 Found 102 hits with Last Name = 'clemens-smith' and Initial = 'a'
Found 102 hits with Last Name = 'clemens-smith' and Initial = 'a' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Glutamate receptor ionotropic, kainate 5
(Homo sapiens (Human)) | BDBM86754
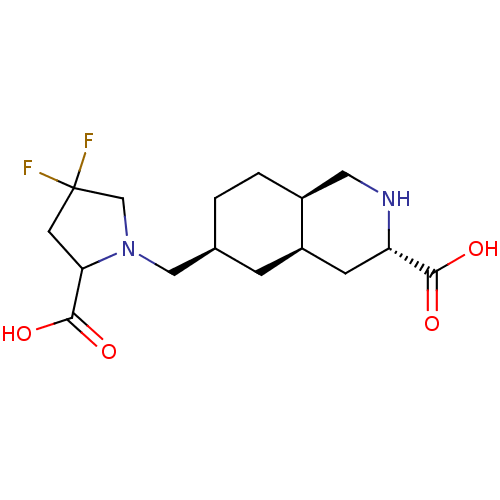
(LY 466195 | LY-466195)Show SMILES OC(=O)C1CC(F)(F)CN1C[C@H]1CC[C@H]2CN[C@@H](C[C@H]2C1)C(O)=O |r| Show InChI InChI=1S/C16H24F2N2O4/c17-16(18)5-13(15(23)24)20(8-16)7-9-1-2-10-6-19-12(14(21)22)4-11(10)3-9/h9-13,19H,1-8H2,(H,21,22)(H,23,24)/t9-,10-,11+,12-,13?/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor 2
(Homo sapiens (Human)) | BDBM50207594
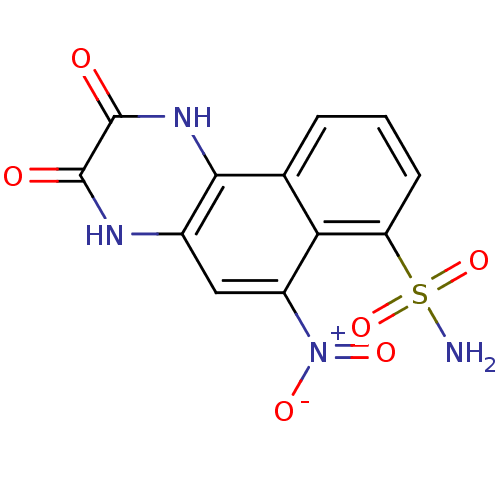
(2,3-Dihydroxy-6-nitro-benzo[f]quinoxaline-7-sulfon...)Show SMILES NS(=O)(=O)c1cccc2c1c(cc1[nH]c(=O)c(=O)[nH]c21)[N+]([O-])=O Show InChI InChI=1S/C12H8N4O6S/c13-23(21,22)8-3-1-2-5-9(8)7(16(19)20)4-6-10(5)15-12(18)11(17)14-6/h1-4H,(H,14,17)(H,15,18)(H2,13,21,22) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human Ionotropic glutamate receptor AMPA 2 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glutamate receptor 4
(Homo sapiens (Human)) | BDBM50207594

(2,3-Dihydroxy-6-nitro-benzo[f]quinoxaline-7-sulfon...)Show SMILES NS(=O)(=O)c1cccc2c1c(cc1[nH]c(=O)c(=O)[nH]c21)[N+]([O-])=O Show InChI InChI=1S/C12H8N4O6S/c13-23(21,22)8-3-1-2-5-9(8)7(16(19)20)4-6-10(5)15-12(18)11(17)14-6/h1-4H,(H,14,17)(H,15,18)(H2,13,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human Ionotropic glutamate receptor AMPA 4 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | |
Glutamate receptor 2
(Homo sapiens (Human)) | BDBM86750
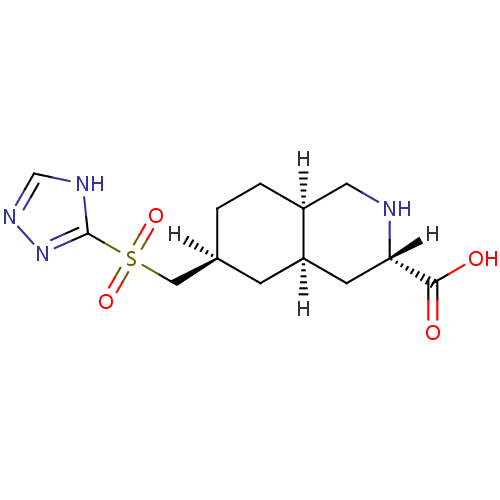
(LY 302679 | LY-302679)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](C[S](=O)(=O)c3nnc[nH]3)CC[C@H]2CN1 Show InChI InChI=1S/C13H20N4O4S/c18-12(19)11-4-10-3-8(1-2-9(10)5-14-11)6-22(20,21)13-15-7-16-17-13/h7-11,14H,1-6H2,(H,18,19)(H,15,16,17)/t8-,9-,10+,11-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 820 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor 3
(Homo sapiens (Human)) | BDBM50207594

