 Found 31 hits with Last Name = 'sashidhara' and Initial = 'kv'
Found 31 hits with Last Name = 'sashidhara' and Initial = 'kv' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM50295172
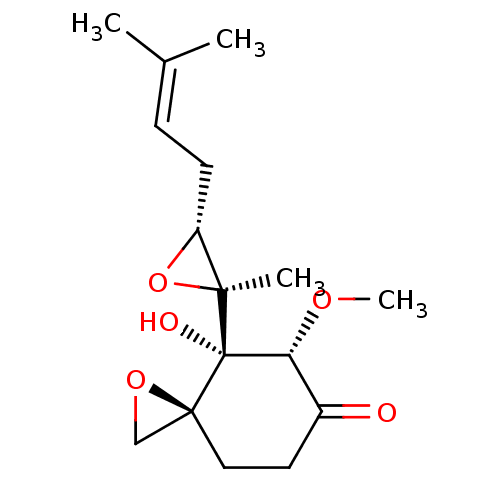
(4-Hydroxy-5-methoxy-4-[(2S,3R)-2-methyl-3-(3-methy...)Show SMILES [#6]-[#8]-[#6@@H]1-[#6](=O)-[#6]-[#6][C@]2([#6]-[#8]2)[C@@]1([#8])[C@@]1([#6])[#8]-[#6@@H]1-[#6]\[#6]=[#6](\[#6])-[#6] |r| Show InChI InChI=1S/C16H24O5/c1-10(2)5-6-12-14(3,21-12)16(18)13(19-4)11(17)7-8-15(16)9-20-15/h5,12-13,18H,6-9H2,1-4H3/t12-,13-,14+,15+,16+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
University of California Santa Cruz
Curated by ChEMBL
| Assay Description
Inhibition of methionine aminopeptidase 2 |
J Nat Prod 72: 588-603 (2010)
Article DOI: 10.1021/np800817y
BindingDB Entry DOI: 10.7270/Q2765G84 |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM17446

((3R,4S,5S,6R)-5-methoxy-4-[(2R,3R)-2-methyl-3-(3-m...)Show SMILES [H][C@@]1([#6@H](-[#8]-[#6])-[#6@@H](-[#6]-[#6][C@]11[#6]-[#8]1)-[#8]-[#6](=O)-[#7]-[#6](=O)-[#6]Cl)[C@@]1([#6])[#8]-[#6@@H]1-[#6]\[#6]=[#6](\[#6])-[#6] |r| Show InChI InChI=1S/C19H28ClNO6/c1-11(2)5-6-13-18(3,27-13)16-15(24-4)12(7-8-19(16)10-25-19)26-17(23)21-14(22)9-20/h5,12-13,15-16H,6-10H2,1-4H3,(H,21,22,23)/t12-,13-,15-,16-,18+,19+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
University of California Santa Cruz
Curated by ChEMBL
| Assay Description
Inhibition of methionine aminopeptidase 2 |
J Nat Prod 72: 588-603 (2010)
Article DOI: 10.1021/np800817y
BindingDB Entry DOI: 10.7270/Q2765G84 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Transient receptor potential cation channel subfamily V member 1
(Rattus norvegicus (rat)) | BDBM50248768

