 Found 66 hits with Last Name = 'hanano' and Initial = 't'
Found 66 hits with Last Name = 'hanano' and Initial = 't' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
5-hydroxytryptamine receptor 1A
(Rattus norvegicus (rat)) | BDBM50088846

(CHEMBL353678 | N-{4-[2-(4-Phenyl-piperazin-1-yl)-e...)Show InChI InChI=1S/C21H27N3O/c1-18(25)22-17-20-9-7-19(8-10-20)11-12-23-13-15-24(16-14-23)21-5-3-2-4-6-21/h2-10H,11-17H2,1H3,(H,22,25) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 2.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards 5-hydroxytryptamine 1A receptor |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Rattus norvegicus (rat)) | BDBM50088844
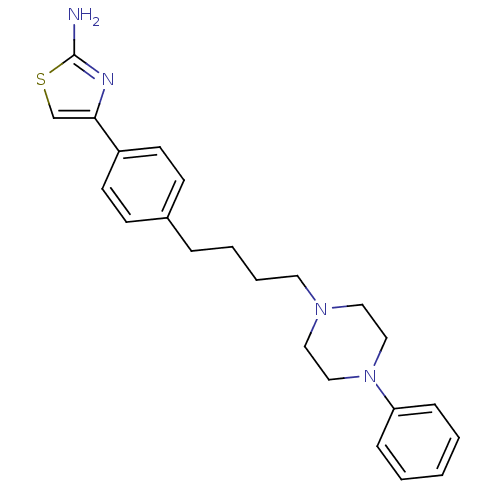
(4-{4-[4-(4-Phenyl-piperazin-1-yl)-butyl]-phenyl}-t...)Show SMILES Nc1nc(cs1)-c1ccc(CCCCN2CCN(CC2)c2ccccc2)cc1 Show InChI InChI=1S/C23H28N4S/c24-23-25-22(18-28-23)20-11-9-19(10-12-20)6-4-5-13-26-14-16-27(17-15-26)21-7-2-1-3-8-21/h1-3,7-12,18H,4-6,13-17H2,(H2,24,25) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 3.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards 5-hydroxytryptamine 1A receptor |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Alpha-1A/Alpha-1B/Alpha-1D adrenergic receptor
(Rattus norvegicus (rat)-Rattus norvegicus (Rat)) | BDBM50088839
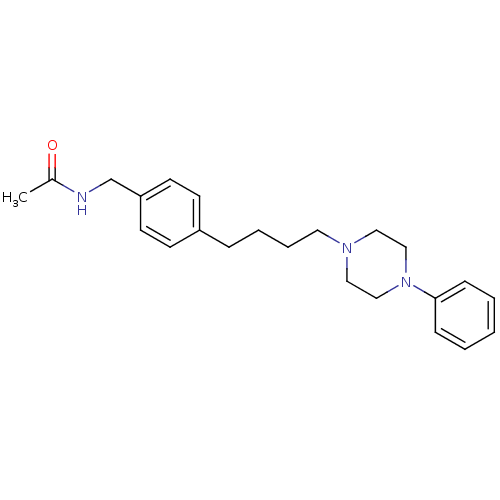
(CHEMBL169702 | N-{4-[4-(4-Phenyl-piperazin-1-yl)-b...)Show InChI InChI=1S/C23H31N3O/c1-20(27)24-19-22-12-10-21(11-13-22)7-5-6-14-25-15-17-26(18-16-25)23-8-3-2-4-9-23/h2-4,8-13H,5-7,14-19H2,1H3,(H,24,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 4.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
In vitro inhibition of human trypsin. |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(BOVINE) | BDBM50088844

(4-{4-[4-(4-Phenyl-piperazin-1-yl)-butyl]-phenyl}-t...)Show SMILES Nc1nc(cs1)-c1ccc(CCCCN2CCN(CC2)c2ccccc2)cc1 Show InChI InChI=1S/C23H28N4S/c24-23-25-22(18-28-23)20-11-9-19(10-12-20)6-4-5-13-26-14-16-27(17-15-26)21-7-2-1-3-8-21/h1-3,7-12,18H,4-6,13-17H2,(H2,24,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 5.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards Dopamine receptor D2 |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Rattus norvegicus (rat)) | BDBM50088839

