

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mu-type opioid receptor (MOUSE) | BDBM50533716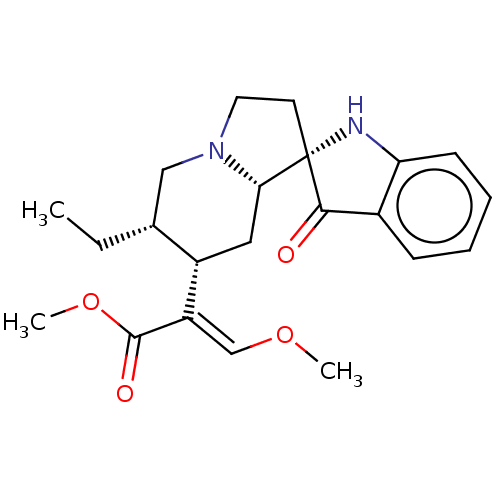 (CHEMBL4453504) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan Kettering Cancer Center Curated by ChEMBL | Assay Description Displacement of [125I]-IBNtxA from mouse mu opioid receptor-1 expressed in CHO cell membranes incubated for 90 mins | J Med Chem 59: 8381-97 (2016) Article DOI: 10.1021/acs.jmedchem.6b00748 BindingDB Entry DOI: 10.7270/Q27H1P2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuropeptide FF receptor 2 (Homo sapiens (Human)) | BDBM50029138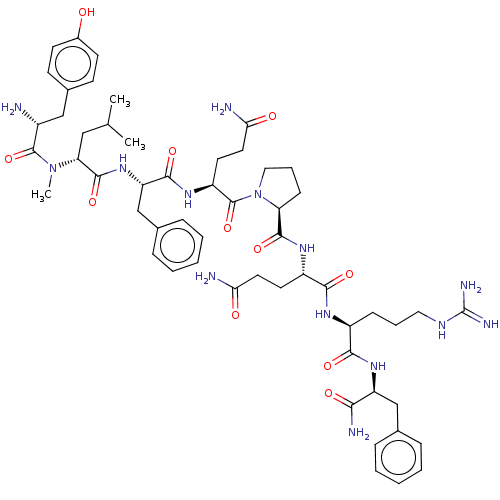 (CHEMBL3360830) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Mississippi Curated by ChEMBL | Assay Description Displacement of [3H]EYF from human NPFF2 receptor expressed in CHO cells after 1 hr by liquid scintillation counting analysis | J Med Chem 57: 8903-27 (2014) Article DOI: 10.1021/jm500989n BindingDB Entry DOI: 10.7270/Q2C24Z18 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071073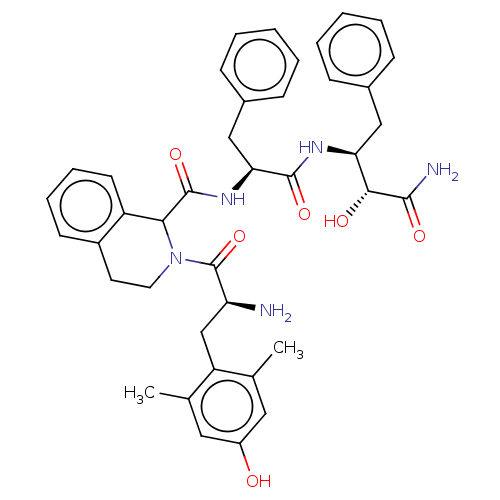 (CHEMBL3409763) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from rat cortex delta opioid receptor after 2.5 hrs by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (MOUSE) | BDBM50533717 (CHEMBL4449284) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan Kettering Cancer Center Curated by ChEMBL | Assay Description Displacement of [125I]-IBNtxA from mouse mu opioid receptor-1 expressed in CHO cell membranes incubated for 90 mins | J Med Chem 59: 8381-97 (2016) Article DOI: 10.1021/acs.jmedchem.6b00748 BindingDB Entry DOI: 10.7270/Q27H1P2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071072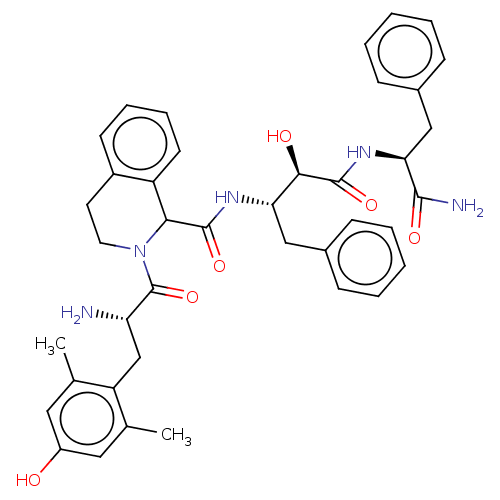 (CHEMBL3409762) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from rat cortex delta opioid receptor after 2.5 hrs by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (MOUSE) | BDBM50474150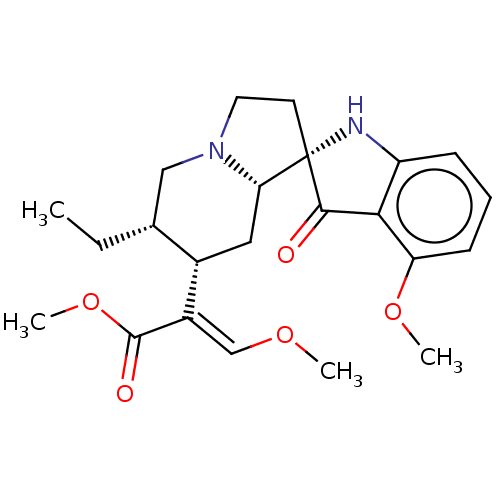 (CHEMBL58362) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem | PDB Article PubMed | 0.75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan Kettering Cancer Center Curated by ChEMBL | Assay Description Displacement of [125I]-IBNtxA from mouse mu opioid receptor-1 expressed in CHO cell membranes incubated for 90 mins | J Med Chem 59: 8381-97 (2016) Article DOI: 