(1,3-dibenzyl urea | CHEMBL504463)Show InChI InChI=1S/C15H16N2O/c18-15(16-11-13-7-3-1-4-8-13)17-12-14-9-5-2-6-10-14/h1-10H,11-12H2,(H2,16,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Antagonist activity at TRPV1 in rat DRG neuron assessed as inhibition of calcium uptake |
Eur J Med Chem 44: 432-6 (2008)
Article DOI: 10.1016/j.ejmech.2007.12.018
BindingDB Entry DOI: 10.7270/Q2FX797G |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM50113436
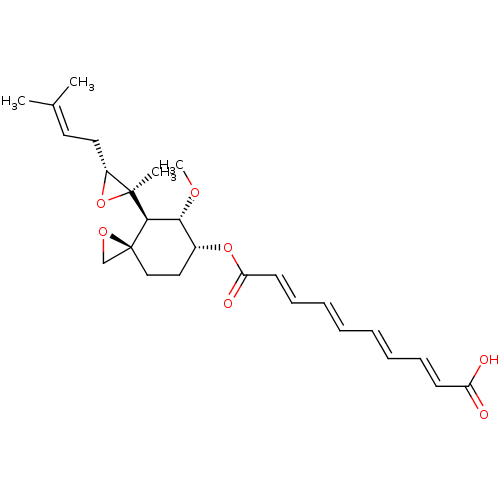
(CHEMBL32838 | fumagillin)Show SMILES [#6]-[#8]-[#6@@H]-1-[#6@@H](-[#6]-[#6][C@]2([#6]-[#8]2)[#6@H]-1[C@@]1([#6])[#8]-[#6@@H]1-[#6]\[#6]=[#6](/[#6])-[#6])-[#8]-[#6](=O)\[#6]=[#6]\[#6]=[#6]\[#6]=[#6]\[#6]=[#6]\[#6](-[#8])=O Show InChI InChI=1S/C26H34O7/c1-18(2)13-14-20-25(3,33-20)24-23(30-4)19(15-16-26(24)17-31-26)32-22(29)12-10-8-6-5-7-9-11-21(27)28/h5-13,19-20,23-24H,14-17H2,1-4H3,(H,27,28)/b7-5+,8-6+,11-9+,12-10+/t19-,20-,23-,24-,25+,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
University of California Santa Cruz
Curated by ChEMBL
| Assay Description
Inhibition of methionine aminopeptidase 2 |
J Nat Prod 72: 588-603 (2010)
Article DOI: 10.1021/np800817y
BindingDB Entry DOI: 10.7270/Q2765G84 |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 1
(Homo sapiens (Human)) | BDBM17847
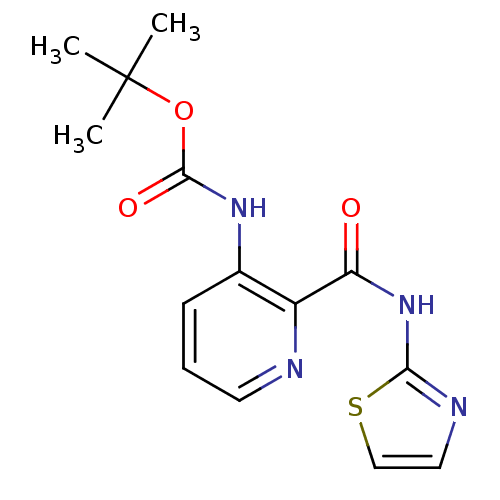
(CHEMBL327579 | pyridine-2-carboxylic acid inhibito...)Show InChI InChI=1S/C14H16N4O3S/c1-14(2,3)21-13(20)17-9-5-4-6-15-10(9)11(19)18-12-16-7-8-22-12/h4-8H,1-3H3,(H,17,20)(H,16,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of California Santa Cruz
Curated by ChEMBL
| Assay Description
Inhibition of methionine aminopeptidase 1 |
J Nat Prod 72: 588-603 (2010)