(CHEMBL169702 | N-{4-[4-(4-Phenyl-piperazin-1-yl)-b...)Show InChI InChI=1S/C23H31N3O/c1-20(27)24-19-22-12-10-21(11-13-22)7-5-6-14-25-15-17-26(18-16-25)23-8-3-2-4-9-23/h2-4,8-13H,5-7,14-19H2,1H3,(H,24,27) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards 5-hydroxytryptamine 1A receptor |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Alpha-1A/Alpha-1B/Alpha-1D adrenergic receptor
(Rattus norvegicus (rat)-Rattus norvegicus (Rat)) | BDBM50088841
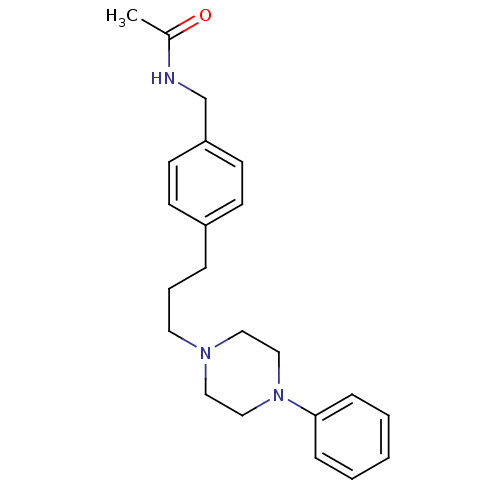
(CHEMBL352617 | N-{4-[3-(4-Phenyl-piperazin-1-yl)-p...)Show InChI InChI=1S/C22H29N3O/c1-19(26)23-18-21-11-9-20(10-12-21)6-5-13-24-14-16-25(17-15-24)22-7-3-2-4-8-22/h2-4,7-12H,5-6,13-18H2,1H3,(H,23,26) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 6.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Coagulation factor X. |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Alpha-1A/Alpha-1B/Alpha-1D adrenergic receptor
(Rattus norvegicus (rat)-Rattus norvegicus (Rat)) | BDBM50088844

(4-{4-[4-(4-Phenyl-piperazin-1-yl)-butyl]-phenyl}-t...)Show SMILES Nc1nc(cs1)-c1ccc(CCCCN2CCN(CC2)c2ccccc2)cc1 Show InChI InChI=1S/C23H28N4S/c24-23-25-22(18-28-23)20-11-9-19(10-12-20)6-4-5-13-26-14-16-27(17-15-26)21-7-2-1-3-8-21/h1-3,7-12,18H,4-6,13-17H2,(H2,24,25) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 7.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of class C beta-lactamase derived from Enterobacter cloacae P99 |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Rattus norvegicus (rat)) | BDBM50088841

(CHEMBL352617 | N-{4-[3-(4-Phenyl-piperazin-1-yl)-p...)Show InChI InChI=1S/C22H29N3O/c1-19(26)23-18-21-11-9-20(10-12-21)6-5-13-24-14-16-25(17-15-24)22-7-3-2-4-8-22/h2-4,7-12H,5-6,13-18H2,1H3,(H,23,26) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 33 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards 5-hydroxytryptamine 1A receptor |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Serotonin 2 (5-HT2) receptor
(Homo sapiens (Human)) | BDBM50088844

(4-{4-[4-(4-Phenyl-piperazin-1-yl)-butyl]-phenyl}-t...)Show SMILES Nc1nc(cs1)-c1ccc(CCCCN2CCN(CC2)c2ccccc2)cc1 Show InChI InChI=1S/C23H28N4S/c24-23-25-22(18-28-23)20-11-9-19(10-12-20)6-4-5-13-26-14-16-27(17-15-26)21-7-2-1-3-8-21/h1-3,7-12,18H,4-6,13-17H2,(H2,24,25) | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of GC1 extended spectrum class C beta-lactamase |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Serotonin 2 (5-HT2) receptor
(Homo sapiens (Human)) | BDBM50088839

(CHEMBL169702 | N-{4-[4-(4-Phenyl-piperazin-1-yl)-b...)Show InChI InChI=1S/C23H31N3O/c1-20(27)24-19-22-12-10-21(11-13-22)7-5-6-14-25-15-17-26(18-16-25)23-8-3-2-4-9-23/h2-4,8-13H,5-7,14-19H2,1H3,(H,24,27) | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 48 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of GC1 extended spectrum class C beta-lactamase |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(BOVINE) | BDBM50088839

(CHEMBL169702 | N-{4-[4-(4-Phenyl-piperazin-1-yl)-b...)Show InChI InChI=1S/C23H31N3O/c1-20(27)24-19-22-12-10-21(11-13-22)7-5-6-14-25-15-17-26(18-16-25)23-8-3-2-4-9-23/h2-4,8-13H,5-7,14-19H2,1H3,(H,24,27) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 49 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards Dopamine receptor D2 |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Alpha-1A/Alpha-1B/Alpha-1D adrenergic receptor
(Rattus norvegicus (rat)-Rattus norvegicus (Rat)) | BDBM50088846

(CHEMBL353678 | N-{4-[2-(4-Phenyl-piperazin-1-yl)-e...)Show InChI InChI=1S/C21H27N3O/c1-18(25)22-17-20-9-7-19(8-10-20)11-12-23-13-15-24(16-14-23)21-5-3-2-4-6-21/h2-10H,11-17H2,1H3,(H,22,25) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 69 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
In vitro inhibition of human trypsin. |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Serotonin 2 (5-HT2) receptor
(Homo sapiens (Human)) | BDBM50088846

