

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50399540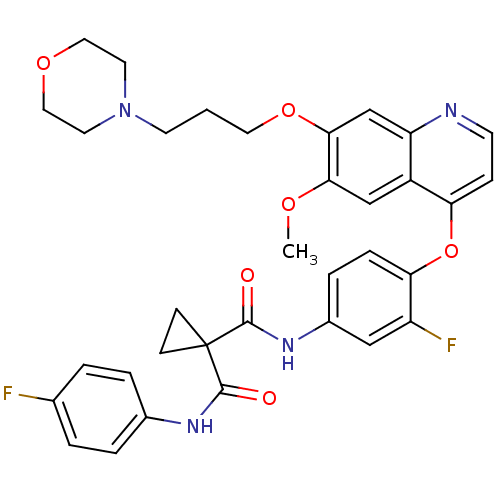 (FORETINIB | US10464902, Foretinib | US10882853, Co...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met (unknown origin) using poly (Glu, Tyr) 4:1 as substrate after 30 mins by HTRF assay | Eur J Med Chem 120: 37-50 (2016) Article DOI: 10.1016/j.ejmech.2016.04.062 BindingDB Entry DOI: 10.7270/Q2VM4F5Q | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50399540 (FORETINIB | US10464902, Foretinib | US10882853, Co...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met kinase (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50147893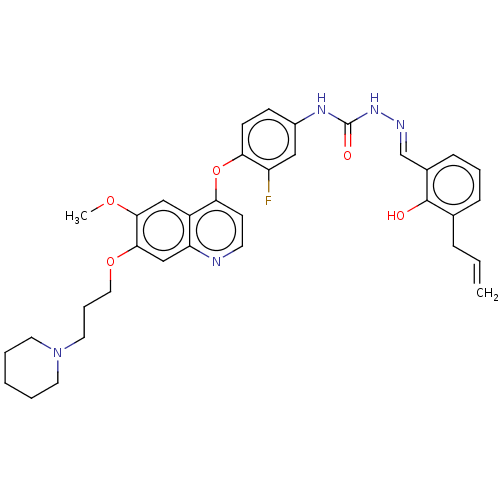 (CHEMBL3764279) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met kinase (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50399540 (FORETINIB | US10464902, Foretinib | US10882853, Co...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met (unknown origin) | Eur J Med Chem 120: 37-50 (2016) Article DOI: 10.1016/j.ejmech.2016.04.062 BindingDB Entry DOI: 10.7270/Q2VM4F5Q | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50190810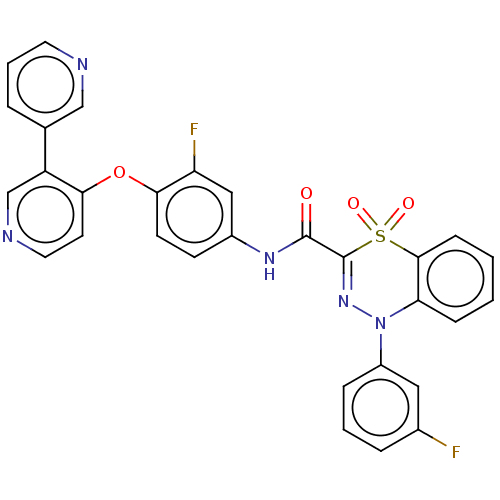 (CHEMBL3975648) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 8.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met (unknown origin) using poly (Glu, Tyr) 4:1 as substrate after 30 mins by HTRF assay | Eur J Med Chem 120: 37-50 (2016) Article DOI: 10.1016/j.ejmech.2016.04.062 BindingDB Entry DOI: 10.7270/Q2VM4F5Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50147911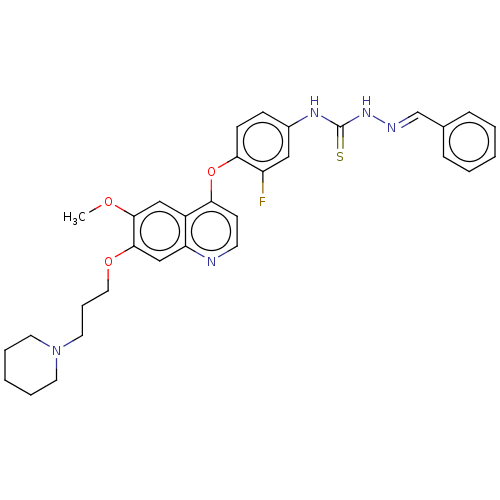 (CHEMBL3764158) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 8.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met kinase (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50147892 (CHEMBL3764241) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met kinase (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage-stimulating protein receptor (Homo sapiens (Human)) | BDBM50147893 (CHEMBL3764279) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of Ron (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50147899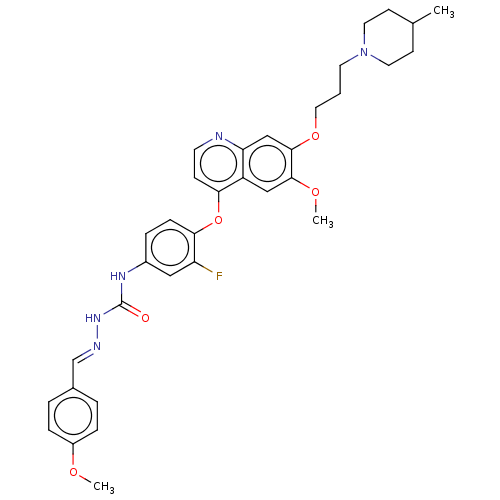 (CHEMBL3765572) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met kinase (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50147914 (CHEMBL3764060) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met kinase (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50190915 (CHEMBL3983663) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met (unknown origin) using poly (Glu, Tyr) 4:1 as substrate after 30 mins by HTRF assay | Eur J Med Chem 120: 37-50 (2016) Article DOI: 10.1016/j.ejmech.2016.04.062 BindingDB Entry DOI: 10.7270/Q2VM4F5Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50190808 (CHEMBL3903767) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met (unknown origin) using poly (Glu, Tyr) 4:1 as substrate after 30 mins by HTRF assay | Eur J Med Chem 120: 37-50 (2016) Article DOI: 10.1016/j.ejmech.2016.04.062 BindingDB Entry DOI: 10.7270/Q2VM4F5Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50190914 (CHEMBL3959949) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met (unknown origin) using poly (Glu, Tyr) 4:1 as substrate after 30 mins by HTRF assay | Eur J Med Chem 120: 37-50 (2016) Article DOI: 10.1016/j.ejmech.2016.04.062 BindingDB Entry DOI: 10.7270/Q2VM4F5Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50147903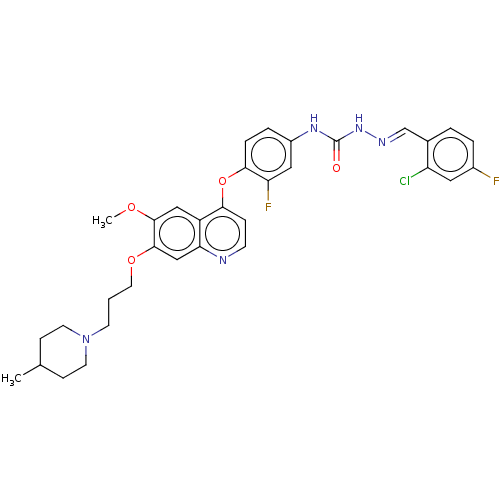 (CHEMBL3763886) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met kinase (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50190912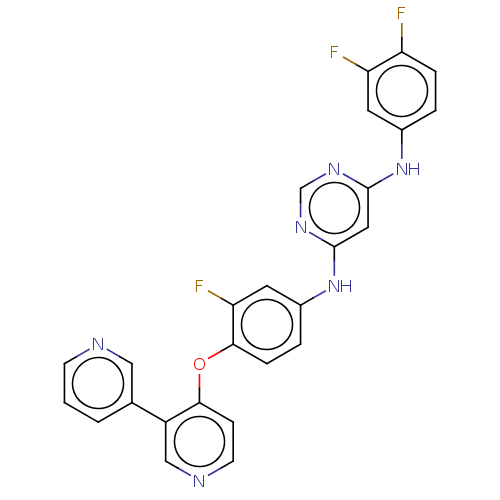 (CHEMBL3945597) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 38 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met (unknown origin) using poly (Glu, Tyr) 4:1 as substrate after 30 mins by HTRF assay | Eur J Med Chem 120: 37-50 (2016) Article DOI: 10.1016/j.ejmech.2016.04.062 BindingDB Entry DOI: 10.7270/Q2VM4F5Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50191065 (CHEMBL3894843) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 47 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met (unknown origin) using poly (Glu, Tyr) 4:1 as substrate after 30 mins by HTRF assay | Eur J Med Chem 120: 37-50 (2016) Article DOI: 10.1016/j.ejmech.2016.04.062 BindingDB Entry DOI: 10.7270/Q2VM4F5Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50190809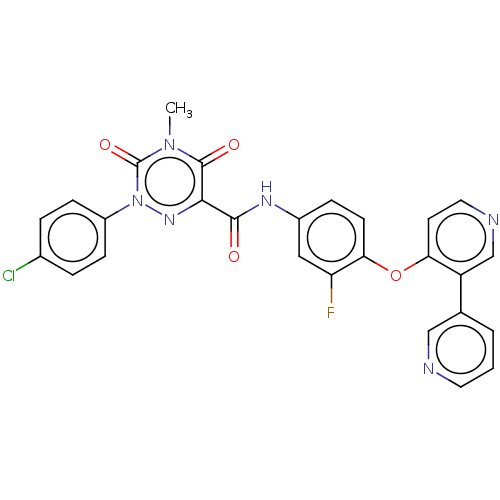 (CHEMBL3942488) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 48 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met (unknown origin) using poly (Glu, Tyr) 4:1 as substrate after 30 mins by HTRF assay | Eur J Med Chem 120: 37-50 (2016) Article DOI: 10.1016/j.ejmech.2016.04.062 BindingDB Entry DOI: 10.7270/Q2VM4F5Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50190916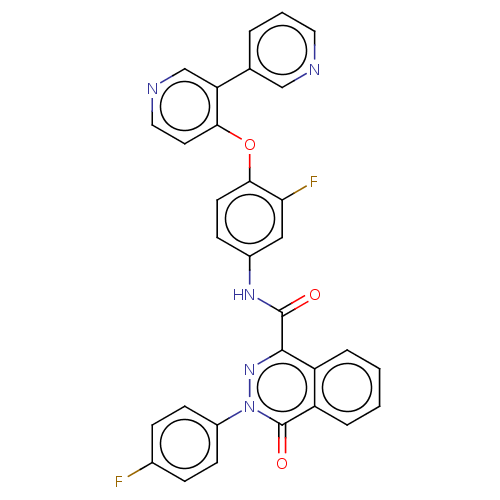 (CHEMBL3894838) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 52 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met (unknown origin) using poly (Glu, Tyr) 4:1 as substrate after 30 mins by HTRF assay | Eur J Med Chem 120: 37-50 (2016) Article DOI: 10.1016/j.ejmech.2016.04.062 BindingDB Entry DOI: 10.7270/Q2VM4F5Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50190909 (CHEMBL3980971) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 53 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met (unknown origin) using poly (Glu, Tyr) 4:1 as substrate after 30 mins by HTRF assay | Eur J Med Chem 120: 37-50 (2016) Article DOI: 10.1016/j.ejmech.2016.04.062 BindingDB Entry DOI: 10.7270/Q2VM4F5Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50147895 (CHEMBL3765124) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 54 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met kinase (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50147888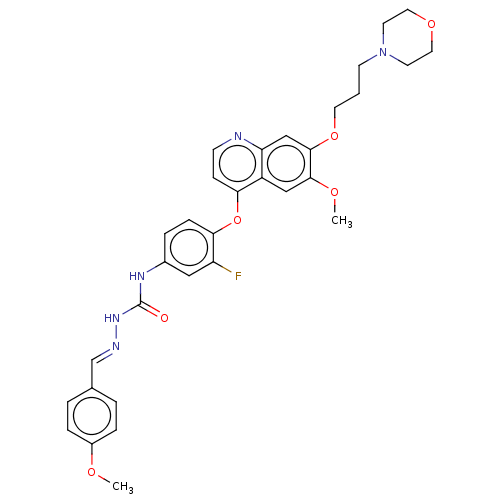 (CHEMBL3764184) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 56 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met kinase (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50191062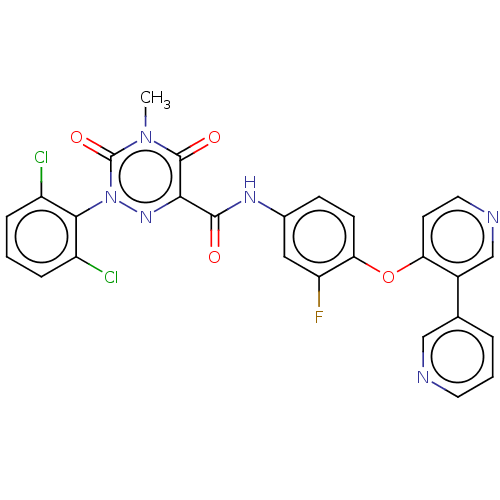 (CHEMBL3951223) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 56 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met (unknown origin) using poly (Glu, Tyr) 4:1 as substrate after 30 mins by HTRF assay | Eur J Med Chem 120: 37-50 (2016) Article DOI: 10.1016/j.ejmech.2016.04.062 BindingDB Entry DOI: 10.7270/Q2VM4F5Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50147919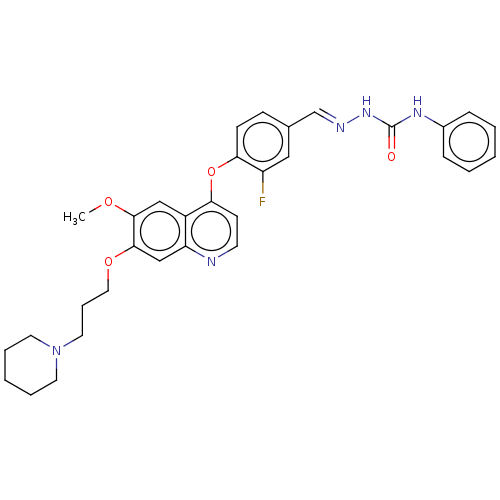 (CHEMBL3765283) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 63 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met kinase (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50147891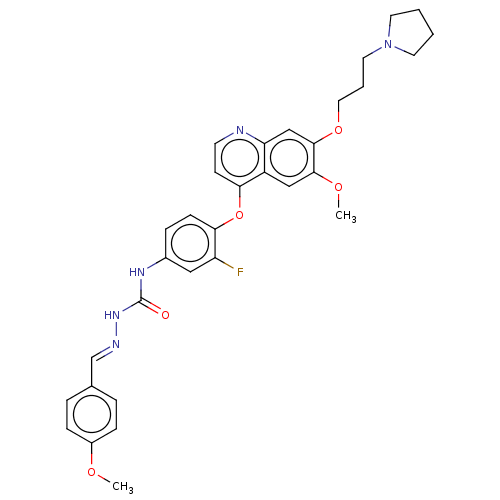 (CHEMBL3764320) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 72 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met kinase (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50190910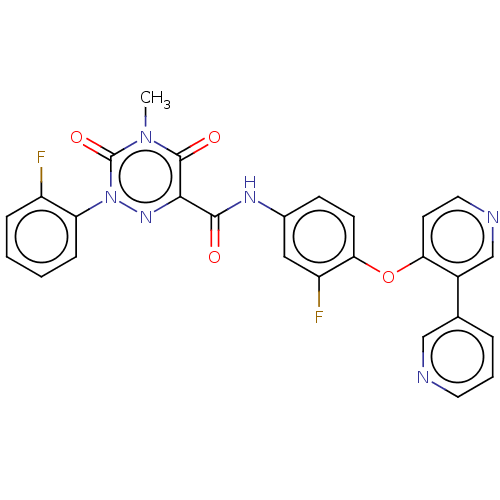 (CHEMBL3941430) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 85 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met (unknown origin) using poly (Glu, Tyr) 4:1 as substrate after 30 mins by HTRF assay | Eur J Med