Article DOI: 10.1021/np800817y
BindingDB Entry DOI: 10.7270/Q2765G84 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Methionine aminopeptidase 1
(Homo sapiens (Human)) | BDBM50295174
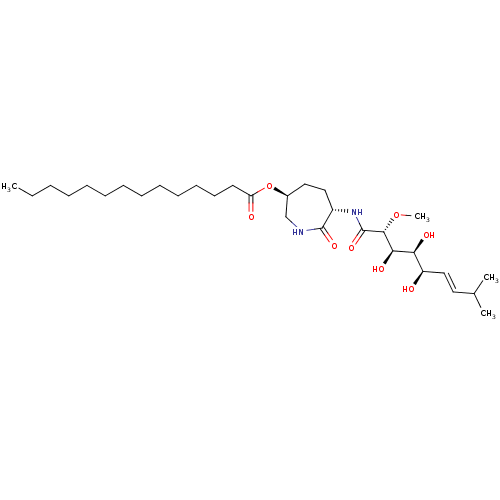
(Bengamide A | CHEMBL256279)Show SMILES CCCCCCCCCCCCCC(=O)O[C@H]1CC[C@H](NC(=O)[C@H](OC)[C@H](O)[C@@H](O)[C@H](O)\C=C\C(C)C)C(=O)NC1 |r| Show InChI InChI=1S/C31H56N2O8/c1-5-6-7-8-9-10-11-12-13-14-15-16-26(35)41-23-18-19-24(30(38)32-21-23)33-31(39)29(40-4)28(37)27(36)25(34)20-17-22(2)3/h17,20,22-25,27-29,34,36-37H,5-16,18-19,21H2,1-4H3,(H,32,38)(H,33,39)/b20-17+/t23-,24-,25+,27-,28+,29+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of California Santa Cruz
Curated by ChEMBL
| Assay Description
Inhibition of methionine aminopeptidase 1 |
J Nat Prod 72: 588-603 (2010)
Article DOI: 10.1021/np800817y
BindingDB Entry DOI: 10.7270/Q2765G84 |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 1
(Homo sapiens (Human)) | BDBM50295173
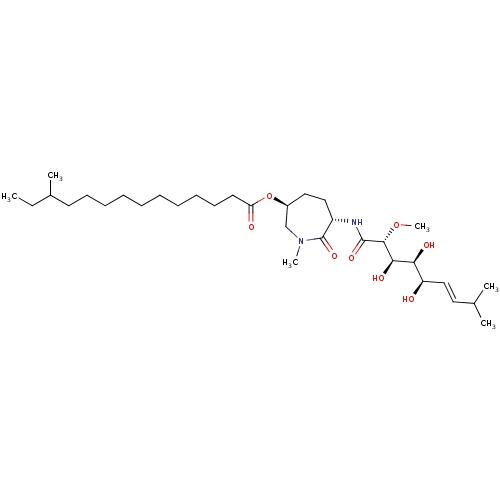
(Bengamide O | CHEMBL539894)Show SMILES CCC(C)CCCCCCCCCCC(=O)O[C@H]1CC[C@H](NC(=O)[C@H](OC)[C@H](O)[C@@H](O)[C@H](O)\C=C\C(C)C)C(=O)N(C)C1 |r| Show InChI InChI=1S/C33H60N2O8/c1-7-24(4)16-14-12-10-8-9-11-13-15-17-28(37)43-25-19-20-26(33(41)35(5)22-25)34-32(40)31(42-6)30(39)29(38)27(36)21-18-23(2)3/h18,21,23-27,29-31,36,38-39H,7-17,19-20,22H2,1-6H3,(H,34,40)/b21-18+/t24?,25-,26-,27+,29-,30+,31+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of California Santa Cruz
Curated by ChEMBL
| Assay Description
Inhibition of methionine aminopeptidase 1 |
J Nat Prod 72: 588-603 (2010)
Article DOI: 10.1021/np800817y
BindingDB Entry DOI: 10.7270/Q2765G84 |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM50295174