10.1021/acs.jmedchem.6b00748 BindingDB Entry DOI: 10.7270/Q27H1P2H | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Neuropeptide FF receptor 2 (Homo sapiens (Human)) | BDBM50029138 (CHEMBL3360830) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Mississippi Curated by ChEMBL | Assay Description Displacement of [125I]EYF from human NPFF2 receptor expressed in CHO cells after 1 hr by liquid scintillation counting analysis | J Med Chem 57: 8903-27 (2014) Article DOI: 10.1021/jm500989n BindingDB Entry DOI: 10.7270/Q2C24Z18 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50528969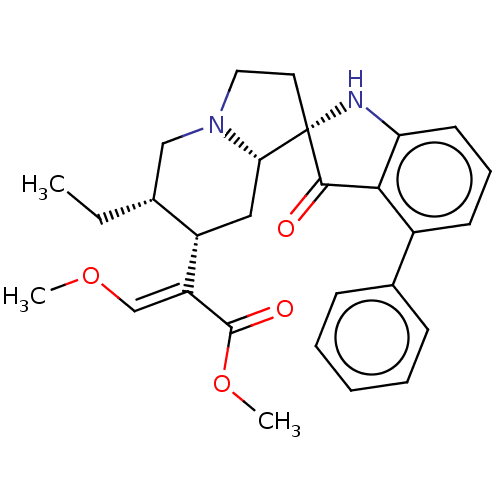 (CHEMBL4458337) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan Kettering Cancer Center Curated by ChEMBL | Assay Description Displacement of [125I]-IBNtxA from mouse delta opioid receptor-1 expressed in CHO cell membranes incubated for 90 mins | J Med Chem 59: 8381-97 (2016) Article DOI: 10.1021/acs.jmedchem.6b00748 BindingDB Entry DOI: 10.7270/Q27H1P2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071071 (CHEMBL3409761) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from rat cortex delta opioid receptor after 2.5 hrs by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (MOUSE) | BDBM50528969 (CHEMBL4458337) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.910 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan Kettering Cancer Center Curated by ChEMBL | Assay Description Displacement of [125I]-IBNtxA from mouse mu opioid receptor-1 expressed in CHO cell membranes incubated for 90 mins | J Med Chem 59: 8381-97 (2016) Article DOI: 10.1021/acs.jmedchem.6b00748 BindingDB Entry DOI: 10.7270/Q27H1P2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (MOUSE) | BDBM50533719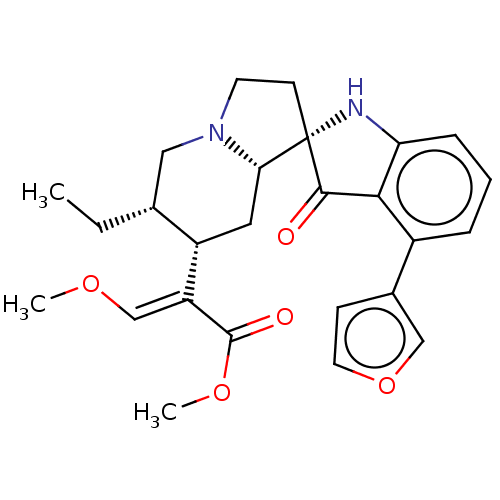 (CHEMBL4441457) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.940 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan Kettering Cancer Center Curated by ChEMBL | Assay Description Displacement of [125I]-IBNtxA from mouse mu opioid receptor-1 expressed in CHO cell membranes incubated for 90 mins | J Med Chem 59: 8381-97 (2016) Article DOI: 10.1021/acs.jmedchem.6b00748 BindingDB Entry DOI: 10.7270/Q27H1P2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071070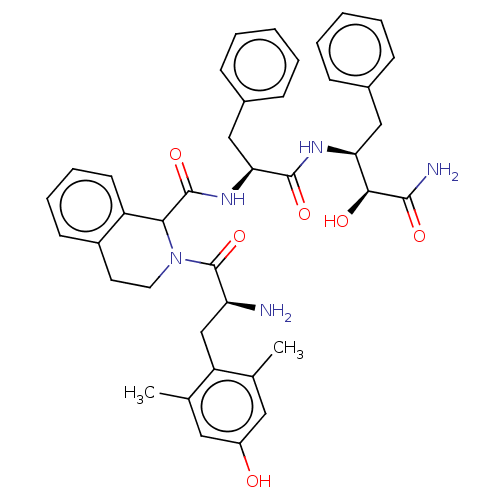 (CHEMBL3409760) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from rat cortex delta