Chem 120: 37-50 (2016) Article DOI: 10.1016/j.ejmech.2016.04.062 BindingDB Entry DOI: 10.7270/Q2VM4F5Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50191070 (CHEMBL3969959) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 89 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met (unknown origin) using poly (Glu, Tyr) 4:1 as substrate after 30 mins by HTRF assay | Eur J Med Chem 120: 37-50 (2016) Article DOI: 10.1016/j.ejmech.2016.04.062 BindingDB Entry DOI: 10.7270/Q2VM4F5Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50191066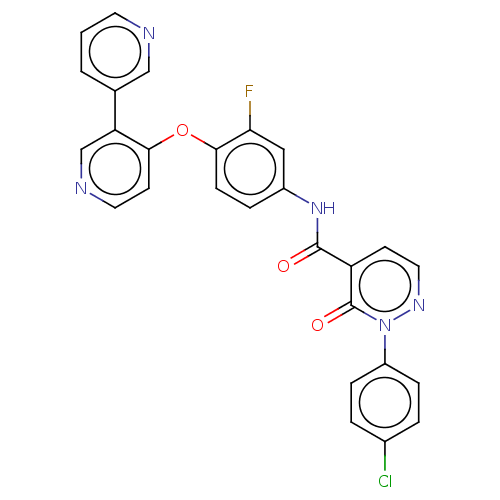 (CHEMBL3946333) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met (unknown origin) using poly (Glu, Tyr) 4:1 as substrate after 30 mins by HTRF assay | Eur J Med Chem 120: 37-50 (2016) Article DOI: 10.1016/j.ejmech.2016.04.062 BindingDB Entry DOI: 10.7270/Q2VM4F5Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50190811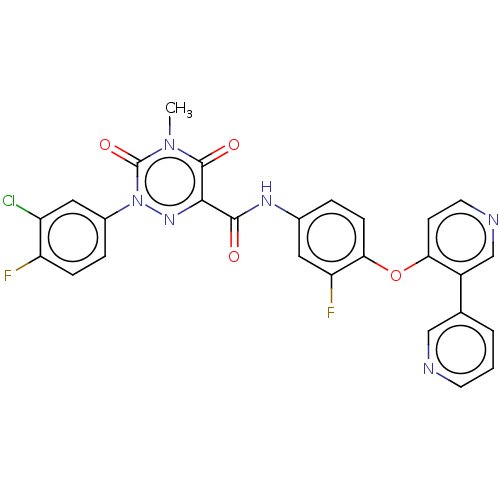 (CHEMBL3923500) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 103 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met (unknown origin) using poly (Glu, Tyr) 4:1 as substrate after 30 mins by HTRF assay | Eur J Med Chem 120: 37-50 (2016) Article DOI: 10.1016/j.ejmech.2016.04.062 BindingDB Entry DOI: 10.7270/Q2VM4F5Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50191071 (CHEMBL3899656) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 106 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met (unknown origin) using poly (Glu, Tyr) 4:1 as substrate after 30 mins by HTRF assay | Eur J Med Chem 120: 37-50 (2016) Article DOI: 10.1016/j.ejmech.2016.04.062 BindingDB Entry DOI: 10.7270/Q2VM4F5Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50147922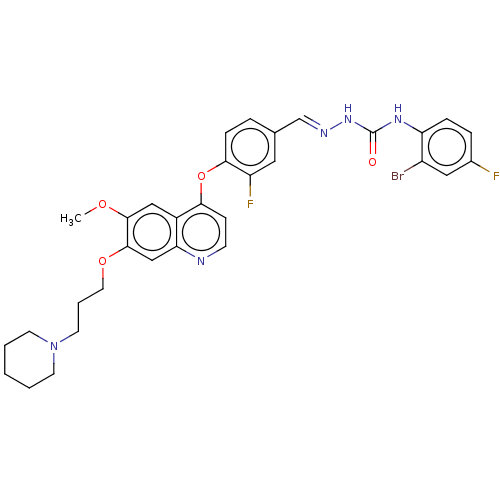 (CHEMBL3763970) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 121 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-Met kinase (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mast/stem cell growth factor receptor Kit (Homo sapiens (Human)) | BDBM50147893 (CHEMBL3764279) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 163 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of c-kit (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vascular endothelial growth factor receptor 2 (Homo sapiens (Human)) | BDBM50147893 (CHEMBL3764279) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 210 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of KDR (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| N-lysine methyltransferase KMT5A (Homo sapiens (Human)) | BDBM50106497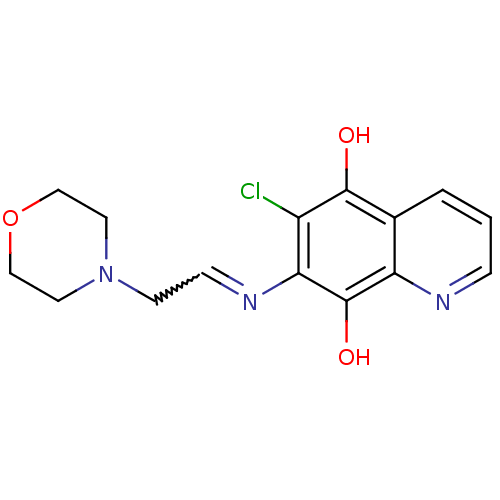 (6-Chloro-7-(2-morpholin-4-yl-ethylamino)-quinoline...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 210 | n/a | n/a | n/a | n/a | n/a | n/a |