(Bengamide A | CHEMBL256279)Show SMILES CCCCCCCCCCCCCC(=O)O[C@H]1CC[C@H](NC(=O)[C@H](OC)[C@H](O)[C@@H](O)[C@H](O)\C=C\C(C)C)C(=O)NC1 |r| Show InChI InChI=1S/C31H56N2O8/c1-5-6-7-8-9-10-11-12-13-14-15-16-26(35)41-23-18-19-24(30(38)32-21-23)33-31(39)29(40-4)28(37)27(36)25(34)20-17-22(2)3/h17,20,22-25,27-29,34,36-37H,5-16,18-19,21H2,1-4H3,(H,32,38)(H,33,39)/b20-17+/t23-,24-,25+,27-,28+,29+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of California Santa Cruz
Curated by ChEMBL
| Assay Description
Inhibition of methionine aminopeptidase 2 |
J Nat Prod 72: 588-603 (2010)
Article DOI: 10.1021/np800817y
BindingDB Entry DOI: 10.7270/Q2765G84 |
More data for this
Ligand-Target Pair | |
Kinesin-like protein KIF11
(Homo sapiens (Human)) | BDBM50216502
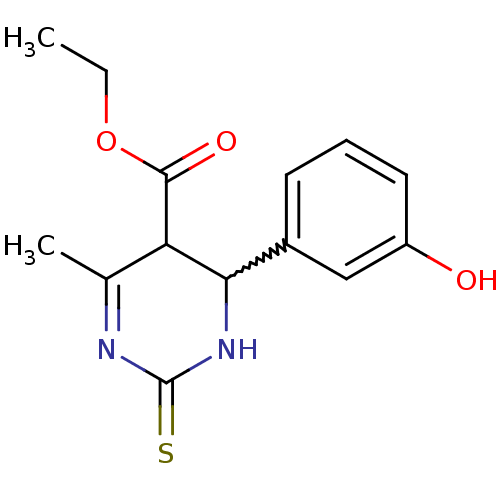
(4-(3-hydroxy-phenyl)-6-methyl-2-thioxo-1,2,3,4-tet...)Show SMILES CCOC(=O)C1C(NC(=S)N=C1C)c1cccc(O)c1 |w:6.13,c:10| Show InChI InChI=1S/C14H16N2O3S/c1-3-19-13(18)11-8(2)15-14(20)16-12(11)9-5-4-6-10(17)7-9/h4-7,11-12,17H,3H2,1-2H3,(H,16,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of Eg5 (unknown origin) |
Eur J Med Chem 60: 120-7 (2013)
Article DOI: 10.1016/j.ejmech.2012.11.044
BindingDB Entry DOI: 10.7270/Q24M95VR |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
3-hydroxy-3-methylglutaryl-coenzyme A reductase
(Homo sapiens (Human)) | BDBM34168
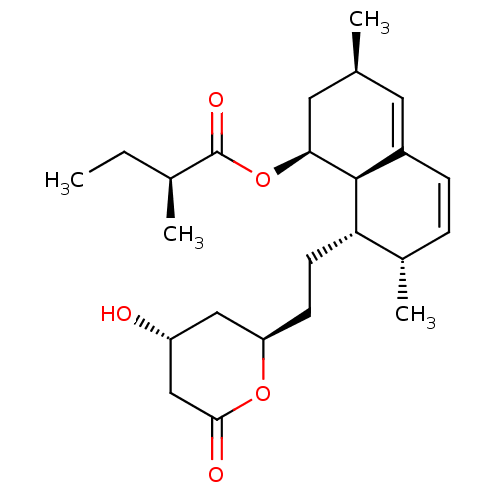
(LOVASTATIN | MLS000069585 | SMR000058779 | US91151...)Show SMILES CC[C@H](C)C(=O)O[C@H]1C[C@@H](C)C=C2C=C[C@H](C)[C@H](CC[C@@H]3C[C@@H](O)CC(=O)O3)[C@@H]12 |c:13,t:11| Show InChI InChI=1S/C24H36O5/c1-5-15(3)24(27)29-21-11-14(2)10-17-7-6-16(4)20(23(17)21)9-8-19-12-18(25)13-22(26)28-19/h6-7,10,14-16,18-21,23,25H,5,8-9,11-13H2,1-4H3/t14-,15-,16-,18+,19+,20-,21-,23-/m0/s1 | PDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.01E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Central Drug Research Institute (CSIR)
Curated by ChEMBL
| Assay Description
Inhibition of HMG-CoA reductase using HMG-CoA as substrate by spectrophotometry in presence of NADPH |
Eur J Med Chem 46: 5206-11 (2011)
Article DOI: 10.1016/j.ejmech.2011.08.012
BindingDB Entry DOI: 10.7270/Q2M32W50 |
More data for this
Ligand-Target Pair | |
3-hydroxy-3-methylglutaryl-coenzyme A reductase
(Homo sapiens (Human)) | BDBM50353742

