

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Prothrombin (Homo sapiens (Human)) | BDBM50075121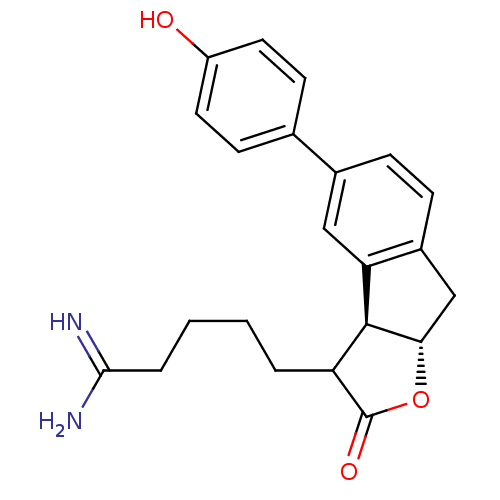 (5-[(3aS,8aS)-5-(4-Hydroxy-phenyl)-2-oxo-3,3a,8,8a-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity of the compound against human thrombin was determined | Bioorg Med Chem Lett 9: 431-6 (1999) BindingDB Entry DOI: 10.7270/Q26T0KS3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50078554 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-6-(3-diethy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of thrombin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50078544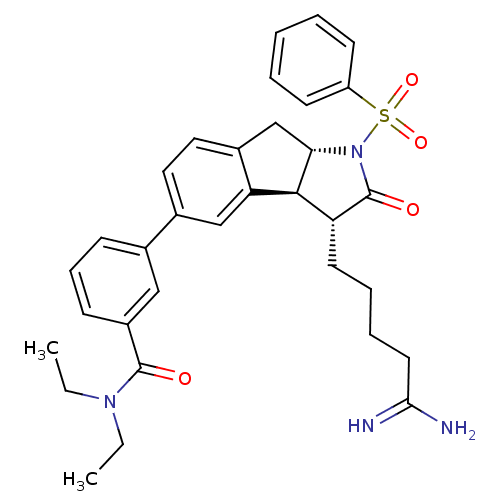 (3-[(3R,3aS,8aS)-1-Benzenesulfonyl-3-(4-carbamimido...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of thrombin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine protease 1 (Homo sapiens (Human)) | BDBM50078554 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-6-(3-diethy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of trypsin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50078553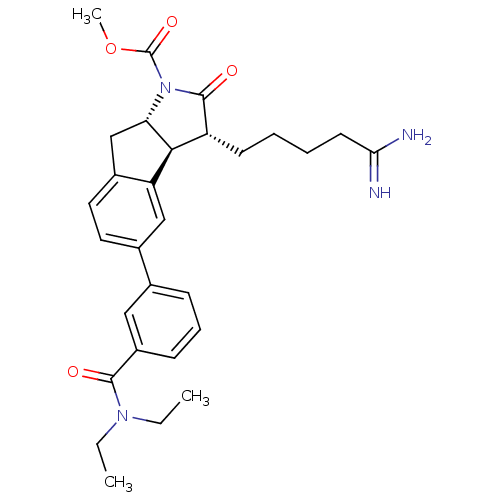 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-5-(3-diethy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of thrombin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine protease 1 (Homo sapiens (Human)) | BDBM50078544 (3-[(3R,3aS,8aS)-1-Benzenesulfonyl-3-(4-carbamimido...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 66 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of trypsin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50078544 (3-[(3R,3aS,8aS)-1-Benzenesulfonyl-3-(4-carbamimido...