(CHEMBL353678 | N-{4-[2-(4-Phenyl-piperazin-1-yl)-e...)Show InChI InChI=1S/C21H27N3O/c1-18(25)22-17-20-9-7-19(8-10-20)11-12-23-13-15-24(16-14-23)21-5-3-2-4-6-21/h2-10H,11-17H2,1H3,(H,22,25) | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
In vitro inhibition of human thrombin. |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Rattus norvegicus (rat)) | BDBM50088845
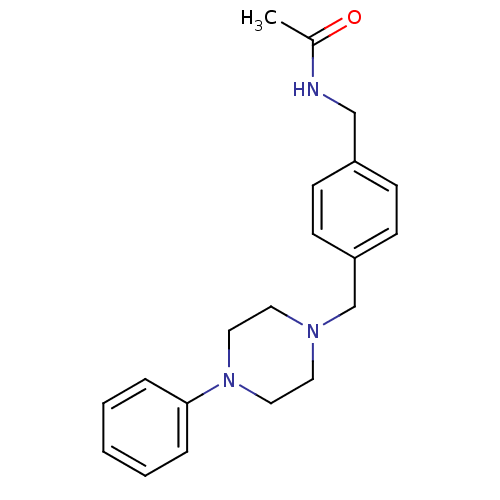
(CHEMBL170307 | N-[4-(4-Phenyl-piperazin-1-ylmethyl...)Show InChI InChI=1S/C20H25N3O/c1-17(24)21-15-18-7-9-19(10-8-18)16-22-11-13-23(14-12-22)20-5-3-2-4-6-20/h2-10H,11-16H2,1H3,(H,21,24) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards 5-hydroxytryptamine 1A receptor |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Serotonin 2 (5-HT2) receptor
(Homo sapiens (Human)) | BDBM50088841

(CHEMBL352617 | N-{4-[3-(4-Phenyl-piperazin-1-yl)-p...)Show InChI InChI=1S/C22H29N3O/c1-19(26)23-18-21-11-9-20(10-12-21)6-5-13-24-14-16-25(17-15-24)22-7-3-2-4-8-22/h2-4,7-12H,5-6,13-18H2,1H3,(H,23,26) | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of GC1 extended spectrum class C beta-lactamase |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(BOVINE) | BDBM50088846

(CHEMBL353678 | N-{4-[2-(4-Phenyl-piperazin-1-yl)-e...)Show InChI InChI=1S/C21H27N3O/c1-18(25)22-17-20-9-7-19(8-10-20)11-12-23-13-15-24(16-14-23)21-5-3-2-4-6-21/h2-10H,11-17H2,1H3,(H,22,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards Dopamine receptor D2 |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(BOVINE) | BDBM50088841