opioid receptor after 2.5 hrs by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071094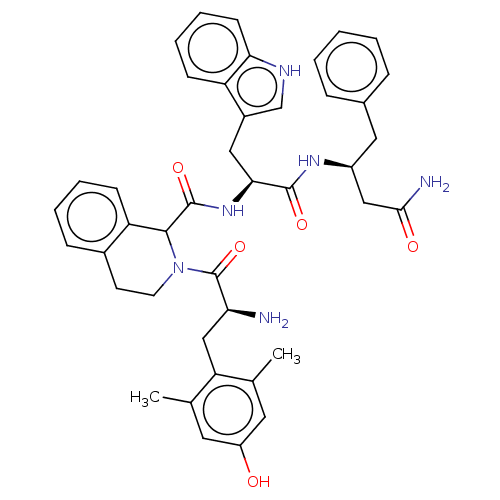 (CHEMBL3409765) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from rat cortex delta opioid receptor after 2.5 hrs by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071093 (CHEMBL3409764) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from rat cortex delta opioid receptor after 2.5 hrs by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071095 (CHEMBL3409766) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from rat cortex delta opioid receptor after 2.5 hrs by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (MOUSE) | BDBM50533718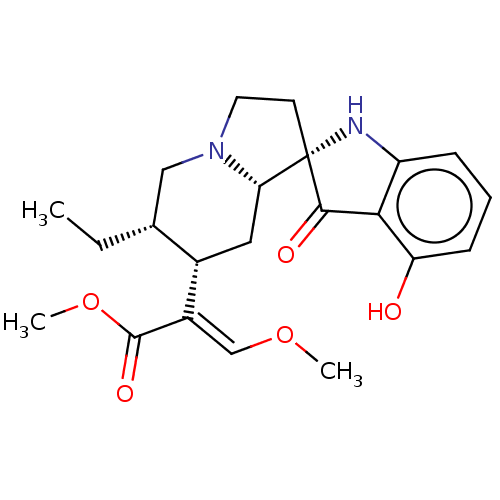 (CHEMBL4530421) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan Kettering Cancer Center Curated by ChEMBL | Assay Description Displacement of [125I]-IBNtxA from mouse mu opioid receptor-1 expressed in CHO cell membranes incubated for 90 mins | J Med Chem 59: 8381-97 (2016) Article DOI: 10.1021/acs.jmedchem.6b00748 BindingDB Entry DOI: 10.7270/Q27H1P2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50533719 (CHEMBL4441457) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan Kettering Cancer Center Curated by ChEMBL | Assay Description Displacement of [125I]-IBNtxA from mouse delta opioid receptor-1 expressed in CHO cell membranes incubated for 90 mins | J Med Chem 59: 8381-97 (2016) Article DOI: 10.1021/acs.jmedchem.6b00748 BindingDB Entry DOI: 10.7270/Q27H1P2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071069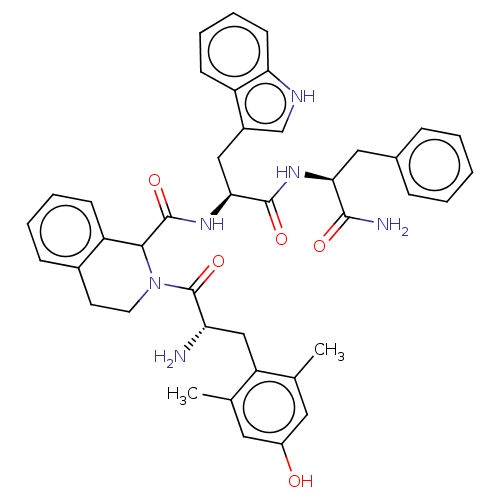 (CHEMBL3409759) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from rat cortex delta opioid receptor after 2.5 hrs by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071068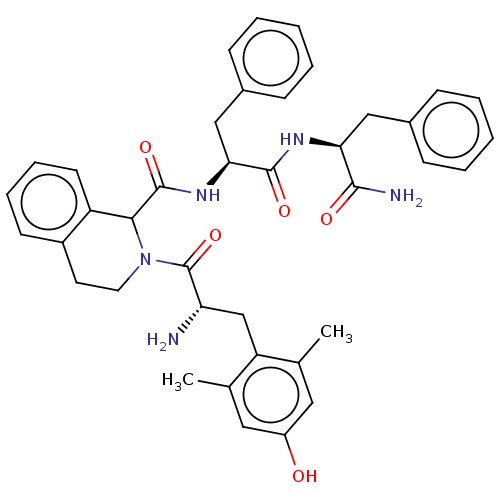 (CHEMBL3409758) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from rat cortex delta opioid receptor after 2.5 hrs by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50533717 (CHEMBL4449284) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan Kettering Cancer Center Curated by ChEMBL | Assay Description Displacement of [125I]-IBNtxA from mouse delta opioid receptor-1 expressed in CHO cell membranes incubated for 90 mins | J Med Chem 59: 8381-97 (2016) Article DOI: 10.1021/acs.jmedchem.6b00748 BindingDB Entry DOI: 