The Center for Combinatorial Chemistry and Drug Discovery of Jilin University Curated by ChEMBL | Assay Description Inhibition of N-terminal His6-tagged human SETD8 (191 to 352 residues) expressed in Escherichia coli Rosseta-2(DE3) cells using histone peptide subst... | Bioorg Med Chem Lett 26: 4436-4440 (2016) Article DOI: 10.1016/j.bmcl.2016.08.004 BindingDB Entry DOI: 10.7270/Q27H1P3Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Platelet-derived growth factor receptor alpha (Homo sapiens (Human)) | BDBM50147893 (CHEMBL3764279) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 212 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of PDGFR-alpha (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Receptor-type tyrosine-protein kinase FLT3 (Homo sapiens (Human)) | BDBM50147893 (CHEMBL3764279) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 367 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of Flt-3 (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| N-lysine methyltransferase KMT5A (Homo sapiens (Human)) | BDBM50075100 (CHEMBL3414625) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
The Center for Combinatorial Chemistry and Drug Discovery of Jilin University Curated by ChEMBL | Assay Description Inhibition of N-terminal His6-tagged human SETD8 (191 to 352 residues) expressed in Escherichia coli Rosseta-2(DE3) cells using histone peptide subst... | Bioorg Med Chem Lett 26: 4436-4440 (2016) Article DOI: 10.1016/j.bmcl.2016.08.004 BindingDB Entry DOI: 10.7270/Q27H1P3Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| N-lysine methyltransferase KMT5A (Homo sapiens (Human)) | BDBM50075099 (CHEMBL580421 | TCMDC-124224) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
The Center for Combinatorial Chemistry and Drug Discovery of Jilin University Curated by ChEMBL | Assay Description Inhibition of N-terminal His6-tagged human SETD8 (191 to 352 residues) expressed in Escherichia coli Rosseta-2(DE3) cells using histone peptide subst... | Bioorg Med Chem Lett 26: 4436-4440 (2016) Article DOI: 10.1016/j.bmcl.2016.08.004 BindingDB Entry DOI: 10.7270/Q27H1P3Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| N-lysine methyltransferase KMT5A (Homo sapiens (Human)) | BDBM50051116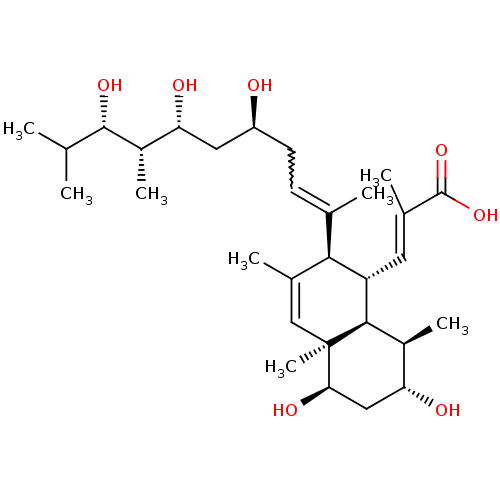 (CHEMBL3318284) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 6.