(2,3-Dihydroxy-6-nitro-benzo[f]quinoxaline-7-sulfon...)Show SMILES NS(=O)(=O)c1cccc2c1c(cc1[nH]c(=O)c(=O)[nH]c21)[N+]([O-])=O Show InChI InChI=1S/C12H8N4O6S/c13-23(21,22)8-3-1-2-5-9(8)7(16(19)20)4-6-10(5)15-12(18)11(17)14-6/h1-4H,(H,14,17)(H,15,18)(H2,13,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human Ionotropic glutamate receptor AMPA 3 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glutamate receptor ionotropic, kainate 5
(Homo sapiens (Human)) | BDBM86749

(LY 457691)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@H](CC[C@H]2CN1)Nc1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C17H22N6O2/c24-17(25)15-8-11-7-12(6-5-10(11)9-18-15)19-14-4-2-1-3-13(14)16-20-22-23-21-16/h1-4,10-12,15,18-19H,5-9H2,(H,24,25)(H,20,21,22,23)/t10-,11+,12-,15-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.55E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor 1
(Homo sapiens (Human)) | BDBM50207594

(2,3-Dihydroxy-6-nitro-benzo[f]quinoxaline-7-sulfon...)Show SMILES NS(=O)(=O)c1cccc2c1c(cc1[nH]c(=O)c(=O)[nH]c21)[N+]([O-])=O Show InChI InChI=1S/C12H8N4O6S/c13-23(21,22)8-3-1-2-5-9(8)7(16(19)20)4-6-10(5)15-12(18)11(17)14-6/h1-4H,(H,14,17)(H,15,18)(H2,13,21,22) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human Ionotropic glutamate receptor AMPA 1 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glutamate receptor ionotropic, kainate 5
(Homo sapiens (Human)) | BDBM86755

(LY 458545 | LY-458545)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@H](CC[C@H]2CN1)Oc1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C17H21N5O3/c23-17(24)14-8-11-7-12(6-5-10(11)9-18-14)25-15-4-2-1-3-13(15)16-19-21-22-20-16/h1-4,10-12,14,18H,5-9H2,(H,23,24)(H,19,20,21,22)/t10-,11+,12-,14-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.69E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 5
(Homo sapiens (Human)) | BDBM86752
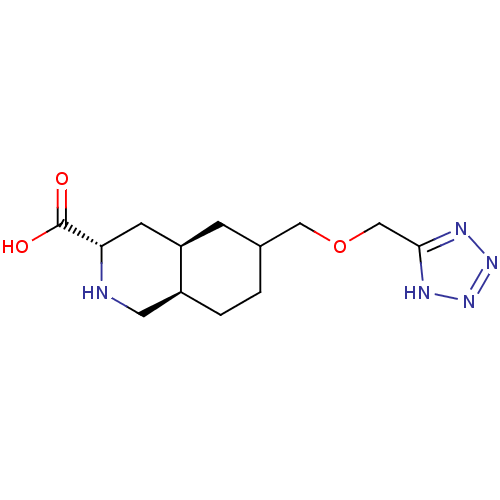
(LY 294486 | LY-294486)Show SMILES OC(=O)[C@@H]1C[C@H]2CC(COCc3nnn[nH]3)CC[C@H]2CN1 Show InChI InChI=1S/C13H21N5O3/c19-13(20)11-4-10-3-8(1-2-9(10)5-14-11)6-21-7-12-15-17-18-16-12/h8-11,14H,1-7H2,(H,19,20)(H,15,16,17,18)/t8?,9-,10+,11-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.06E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor 2
(Homo sapiens (Human)) | BDBM86751
(CHEMBL14935 | LY 293558 | LY-293558)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](CCc3nnn[nH]3)CC[C@H]2CN1 Show InChI InChI=1S/C13H21N5O2/c19-13(20)11-6-10-5-8(1-3-9(10)7-14-11)2-4-12-15-17-18-16-12/h8-11,14H,1-7H2,(H,19,20)(H,15,16,17,18)/t8-,9+,10-,11+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor 2
(Homo sapiens (Human)) | BDBM86751