(CHEMBL352617 | N-{4-[3-(4-Phenyl-piperazin-1-yl)-p...)Show InChI InChI=1S/C22H29N3O/c1-19(26)23-18-21-11-9-20(10-12-21)6-5-13-24-14-16-25(17-15-24)22-7-3-2-4-8-22/h2-4,7-12H,5-6,13-18H2,1H3,(H,23,26) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards Dopamine receptor D2 |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Rattus norvegicus (rat)) | BDBM50088840
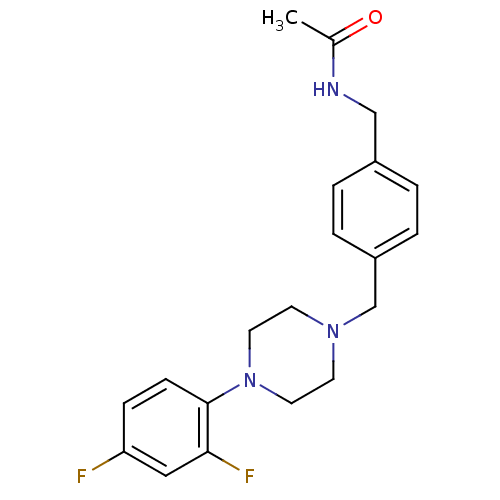
(CHEMBL352576 | N-{4-[4-(2,4-Difluoro-phenyl)-piper...)Show InChI InChI=1S/C20H23F2N3O/c1-15(26)23-13-16-2-4-17(5-3-16)14-24-8-10-25(11-9-24)20-7-6-18(21)12-19(20)22/h2-7,12H,8-11,13-14H2,1H3,(H,23,26) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards 5-hydroxytryptamine 1A receptor |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(BOVINE) | BDBM50088842
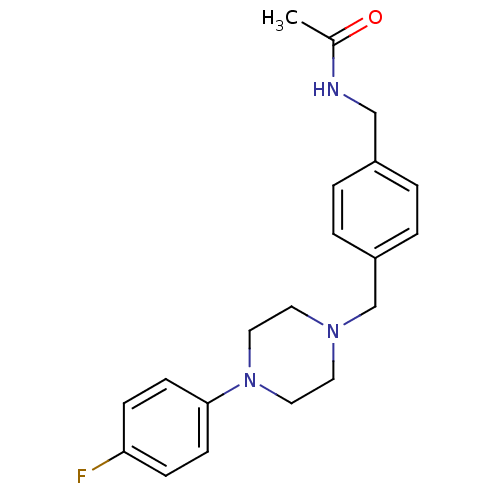
(CHEMBL171052 | N-{4-[4-(4-Fluoro-phenyl)-piperazin...)Show InChI InChI=1S/C20H24FN3O/c1-16(25)22-14-17-2-4-18(5-3-17)15-23-10-12-24(13-11-23)20-8-6-19(21)7-9-20/h2-9H,10-15H2,1H3,(H,22,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards Dopamine receptor D2 |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(BOVINE) | BDBM50088843
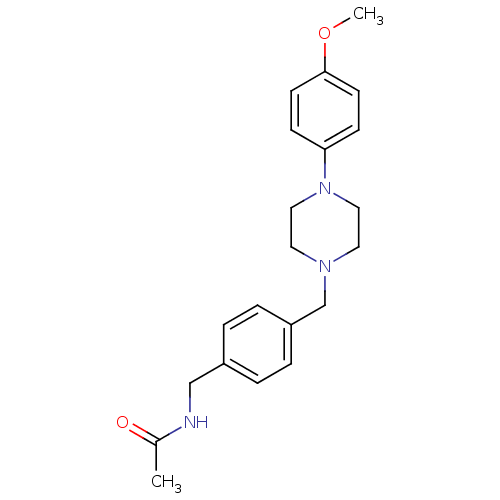
(CHEMBL171115 | N-{4-[4-(4-Methoxy-phenyl)-piperazi...)Show InChI InChI=1S/C21H27N3O2/c1-17(25)22-15-18-3-5-19(6-4-18)16-23-11-13-24(14-12-23)20-7-9-21(26-2)10-8-20/h3-10H,11-16H2,1-2H3,(H,22,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards Dopamine receptor D2 |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Rattus norvegicus (rat)) | BDBM50088843

(CHEMBL171115 | N-{4-[4-(4-Methoxy-phenyl)-piperazi...)Show InChI InChI=1S/C21H27N3O2/c1-17(25)22-15-18-3-5-19(6-4-18)16-23-11-13-24(14-12-23)20-7-9-21(26-2)10-8-20/h3-10H,11-16H2,1-2H3,(H,22,25) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards 5-hydroxytryptamine 1A receptor |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(BOVINE) | BDBM50088845

(CHEMBL170307 | N-[4-(4-Phenyl-piperazin-1-ylmethyl...)Show InChI InChI=1S/C20H25N3O/c1-17(24)21-15-18-7-9-19(10-8-18)16-22-11-13-23(14-12-22)20-5-3-2-4-6-20/h2-10H,11-16H2,1H3,(H,21,24) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards Dopamine receptor D2 |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(BOVINE) | BDBM50088840

(CHEMBL352576 | N-{4-[4-(2,4-Difluoro-phenyl)-piper...)Show InChI InChI=1S/C20H23F2N3O/c1-15(26)23-13-16-2-4-17(5-3-16)14-24-8-10-25(11-9-24)20-7-6-18(21)12-19(20)22/h2-7,12H,8-11,13-14H2,1H3,(H,23,26) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards Dopamine receptor D2 |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(BOVINE) | BDBM50088847

(CHEMBL171345 | N-{4-[4-(4-Chloro-phenyl)-piperazin...)Show InChI InChI=1S/C20H24ClN3O/c1-16(25)22-14-17-2-4-18(5-3-17)15-23-10-12-24(13-11-23)20-8-6-19(21)7-9-20/h2-9H,10-15H2,1H3,(H,22,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards Dopamine receptor D2 |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Rattus norvegicus (rat)) | BDBM50088847

(CHEMBL171345 | N-{4-[4-(4-Chloro-phenyl)-piperazin...)Show InChI InChI=1S/C20H24ClN3O/c1-16(25)22-14-17-2-4-18(5-3-17)15-23-10-12-24(13-11-23)20-8-6-19(21)7-9-20/h2-9H,10-15H2,1H3,(H,22,25) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards 5-hydroxytryptamine 1A receptor |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Rattus norvegicus (rat)) | BDBM50088842