(CHEMBL486985)Show SMILES C[C@@H]1CC[C@]2(C)[C@H](CCC=C2C)[C@@]1(C)CCC1=CC(=O)O[C@@H]1O |r,c:9,t:18| Show InChI InChI=1S/C20H30O3/c1-13-6-5-7-16-19(13,3)10-8-14(2)20(16,4)11-9-15-12-17(21)23-18(15)22/h6,12,14,16,18,22H,5,7-11H2,1-4H3/t14-,16+,18+,19+,20+/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.02E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Central Drug Research Institute (CSIR)
Curated by ChEMBL
| Assay Description
Inhibition of HMG-CoA reductase using HMG-CoA as substrate by spectrophotometry in presence of NADPH |
Eur J Med Chem 46: 5206-11 (2011)
Article DOI: 10.1016/j.ejmech.2011.08.012
BindingDB Entry DOI: 10.7270/Q2M32W50 |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM50295173

(Bengamide O | CHEMBL539894)Show SMILES CCC(C)CCCCCCCCCCC(=O)O[C@H]1CC[C@H](NC(=O)[C@H](OC)[C@H](O)[C@@H](O)[C@H](O)\C=C\C(C)C)C(=O)N(C)C1 |r| Show InChI InChI=1S/C33H60N2O8/c1-7-24(4)16-14-12-10-8-9-11-13-15-17-28(37)43-25-19-20-26(33(41)35(5)22-25)34-32(40)31(42-6)30(39)29(38)27(36)21-18-23(2)3/h18,21,23-27,29-31,36,38-39H,7-17,19-20,22H2,1-6H3,(H,34,40)/b21-18+/t24?,25-,26-,27+,29-,30+,31+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of California Santa Cruz
Curated by ChEMBL
| Assay Description
Inhibition of methionine aminopeptidase 2 |
J Nat Prod 72: 588-603 (2010)
Article DOI: 10.1021/np800817y
BindingDB Entry DOI: 10.7270/Q2765G84 |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM17847