(CHEMBL14935 | LY 293558 | LY-293558)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](CCc3nnn[nH]3)CC[C@H]2CN1 Show InChI InChI=1S/C13H21N5O2/c19-13(20)11-6-10-5-8(1-3-9(10)7-14-11)2-4-12-15-17-18-16-12/h8-11,14H,1-7H2,(H,19,20)(H,15,16,17,18)/t8-,9+,10-,11+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human Ionotropic glutamate receptor AMPA 2 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 5
(Homo sapiens (Human)) | BDBM86750

(LY 302679 | LY-302679)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](C[S](=O)(=O)c3nnc[nH]3)CC[C@H]2CN1 Show InChI InChI=1S/C13H20N4O4S/c18-12(19)11-4-10-3-8(1-2-9(10)5-14-11)6-22(20,21)13-15-7-16-17-13/h7-11,14H,1-6H2,(H,18,19)(H,15,16,17)/t8-,9-,10+,11-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.41E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 5
(Homo sapiens (Human)) | BDBM50168962

((3S,4aR,6S,8aR)-6-(4-Carboxy-benzyl)-decahydro-iso...)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](Cc3ccc(cc3)C(O)=O)CC[C@H]2CN1 Show InChI InChI=1S/C18H23NO4/c20-17(21)13-4-1-11(2-5-13)7-12-3-6-14-10-19-16(18(22)23)9-15(14)8-12/h1-2,4-5,12,14-16,19H,3,6-10H2,(H,20,21)(H,22,23)/t12-,14+,15-,16+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 5
(Homo sapiens (Human)) | BDBM86751

(CHEMBL14935 | LY 293558 | LY-293558)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](CCc3nnn[nH]3)CC[C@H]2CN1 Show InChI InChI=1S/C13H21N5O2/c19-13(20)11-6-10-5-8(1-3-9(10)7-14-11)2-4-12-15-17-18-16-12/h8-11,14H,1-7H2,(H,19,20)(H,15,16,17,18)/t8-,9+,10-,11+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor 2
(Homo sapiens (Human)) | BDBM86749

(LY 457691)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@H](CC[C@H]2CN1)Nc1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C17H22N6O2/c24-17(25)15-8-11-7-12(6-5-10(11)9-18-15)19-14-4-2-1-3-13(14)16-20-22-23-21-16/h1-4,10-12,15,18-19H,5-9H2,(H,24,25)(H,20,21,22,23)/t10-,11+,12-,15-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 5.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 5
(Homo sapiens (Human)) | BDBM86756

(LY 467711 | LY-467711)Show SMILES CCC(C)OC(=O)[C@@H]1C[C@H]2C[C@H](CC[C@H]2CN1)Oc1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C21H29N5O3/c1-3-13(2)28-21(27)18-11-15-10-16(9-8-14(15)12-22-18)29-19-7-5-4-6-17(19)20-23-25-26-24-20/h4-7,13-16,18,22H,3,8-12H2,1-2H3,(H,23,24,25,26)/t13?,14-,15+,16-,18-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor 2
(Homo sapiens (Human)) | BDBM86755

