 Found 59 hits with Last Name = 'nawano' and Initial = 'm'
Found 59 hits with Last Name = 'nawano' and Initial = 'm' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50543892
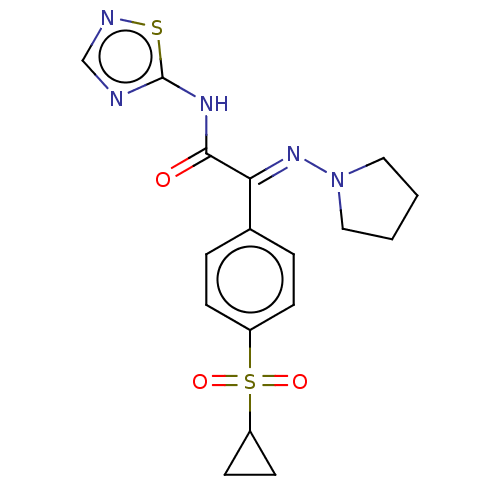
(CHEMBL4649482)Show SMILES O=C(Nc1ncns1)C(=N\N1CCCC1)\c1ccc(cc1)S(=O)(=O)C1CC1 Show InChI InChI=1S/C17H19N5O3S2/c23-16(20-17-18-11-19-26-17)15(21-22-9-1-2-10-22)12-3-5-13(6-4-12)27(24,25)14-7-8-14/h3-6,11,14H,1-2,7-10H2,(H,18,19,20,23)/b21-15+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Chiba University
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
Bioorg Med Chem Lett 30: (2020)
Article DOI: 10.1016/j.bmcl.2020.127249
BindingDB Entry DOI: 10.7270/Q2P84GG5 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50543890

(CHEMBL4641678)Show SMILES Clc1cnc(NC(=O)C(=N\N2CCCC2)\c2ccc(cc2)S(=O)(=O)C2CC2)s1 Show InChI InChI=1S/C18H19ClN4O3S2/c19-15-11-20-18(27-15)21-17(24)16(22-23-9-1-2-10-23)12-3-5-13(6-4-12)28(25,26)14-7-8-14/h3-6,11,14H,1-2,7-10H2,(H,20,21,24)/b22-16+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.67E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Chiba University
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
Bioorg Med Chem Lett 30: (2020)
Article DOI: 10.1016/j.bmcl.2020.127249
BindingDB Entry DOI: 10.7270/Q2P84GG5 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50543895
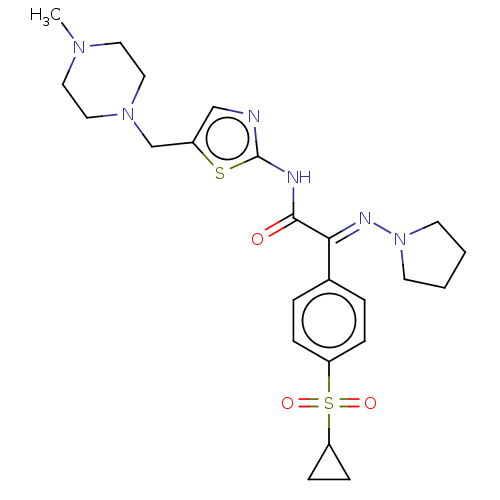
(CHEMBL4646396)Show SMILES CN1CCN(Cc2cnc(NC(=O)C(=N\N3CCCC3)\c3ccc(cc3)S(=O)(=O)C3CC3)s2)CC1 Show InChI InChI=1S/C24H32N6O3S2/c1-28-12-14-29(15-13-28)17-19-16-25-24(34-19)26-23(31)22(27-30-10-2-3-11-30)18-4-6-20(7-5-18)35(32,33)21-8-9-21/h4-7,16,21H,2-3,8-15,17H2,1H3,(H,25,26,31)/b27-22+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.69E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Chiba University
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
Bioorg Med Chem Lett 30: (2020)
Article DOI: 10.1016/j.bmcl.2020.127249
BindingDB Entry DOI: 10.7270/Q2P84GG5 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50543889
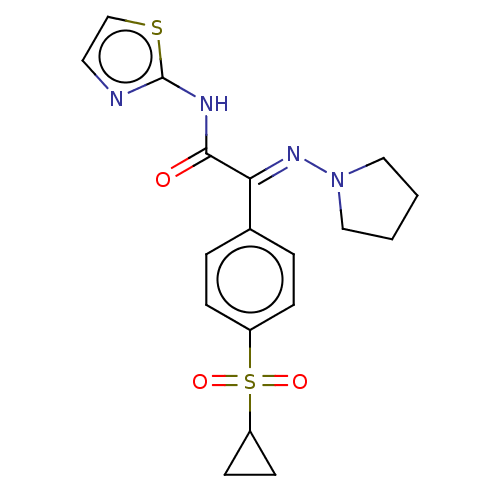
(CHEMBL4645293)Show SMILES O=C(Nc1nccs1)C(=N\N1CCCC1)\c1ccc(cc1)S(=O)(=O)C1CC1 Show InChI InChI=1S/C18H20N4O3S2/c23-17(20-18-19-9-12-26-18)16(21-22-10-1-2-11-22)13-3-5-14(6-4-13)27(24,25)15-7-8-15/h3-6,9,12,15H,1-2,7-8,10-11H2,(H,19,20,23)/b21-16+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.75E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Chiba University
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
Bioorg Med Chem Lett 30: (2020)
Article DOI: 10.1016/j.bmcl.2020.127249
BindingDB Entry DOI: 10.7270/Q2P84GG5 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50543894
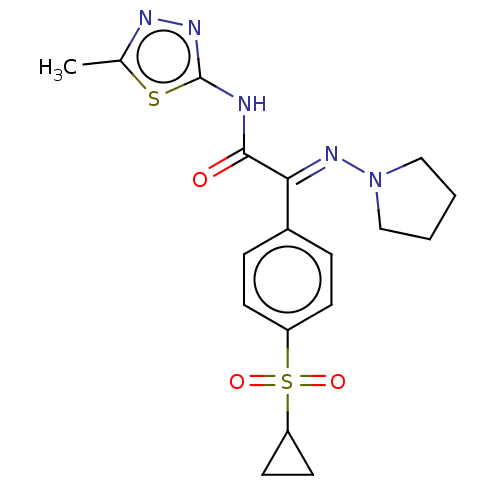
(CHEMBL4637653)Show SMILES Cc1nnc(NC(=O)C(=N\N2CCCC2)\c2ccc(cc2)S(=O)(=O)C2CC2)s1 Show InChI InChI=1S/C18H21N5O3S2/c1-12-20-21-18(27-12)19-17(24)16(22-23-10-2-3-11-23)13-4-6-14(7-5-13)28(25,26)15-8-9-15/h4-7,15H,2-3,8-11H2,1H3,(H,19,21,24)/b22-16+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Chiba University
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
Bioorg Med Chem Lett 30: (2020)
Article DOI: 10.1016/j.bmcl.2020.127249
BindingDB Entry DOI: 10.7270/Q2P84GG5 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50543893
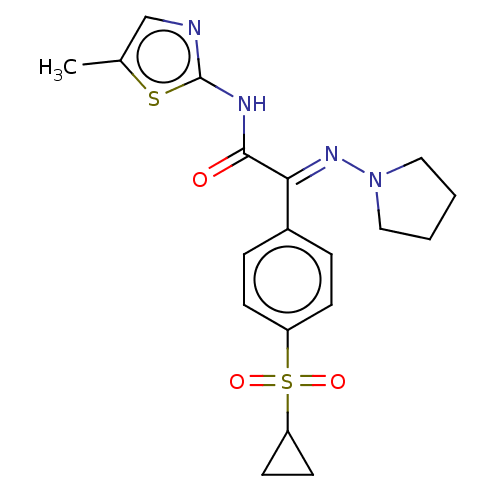
(CHEMBL4632835)Show SMILES Cc1cnc(NC(=O)C(=N\N2CCCC2)\c2ccc(cc2)S(=O)(=O)C2CC2)s1 Show InChI InChI=1S/C19H22N4O3S2/c1-13-12-20-19(27-13)21-18(24)17(22-23-10-2-3-11-23)14-4-6-15(7-5-14)28(25,26)16-8-9-16/h4-7,12,16H,2-3,8-11H2,1H3,(H,20,21,24)/b22-17+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Chiba University
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
Bioorg Med Chem Lett 30: (2020)
Article DOI: 10.1016/j.bmcl.2020.127249
BindingDB Entry DOI: 10.7270/Q2P84GG5 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50543896
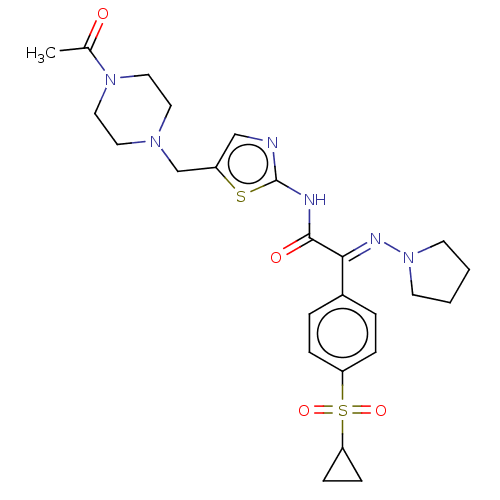
(CHEMBL4644080)Show SMILES CC(=O)N1CCN(Cc2cnc(NC(=O)C(=N\N3CCCC3)\c3ccc(cc3)S(=O)(=O)C3CC3)s2)CC1 Show InChI InChI=1S/C25H32N6O4S2/c1-18(32)30-14-12-29(13-15-30)17-20-16-26-25(36-20)27-24(33)23(28-31-10-2-3-11-31)19-4-6-21(7-5-19)37(34,35)22-8-9-22/h4-7,16,22H,2-3,8-15,17H2,1H3,(H,26,27,33)/b28-23+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Chiba University
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
Bioorg Med Chem Lett 30: (2020)
Article DOI: 10.1016/j.bmcl.2020.127249
BindingDB Entry DOI: 10.7270/Q2P84GG5 |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161677