(CHEMBL327579 | pyridine-2-carboxylic acid inhibito...)Show InChI InChI=1S/C14H16N4O3S/c1-14(2,3)21-13(20)17-9-5-4-6-15-10(9)11(19)18-12-16-7-8-22-12/h4-8H,1-3H3,(H,17,20)(H,16,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of California Santa Cruz
Curated by ChEMBL
| Assay Description
Inhibition of methionine aminopeptidase 2 |
J Nat Prod 72: 588-603 (2010)
Article DOI: 10.1021/np800817y
BindingDB Entry DOI: 10.7270/Q2765G84 |
More data for this
Ligand-Target Pair | |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM50405697
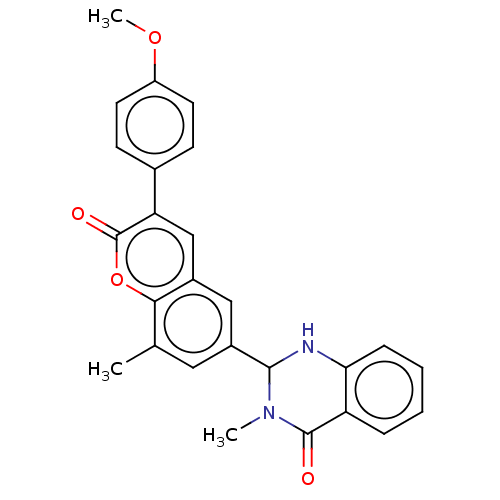
(CHEMBL4164140)Show SMILES COc1ccc(cc1)-c1cc2cc(cc(C)c2oc1=O)C1Nc2ccccc2C(=O)N1C Show InChI InChI=1S/C15H25NO3S/c1-10(9-20)14(17)16-8-12(7-13(16)15(18)19)11-5-3-2-4-6-11/h10-13,20H,2-9H2,1H3,(H,18,19)/t10-,12+,13+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HTLA cells assessed as increase in beta-arrestin2 recruitment after overnight incubation by Tango assa... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM50405699
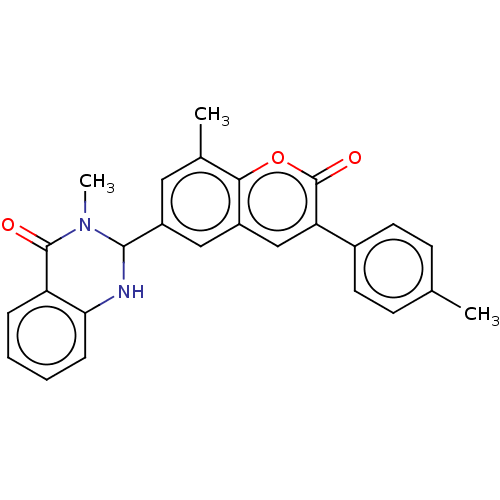
(CHEMBL4174798)Show SMILES CN1C(Nc2ccccc2C1=O)c1cc(C)c2oc(=O)c(cc2c1)-c1ccc(C)cc1 Show InChI InChI=1S/C11H19NO5S/c1-7(5-18)9(13)12-6-11(16-2,17-3)4-8(12)10(14)15/h7-8,18H,4-6H2,1-3H3,(H,14,15)/t7-,8+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 234 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HTLA cells assessed as increase in beta-arrestin2 recruitment after overnight incubation by Tango assa... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM50405700
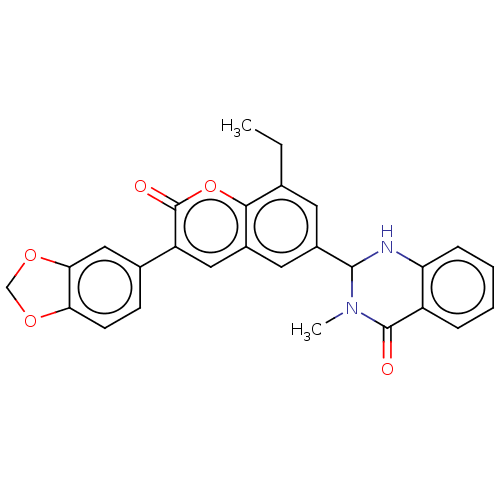
(CHEMBL4163051)Show SMILES CCc1cc(cc2cc(-c3ccc4OCOc4c3)c(=O)oc12)C1Nc2ccccc2C(=O)N1C Show InChI InChI=1S/C9H15NO4S/c1-5(4-15)8(12)10-3-6(11)2-7(10)9(13)14/h5-7,11,15H,2-4H2,1H3,(H,13,14)/t5-,6-,7+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 276 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HTLA cells assessed as increase in beta-arrestin2 recruitment after overnight incubation by Tango assa... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM50405701
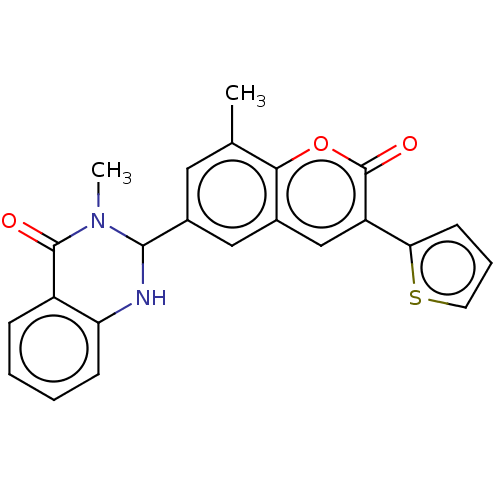
(CHEMBL4167588)Show SMILES CN1C(Nc2ccccc2C1=O)c1cc(C)c2oc(=O)c(cc2c1)-c1cccs1 Show InChI InChI=1S/C15H19NO5S2/c1-10(9-22)14(17)16-8-12(7-13(16)15(18)19)23(20,21)11-5-3-2-4-6-11/h2-6,10,12-13,22H,7-9H2,1H3,(H,18,19)/t10-,12+,13+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.01E+3 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HTLA cells assessed as increase in beta-arrestin2 recruitment after overnight incubation by Tango assa... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM50405701

(CHEMBL4167588)Show SMILES CN1C(Nc2ccccc2C1=O)c1cc(C)c2oc(=O)c(cc2c1)-c1cccs1 Show InChI InChI=1S/C15H19NO5S2/c1-10(9-22)14(17)16-8-12(7-13(16)15(18)19)23(20,21)11-5-3-2-4-6-11/h2-6,10,12-13,22H,7-9H2,1H3,(H,18,19)/t10-,12+,13+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.02E+3 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HTLA cells assessed as increase in beta-arrestin2 recruitment after overnight incubation by Tango assa... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM50405700

(CHEMBL4163051)Show SMILES CCc1cc(cc2cc(-c3ccc4OCOc4c3)c(=O)oc12)C1Nc2ccccc2C(=O)N1C Show InChI InChI=1S/C9H15NO4S/c1-5(4-15)8(12)10-3-6(11)2-7(10)9(13)14/h5-7,11,15H,2-4H2,1H3,(H,13,14)/t5-,6-,7+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 275 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HTLA cells assessed as increase in beta-arrestin2 recruitment after overnight incubation by Tango assa... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM50405699