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Center for Combinatorial Chemistry and Drug Discovery of Jilin University Curated by ChEMBL | Assay Description Inhibition of SETD8 (unknown origin) using biotin-labeled histone H4 (1 to 24 residues) as substrate incubated for 1 hr in presence of [3H-Me]SAM by ... | Bioorg Med Chem Lett 26: 4436-4440 (2016) Article DOI: 10.1016/j.bmcl.2016.08.004 BindingDB Entry DOI: 10.7270/Q27H1P3Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| N-lysine methyltransferase KMT5A (Homo sapiens (Human)) | BDBM50536815 (CHEMBL4568659) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Center for Combinatorial Chemistry and Drug Discovery of Jilin University Curated by ChEMBL | Assay Description Inhibition of SETD8 (unknown origin) using biotin-labeled histone H4 (1 to 24 residues) as substrate in presence of [3H-Me]SAM by scintillation proxi... | Bioorg Med Chem Lett 26: 4436-4440 (2016) Article DOI: 10.1016/j.bmcl.2016.08.004 BindingDB Entry DOI: 10.7270/Q27H1P3Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50147893 (CHEMBL3764279) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of EGFR (unknown origin) using poly(glu,Tyr)4:1 as substrate incubated for 30 mins by homogeneous time-resolved fluorescence assay | Bioorg Med Chem 24: 1331-45 (2016) Article DOI: 10.1016/j.bmc.2016.02.003 BindingDB Entry DOI: 10.7270/Q2V989XB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| N-lysine methyltransferase KMT5A (Homo sapiens (Human)) | BDBM50536813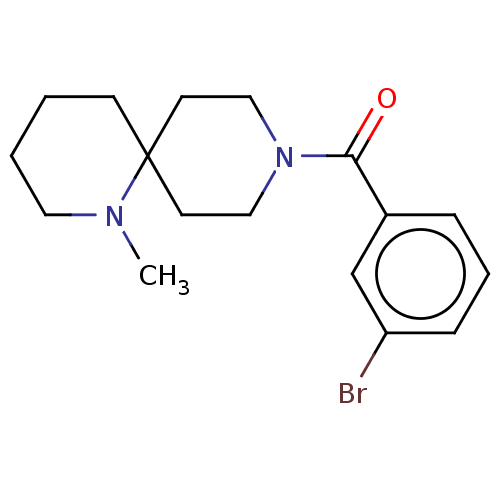 (CHEMBL4556920) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
The Center for Combinatorial Chemistry and Drug Discovery of Jilin University Curated by ChEMBL | Assay Description Inhibition of SETD8 (unknown origin) using biotin-labeled histone H4 (1 to 24 residues) as substrate in presence of [3H-Me]SAM by scintillation proxi... | Bioorg Med Chem Lett 26: 4436-4440 (2016) Article DOI: 10.1016/j.bmcl.2016.08.004 BindingDB Entry DOI: 10.7270/Q27H1P3Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| N-lysine methyltransferase KMT5A (Homo sapiens (Human)) | BDBM50536814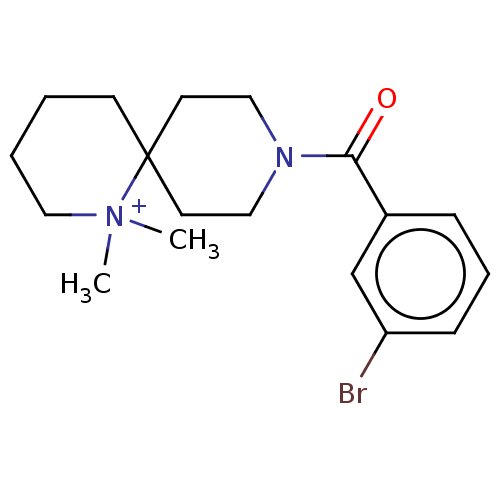 (CHEMBL4483751) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