(4-[3-(5-Chloro-thiazol-2-yl)-1-cyclopentylmethyl-u...)Show SMILES NS(=O)(=O)c1ccc(cc1)N(CC1CCCC1)C(=O)Nc1ncc(Cl)s1 Show InChI InChI=1S/C16H19ClN4O3S2/c17-14-9-19-15(25-14)20-16(22)21(10-11-3-1-2-4-11)12-5-7-13(8-6-12)26(18,23)24/h5-9,11H,1-4,10H2,(H2,18,23,24)(H,19,20,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161675
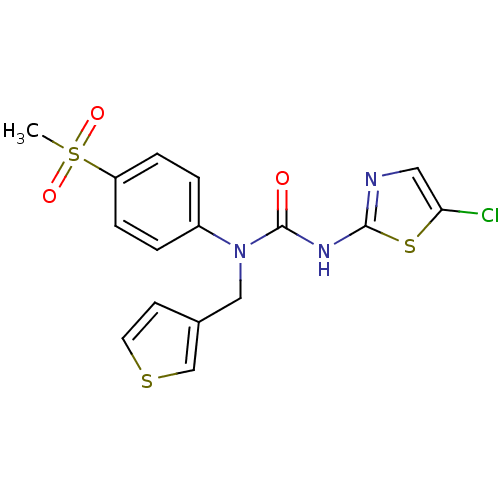
(3-(5-Chloro-thiazol-2-yl)-1-(4-methanesulfonyl-phe...)Show SMILES CS(=O)(=O)c1ccc(cc1)N(Cc1ccsc1)C(=O)Nc1ncc(Cl)s1 Show InChI InChI=1S/C16H14ClN3O3S3/c1-26(22,23)13-4-2-12(3-5-13)20(9-11-6-7-24-10-11)16(21)19-15-18-8-14(17)25-15/h2-8,10H,9H2,1H3,(H,18,19,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 6.80E+3 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161680
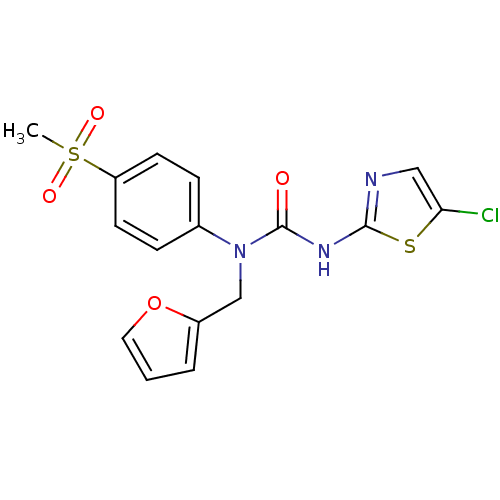
(3-(5-Chloro-thiazol-2-yl)-1-furan-2-ylmethyl-1-(4-...)Show SMILES CS(=O)(=O)c1ccc(cc1)N(Cc1ccco1)C(=O)Nc1ncc(Cl)s1 Show InChI InChI=1S/C16H14ClN3O4S2/c1-26(22,23)13-6-4-11(5-7-13)20(10-12-3-2-8-24-12)16(21)19-15-18-9-14(17)25-15/h2-9H,10H2,1H3,(H,18,19,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.15E+4 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161681

(3-(5-Chloro-thiazol-2-yl)-1-(6-cyano-pyridin-3-yl)...)Show InChI InChI=1S/C16H16ClN5OS/c17-14-9-20-15(24-14)21-16(23)22(10-11-3-1-2-4-11)13-6-5-12(7-18)19-8-13/h5-6,8-9,11H,1-4,10H2,(H,20,21,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 7.70E+3 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161682

(3-(5-Chloro-thiazol-2-yl)-1-cyclopentylmethyl-1-(3...)Show InChI InChI=1S/C17H20ClN3O2S/c1-23-14-8-4-7-13(9-14)21(11-12-5-2-3-6-12)17(22)20-16-19-10-15(18)24-16/h4,7-10,12H,2-3,5-6,11H2,1H3,(H,19,20,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161678