(CHEMBL171052 | N-{4-[4-(4-Fluoro-phenyl)-piperazin...)Show InChI InChI=1S/C20H24FN3O/c1-16(25)22-14-17-2-4-18(5-3-17)15-23-10-12-24(13-11-23)20-8-6-19(21)7-9-20/h2-9H,10-15H2,1H3,(H,22,25) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards 5-hydroxytryptamine 1A receptor |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Alpha-1A/Alpha-1B/Alpha-1D adrenergic receptor
(Rattus norvegicus (rat)-Rattus norvegicus (Rat)) | BDBM50088847

(CHEMBL171345 | N-{4-[4-(4-Chloro-phenyl)-piperazin...)Show InChI InChI=1S/C20H24ClN3O/c1-16(25)22-14-17-2-4-18(5-3-17)15-23-10-12-24(13-11-23)20-8-6-19(21)7-9-20/h2-9H,10-15H2,1H3,(H,22,25) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of class C beta-lactamase derived from Enterobacter cloacae P99 |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Alpha-1A/Alpha-1B/Alpha-1D adrenergic receptor
(Rattus norvegicus (rat)-Rattus norvegicus (Rat)) | BDBM50088843

(CHEMBL171115 | N-{4-[4-(4-Methoxy-phenyl)-piperazi...)Show InChI InChI=1S/C21H27N3O2/c1-17(25)22-15-18-3-5-19(6-4-18)16-23-11-13-24(14-12-23)20-7-9-21(26-2)10-8-20/h3-10H,11-16H2,1-2H3,(H,22,25) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards Alpha-1 adrenergic receptor |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Serotonin 2 (5-HT2) receptor
(Homo sapiens (Human)) | BDBM50088843

(CHEMBL171115 | N-{4-[4-(4-Methoxy-phenyl)-piperazi...)Show InChI InChI=1S/C21H27N3O2/c1-17(25)22-15-18-3-5-19(6-4-18)16-23-11-13-24(14-12-23)20-7-9-21(26-2)10-8-20/h3-10H,11-16H2,1-2H3,(H,22,25) | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Coagulation factor X. |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Serotonin 2 (5-HT2) receptor
(Homo sapiens (Human)) | BDBM50088840

(CHEMBL352576 | N-{4-[4-(2,4-Difluoro-phenyl)-piper...)Show InChI InChI=1S/C20H23F2N3O/c1-15(26)23-13-16-2-4-17(5-3-16)14-24-8-10-25(11-9-24)20-7-6-18(21)12-19(20)22/h2-7,12H,8-11,13-14H2,1H3,(H,23,26) | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
In vitro inhibition of human thrombin. |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Alpha-1A/Alpha-1B/Alpha-1D adrenergic receptor
(Rattus norvegicus (rat)-Rattus norvegicus (Rat)) | BDBM50088845

(CHEMBL170307 | N-[4-(4-Phenyl-piperazin-1-ylmethyl...)Show InChI InChI=1S/C20H25N3O/c1-17(24)21-15-18-7-9-19(10-8-18)16-22-11-13-23(14-12-22)20-5-3-2-4-6-20/h2-10H,11-16H2,1H3,(H,21,24) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
In vitro inhibition of human plasmin. |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Alpha-1A/Alpha-1B/Alpha-1D adrenergic receptor
(Rattus norvegicus (rat)-Rattus norvegicus (Rat)) | BDBM50088842

(CHEMBL171052 | N-{4-[4-(4-Fluoro-phenyl)-piperazin...)Show InChI InChI=1S/C20H24FN3O/c1-16(25)22-14-17-2-4-18(5-3-17)15-23-10-12-24(13-11-23)20-8-6-19(21)7-9-20/h2-9H,10-15H2,1H3,(H,22,25) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Coagulation factor X. |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Serotonin 2 (5-HT2) receptor
(Homo sapiens (Human)) | BDBM50088847

(CHEMBL171345 | N-{4-[4-(4-Chloro-phenyl)-piperazin...)Show InChI InChI=1S/C20H24ClN3O/c1-16(25)22-14-17-2-4-18(5-3-17)15-23-10-12-24(13-11-23)20-8-6-19(21)7-9-20/h2-9H,10-15H2,1H3,(H,22,25) | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of class C beta-lactamase derived from Enterobacter cloacae P99 |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Serotonin 2 (5-HT2) receptor
(Homo sapiens (Human)) | BDBM50088845

(CHEMBL170307 | N-[4-(4-Phenyl-piperazin-1-ylmethyl...)Show InChI InChI=1S/C20H25N3O/c1-17(24)21-15-18-7-9-19(10-8-18)16-22-11-13-23(14-12-22)20-5-3-2-4-6-20/h2-10H,11-16H2,1H3,(H,21,24) | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of GC1 extended spectrum class C beta-lactamase |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Serotonin 2 (5-HT2) receptor
(Homo sapiens (Human)) | BDBM50088842