10.7270/Q27H1P2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (MOUSE) | BDBM50533720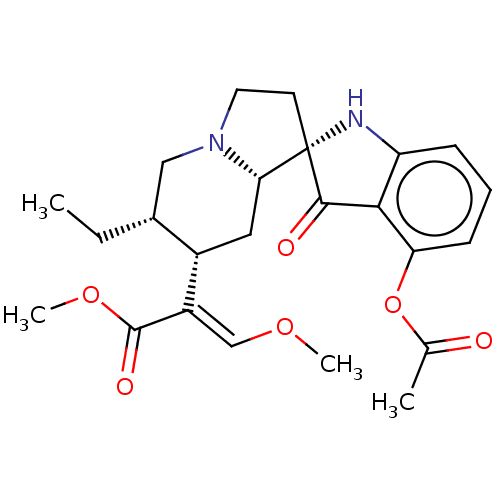 (CHEMBL4557905) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan Kettering Cancer Center Curated by ChEMBL | Assay Description Displacement of [125I]-IBNtxA from mouse mu opioid receptor-1 expressed in CHO cell membranes incubated for 90 mins | J Med Chem 59: 8381-97 (2016) Article DOI: 10.1021/acs.jmedchem.6b00748 BindingDB Entry DOI: 10.7270/Q27H1P2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50533716 (CHEMBL4453504) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan Kettering Cancer Center Curated by ChEMBL | Assay Description Displacement of [125I]-IBNtxA from mouse delta opioid receptor-1 expressed in CHO cell membranes incubated for 90 mins | J Med Chem 59: 8381-97 (2016) Article DOI: 10.1021/acs.jmedchem.6b00748 BindingDB Entry DOI: 10.7270/Q27H1P2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50474150 (CHEMBL58362) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem | Article PubMed | 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan Kettering Cancer Center Curated by ChEMBL | Assay Description Displacement of [125I]-IBNtxA from mouse delta opioid receptor-1 expressed in CHO cell membranes incubated for 90 mins | J Med Chem 59: 8381-97 (2016) Article DOI: 10.1021/acs.jmedchem.6b00748 BindingDB Entry DOI: 10.7270/Q27H1P2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50139013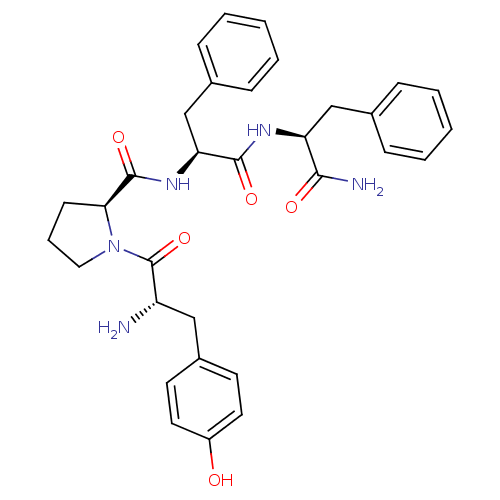 ((S)-1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat cortex mu opioid receptor after 30 mins by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071071 (CHEMBL3409761) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat cortex mu opioid receptor after 30 mins by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50029183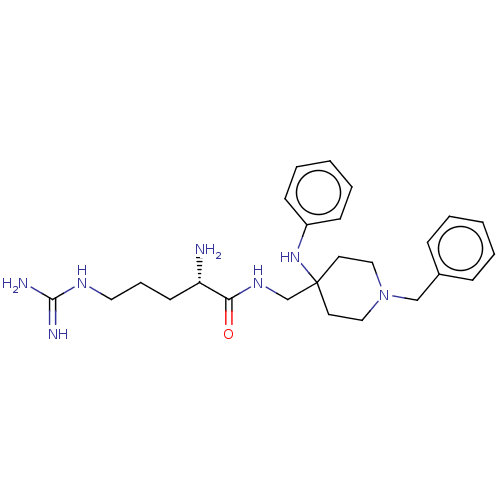 (CHEMBL3360832) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 3.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Mississippi Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from human MOR expressed in HEK293 cells after 60 mins by scintillation counting analysis | J Med Chem 57: 8903-27 (2014) Article DOI: 10.1021/jm500989n BindingDB Entry DOI: 10.7270/Q2C24Z18 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuropeptide FF receptor 1 (Homo sapiens (Human)) | BDBM50029188 (CHEMBL2165920) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 3.