(3-(5-Chloro-thiazol-2-yl)-1-cyclopentylmethyl-1-(6...)Show InChI InChI=1S/C16H19ClN4O2S/c1-23-14-7-6-12(8-18-14)21(10-11-4-2-3-5-11)16(22)20-15-19-9-13(17)24-15/h6-9,11H,2-5,10H2,1H3,(H,19,20,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161679
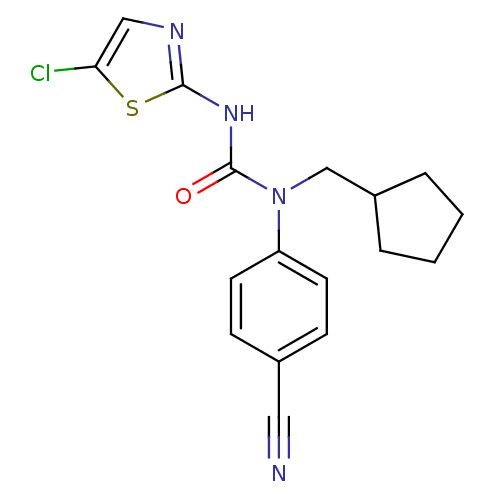
(3-(5-Chloro-thiazol-2-yl)-1-(4-cyano-phenyl)-1-cyc...)Show InChI InChI=1S/C17H17ClN4OS/c18-15-10-20-16(24-15)21-17(23)22(11-13-3-1-2-4-13)14-7-5-12(9-19)6-8-14/h5-8,10,13H,1-4,11H2,(H,20,21,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161684
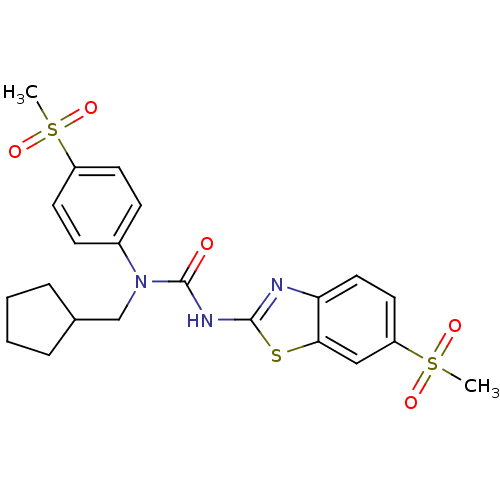
(1-Cyclopentylmethyl-3-(6-methanesulfonyl-benzothia...)Show SMILES CS(=O)(=O)c1ccc(cc1)N(CC1CCCC1)C(=O)Nc1nc2ccc(cc2s1)S(C)(=O)=O Show InChI InChI=1S/C22H25N3O5S3/c1-32(27,28)17-9-7-16(8-10-17)25(14-15-5-3-4-6-15)22(26)24-21-23-19-12-11-18(33(2,29)30)13-20(19)31-21/h7-13,15H,3-6,14H2,1-2H3,(H,23,24,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 3.29E+4 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161685
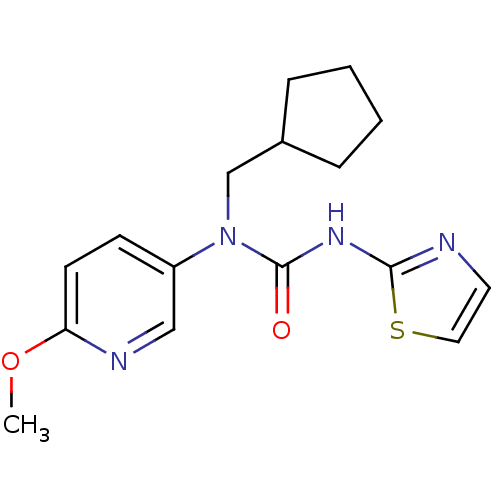
(1-Cyclopentylmethyl-1-(6-methoxy-pyridin-3-yl)-3-t...)Show InChI InChI=1S/C16H20N4O2S/c1-22-14-7-6-13(10-18-14)20(11-12-4-2-3-5-12)16(21)19-15-17-8-9-23-15/h6-10,12H,2-5,11H2,1H3,(H,17,19,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.92E+4 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161683
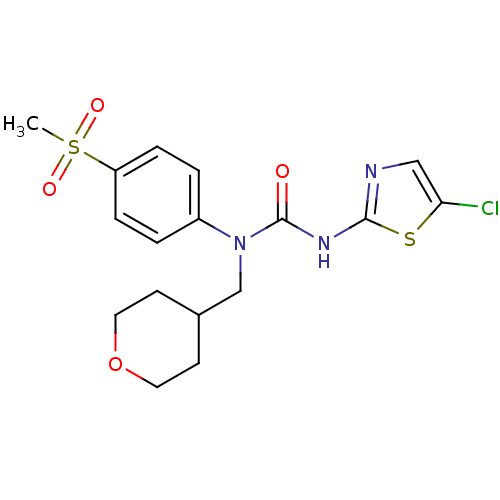
(3-(5-Chloro-thiazol-2-yl)-1-(4-methanesulfonyl-phe...)Show SMILES CS(=O)(=O)c1ccc(cc1)N(CC1CCOCC1)C(=O)Nc1ncc(Cl)s1 Show InChI InChI=1S/C17H20ClN3O4S2/c1-27(23,24)14-4-2-13(3-5-14)21(11-12-6-8-25-9-7-12)17(22)20-16-19-10-15(18)26-16/h2-5,10,12H,6-9,11H2,1H3,(H,19,20,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 4.90E+3 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161674
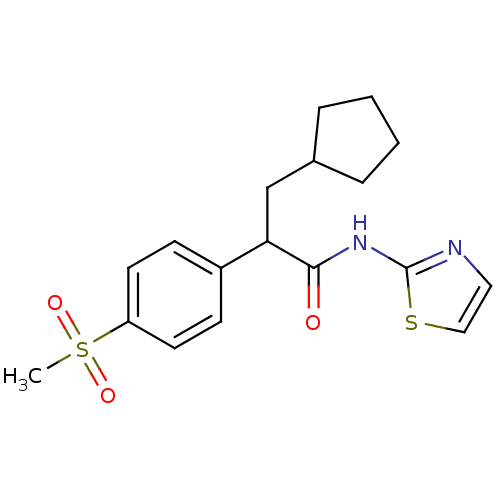
(3-Cyclopentyl-2-(4-methanesulfonyl-phenyl)-N-thiaz...)Show SMILES CS(=O)(=O)c1ccc(cc1)C(CC1CCCC1)C(=O)Nc1nccs1 Show InChI InChI=1S/C18H22N2O3S2/c1-25(22,23)15-8-6-14(7-9-15)16(12-13-4-2-3-5-13)17(21)20-18-19-10-11-24-18/h6-11,13,16H,2-5,12H2,1H3,(H,19,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 2.20E+3 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hexokinase-4
(Rattus norvegicus) | BDBM50161686

(3-(5-Chloro-thiazol-2-yl)-1-cyclopentylmethyl-1-(4...)Show SMILES FC(F)(F)c1ccc(cc1)N(CC1CCCC1)C(=O)Nc1ncc(Cl)s1 Show InChI InChI=1S/C17H17ClF3N3OS/c18-14-9-22-15(26-14)23-16(25)24(10-11-3-1-2-4-11)13-7-5-12(6-8-13)17(19,20)21/h5-9,11H,1-4,10H2,(H,22,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161687
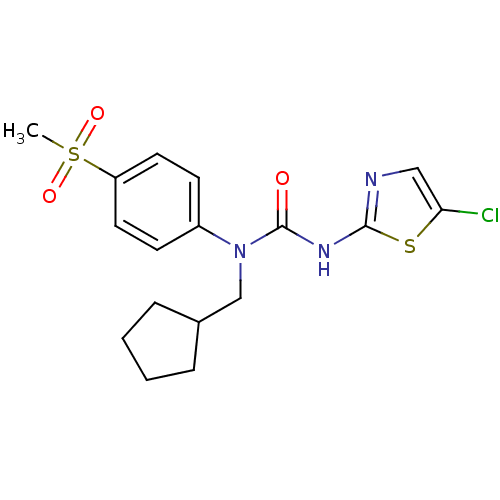
(3-(5-Chloro-thiazol-2-yl)-1-cyclopentylmethyl-1-(4...)Show SMILES CS(=O)(=O)c1ccc(cc1)N(CC1CCCC1)C(=O)Nc1ncc(Cl)s1 Show InChI InChI=1S/C17H20ClN3O3S2/c1-26(23,24)14-8-6-13(7-9-14)21(11-12-4-2-3-5-12)17(22)20-16-19-10-15(18)25-16/h6-10,12H,2-5,11H2,1H3,(H,19,20,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161688