(CHEMBL171052 | N-{4-[4-(4-Fluoro-phenyl)-piperazin...)Show InChI InChI=1S/C20H24FN3O/c1-16(25)22-14-17-2-4-18(5-3-17)15-23-10-12-24(13-11-23)20-8-6-19(21)7-9-20/h2-9H,10-15H2,1H3,(H,22,25) | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards 5-hydroxytryptamine 2 receptor |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Alpha-1A/Alpha-1B/Alpha-1D adrenergic receptor
(Rattus norvegicus (rat)-Rattus norvegicus (Rat)) | BDBM50088840

(CHEMBL352576 | N-{4-[4-(2,4-Difluoro-phenyl)-piper...)Show InChI InChI=1S/C20H23F2N3O/c1-15(26)23-13-16-2-4-17(5-3-16)14-24-8-10-25(11-9-24)20-7-6-18(21)12-19(20)22/h2-7,12H,8-11,13-14H2,1H3,(H,23,26) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yoshitomi Pharmaceutical Industries, Ltd.
Curated by ChEMBL
| Assay Description
Binding affinity towards Alpha-1 adrenergic receptor |
Bioorg Med Chem Lett 10: 875-9 (2000)
BindingDB Entry DOI: 10.7270/Q2CZ36C7 |
More data for this
Ligand-Target Pair | |
Casein kinase I isoform alpha
(Homo sapiens (Human)) | BDBM50443871
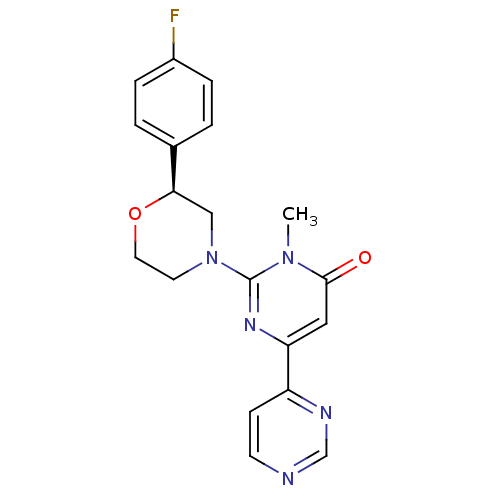
(CHEMBL3091536)Show SMILES Cn1c(nc(cc1=O)-c1ccncn1)N1CCO[C@H](C1)c1ccc(F)cc1 |r| Show InChI InChI=1S/C19H18FN5O2/c1-24-18(26)10-16(15-6-7-21-12-22-15)23-19(24)25-8-9-27-17(11-25)13-2-4-14(20)5-3-13/h2-7,10,12,17H,8-9,11H2,1H3/t17-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Tanabe Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CK1 (unknown origin) |
Bioorg Med Chem Lett 23: 6933-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.020
BindingDB Entry DOI: 10.7270/Q2VT1TJ4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50443880
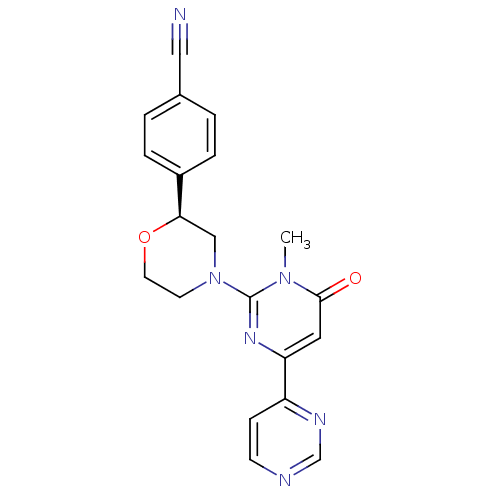
(CHEMBL3091537)Show SMILES Cn1c(nc(cc1=O)-c1ccncn1)N1CCO[C@H](C1)c1ccc(cc1)C#N |r| Show InChI InChI=1S/C20H18N6O2/c1-25-19(27)10-17(16-6-7-22-13-23-16)24-20(25)26-8-9-28-18(12-26)15-4-2-14(11-21)3-5-15/h2-7,10,13,18H,8-9,12H2,1H3/t18-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Tanabe Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human CYP2D6 |
Bioorg Med Chem Lett 23: 6933-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.020
BindingDB Entry DOI: 10.7270/Q2VT1TJ4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50443881
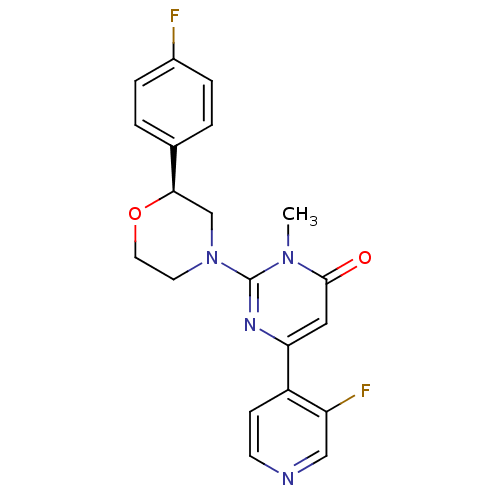
(CHEMBL3091531)Show SMILES Cn1c(nc(cc1=O)-c1ccncc1F)N1CCO[C@H](C1)c1ccc(F)cc1 |r| Show InChI InChI=1S/C20H18F2N4O2/c1-25-19(27)10-17(15-6-7-23-11-16(15)22)24-20(25)26-8-9-28-18(12-26)13-2-4-14(21)5-3-13/h2-7,10-11,18H,8-9,12H2,1H3/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Tanabe Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human CYP1A2 |
Bioorg Med Chem Lett 23: 6933-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.020
BindingDB Entry DOI: 10.7270/Q2VT1TJ4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50443879
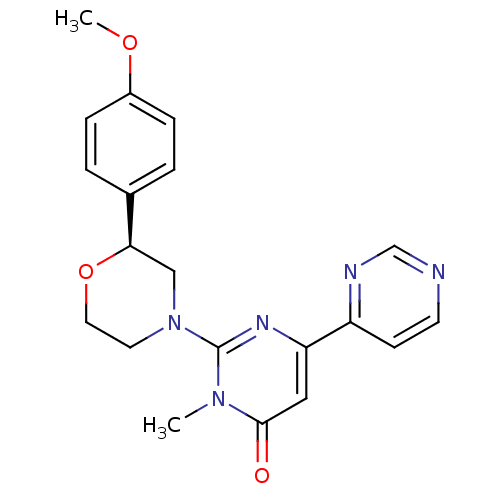
(CHEMBL3091538)Show SMILES COc1ccc(cc1)[C@H]1CN(CCO1)c1nc(cc(=O)n1C)-c1ccncn1 |r| Show InChI InChI=1S/C20H21N5O3/c1-24-19(26)11-17(16-7-8-21-13-22-16)23-20(24)25-9-10-28-18(12-25)14-3-5-15(27-2)6-4-14/h3-8,11,13,18H,9-10,12H2,1-2H3/t18-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Tanabe Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human CYP2D6 |
Bioorg Med Chem Lett 23: 6933-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.020
BindingDB Entry DOI: 10.7270/Q2VT1TJ4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50443876