(CHEMBL4174798)Show SMILES CN1C(Nc2ccccc2C1=O)c1cc(C)c2oc(=O)c(cc2c1)-c1ccc(C)cc1 Show InChI InChI=1S/C11H19NO5S/c1-7(5-18)9(13)12-6-11(16-2,17-3)4-8(12)10(14)15/h7-8,18H,4-6H2,1-3H3,(H,14,15)/t7-,8+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 31 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HEK293 cells harboring glosensor-22F cAMP plasmid DNA assessed as inhibition of forskolin-stimulated c... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM50405697

(CHEMBL4164140)Show SMILES COc1ccc(cc1)-c1cc2cc(cc(C)c2oc1=O)C1Nc2ccccc2C(=O)N1C Show InChI InChI=1S/C15H25NO3S/c1-10(9-20)14(17)16-8-12(7-13(16)15(18)19)11-5-3-2-4-6-11/h10-13,20H,2-9H2,1H3,(H,18,19)/t10-,12+,13+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.69E+3 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HTLA cells assessed as increase in beta-arrestin2 recruitment after overnight incubation by Tango assa... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM50405698
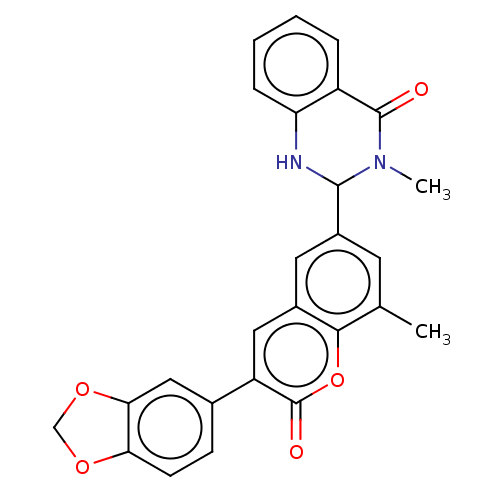
(CHEMBL4159670)Show SMILES CN1C(Nc2ccccc2C1=O)c1cc(C)c2oc(=O)c(cc2c1)-c1ccc2OCOc2c1 Show InChI InChI=1S/C24H28N2O5S/c1-16(25-20(23(28)29)13-12-17-8-4-2-5-9-17)22(27)26-15-19(14-21(26)24(30)31)32-18-10-6-3-7-11-18/h2-11,16,19-21,25H,12-15H2,1H3,(H,28,29)(H,30,31)/t16-,19+,20-,21-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 454 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HTLA cells assessed as increase in beta-arrestin2 recruitment after overnight incubation by Tango assa... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM23515
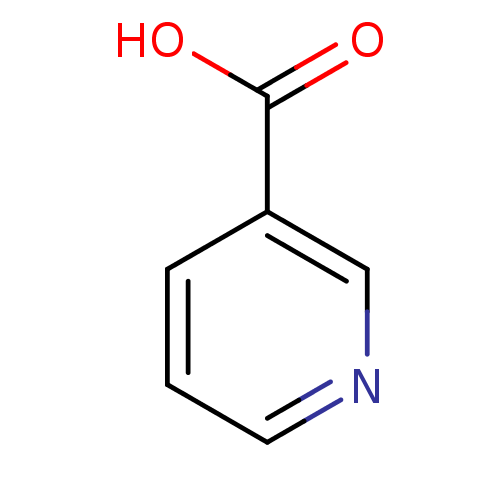
(CHEMBL573 | Niacin | Nicotinic Acid | [5, 6-3H]-ni...)Show InChI InChI=1S/C6H5NO2/c8-6(9)5-2-1-3-7-4-5/h1-4H,(H,8,9) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 21 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HEK293 cells harboring glosensor-22F cAMP plasmid DNA assessed as inhibition of forskolin-stimulated c... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM50405699

(CHEMBL4174798)Show SMILES CN1C(Nc2ccccc2C1=O)c1cc(C)c2oc(=O)c(cc2c1)-c1ccc(C)cc1 Show InChI InChI=1S/C11H19NO5S/c1-7(5-18)9(13)12-6-11(16-2,17-3)4-8(12)10(14)15/h7-8,18H,4-6H2,1-3H3,(H,14,15)/t7-,8+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 30 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HEK293 cells harboring glosensor-22F cAMP plasmid DNA assessed as inhibition of forskolin-stimulated c... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM50405697