(3-(5-Chloro-thiazol-2-yl)-1-(4-methanesulfonyl-phe...)Show SMILES CS(=O)(=O)c1ccc(cc1)N(Cc1cccs1)C(=O)Nc1ncc(Cl)s1 Show InChI InChI=1S/C16H14ClN3O3S3/c1-26(22,23)13-6-4-11(5-7-13)20(10-12-3-2-8-24-12)16(21)19-15-18-9-14(17)25-15/h2-9H,10H2,1H3,(H,18,19,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 6.60E+3 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161689
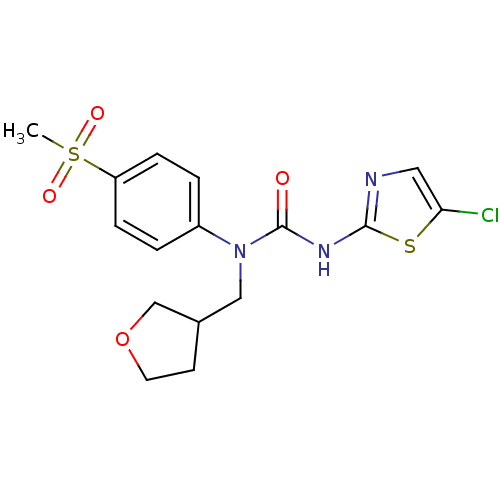
(3-(5-Chloro-thiazol-2-yl)-1-(4-methanesulfonyl-phe...)Show SMILES CS(=O)(=O)c1ccc(cc1)N(CC1CCOC1)C(=O)Nc1ncc(Cl)s1 Show InChI InChI=1S/C16H18ClN3O4S2/c1-26(22,23)13-4-2-12(3-5-13)20(9-11-6-7-24-10-11)16(21)19-15-18-8-14(17)25-15/h2-5,8,11H,6-7,9-10H2,1H3,(H,18,19,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.19E+4 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161690
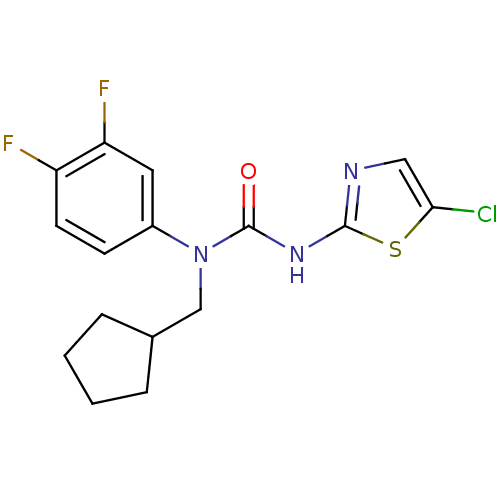
(3-(5-Chloro-thiazol-2-yl)-1-cyclopentylmethyl-1-(3...)Show InChI InChI=1S/C16H16ClF2N3OS/c17-14-8-20-15(24-14)21-16(23)22(9-10-3-1-2-4-10)11-5-6-12(18)13(19)7-11/h5-8,10H,1-4,9H2,(H,20,21,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161691