(CHEMBL3091540)Show SMILES COc1ccccc1[C@H]1CN(CCO1)c1nc(cc(=O)n1C)-c1ccncn1 |r| Show InChI InChI=1S/C20H21N5O3/c1-24-19(26)11-16(15-7-8-21-13-22-15)23-20(24)25-9-10-28-18(12-25)14-5-3-4-6-17(14)27-2/h3-8,11,13,18H,9-10,12H2,1-2H3/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.57E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Tanabe Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human CYP1A2 |
Bioorg Med Chem Lett 23: 6933-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.020
BindingDB Entry DOI: 10.7270/Q2VT1TJ4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50443876

(CHEMBL3091540)Show SMILES COc1ccccc1[C@H]1CN(CCO1)c1nc(cc(=O)n1C)-c1ccncn1 |r| Show InChI InChI=1S/C20H21N5O3/c1-24-19(26)11-16(15-7-8-21-13-22-15)23-20(24)25-9-10-28-18(12-25)14-5-3-4-6-17(14)27-2/h3-8,11,13,18H,9-10,12H2,1-2H3/t18-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Tanabe Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human CYP2D6 |
Bioorg Med Chem Lett 23: 6933-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.020
BindingDB Entry DOI: 10.7270/Q2VT1TJ4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50443875

(CHEMBL3091541)Show SMILES COc1cc(F)ccc1[C@H]1CN(CCO1)c1nc(cc(=O)n1C)-c1ccncn1 |r| Show InChI InChI=1S/C20H20FN5O3/c1-25-19(27)10-16(15-5-6-22-12-23-15)24-20(25)26-7-8-29-18(11-26)14-4-3-13(21)9-17(14)28-2/h3-6,9-10,12,18H,7-8,11H2,1-2H3/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.65E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Tanabe Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human CYP1A2 |
Bioorg Med Chem Lett 23: 6933-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.020
BindingDB Entry DOI: 10.7270/Q2VT1TJ4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50443882

(CHEMBL3091529)Show SMILES Cn1c(nc(cc1=O)-c1ccncc1)N1CCO[C@H](C1)c1ccc(F)cc1 |r| Show InChI InChI=1S/C20H19FN4O2/c1-24-19(26)12-17(14-6-8-22-9-7-14)23-20(24)25-10-11-27-18(13-25)15-2-4-16(21)5-3-15/h2-9,12,18H,10-11,13H2,1H3/t18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.71E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Tanabe Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human CYP1A2 |
Bioorg Med Chem Lett 23: 6933-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.020
BindingDB Entry DOI: 10.7270/Q2VT1TJ4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50443875