The Center for Combinatorial Chemistry and Drug Discovery of Jilin University Curated by ChEMBL | Assay Description Inhibition of SETD8 (unknown origin) using biotin-labeled histone H4 (1 to 24 residues) as substrate in presence of [3H-Me]SAM by scintillation proxi... | Bioorg Med Chem Lett 26: 4436-4440 (2016) Article DOI: 10.1016/j.bmcl.2016.08.004 BindingDB Entry DOI: 10.7270/Q27H1P3Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| N-lysine methyltransferase KMT5A (Homo sapiens (Human)) | BDBM50075100 (CHEMBL3414625) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | <1.00E+3 | n/a | n/a | n/a | n/a |
The Center for Combinatorial Chemistry and Drug Discovery of Jilin University Curated by ChEMBL | Assay Description Inhibition of SETD8 in HEK293T cells assessed as reduction in H4K20me level after 2 to 3 days by Western blot analysis | Bioorg Med Chem Lett 26: 4436-4440 (2016) Article DOI: 10.1016/j.bmcl.2016.08.004 BindingDB Entry DOI: 10.7270/Q27H1P3Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nuclear receptor subfamily 4 group A member 1 (Homo sapiens (Human)) | BDBM50570058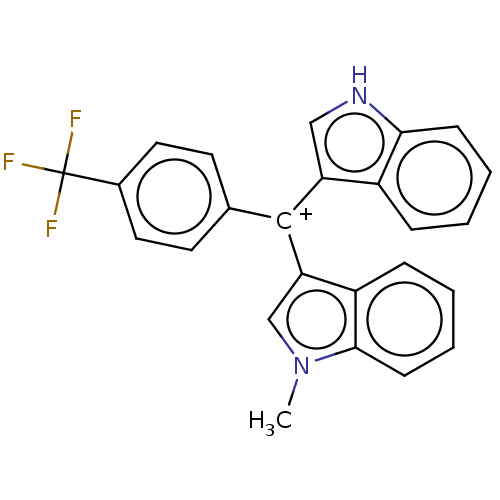 (CHEMBL4867199) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Binding affinity to recombinant human N-terminal His-tagged Nur77-LBD (367 to 598 residues) expressed in Escherichia coli BL21 DE3 by surface plasmon... | Citation and Details Article DOI: 10.1016/j.ejmech.2020.113020 BindingDB Entry DOI: 10.7270/Q23F4TF9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| N-lysine methyltransferase KMT5A (Homo sapiens (Human)) | BDBM50106497 (6-Chloro-7-(2-morpholin-4-yl-ethylamino)-quinoline...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | <5.00E+3 | n/a | n/a | n/a | n/a |
The Center for Combinatorial Chemistry and Drug Discovery of Jilin University Curated by ChEMBL | Assay Description Inhibition of SETD8 in HEK293T cells assessed as reduction in H4K20me level after 2 to 3 days by Western blot analysis | Bioorg Med Chem Lett 26: 4436-4440 (2016) Article DOI: 10.1016/j.bmcl.2016.08.004 BindingDB Entry DOI: 10.7270/Q27H1P3Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nuclear receptor subfamily 4 group A member 1 (Homo sapiens (Human)) | BDBM50570056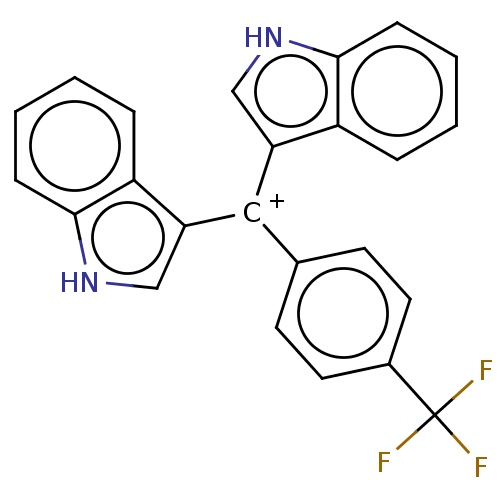 (CHEMBL4877512) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | 217 | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Binding affinity to recombinant human N-terminal His-tagged Nur77-LBD (367 to 598 residues) expressed in Escherichia coli BL21 DE3 by flourescence qu... | Citation and Details Article DOI: 10.1016/j.ejmech.2020.113020 BindingDB Entry DOI: 10.7270/Q23F4TF9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| N-lysine methyltransferase KMT5A (Homo sapiens (Human)) | BDBM50075099 (CHEMBL580421 | TCMDC-124224) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | <5.00E+3 | n/a | n/a | n/a | n/a |
The Center for Combinatorial Chemistry and Drug Discovery of Jilin University Curated by ChEMBL | Assay Description Inhibition of SETD8 in HEK293T cells assessed as reduction in H4K20me level after 2 to 3 days by Western blot analysis | Bioorg Med Chem Lett 26: 4436-4440 (2016) Article DOI: 10.1016/j.bmcl.2016.08.004 BindingDB Entry DOI: 10.7270/Q27H1P3Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nuclear receptor subfamily 4 group A member 1 (Homo sapiens (Human)) | BDBM50570058 (CHEMBL4867199) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | 66 | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Binding affinity to recombinant human N-terminal His-tagged Nur77-LBD (367 to 598 residues) expressed in Escherichia coli BL21 DE3 by flourescence qu... | Citation and Details Article DOI: 10.1016/j.ejmech.2020.113020 BindingDB Entry DOI: 10.7270/Q23F4TF9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nuclear receptor subfamily 4 group A member 1 (Homo sapiens (Human)) | BDBM50570056 (CHEMBL4877512) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Binding affinity to recombinant human N-terminal His-tagged Nur77-LBD (367 to 598 residues) expressed in Escherichia coli BL21 DE3 by surface plasmon... | Citation and Details Article DOI: 10.1016/j.ejmech.2020.113020 BindingDB Entry DOI: 10.7270/Q23F4TF9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nuclear receptor subfamily 4 group A member 1 (Homo sapiens (Human)) | BDBM50570057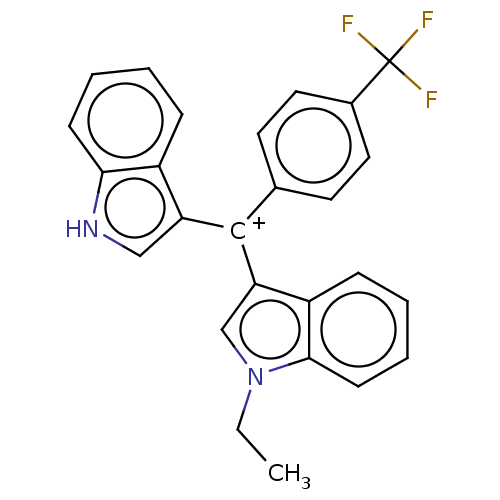 (CHEMBL4857769) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Binding affinity to recombinant human N-terminal His-tagged Nur77-LBD (367 to 598 residues) expressed in Escherichia coli BL21 DE3 by surface plasmon... | Citation and Details Article DOI: 10.1016/j.ejmech.2020.113020 BindingDB Entry DOI: 10.7270/Q23F4TF9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 51 total ) | Next | Last >> |