(CHEMBL180172 | N-{3-[3-(5-Chloro-thiazol-2-yl)-1-c...)Show SMILES CC(=O)Nc1cccc(c1)N(CC1CCCC1)C(=O)Nc1ncc(Cl)s1 Show InChI InChI=1S/C18H21ClN4O2S/c1-12(24)21-14-7-4-8-15(9-14)23(11-13-5-2-3-6-13)18(25)22-17-20-10-16(19)26-17/h4,7-10,13H,2-3,5-6,11H2,1H3,(H,21,24)(H,20,22,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 300 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161694
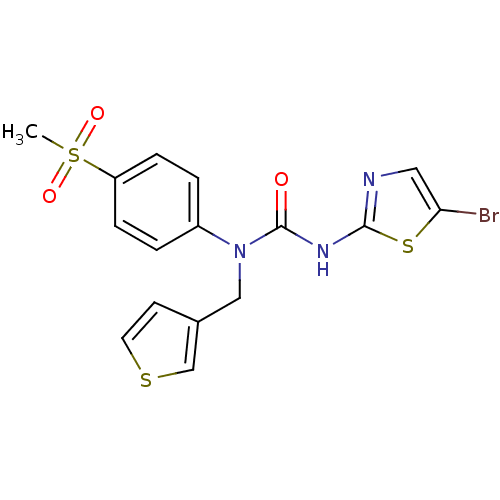
(3-(5-Bromo-thiazol-2-yl)-1-(4-methanesulfonyl-phen...)Show SMILES CS(=O)(=O)c1ccc(cc1)N(Cc1ccsc1)C(=O)Nc1ncc(Br)s1 Show InChI InChI=1S/C16H14BrN3O3S3/c1-26(22,23)13-4-2-12(3-5-13)20(9-11-6-7-24-10-11)16(21)19-15-18-8-14(17)25-15/h2-8,10H,9H2,1H3,(H,18,19,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 6.50E+3 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161693
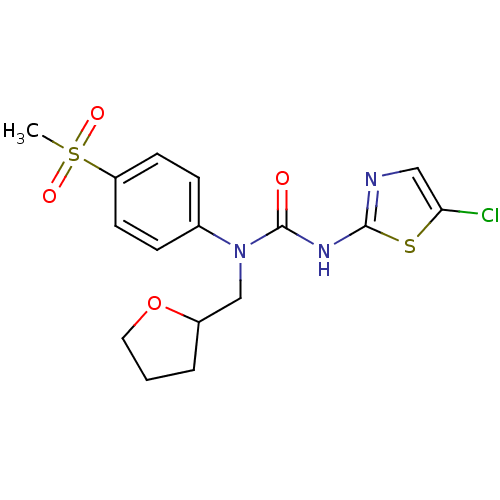
(3-(5-Chloro-thiazol-2-yl)-1-(4-methanesulfonyl-phe...)Show SMILES CS(=O)(=O)c1ccc(cc1)N(CC1CCCO1)C(=O)Nc1ncc(Cl)s1 Show InChI InChI=1S/C16H18ClN3O4S2/c1-26(22,23)13-6-4-11(5-7-13)20(10-12-3-2-8-24-12)16(21)19-15-18-9-14(17)25-15/h4-7,9,12H,2-3,8,10H2,1H3,(H,18,19,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.16E+4 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161668
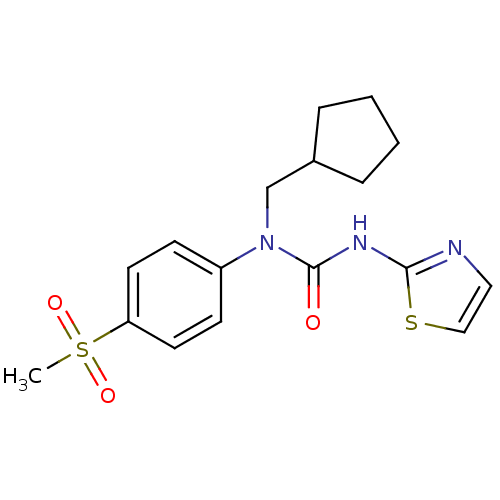
(1-Cyclopentylmethyl-1-(4-methanesulfonyl-phenyl)-3...)Show SMILES CS(=O)(=O)c1ccc(cc1)N(CC1CCCC1)C(=O)Nc1nccs1 Show InChI InChI=1S/C17H21N3O3S2/c1-25(22,23)15-8-6-14(7-9-15)20(12-13-4-2-3-5-13)17(21)19-16-18-10-11-24-16/h6-11,13H,2-5,12H2,1H3,(H,18,19,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 9.70E+3 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 15 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161695
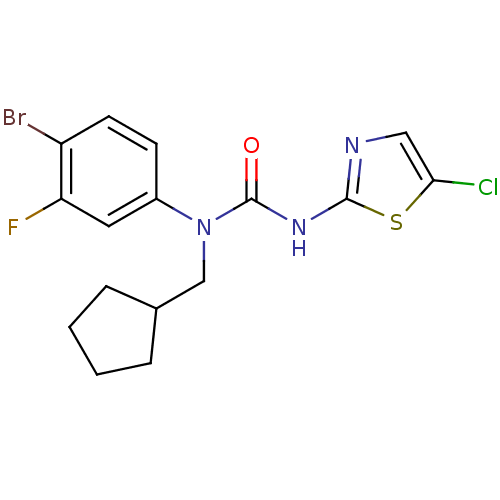
(1-(4-Bromo-3-fluoro-phenyl)-3-(5-chloro-thiazol-2-...)Show InChI InChI=1S/C16H16BrClFN3OS/c17-12-6-5-11(7-13(12)19)22(9-10-3-1-2-4-10)16(23)21-15-20-8-14(18)24-15/h5-8,10H,1-4,9H2,(H,20,21,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 700 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Rattus norvegicus) | BDBM50161692
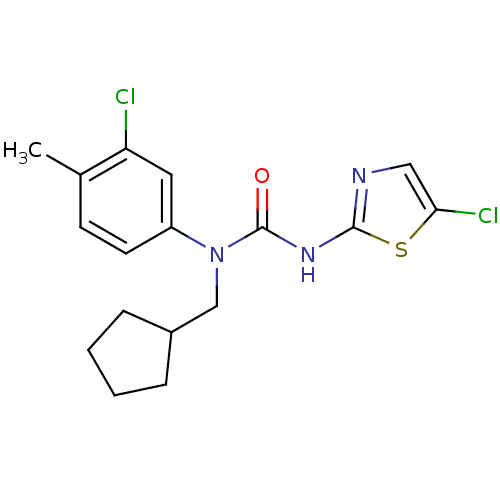
(1-(3-Chloro-4-methyl-phenyl)-3-(5-chloro-thiazol-2...)Show InChI InChI=1S/C17H19Cl2N3OS/c1-11-6-7-13(8-14(11)18)22(10-12-4-2-3-5-12)17(23)21-16-20-9-15(19)24-16/h6-9,12H,2-5,10H2,1H3,(H,20,21,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 400 | n/a | n/a | n/a | n/a |
OSI Pharmaceuticals
Curated by ChEMBL
| Assay Description
Effective concentration for glucokinase activation with 5 mM glucose |
Bioorg Med Chem Lett 15: 1501-4 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.083
BindingDB Entry DOI: 10.7270/Q2HT2NVC |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50251802
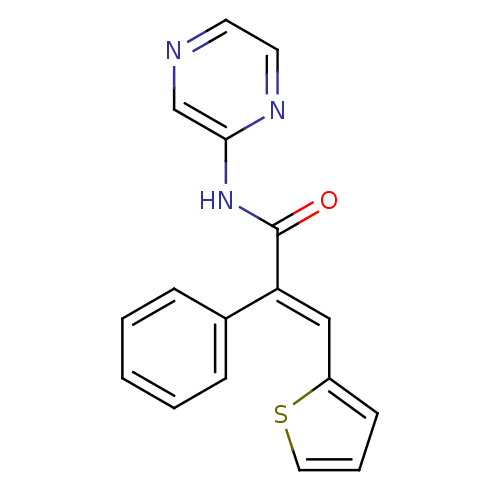
((E)-2-phenyl-N-(pyrazin-2-yl)-3-(thiophen-2-yl)acr...)Show InChI InChI=1S/C17H13N3OS/c21-17(20-16-12-18-8-9-19-16)15(11-14-7-4-10-22-14)13-5-2-1-3-6-13/h1-12H,(H,19,20,21)/b15-11+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.60E+4 | n/a | n/a | n/a | n/a |
Oxford OX4 6LT
Curated by ChEMBL
| Assay Description
Activation of GST fused human liver glucokinase assessed as generation of NADPH by G6PDH coupled assay |
J Med Chem 51: 4340-5 (2008)
Article DOI: 10.1021/jm8003202
BindingDB Entry DOI: 10.7270/Q2R49QKJ |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50251450
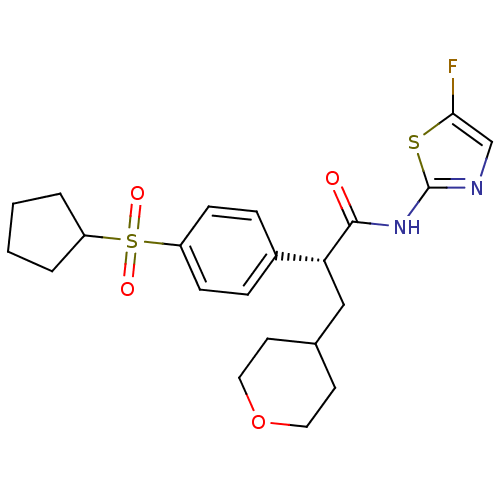
((R)-2-(4-(cyclopentylsulfonyl)phenyl)-N-(5-fluorot...)Show SMILES Fc1cnc(NC(=O)[C@H](CC2CCOCC2)c2ccc(cc2)S(=O)(=O)C2CCCC2)s1 |r| Show InChI InChI=1S/C22H27FN2O4S2/c23-20-14-24-22(30-20)25-21(26)19(13-15-9-11-29-12-10-15)16-5-7-18(8-6-16)31(27,28)17-3-1-2-4-17/h5-8,14-15,17,19H,1-4,9-13H2,(H,24,25,26)/t19-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 70 | n/a | n/a | n/a | n/a |
Oxford OX4 6LT
Curated by ChEMBL
| Assay Description
Activation of GST fused human liver glucokinase assessed as generation of NADPH by G6PDH coupled assay |
J Med Chem 51: 4340-5 (2008)
Article DOI: 10.1021/jm8003202
BindingDB Entry DOI: 10.7270/Q2R49QKJ |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50251421
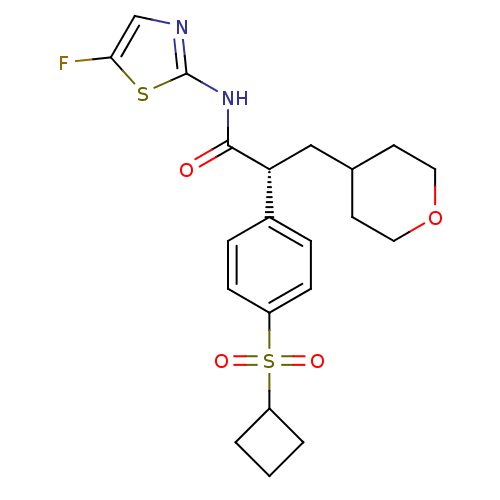
((R)-2-(4-(cyclobutylsulfonyl)phenyl)-N-(5-fluoroth...)Show SMILES Fc1cnc(NC(=O)[C@H](CC2CCOCC2)c2ccc(cc2)S(=O)(=O)C2CCC2)s1 |r| Show InChI InChI=1S/C21H25FN2O4S2/c22-19-13-23-21(29-19)24-20(25)18(12-14-8-10-28-11-9-14)15-4-6-17(7-5-15)30(26,27)16-2-1-3-16/h4-7,13-14,16,18H,1-3,8-12H2,(H,23,24,25)/t18-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 140 | n/a | n/a | n/a | n/a |
Oxford OX4 6LT
Curated by ChEMBL
| Assay Description
Activation of GST fused human liver glucokinase assessed as generation of NADPH by G6PDH coupled assay |
J Med Chem 51: 4340-5 (2008)
Article DOI: 10.1021/jm8003202
BindingDB Entry DOI: 10.7270/Q2R49QKJ |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50251392
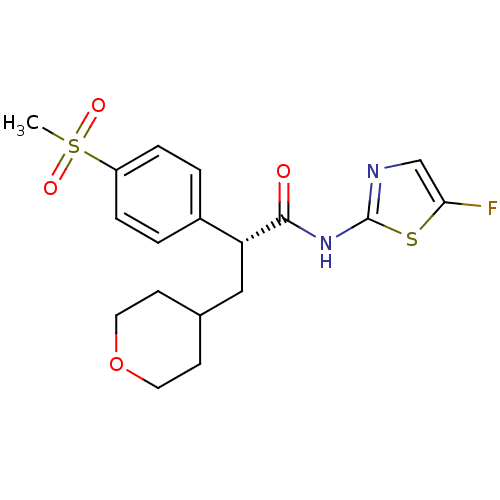
((R)-N-(5-fluorothiazol-2-yl)-2-(4-(methylsulfonyl)...)Show SMILES CS(=O)(=O)c1ccc(cc1)[C@@H](CC1CCOCC1)C(=O)Nc1ncc(F)s1 |r| Show InChI InChI=1S/C18H21FN2O4S2/c1-27(23,24)14-4-2-13(3-5-14)15(10-12-6-8-25-9-7-12)17(22)21-18-20-11-16(19)26-18/h2-5,11-12,15H,6-10H2,1H3,(H,20,21,22)/t15-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.43E+3 | n/a | n/a | n/a | n/a |
Oxford OX4 6LT
Curated by ChEMBL
| Assay Description
Activation of GST fused human liver glucokinase assessed as generation of NADPH by G6PDH coupled assay |
J Med Chem 51: 4340-5 (2008)
Article DOI: 10.1021/jm8003202
BindingDB Entry DOI: 10.7270/Q2R49QKJ |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50251391
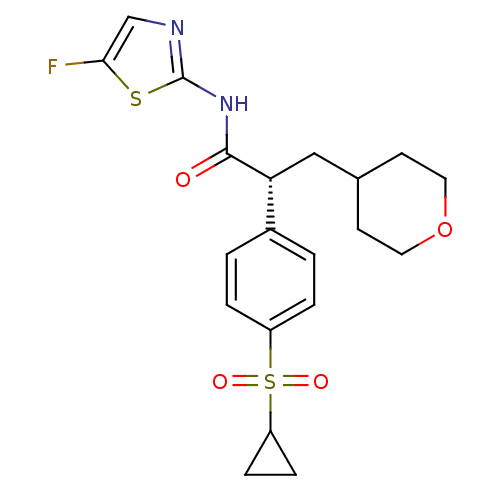
((2R)-2-(4-Cyclopropanesulfonylphenyl)-N-(5-fluorot...)Show SMILES Fc1cnc(NC(=O)[C@H](CC2CCOCC2)c2ccc(cc2)S(=O)(=O)C2CC2)s1 |r| Show InChI InChI=1S/C20H23FN2O4S2/c21-18-12-22-20(28-18)23-19(24)17(11-13-7-9-27-10-8-13)14-1-3-15(4-2-14)29(25,26)16-5-6-16/h1-4,12-13,16-17H,5-11H2,(H,22,23,24)/t17-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 130 | n/a | n/a | n/a | n/a |
Oxford OX4 6LT
Curated by ChEMBL
| Assay Description
Activation of GST fused human liver glucokinase assessed as generation of NADPH by G6PDH coupled assay |
J Med Chem 51: 4340-5 (2008)
Article DOI: 10.1021/jm8003202
BindingDB Entry DOI: 10.7270/Q2R49QKJ |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50251390