(CHEMBL3091541)Show SMILES COc1cc(F)ccc1[C@H]1CN(CCO1)c1nc(cc(=O)n1C)-c1ccncn1 |r| Show InChI InChI=1S/C20H20FN5O3/c1-25-19(27)10-16(15-5-6-22-12-23-15)24-20(25)26-7-8-29-18(11-26)14-4-3-13(21)9-17(14)28-2/h3-6,9-10,12,18H,7-8,11H2,1-2H3/t18-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Tanabe Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human CYP2D6 |
Bioorg Med Chem Lett 23: 6933-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.020
BindingDB Entry DOI: 10.7270/Q2VT1TJ4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50443872

(CHEMBL3091544)Show SMILES Cn1c(nc(cc1=O)-c1ccncn1)N1CCO[C@H](C1)c1c(F)cccc1Cl |r| Show InChI InChI=1S/C19H17ClFN5O2/c1-25-17(27)9-15(14-5-6-22-11-23-14)24-19(25)26-7-8-28-16(10-26)18-12(20)3-2-4-13(18)21/h2-6,9,11,16H,7-8,10H2,1H3/t16-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.95E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Tanabe Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human CYP1A2 |
Bioorg Med Chem Lett 23: 6933-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.020
BindingDB Entry DOI: 10.7270/Q2VT1TJ4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50443881

(CHEMBL3091531)Show SMILES Cn1c(nc(cc1=O)-c1ccncc1F)N1CCO[C@H](C1)c1ccc(F)cc1 |r| Show InChI InChI=1S/C20H18F2N4O2/c1-25-19(27)10-17(15-6-7-23-11-16(15)22)24-20(25)26-8-9-28-18(12-26)13-2-4-14(21)5-3-13/h2-7,10-11,18H,8-9,12H2,1H3/t18-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.21E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Tanabe Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human CYP2D6 |
Bioorg Med Chem Lett 23: 6933-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.020
BindingDB Entry DOI: 10.7270/Q2VT1TJ4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50443884

(CHEMBL3091546)Show InChI InChI=1S/C20H20N4O2/c1-23-19(25)13-17(15-7-9-21-10-8-15)22-20(23)24-11-12-26-18(14-24)16-5-3-2-4-6-16/h2-10,13,18H,11-12,14H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.31E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Tanabe Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human CYP1A2 |
Bioorg Med Chem Lett 23: 6933-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.020
BindingDB Entry DOI: 10.7270/Q2VT1TJ4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50443877

(CHEMBL3091539)Show SMILES Cn1c(nc(cc1=O)-c1ccncn1)N1CCO[C@H](C1)c1ccccc1F |r| Show InChI InChI=1S/C19H18FN5O2/c1-24-18(26)10-16(15-6-7-21-12-22-15)23-19(24)25-8-9-27-17(11-25)13-4-2-3-5-14(13)20/h2-7,10,12,17H,8-9,11H2,1H3/t17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.33E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Tanabe Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human CYP1A2 |
Bioorg Med Chem Lett 23: 6933-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.020
BindingDB Entry DOI: 10.7270/Q2VT1TJ4 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50443883
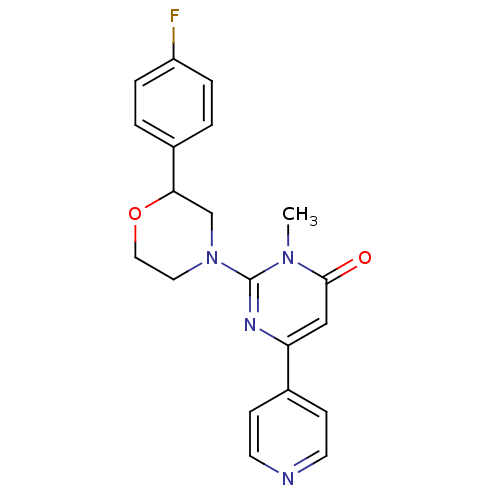
(CHEMBL3091547)Show SMILES Cn1c(nc(cc1=O)-c1ccncc1)N1CCOC(C1)c1ccc(F)cc1 Show InChI InChI=1S/C20H19FN4O2/c1-24-19(26)12-17(14-6-8-22-9-7-14)23-20(24)25-10-11-27-18(13-25)15-2-4-16(21)5-3-15/h2-9,12,18H,10-11,13H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.39E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Mitsubishi Tanabe Pharma Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human CYP1A2 |
Bioorg Med Chem Lett 23: 6933-7 (2013)
Article DOI: 10.1016/j.bmcl.2013.09.020
BindingDB Entry DOI: 10.7270/Q2VT1TJ4 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data