

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM16047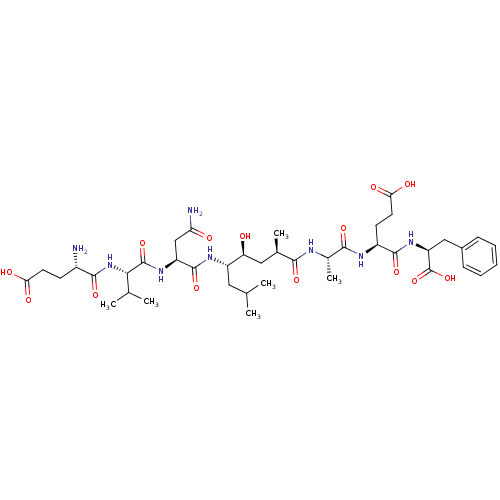 ((4S)-4-[(2S)-2-[(2R,4S,5S)-5-[(2S)-2-[(2S)-2-[(4S)...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB PubMed | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company Curated by ChEMBL | Assay Description Inhibition of human beta-secretase | Bioorg Med Chem Lett 13: 4335-9 (2003) BindingDB Entry DOI: 10.7270/Q2513ZR3 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gamma-aminobutyric acid receptor subunit alpha-6/beta-2/gamma-2 (RAT-Rattus norvegicus (Rat)) | BDBM50048855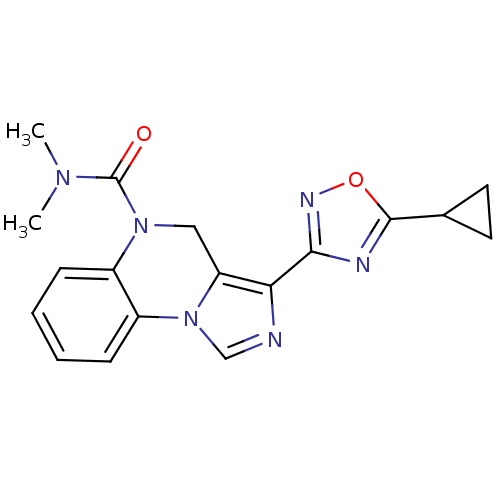 (3-(5-Cyclopropyl-[1,2,4]oxadiazol-3-yl)-4H-imidazo...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 13.2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Upjohn Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-Ro- 15-4513 from alpha6 beta2 gamma2 subunits of Rat GABA-A receptor expressed in Sf-9 insect cell membranes | J Med Chem 39: 4654-66 (1996) Article DOI: 10.1021/jm960401i BindingDB Entry DOI: 10.7270/Q20K27NN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutamate racemase (Lactobacillus fermentum) | BDBM50118891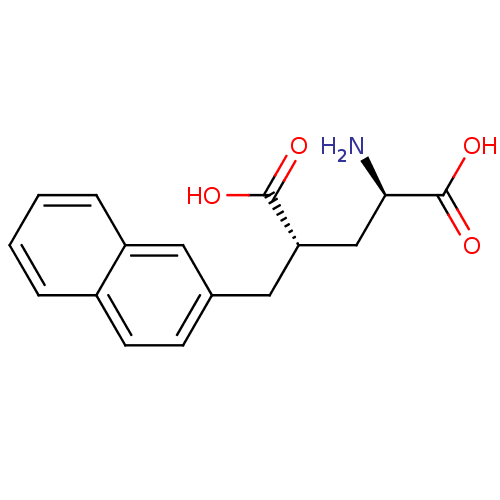 ((4S)-4-(2-NAPHTHYLMETHYL)-D-GLUTAMIC ACID | 2-Amin...) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Similars | PubMed | 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Co. Curated by ChEMBL | Assay Description Binding affinity against Glutamate Racemase | J Med Chem 45: 4559-70 (2002) BindingDB Entry DOI: 10.7270/Q2B857F3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gamma-aminobutyric acid receptor subunit alpha-6/beta-2/gamma-2 (RAT-Rattus norvegicus (Rat)) | BDBM50054662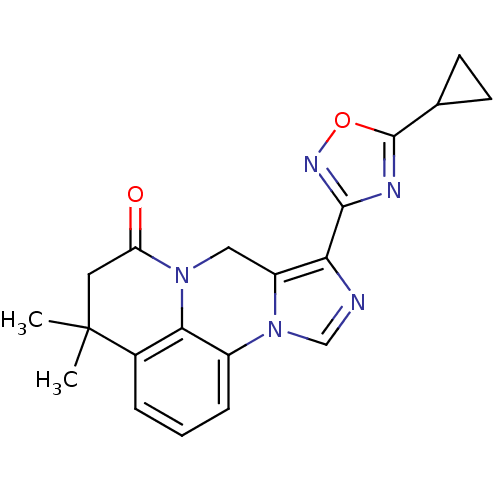 (8-(5-Cyclopropyl-[1,2,4]oxadiazol-3-yl)-4,4-dimeth...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >589 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Upjohn Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-Ro- 15-4513 from alpha6 beta2 gamma2 subunits of Rat GABA-A receptor expressed in Sf-9 insect cell membranes | J Med Chem 39: 4654-66 (1996) Article DOI: 10.1021/jm960401i BindingDB Entry DOI: 10.7270/Q20K27NN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gamma-aminobutyric acid receptor subunit alpha-6/beta-2/gamma-2 (RAT-Rattus norvegicus (Rat)) | BDBM50054659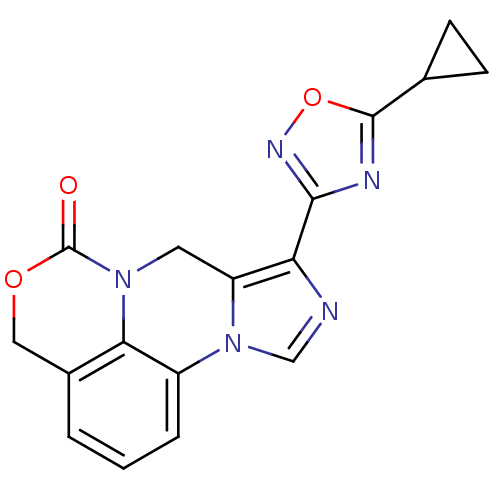 (8-(5-Cyclopropyl-[1,2,4]oxadiazol-3-yl)-4H,7H-5-ox...