(LY 458545 | LY-458545)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@H](CC[C@H]2CN1)Oc1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C17H21N5O3/c23-17(24)14-8-11-7-12(6-5-10(11)9-18-14)25-15-4-2-1-3-13(15)16-19-21-22-20-16/h1-4,10-12,14,18H,5-9H2,(H,23,24)(H,19,20,21,22)/t10-,11+,12-,14-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 8.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 3
(Homo sapiens (Human)) | BDBM86754

(LY 466195 | LY-466195)Show SMILES OC(=O)C1CC(F)(F)CN1C[C@H]1CC[C@H]2CN[C@@H](C[C@H]2C1)C(O)=O |r| Show InChI InChI=1S/C16H24F2N2O4/c17-16(18)5-13(15(23)24)20(8-16)7-9-1-2-10-6-19-12(14(21)22)4-11(10)3-9/h9-13,19H,1-8H2,(H,21,22)(H,23,24)/t9-,10-,11+,12-,13?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 8.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor 1
(Homo sapiens (Human)) | BDBM86751

(CHEMBL14935 | LY 293558 | LY-293558)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](CCc3nnn[nH]3)CC[C@H]2CN1 Show InChI InChI=1S/C13H21N5O2/c19-13(20)11-6-10-5-8(1-3-9(10)7-14-11)2-4-12-15-17-18-16-12/h8-11,14H,1-7H2,(H,19,20)(H,15,16,17,18)/t8-,9+,10-,11+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 9.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human Ionotropic glutamate receptor AMPA 1 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | |
Glutamate receptor 1
(Homo sapiens (Human)) | BDBM50168962

((3S,4aR,6S,8aR)-6-(4-Carboxy-benzyl)-decahydro-iso...)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](Cc3ccc(cc3)C(O)=O)CC[C@H]2CN1 Show InChI InChI=1S/C18H23NO4/c20-17(21)13-4-1-11(2-5-13)7-12-3-6-14-10-19-16(18(22)23)9-15(14)8-12/h1-2,4-5,12,14-16,19H,3,6-10H2,(H,20,21)(H,22,23)/t12-,14+,15-,16+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor 1
(Homo sapiens (Human)) | BDBM86754

(LY 466195 | LY-466195)Show SMILES OC(=O)C1CC(F)(F)CN1C[C@H]1CC[C@H]2CN[C@@H](C[C@H]2C1)C(O)=O |r| Show InChI InChI=1S/C16H24F2N2O4/c17-16(18)5-13(15(23)24)20(8-16)7-9-1-2-10-6-19-12(14(21)22)4-11(10)3-9/h9-13,19H,1-8H2,(H,21,22)(H,23,24)/t9-,10-,11+,12-,13?/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor 2
(Homo sapiens (Human)) | BDBM86752

(LY 294486 | LY-294486)Show SMILES OC(=O)[C@@H]1C[C@H]2CC(COCc3nnn[nH]3)CC[C@H]2CN1 Show InChI InChI=1S/C13H21N5O3/c19-13(20)11-4-10-3-8(1-2-9(10)5-14-11)6-21-7-12-15-17-18-16-12/h8-11,14H,1-7H2,(H,19,20)(H,15,16,17,18)/t8?,9-,10+,11-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor 2
(Homo sapiens (Human)) | BDBM50168962

((3S,4aR,6S,8aR)-6-(4-Carboxy-benzyl)-decahydro-iso...)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](Cc3ccc(cc3)C(O)=O)CC[C@H]2CN1 Show InChI InChI=1S/C18H23NO4/c20-17(21)13-4-1-11(2-5-13)7-12-3-6-14-10-19-16(18(22)23)9-15(14)8-12/h1-2,4-5,12,14-16,19H,3,6-10H2,(H,20,21)(H,22,23)/t12-,14+,15-,16+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor 2
(Homo sapiens (Human)) | BDBM86754

(LY 466195 | LY-466195)Show SMILES OC(=O)C1CC(F)(F)CN1C[C@H]1CC[C@H]2CN[C@@H](C[C@H]2C1)C(O)=O |r| Show InChI InChI=1S/C16H24F2N2O4/c17-16(18)5-13(15(23)24)20(8-16)7-9-1-2-10-6-19-12(14(21)22)4-11(10)3-9/h9-13,19H,1-8H2,(H,21,22)(H,23,24)/t9-,10-,11+,12-,13?/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor 2
(Homo sapiens (Human)) | BDBM86756