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Mississippi Curated by ChEMBL | Assay Description Displacement of [3H]NPVF from human NPFF1 receptor expressed in CHO cells after 1 hr by liquid scintillation counting analysis | J Med Chem 57: 8903-27 (2014) Article DOI: 10.1021/jm500989n BindingDB Entry DOI: 10.7270/Q2C24Z18 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuropeptide FF receptor 1 (Homo sapiens (Human)) | BDBM50029138 (CHEMBL3360830) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 3.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Mississippi Curated by ChEMBL | Assay Description Displacement of [3H]NPVF from human NPFF1 receptor expressed in CHO cells after 1 hr by liquid scintillation counting analysis | J Med Chem 57: 8903-27 (2014) Article DOI: 10.1021/jm500989n BindingDB Entry DOI: 10.7270/Q2C24Z18 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50095155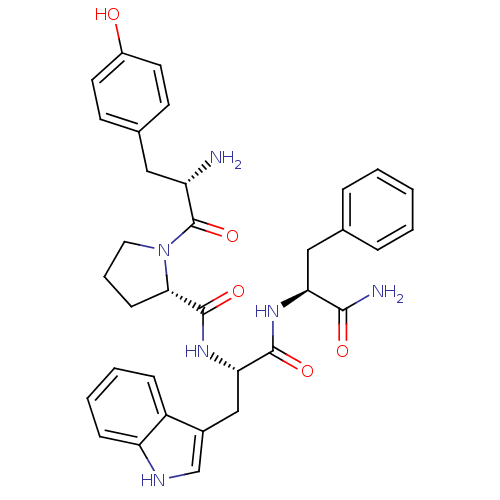 ((S)-1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat cortex mu opioid receptor after 30 mins by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuropeptide FF receptor 1 (Homo sapiens (Human)) | BDBM50029138 (CHEMBL3360830) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Mississippi Curated by ChEMBL | Assay Description Displacement of [125I]YVP from human NPFF1 receptor expressed in CHO cells after 1 hr by liquid scintillation counting analysis | J Med Chem 57: 8903-27 (2014) Article DOI: 10.1021/jm500989n BindingDB Entry DOI: 10.7270/Q2C24Z18 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50029183 (CHEMBL3360832) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 4.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Mississippi Curated by ChEMBL | Assay Description Displacement of [3H]U-69593 from human KOR expressed in HEK293 cells after 60 mins by scintillation counting analysis | J Med Chem 57: 8903-27 (2014) Article DOI: 10.1021/jm500989n BindingDB Entry DOI: 10.7270/Q2C24Z18 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071068 (CHEMBL3409758) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat cortex mu opioid receptor after 30 mins by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071069 (CHEMBL3409759) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat cortex mu opioid receptor after 30 mins