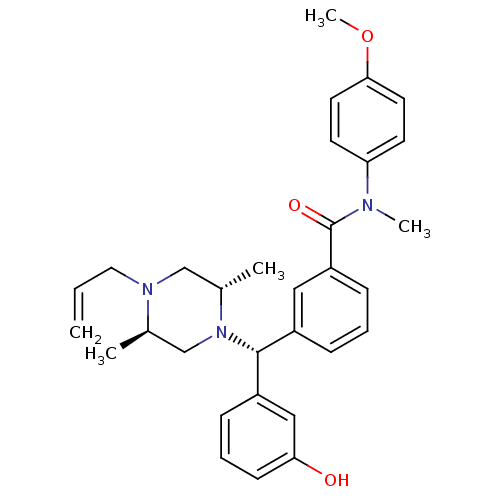 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.01 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor delta 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123652 (3-[(S)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor delta 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123657 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor mu 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123650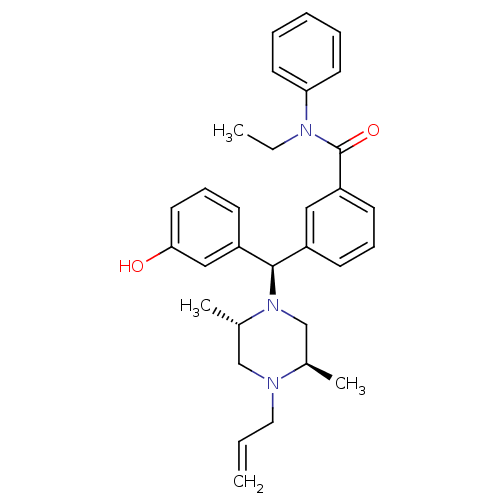 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor delta 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123662 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor mu 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471954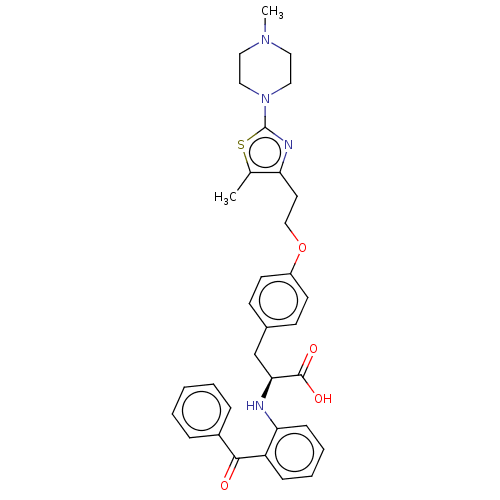 (CHEMBL358325) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123648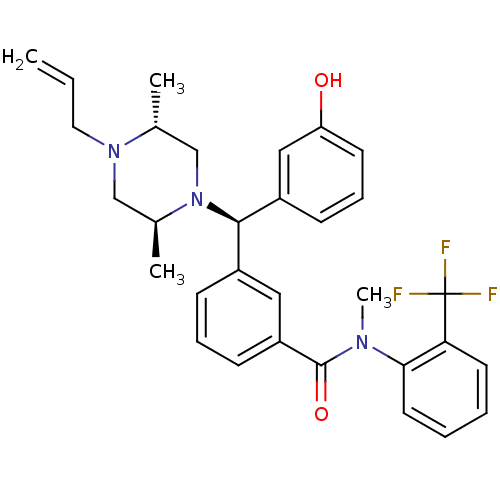 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.31 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor delta 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123656 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.35 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor mu 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123660 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor mu 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123653 (3-[(R)-((2S,5R)-4-Allyl-2,5-dimethyl-piperazin-1-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.41 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor delta 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50123654 (CHEMBL157660 | {3-[(R)-((2S,5R)-4-Allyl-2,5-dimeth...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity of compound evaluated for Opioid receptor mu 1 isolated from rat brain | J Med Chem 46: 623-33 (2003) Article DOI: 10.1021/jm020395s BindingDB Entry DOI: 10.7270/Q2DZ0915 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471989 (CHEMBL149835) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471963 (CHEMBL359285) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Peroxisome proliferator-activated receptor gamma (Homo sapiens (Human)) | BDBM50471991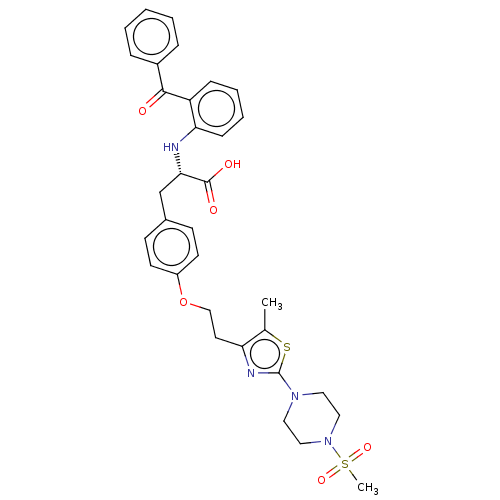 (CHEMBL146650) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition by 50% of in vitro binding to Peroxisome proliferator activated receptor gamma | J Med Chem 41: 5037-54 (1998) Article DOI: 10.1021/jm980413z BindingDB Entry DOI: 10.7270/Q2J38W9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 276 total ) | Next | Last >> |