(CHEMBL4164140)Show SMILES COc1ccc(cc1)-c1cc2cc(cc(C)c2oc1=O)C1Nc2ccccc2C(=O)N1C Show InChI InChI=1S/C15H25NO3S/c1-10(9-20)14(17)16-8-12(7-13(16)15(18)19)11-5-3-2-4-6-11/h10-13,20H,2-9H2,1H3,(H,18,19)/t10-,12+,13+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 11 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HEK293 cells harboring glosensor-22F cAMP plasmid DNA assessed as inhibition of forskolin-stimulated c... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM23515

(CHEMBL573 | Niacin | Nicotinic Acid | [5, 6-3H]-ni...)Show InChI InChI=1S/C6H5NO2/c8-6(9)5-2-1-3-7-4-5/h1-4H,(H,8,9) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 2.73E+3 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HTLA cells assessed as increase in beta-arrestin2 recruitment after overnight incubation by Tango assa... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM50405699

(CHEMBL4174798)Show SMILES CN1C(Nc2ccccc2C1=O)c1cc(C)c2oc(=O)c(cc2c1)-c1ccc(C)cc1 Show InChI InChI=1S/C11H19NO5S/c1-7(5-18)9(13)12-6-11(16-2,17-3)4-8(12)10(14)15/h7-8,18H,4-6H2,1-3H3,(H,14,15)/t7-,8+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 232 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HTLA cells assessed as increase in beta-arrestin2 recruitment after overnight incubation by Tango assa... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM23515

(CHEMBL573 | Niacin | Nicotinic Acid | [5, 6-3H]-ni...)Show InChI InChI=1S/C6H5NO2/c8-6(9)5-2-1-3-7-4-5/h1-4H,(H,8,9) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 21 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HEK293 cells harboring glosensor-22F cAMP plasmid DNA assessed as inhibition of forskolin-stimulated c... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM50405697

(CHEMBL4164140)Show SMILES COc1ccc(cc1)-c1cc2cc(cc(C)c2oc1=O)C1Nc2ccccc2C(=O)N1C Show InChI InChI=1S/C15H25NO3S/c1-10(9-20)14(17)16-8-12(7-13(16)15(18)19)11-5-3-2-4-6-11/h10-13,20H,2-9H2,1H3,(H,18,19)/t10-,12+,13+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 11 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HEK293 cells harboring glosensor-22F cAMP plasmid DNA assessed as inhibition of forskolin-stimulated c... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM23515

(CHEMBL573 | Niacin | Nicotinic Acid | [5, 6-3H]-ni...)Show InChI InChI=1S/C6H5NO2/c8-6(9)5-2-1-3-7-4-5/h1-4H,(H,8,9) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 2.75E+3 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HTLA cells assessed as increase in beta-arrestin2 recruitment after overnight incubation by Tango assa... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hydroxycarboxylic acid receptor 2
(Homo sapiens (Human)) | BDBM50405698

(CHEMBL4159670)Show SMILES CN1C(Nc2ccccc2C1=O)c1cc(C)c2oc(=O)c(cc2c1)-c1ccc2OCOc2c1 Show InChI InChI=1S/C24H28N2O5S/c1-16(25-20(23(28)29)13-12-17-8-4-2-5-9-17)22(27)26-15-19(14-21(26)24(30)31)32-18-10-6-3-7-11-18/h2-11,16,19-21,25H,12-15H2,1H3,(H,28,29)(H,30,31)/t16-,19+,20-,21-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 457 | n/a | n/a | n/a | n/a |
CSIR-Central Drug Research Institute
Curated by ChEMBL
| Assay Description
Agonist activity at human GPR109a expressed in HTLA cells assessed as increase in beta-arrestin2 recruitment after overnight incubation by Tango assa... |
Eur J Med Chem 152: 208-222 (2018)
Article DOI: 10.1016/j.ejmech.2018.04.037
BindingDB Entry DOI: 10.7270/Q2B27XT0 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data