((R)-2-(4-(cyclopropylsulfonyl)phenyl)-N-(5-methylt...)Show SMILES Cc1cnc(NC(=O)[C@H](CC2CCOCC2)c2ccc(cc2)S(=O)(=O)C2CC2)s1 |r| Show InChI InChI=1S/C21H26N2O4S2/c1-14-13-22-21(28-14)23-20(24)19(12-15-8-10-27-11-9-15)16-2-4-17(5-3-16)29(25,26)18-6-7-18/h2-5,13,15,18-19H,6-12H2,1H3,(H,22,23,24)/t19-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 580 | n/a | n/a | n/a | n/a |
Oxford OX4 6LT
Curated by ChEMBL
| Assay Description
Activation of GST fused human liver glucokinase assessed as generation of NADPH by G6PDH coupled assay |
J Med Chem 51: 4340-5 (2008)
Article DOI: 10.1021/jm8003202
BindingDB Entry DOI: 10.7270/Q2R49QKJ |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50251389
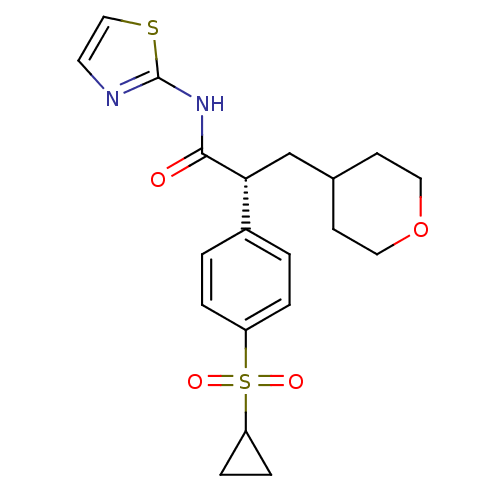
((R)-2-(4-(cyclopropylsulfonyl)phenyl)-3-(tetrahydr...)Show SMILES O=C(Nc1nccs1)[C@H](CC1CCOCC1)c1ccc(cc1)S(=O)(=O)C1CC1 |r| Show InChI InChI=1S/C20H24N2O4S2/c23-19(22-20-21-9-12-27-20)18(13-14-7-10-26-11-8-14)15-1-3-16(4-2-15)28(24,25)17-5-6-17/h1-4,9,12,14,17-18H,5-8,10-11,13H2,(H,21,22,23)/t18-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 570 | n/a | n/a | n/a | n/a |
Oxford OX4 6LT
Curated by ChEMBL
| Assay Description
Activation of GST fused human liver glucokinase assessed as generation of NADPH by G6PDH coupled assay |
J Med Chem 51: 4340-5 (2008)
Article DOI: 10.1021/jm8003202
BindingDB Entry DOI: 10.7270/Q2R49QKJ |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50251388
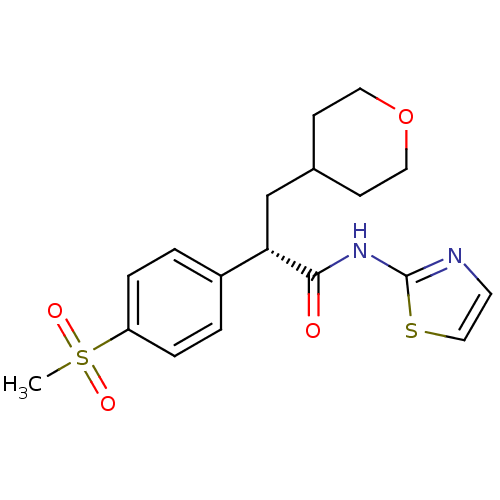
((S)-2-(4-(methylsulfonyl)phenyl)-3-(tetrahydro-2H-...)Show SMILES CS(=O)(=O)c1ccc(cc1)[C@H](CC1CCOCC1)C(=O)Nc1nccs1 |r| Show InChI InChI=1S/C18H22N2O4S2/c1-26(22,23)15-4-2-14(3-5-15)16(12-13-6-9-24-10-7-13)17(21)20-18-19-8-11-25-18/h2-5,8,11,13,16H,6-7,9-10,12H2,1H3,(H,19,20,21)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a |
Oxford OX4 6LT
Curated by ChEMBL
| Assay Description
Activation of GST fused human liver glucokinase assessed as generation of NADPH by G6PDH coupled assay |
J Med Chem 51: 4340-5 (2008)
Article DOI: 10.1021/jm8003202
BindingDB Entry DOI: 10.7270/Q2R49QKJ |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50251833
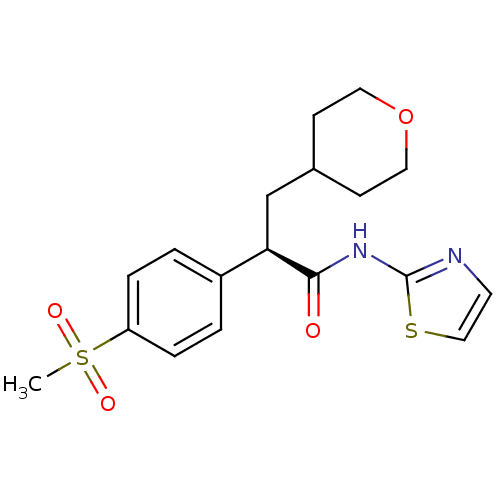
((R)-2-(4-(methylsulfonyl)phenyl)-3-(tetrahydro-2H-...)Show SMILES CS(=O)(=O)c1ccc(cc1)[C@@H](CC1CCOCC1)C(=O)Nc1nccs1 |r| Show InChI InChI=1S/C18H22N2O4S2/c1-26(22,23)15-4-2-14(3-5-15)16(12-13-6-9-24-10-7-13)17(21)20-18-19-8-11-25-18/h2-5,8,11,13,16H,6-7,9-10,12H2,1H3,(H,19,20,21)/t16-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 3.47E+3 | n/a | n/a | n/a | n/a |
Oxford OX4 6LT
Curated by ChEMBL
| Assay Description
Activation of GST fused human liver glucokinase assessed as generation of NADPH by G6PDH coupled assay |
J Med Chem 51: 4340-5 (2008)
Article DOI: 10.1021/jm8003202
BindingDB Entry DOI: 10.7270/Q2R49QKJ |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50251832
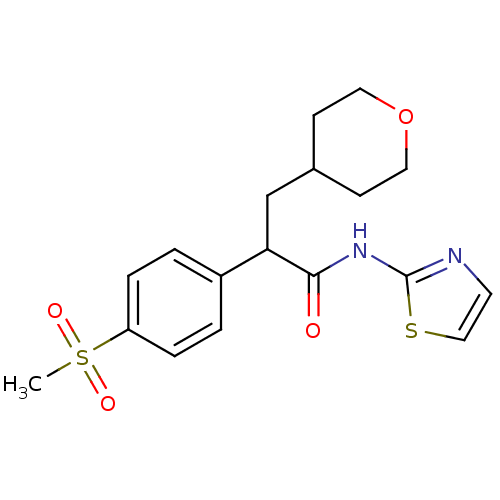
(2-(4-(methylsulfonyl)phenyl)-3-(tetrahydro-2H-pyra...)Show SMILES CS(=O)(=O)c1ccc(cc1)C(CC1CCOCC1)C(=O)Nc1nccs1 Show InChI InChI=1S/C18H22N2O4S2/c1-26(22,23)15-4-2-14(3-5-15)16(12-13-6-9-24-10-7-13)17(21)20-18-19-8-11-25-18/h2-5,8,11,13,16H,6-7,9-10,12H2,1H3,(H,19,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 8.93E+3 | n/a | n/a | n/a | n/a |
Oxford OX4 6LT
Curated by ChEMBL
| Assay Description
Activation of GST fused human liver glucokinase assessed as generation of NADPH by G6PDH coupled assay |
J Med Chem 51: 4340-5 (2008)
Article DOI: 10.1021/jm8003202
BindingDB Entry DOI: 10.7270/Q2R49QKJ |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50251831
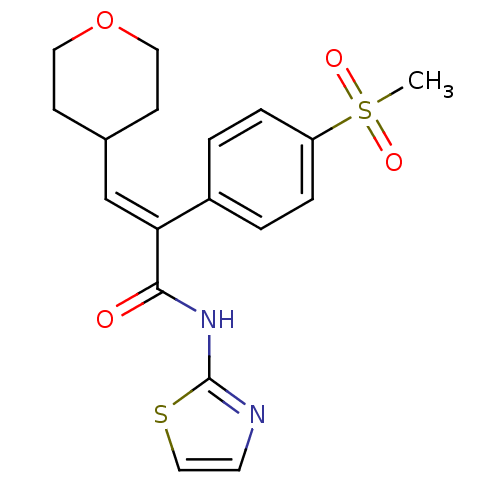
(2-(4-(methylsulfonyl)phenyl)-3-(tetrahydro-2H-pyra...)Show SMILES CS(=O)(=O)c1ccc(cc1)C(=C/C1CCOCC1)\C(=O)Nc1nccs1 Show InChI InChI=1S/C18H20N2O4S2/c1-26(22,23)15-4-2-14(3-5-15)16(12-13-6-9-24-10-7-13)17(21)20-18-19-8-11-25-18/h2-5,8,11-13H,6-7,9-10H2,1H3,(H,19,20,21)/b16-12+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.30E+3 | n/a | n/a | n/a | n/a |
Oxford OX4 6LT
Curated by ChEMBL
| Assay Description
Activation of GST fused human liver glucokinase assessed as generation of NADPH by G6PDH coupled assay |
J Med Chem 51: 4340-5 (2008)
Article DOI: 10.1021/jm8003202
BindingDB Entry DOI: 10.7270/Q2R49QKJ |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50251830