by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50580195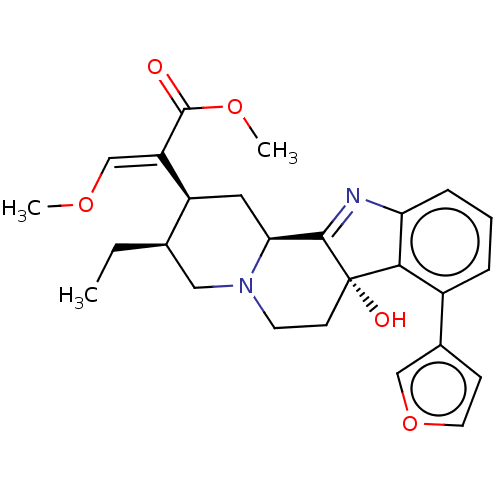 (CHEMBL5071286) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Displacement of [3H]-DAMGO from mu opioid receptor (unknown origin) expressed in CHO cells at 10 uM | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c01273 BindingDB Entry DOI: 10.7270/Q25D8WQH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071095 (CHEMBL3409766) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat cortex mu opioid receptor after 30 mins by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50533718 (CHEMBL4530421) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 6.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan Kettering Cancer Center Curated by ChEMBL | Assay Description Displacement of [125I]-IBNtxA from mouse delta opioid receptor-1 expressed in CHO cell membranes incubated for 90 mins | J Med Chem 59: 8381-97 (2016) Article DOI: 10.1021/acs.jmedchem.6b00748 BindingDB Entry DOI: 10.7270/Q27H1P2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071072 (CHEMBL3409762) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat cortex mu opioid receptor after 30 mins by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071076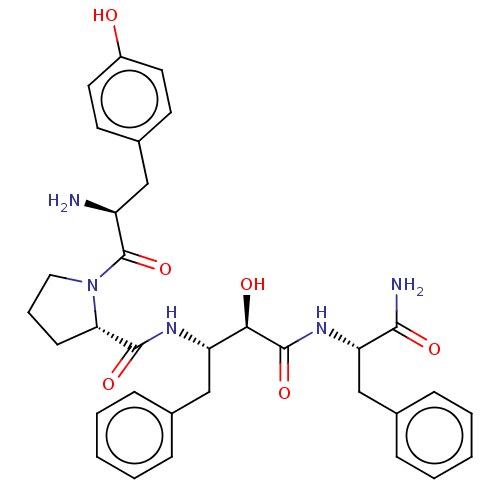 (CHEMBL3409749) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat cortex mu opioid receptor after 30 mins by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071086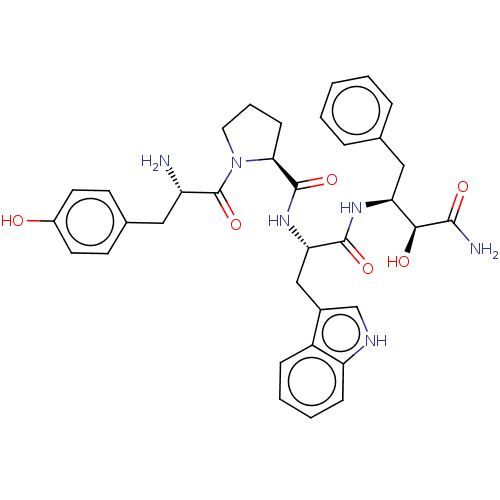 (CHEMBL3409741) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat cortex mu opioid receptor after 30 mins by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50114010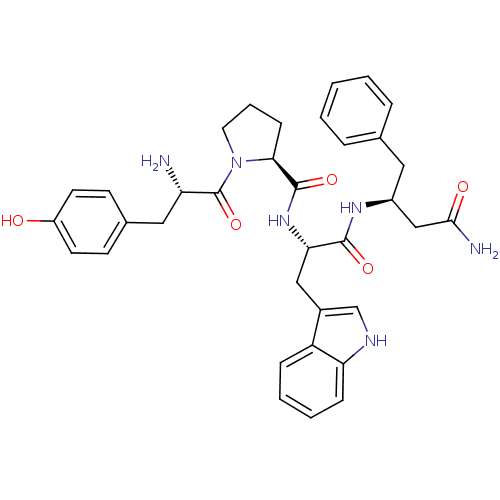 (1-[2-Amino-3-(4-hydroxy-phenyl)-propionyl]-pyrroli...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat cortex mu opioid receptor after 30 mins by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071070 (CHEMBL3409760) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat cortex mu opioid receptor after 30 mins by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071092 (CHEMBL3409755) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat cortex mu opioid receptor after 30 mins by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071094 (CHEMBL3409765) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat cortex mu opioid receptor after 30 mins