(LY 467711 | LY-467711)Show SMILES CCC(C)OC(=O)[C@@H]1C[C@H]2C[C@H](CC[C@H]2CN1)Oc1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C21H29N5O3/c1-3-13(2)28-21(27)18-11-15-10-16(9-8-14(15)12-22-18)29-19-7-5-4-6-17(19)20-23-25-26-24-20/h4-7,13-16,18,22H,3,8-12H2,1-2H3,(H,23,24,25,26)/t13?,14-,15+,16-,18-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
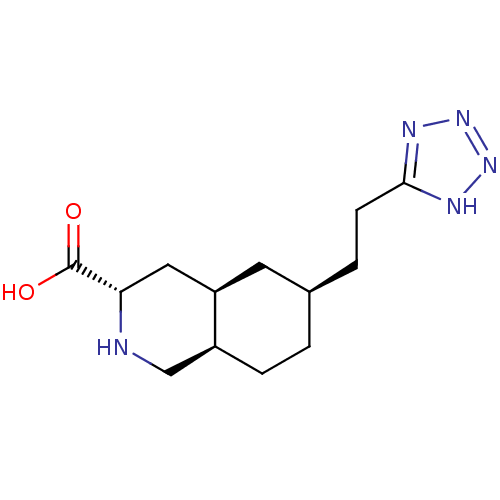
Glutamate receptor 2
(Homo sapiens (Human)) | BDBM86753
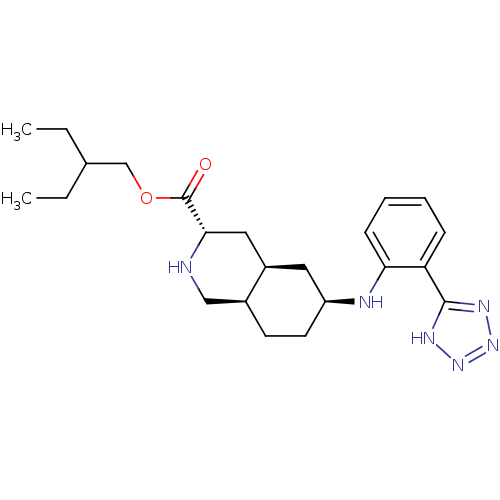
(LY 525327 | LY-525327)Show SMILES CCC(CC)COC(=O)[C@@H]1C[C@H]2C[C@H](CC[C@H]2CN1)Nc1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C23H34N6O2/c1-3-15(4-2)14-31-23(30)21-12-17-11-18(10-9-16(17)13-24-21)25-20-8-6-5-7-19(20)22-26-28-29-27-22/h5-8,15-18,21,24-25H,3-4,9-14H2,1-2H3,(H,26,27,28,29)/t16-,17+,18-,21-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor 4
(Homo sapiens (Human)) | BDBM86751

(CHEMBL14935 | LY 293558 | LY-293558)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](CCc3nnn[nH]3)CC[C@H]2CN1 Show InChI InChI=1S/C13H21N5O2/c19-13(20)11-6-10-5-8(1-3-9(10)7-14-11)2-4-12-15-17-18-16-12/h8-11,14H,1-7H2,(H,19,20)(H,15,16,17,18)/t8-,9+,10-,11+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor 4
(Homo sapiens (Human)) | BDBM50168962

((3S,4aR,6S,8aR)-6-(4-Carboxy-benzyl)-decahydro-iso...)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](Cc3ccc(cc3)C(O)=O)CC[C@H]2CN1 Show InChI InChI=1S/C18H23NO4/c20-17(21)13-4-1-11(2-5-13)7-12-3-6-14-10-19-16(18(22)23)9-15(14)8-12/h1-2,4-5,12,14-16,19H,3,6-10H2,(H,20,21)(H,22,23)/t12-,14+,15-,16+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor 4
(Homo sapiens (Human)) | BDBM86754