((Z)-2-(4-(methylsulfonyl)phenyl)-N-(thiazol-2-yl)-...)Show SMILES CS(=O)(=O)c1ccc(cc1)C(=C/c1cccs1)\C(=O)Nc1nccs1 Show InChI InChI=1S/C17H14N2O3S3/c1-25(21,22)14-6-4-12(5-7-14)15(11-13-3-2-9-23-13)16(20)19-17-18-8-10-24-17/h2-11H,1H3,(H,18,19,20)/b15-11+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 560 | n/a | n/a | n/a | n/a |
Oxford OX4 6LT
Curated by ChEMBL
| Assay Description
Activation of GST fused human liver glucokinase assessed as generation of NADPH by G6PDH coupled assay |
J Med Chem 51: 4340-5 (2008)
Article DOI: 10.1021/jm8003202
BindingDB Entry DOI: 10.7270/Q2R49QKJ |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50251829
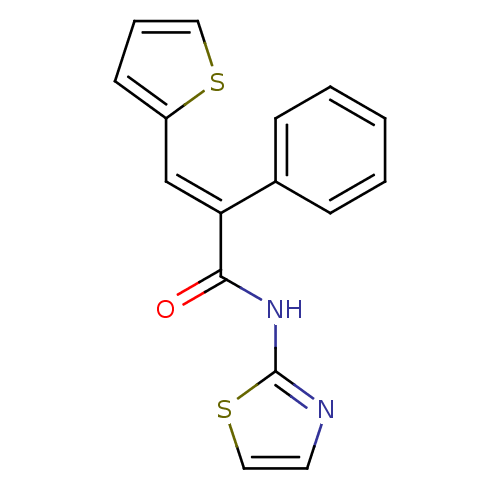
(2-phenyl-N-(thiazol-2-yl)-3-(thiophen-2-yl)acrylam...)Show InChI InChI=1S/C16H12N2OS2/c19-15(18-16-17-8-10-21-16)14(11-13-7-4-9-20-13)12-5-2-1-3-6-12/h1-11H,(H,17,18,19)/b14-11+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.08E+4 | n/a | n/a | n/a | n/a |
Oxford OX4 6LT
Curated by ChEMBL
| Assay Description
Activation of GST fused human liver glucokinase assessed as generation of NADPH by G6PDH coupled assay |
J Med Chem 51: 4340-5 (2008)
Article DOI: 10.1021/jm8003202
BindingDB Entry DOI: 10.7270/Q2R49QKJ |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50543888
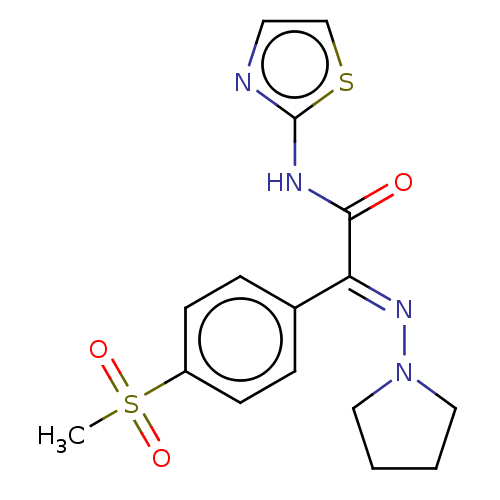
(CHEMBL4641724)Show SMILES CS(=O)(=O)c1ccc(cc1)C(=N/N1CCCC1)\C(=O)Nc1nccs1 Show InChI InChI=1S/C16H18N4O3S2/c1-25(22,23)13-6-4-12(5-7-13)14(19-20-9-2-3-10-20)15(21)18-16-17-8-11-24-16/h4-8,11H,2-3,9-10H2,1H3,(H,17,18,21)/b19-14+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 5.60E+3 | n/a | n/a | n/a | n/a |
Chiba University
Curated by ChEMBL
| Assay Description
Activation of human glucokinase |
Bioorg Med Chem Lett 30: (2020)
Article DOI: 10.1016/j.bmcl.2020.127249
BindingDB Entry DOI: 10.7270/Q2P84GG5 |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50543889