by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50580193 (CHEMBL5086786) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Displacement of [3H]-DAMGO from mu opioid receptor (unknown origin) expressed in CHO cells at 10 uM | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c01273 BindingDB Entry DOI: 10.7270/Q25D8WQH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50580194 (CHEMBL5074977) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Displacement of [3H]-DAMGO from mu opioid receptor (unknown origin) expressed in CHO cells at 10 uM | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c01273 BindingDB Entry DOI: 10.7270/Q25D8WQH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071093 (CHEMBL3409764) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat cortex mu opioid receptor after 30 mins by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071073 (CHEMBL3409763) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat cortex mu opioid receptor after 30 mins by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50071090 (CHEMBL3409745) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhejiang University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from rat cortex mu opioid receptor after 30 mins by liquid scintillation counting analysis | Eur J Med Chem 92: 270-81 (2015) Article DOI: 10.1016/j.ejmech.2014.12.049 BindingDB Entry DOI: 10.7270/Q22B90RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Mus musculus (Mouse)) | BDBM50533716 (CHEMBL4453504) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan Kettering Cancer Center Curated by ChEMBL | Assay Description Displacement of [125I]-IBNtxA from mouse kappa opioid receptor-1 expressed in CHO cell membranes incubated for 90 mins | J Med Chem 59: 8381-97 (2016) Article DOI: 10.1021/acs.jmedchem.6b00748 BindingDB Entry DOI: 10.7270/Q27H1P2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (MOUSE) | BDBM50533720 (CHEMBL4557905) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan Kettering Cancer Center Curated by ChEMBL | Assay Description Displacement of [125I]-IBNtxA from mouse delta opioid receptor-1 expressed in CHO cell membranes incubated for 90 mins | J Med Chem 59: 8381-97 (2016) Article DOI: 10.1021/acs.jmedchem.6b00748 BindingDB Entry DOI: 10.7270/Q27H1P2H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 425 total ) | Next | Last >> |