(LY 466195 | LY-466195)Show SMILES OC(=O)C1CC(F)(F)CN1C[C@H]1CC[C@H]2CN[C@@H](C[C@H]2C1)C(O)=O |r| Show InChI InChI=1S/C16H24F2N2O4/c17-16(18)5-13(15(23)24)20(8-16)7-9-1-2-10-6-19-12(14(21)22)4-11(10)3-9/h9-13,19H,1-8H2,(H,21,22)(H,23,24)/t9-,10-,11+,12-,13?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 5
(Homo sapiens (Human)) | BDBM86753

(LY 525327 | LY-525327)Show SMILES CCC(CC)COC(=O)[C@@H]1C[C@H]2C[C@H](CC[C@H]2CN1)Nc1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C23H34N6O2/c1-3-15(4-2)14-31-23(30)21-12-17-11-18(10-9-16(17)13-24-21)25-20-8-6-5-7-19(20)22-26-28-29-27-22/h5-8,15-18,21,24-25H,3-4,9-14H2,1-2H3,(H,26,27,28,29)/t16-,17+,18-,21-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 3
(Homo sapiens (Human)) | BDBM86751

(CHEMBL14935 | LY 293558 | LY-293558)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](CCc3nnn[nH]3)CC[C@H]2CN1 Show InChI InChI=1S/C13H21N5O2/c19-13(20)11-6-10-5-8(1-3-9(10)7-14-11)2-4-12-15-17-18-16-12/h8-11,14H,1-7H2,(H,19,20)(H,15,16,17,18)/t8-,9+,10-,11+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 3
(Homo sapiens (Human)) | BDBM50168962

((3S,4aR,6S,8aR)-6-(4-Carboxy-benzyl)-decahydro-iso...)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](Cc3ccc(cc3)C(O)=O)CC[C@H]2CN1 Show InChI InChI=1S/C18H23NO4/c20-17(21)13-4-1-11(2-5-13)7-12-3-6-14-10-19-16(18(22)23)9-15(14)8-12/h1-2,4-5,12,14-16,19H,3,6-10H2,(H,20,21)(H,22,23)/t12-,14+,15-,16+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Homo sapiens (Human)) | BDBM86751

(CHEMBL14935 | LY 293558 | LY-293558)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](CCc3nnn[nH]3)CC[C@H]2CN1 Show InChI InChI=1S/C13H21N5O2/c19-13(20)11-6-10-5-8(1-3-9(10)7-14-11)2-4-12-15-17-18-16-12/h8-11,14H,1-7H2,(H,19,20)(H,15,16,17,18)/t8-,9+,10-,11+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Homo sapiens (Human)) | BDBM50168962

((3S,4aR,6S,8aR)-6-(4-Carboxy-benzyl)-decahydro-iso...)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](Cc3ccc(cc3)C(O)=O)CC[C@H]2CN1 Show InChI InChI=1S/C18H23NO4/c20-17(21)13-4-1-11(2-5-13)7-12-3-6-14-10-19-16(18(22)23)9-15(14)8-12/h1-2,4-5,12,14-16,19H,3,6-10H2,(H,20,21)(H,22,23)/t12-,14+,15-,16+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Homo sapiens (Human)) | BDBM86754

(LY 466195 | LY-466195)Show SMILES OC(=O)C1CC(F)(F)CN1C[C@H]1CC[C@H]2CN[C@@H](C[C@H]2C1)C(O)=O |r| Show InChI InChI=1S/C16H24F2N2O4/c17-16(18)5-13(15(23)24)20(8-16)7-9-1-2-10-6-19-12(14(21)22)4-11(10)3-9/h9-13,19H,1-8H2,(H,21,22)(H,23,24)/t9-,10-,11+,12-,13?/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 318: 772-81 (2006)
Article DOI: 10.1124/jpet.106.101428
BindingDB Entry DOI: 10.7270/Q2NP230D |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Homo sapiens (Human)) | BDBM50207594

(2,3-Dihydroxy-6-nitro-benzo[f]quinoxaline-7-sulfon...)Show SMILES NS(=O)(=O)c1cccc2c1c(cc1[nH]c(=O)c(=O)[nH]c21)[N+]([O-])=O Show InChI InChI=1S/C12H8N4O6S/c13-23(21,22)8-3-1-2-5-9(8)7(16(19)20)4-6-10(5)15-12(18)11(17)14-6/h1-4H,(H,14,17)(H,15,18)(H2,13,21,22) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 1.32E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human AMPA receptor Ionotropic glutamate receptor ionotropic kainate 2 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | |
Glutamate receptor 3
(Homo sapiens (Human)) | BDBM86751