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >589 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Upjohn Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-Ro- 15-4513 from alpha6 beta2 gamma2 subunits of Rat GABA-A receptor expressed in Sf-9 insect cell membranes | J Med Chem 39: 4654-66 (1996) Article DOI: 10.1021/jm960401i BindingDB Entry DOI: 10.7270/Q20K27NN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gamma-aminobutyric acid receptor subunit alpha-6/beta-2/gamma-2 (RAT-Rattus norvegicus (Rat)) | BDBM50054656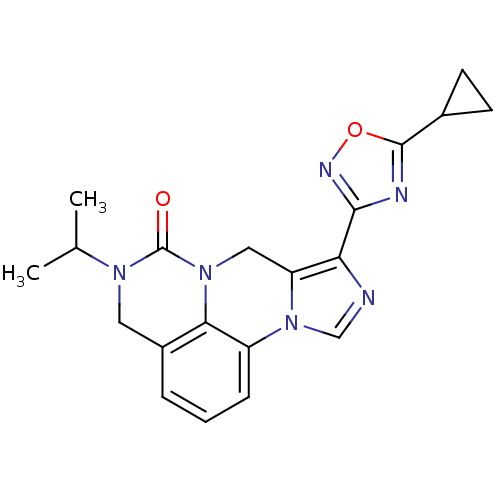 (8-(5-Cyclopropyl-[1,2,4]oxadiazol-3-yl)-5-isopropy...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >589 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Upjohn Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-Ro- 15-4513 from alpha6 beta2 gamma2 subunits of Rat GABA-A receptor expressed in Sf-9 insect cell membranes | J Med Chem 39: 4654-66 (1996) Article DOI: 10.1021/jm960401i BindingDB Entry DOI: 10.7270/Q20K27NN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gamma-aminobutyric acid receptor subunit alpha-6/beta-2/gamma-2 (RAT-Rattus norvegicus (Rat)) | BDBM50054660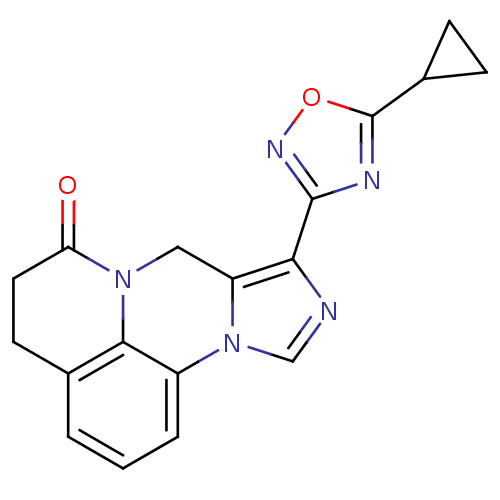 (8-(5-Cyclopropyl-[1,2,4]oxadiazol-3-yl)-4,5-dihydr...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >589 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Upjohn Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-Ro- 15-4513 from alpha6 beta2 gamma2 subunits of Rat GABA-A receptor expressed in Sf-9 insect cell membranes | J Med Chem 39: 4654-66 (1996) Article DOI: 10.1021/jm960401i BindingDB Entry DOI: 10.7270/Q20K27NN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gamma-aminobutyric acid receptor subunit alpha-6/beta-2/gamma-2 (RAT-Rattus norvegicus (Rat)) | BDBM50054663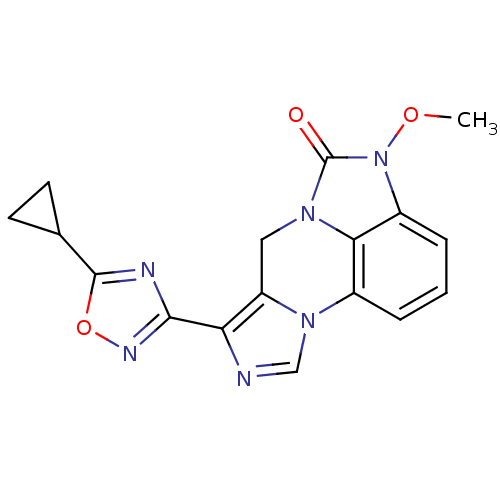 (7-(5-Cyclopropyl-[1,2,4]oxadiazol-3-yl)-4-methoxy-...