(CHEMBL4645293)Show SMILES O=C(Nc1nccs1)C(=N\N1CCCC1)\c1ccc(cc1)S(=O)(=O)C1CC1 Show InChI InChI=1S/C18H20N4O3S2/c23-17(20-18-19-9-12-26-18)16(21-22-10-1-2-11-22)13-3-5-14(6-4-13)27(24,25)15-7-8-15/h3-6,9,12,15H,1-2,7-8,10-11H2,(H,19,20,23)/b21-16+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 420 | n/a | n/a | n/a | n/a |
Chiba University
Curated by ChEMBL
| Assay Description
Activation of human glucokinase |
Bioorg Med Chem Lett 30: (2020)
Article DOI: 10.1016/j.bmcl.2020.127249
BindingDB Entry DOI: 10.7270/Q2P84GG5 |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50543890

(CHEMBL4641678)Show SMILES Clc1cnc(NC(=O)C(=N\N2CCCC2)\c2ccc(cc2)S(=O)(=O)C2CC2)s1 Show InChI InChI=1S/C18H19ClN4O3S2/c19-15-11-20-18(27-15)21-17(24)16(22-23-9-1-2-10-23)12-3-5-13(6-4-12)28(25,26)14-7-8-14/h3-6,11,14H,1-2,7-10H2,(H,20,21,24)/b22-16+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 41 | n/a | n/a | n/a | n/a |
Chiba University
Curated by ChEMBL
| Assay Description
Activation of human glucokinase |
Bioorg Med Chem Lett 30: (2020)
Article DOI: 10.1016/j.bmcl.2020.127249
BindingDB Entry DOI: 10.7270/Q2P84GG5 |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50543891
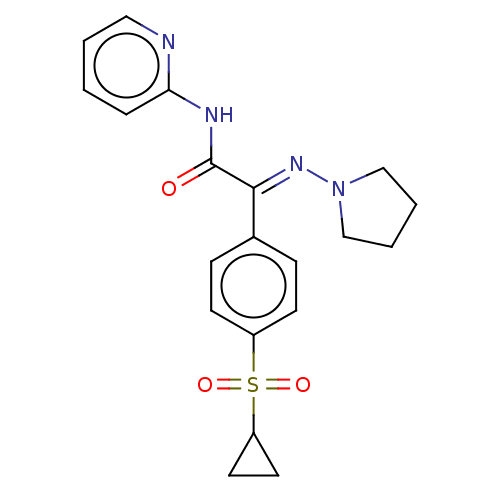
(CHEMBL4638344)Show SMILES O=C(Nc1ccccn1)C(=N\N1CCCC1)\c1ccc(cc1)S(=O)(=O)C1CC1 Show InChI InChI=1S/C20H22N4O3S/c25-20(22-18-5-1-2-12-21-18)19(23-24-13-3-4-14-24)15-6-8-16(9-7-15)28(26,27)17-10-11-17/h1-2,5-9,12,17H,3-4,10-11,13-14H2,(H,21,22,25)/b23-19+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a |
Chiba University
Curated by ChEMBL
| Assay Description
Activation of human glucokinase |
Bioorg Med Chem Lett 30: (2020)
Article DOI: 10.1016/j.bmcl.2020.127249
BindingDB Entry DOI: 10.7270/Q2P84GG5 |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50543892

(CHEMBL4649482)Show SMILES O=C(Nc1ncns1)C(=N\N1CCCC1)\c1ccc(cc1)S(=O)(=O)C1CC1 Show InChI InChI=1S/C17H19N5O3S2/c23-16(20-17-18-11-19-26-17)15(21-22-9-1-2-10-22)12-3-5-13(6-4-12)27(24,25)14-7-8-14/h3-6,11,14H,1-2,7-10H2,(H,18,19,20,23)/b21-15+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 160 | n/a | n/a | n/a | n/a |
Chiba University
Curated by ChEMBL
| Assay Description
Activation of human glucokinase |
Bioorg Med Chem Lett 30: (2020)
Article DOI: 10.1016/j.bmcl.2020.127249
BindingDB Entry DOI: 10.7270/Q2P84GG5 |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50543893

(CHEMBL4632835)Show SMILES Cc1cnc(NC(=O)C(=N\N2CCCC2)\c2ccc(cc2)S(=O)(=O)C2CC2)s1 Show InChI InChI=1S/C19H22N4O3S2/c1-13-12-20-19(27-13)21-18(24)17(22-23-10-2-3-11-23)14-4-6-15(7-5-14)28(25,26)16-8-9-16/h4-7,12,16H,2-3,8-11H2,1H3,(H,20,21,24)/b22-17+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 220 | n/a | n/a | n/a | n/a |
Chiba University
Curated by ChEMBL
| Assay Description
Activation of human glucokinase |
Bioorg Med Chem Lett 30: (2020)
Article DOI: 10.1016/j.bmcl.2020.127249
BindingDB Entry DOI: 10.7270/Q2P84GG5 |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50543894

(CHEMBL4637653)Show SMILES Cc1nnc(NC(=O)C(=N\N2CCCC2)\c2ccc(cc2)S(=O)(=O)C2CC2)s1 Show InChI InChI=1S/C18H21N5O3S2/c1-12-20-21-18(27-12)19-17(24)16(22-23-10-2-3-11-23)13-4-6-14(7-5-13)28(25,26)15-8-9-15/h4-7,15H,2-3,8-11H2,1H3,(H,19,21,24)/b22-16+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.17E+3 | n/a | n/a | n/a | n/a |
Chiba University
Curated by ChEMBL
| Assay Description
Activation of human glucokinase |
Bioorg Med Chem Lett 30: (2020)
Article DOI: 10.1016/j.bmcl.2020.127249
BindingDB Entry DOI: 10.7270/Q2P84GG5 |
More data for this
Ligand-Target Pair | |
Hexokinase-4
(Homo sapiens (Human)) | BDBM50543895

(CHEMBL4646396)Show SMILES CN1CCN(Cc2cnc(NC(=O)C(=N\N3CCCC3)\c3ccc(cc3)S(=O)(=O)C3CC3)s2)CC1 Show InChI InChI=1S/C24H32N6O3S2/c1-28-12-14-29(15-13-28)17-19-16-25-24(34-19)26-23(31)22(27-30-10-2-3-11-30)18-4-6-20(7-5-18)35(32,33)21-8-9-21/h4-7,16,21H,2-3,8-15,17H2,1H3,(H,25,26,31)/b27-22+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 700 | n/a | n/a | n/a | n/a |
Chiba University
Curated by ChEMBL
| Assay Description
Activation of human glucokinase |
Bioorg Med Chem Lett 30: (2020)
Article DOI: 10.1016/j.bmcl.2020.127249
BindingDB Entry DOI: 10.7270/Q2P84GG5 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data