(CHEMBL14935 | LY 293558 | LY-293558)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](CCc3nnn[nH]3)CC[C@H]2CN1 Show InChI InChI=1S/C13H21N5O2/c19-13(20)11-6-10-5-8(1-3-9(10)7-14-11)2-4-12-15-17-18-16-12/h8-11,14H,1-7H2,(H,19,20)(H,15,16,17,18)/t8-,9+,10-,11+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 3.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human Ionotropic glutamate receptor AMPA 3 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | |
Glutamate receptor 4
(Homo sapiens (Human)) | BDBM86751

(CHEMBL14935 | LY 293558 | LY-293558)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](CCc3nnn[nH]3)CC[C@H]2CN1 Show InChI InChI=1S/C13H21N5O2/c19-13(20)11-6-10-5-8(1-3-9(10)7-14-11)2-4-12-15-17-18-16-12/h8-11,14H,1-7H2,(H,19,20)(H,15,16,17,18)/t8-,9+,10-,11+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human Ionotropic glutamate receptor AMPA 4 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Homo sapiens (Human)) | BDBM50207594

(2,3-Dihydroxy-6-nitro-benzo[f]quinoxaline-7-sulfon...)Show SMILES NS(=O)(=O)c1cccc2c1c(cc1[nH]c(=O)c(=O)[nH]c21)[N+]([O-])=O Show InChI InChI=1S/C12H8N4O6S/c13-23(21,22)8-3-1-2-5-9(8)7(16(19)20)4-6-10(5)15-12(18)11(17)14-6/h1-4H,(H,14,17)(H,15,18)(H2,13,21,22) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]KA to recombinant human KA receptor Ionotropic glutamate receptor ionotropic kainate 2 expressed in EK 293 cell me... |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Homo sapiens (Human)) | BDBM86751

(CHEMBL14935 | LY 293558 | LY-293558)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](CCc3nnn[nH]3)CC[C@H]2CN1 Show InChI InChI=1S/C13H21N5O2/c19-13(20)11-6-10-5-8(1-3-9(10)7-14-11)2-4-12-15-17-18-16-12/h8-11,14H,1-7H2,(H,19,20)(H,15,16,17,18)/t8-,9+,10-,11+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human AMPA receptor Ionotropic glutamate receptor ionotropic kainate 2 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Homo sapiens (Human)) | BDBM86751

(CHEMBL14935 | LY 293558 | LY-293558)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](CCc3nnn[nH]3)CC[C@H]2CN1 Show InChI InChI=1S/C13H21N5O2/c19-13(20)11-6-10-5-8(1-3-9(10)7-14-11)2-4-12-15-17-18-16-12/h8-11,14H,1-7H2,(H,19,20)(H,15,16,17,18)/t8-,9+,10-,11+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]KA to recombinant human KA receptor Ionotropic glutamate receptor ionotropic kainate 2 expressed in EK 293 cell me... |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | |
Glutamate receptor 2
(Homo sapiens (Human)) | BDBM50118701
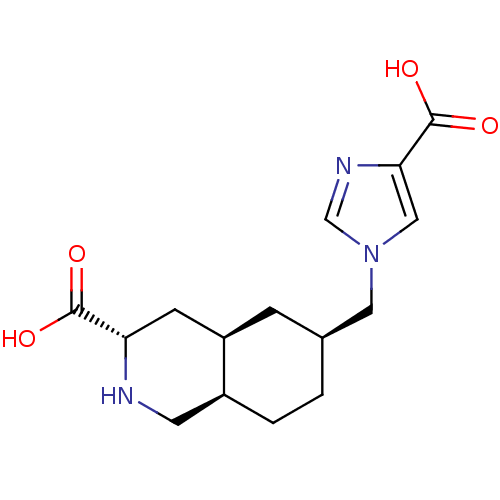
(6-(4-Carboxy-imidazol-1-ylmethyl)-decahydro-isoqui...)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](Cn3cnc(c3)C(O)=O)CC[C@H]2CN1 Show InChI InChI=1S/C15H21N3O4/c19-14(20)12-4-11-3-9(1-2-10(11)5-16-12)6-18-7-13(15(21)22)17-8-18/h7-12,16H,1-6H2,(H,19,20)(H,21,22)/t9-,10-,11+,12-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 1.17E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human Ionotropic glutamate receptor AMPA 2 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | |
Glutamate receptor 1
(Homo sapiens (Human)) | BDBM50118701