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | >589 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Upjohn Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-Ro- 15-4513 from alpha6 beta2 gamma2 subunits of Rat GABA-A receptor expressed in Sf-9 insect cell membranes | J Med Chem 39: 4654-66 (1996) Article DOI: 10.1021/jm960401i BindingDB Entry DOI: 10.7270/Q20K27NN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gamma-aminobutyric acid receptor subunit alpha-6/beta-2/gamma-2 (RAT-Rattus norvegicus (Rat)) | BDBM50054661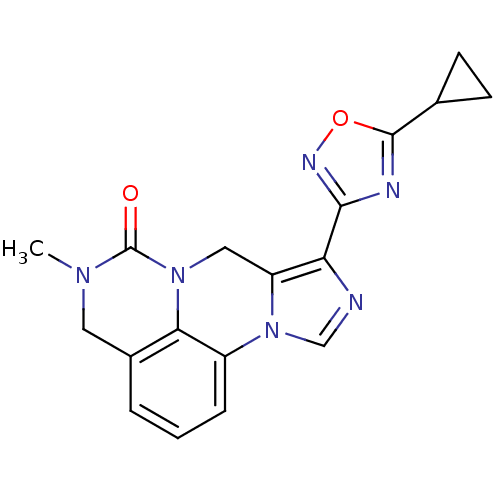 (8-(5-Cyclopropyl-[1,2,4]oxadiazol-3-yl)-5-methyl-4...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >589 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Upjohn Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-Ro- 15-4513 from alpha6 beta2 gamma2 subunits of Rat GABA-A receptor expressed in Sf-9 insect cell membranes | J Med Chem 39: 4654-66 (1996) Article DOI: 10.1021/jm960401i BindingDB Entry DOI: 10.7270/Q20K27NN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50194190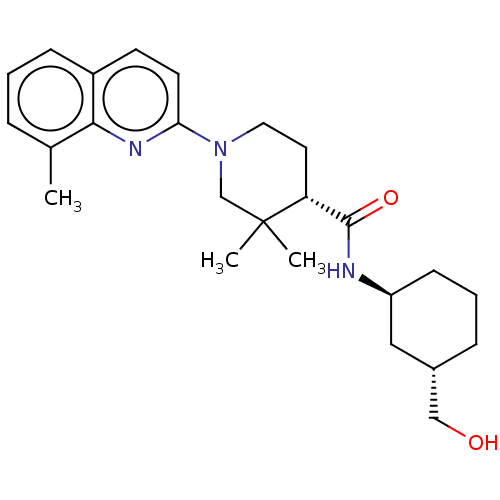 (CHEMBL3956184) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES-1 expressed in 293E cells assessed as reduction in conversion of PGH2 to PGE2 after 1.5 min by LC/MS analysis | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233277 (CHEMBL4085873) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233278 (CHEMBL4064335) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233283 (CHEMBL4102262) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233285 (CHEMBL4101413) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233283 (CHEMBL4102262) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS addition meas... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50194190 (CHEMBL3956184) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES-1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS stimulation ... | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233281 (CHEMBL4084995) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50194141 (CHEMBL3938686) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES-1 expressed in 293E cells assessed as reduction in conversion of PGH2 to PGE2 after 1.5 min by LC/MS analysis | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233284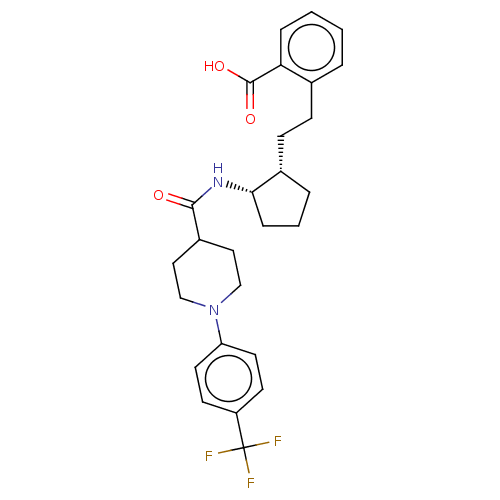 (CHEMBL4071976) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233276 (CHEMBL4078000) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50194141 (CHEMBL3938686) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES-1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS stimulation ... | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50194141 (CHEMBL3938686) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES-1 in human A549 cells assessed as reduction in recombinant human interleukin-1 beta-induced PGE2 production preincubated for 30 m... | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233274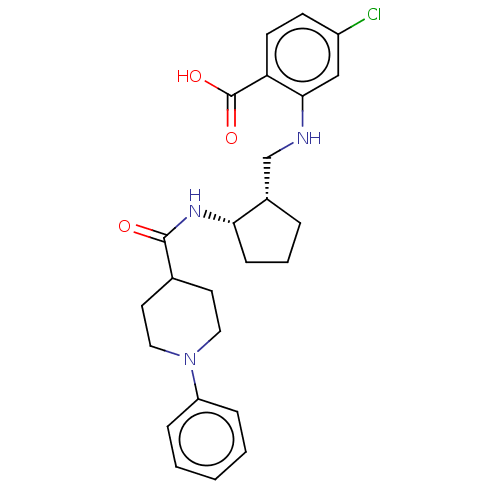 (CHEMBL4067045) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233280 (CHEMBL4063350) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50194138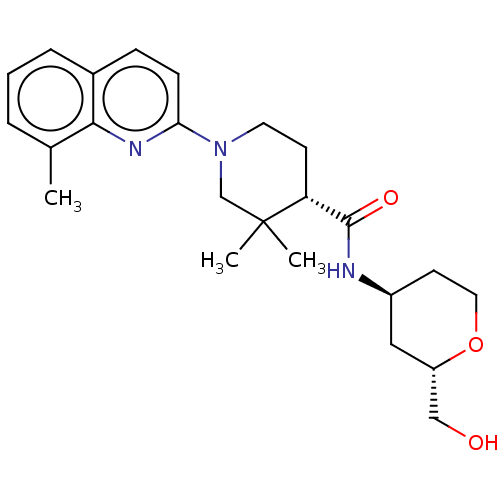 (CHEMBL3928608) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES-1 expressed in 293E cells assessed as reduction in conversion of PGH2 to PGE2 after 1.5 min by LC/MS analysis | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50194140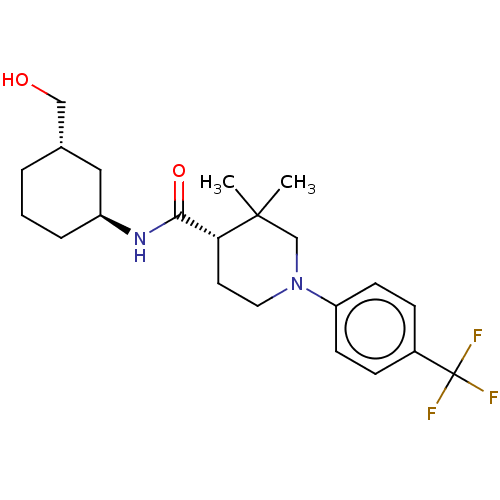 (CHEMBL3947494) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES-1 expressed in 293E cells assessed as reduction in conversion of PGH2 to PGE2 after 1.5 min by LC/MS analysis | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50194190 (CHEMBL3956184) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES-1 in human A549 cells assessed as reduction in recombinant human interleukin-1 beta-induced PGE2 production preincubated for 30 m... | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233277 (CHEMBL4085873) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS addition meas... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233275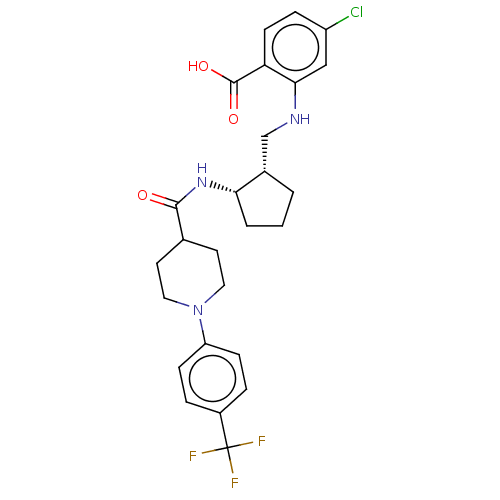 (CHEMBL4094518) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS addition meas... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233275 (CHEMBL4094518) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES1 in human A549 cells assessed as reduction in IL-1beta-induced PGE2 production preincubated for 30 mins followed by IL-1beta addi... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233275 (CHEMBL4094518) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50194137 (CHEMBL3922684) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES-1 expressed in 293E cells assessed as reduction in conversion of PGH2 to PGE2 after 1.5 min by LC/MS analysis | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50194138 (CHEMBL3928608) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES-1 in human A549 cells assessed as reduction in recombinant human interleukin-1 beta-induced PGE2 production preincubated for 30 m... | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50194140 (CHEMBL3947494) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES-1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS stimulation ... | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233284 (CHEMBL4071976) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS addition meas... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233285 (CHEMBL4101413) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS addition meas... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50194193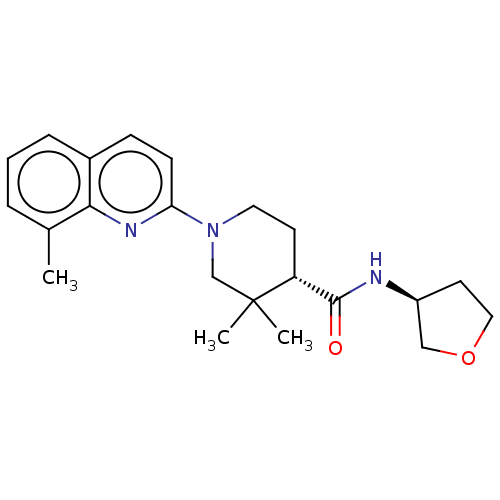 (CHEMBL3926051) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES-1 expressed in 293E cells assessed as reduction in conversion of PGH2 to PGE2 after 1.5 min by LC/MS analysis | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233279 (CHEMBL4092750) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES1 expressed in HEK293 microsomes assessed as reduction in PGE2 production using PGH2 as substrate after 2.5 mins by LC-MS an... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233278 (CHEMBL4064335) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS addition meas... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50194192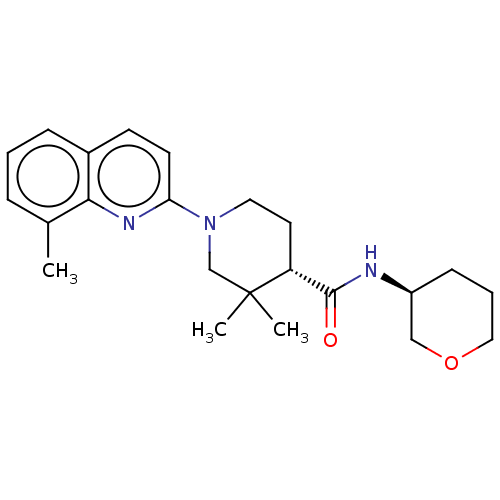 (CHEMBL3910746) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES-1 expressed in 293E cells assessed as reduction in conversion of PGH2 to PGE2 after 1.5 min by LC/MS analysis | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50194138 (CHEMBL3928608) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES-1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS stimulation ... | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233276 (CHEMBL4078000) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS addition meas... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233281 (CHEMBL4084995) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS addition meas... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50194140 (CHEMBL3947494) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES-1 in human A549 cells assessed as reduction in recombinant human interleukin-1 beta-induced PGE2 production preincubated for 30 m... | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Canis familiaris) | BDBM50194138 (CHEMBL3928608) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of dog mPGES-1 | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50194142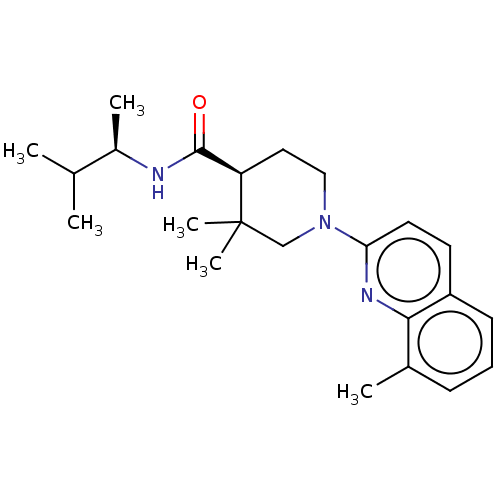 (CHEMBL3947843) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of human mPGES-1 expressed in 293E cells assessed as reduction in conversion of PGH2 to PGE2 after 1.5 min by LC/MS analysis | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233274 (CHEMBL4067045) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES1 in human A549 cells assessed as reduction in IL-1beta-induced PGE2 production preincubated for 30 mins followed by IL-1beta addi... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50233279 (CHEMBL4092750) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS addition meas... | Bioorg Med Chem Lett 27: 1478-1483 (2017) Article DOI: 10.1016/j.bmcl.2016.11.011 BindingDB Entry DOI: 10.7270/Q28P62RZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Homo sapiens (Human)) | BDBM50194137 (CHEMBL3922684) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES-1 in human whole blood assessed as reduction in LPS-induced PGE2 production preincubated for 30 mins followed by LPS stimulation ... | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E synthase (Canis familiaris) | BDBM50194138 (CHEMBL3928608) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories Curated by ChEMBL | Assay Description Inhibition of mPGES-1 in dog whole blood | Bioorg Med Chem Lett 26: 4824-4828 (2016) Article DOI: 10.1016/j.bmcl.2016.08.023 BindingDB Entry DOI: 10.7270/Q2125VMX | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Displayed 1 to 50 (of 97 total ) | Next | Last >> |