(6-(4-Carboxy-imidazol-1-ylmethyl)-decahydro-isoqui...)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](Cn3cnc(c3)C(O)=O)CC[C@H]2CN1 Show InChI InChI=1S/C15H21N3O4/c19-14(20)12-4-11-3-9(1-2-10(11)5-16-12)6-18-7-13(15(21)22)17-8-18/h7-12,16H,1-6H2,(H,19,20)(H,21,22)/t9-,10-,11+,12-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 1.34E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human Ionotropic glutamate receptor AMPA 1 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Homo sapiens (Human)) | BDBM50118701

(6-(4-Carboxy-imidazol-1-ylmethyl)-decahydro-isoqui...)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](Cn3cnc(c3)C(O)=O)CC[C@H]2CN1 Show InChI InChI=1S/C15H21N3O4/c19-14(20)12-4-11-3-9(1-2-10(11)5-16-12)6-18-7-13(15(21)22)17-8-18/h7-12,16H,1-6H2,(H,19,20)(H,21,22)/t9-,10-,11+,12-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 2.36E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]KA to recombinant human KA receptor Ionotropic glutamate receptor ionotropic kainate 2 expressed in EK 293 cell me... |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | |
Glutamate receptor 3
(Homo sapiens (Human)) | BDBM50118701

(6-(4-Carboxy-imidazol-1-ylmethyl)-decahydro-isoqui...)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](Cn3cnc(c3)C(O)=O)CC[C@H]2CN1 Show InChI InChI=1S/C15H21N3O4/c19-14(20)12-4-11-3-9(1-2-10(11)5-16-12)6-18-7-13(15(21)22)17-8-18/h7-12,16H,1-6H2,(H,19,20)(H,21,22)/t9-,10-,11+,12-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 2.47E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human Ionotropic glutamate receptor AMPA 3 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | |
Glutamate receptor 4
(Homo sapiens (Human)) | BDBM50118701

(6-(4-Carboxy-imidazol-1-ylmethyl)-decahydro-isoqui...)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](Cn3cnc(c3)C(O)=O)CC[C@H]2CN1 Show InChI InChI=1S/C15H21N3O4/c19-14(20)12-4-11-3-9(1-2-10(11)5-16-12)6-18-7-13(15(21)22)17-8-18/h7-12,16H,1-6H2,(H,19,20)(H,21,22)/t9-,10-,11+,12-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 2.62E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human Ionotropic glutamate receptor AMPA 4 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | |
Glutamate receptor ionotropic, kainate 2
(Homo sapiens (Human)) | BDBM50118701

(6-(4-Carboxy-imidazol-1-ylmethyl)-decahydro-isoqui...)Show SMILES OC(=O)[C@@H]1C[C@H]2C[C@@H](Cn3cnc(c3)C(O)=O)CC[C@H]2CN1 Show InChI InChI=1S/C15H21N3O4/c19-14(20)12-4-11-3-9(1-2-10(11)5-16-12)6-18-7-13(15(21)22)17-8-18/h7-12,16H,1-6H2,(H,19,20)(H,21,22)/t9-,10-,11+,12-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace binding of [3H]AMPA to recombinant human AMPA receptor Ionotropic glutamate receptor ionotropic kainate 2 |
J Med Chem 45: 4383-6 (2002)
BindingDB Entry DOI: 10.7270/Q2XP75N4 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.79E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Displacement of [3H]LSD from human 5HT6 receptor expressed in CHO cells after 120 mins by scintillation counting method |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1D
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >2.40E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Activity at human 5HT1D receptor |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1F
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >2.97E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Activity at human 5HT1F receptor |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data