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 74 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of activated Coagulation factor X | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50075122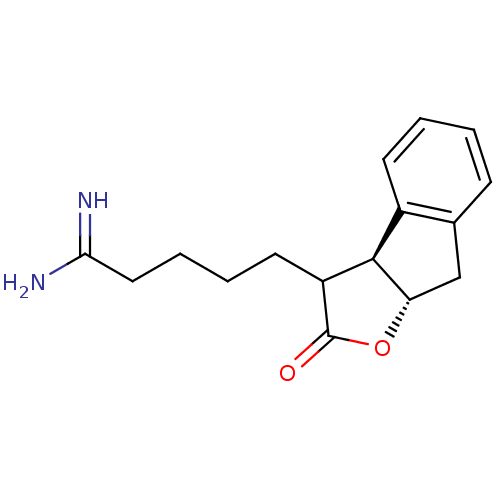 (5-((3aS,8aS)-2-Oxo-3,3a,8,8a-tetrahydro-2H-indeno[...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 77 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity of the compound against human thrombin was determined | Bioorg Med Chem Lett 9: 431-6 (1999) BindingDB Entry DOI: 10.7270/Q26T0KS3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50078554 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-6-(3-diethy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 120 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of activated Coagulation factor X | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50078552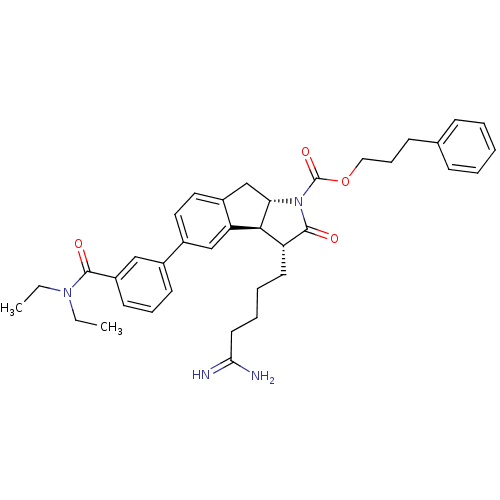 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-5-(3-diethy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of thrombin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50072293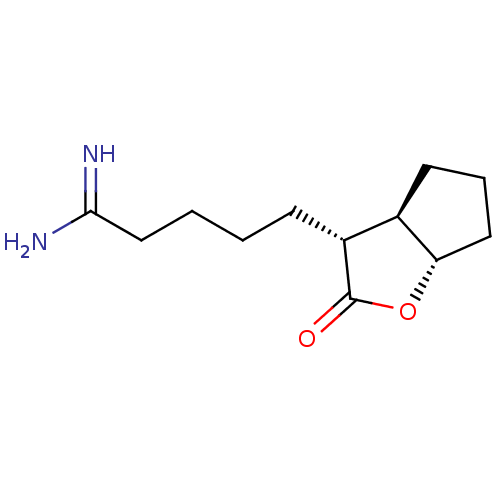 (5-((3R,3aR,6aS)-2-Oxo-hexahydro-cyclopenta[b]furan...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity of the compound against human thrombin was determined | Bioorg Med Chem Lett 9: 431-6 (1999) BindingDB Entry DOI: 10.7270/Q26T0KS3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50078556 (3-[(3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-2-oxo-1-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 380 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of thrombin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50075123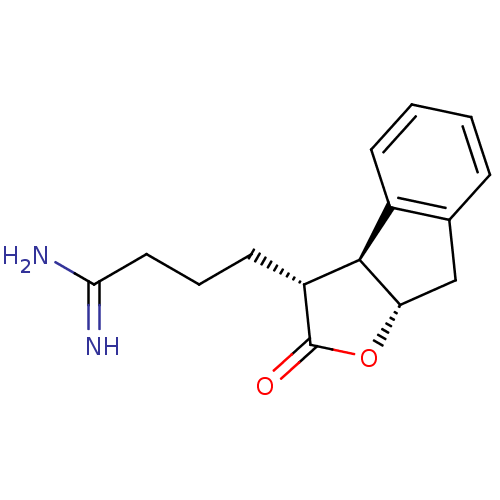 (4-((3R,3aS,8aS)-2-Oxo-3,3a,8,8a-tetrahydro-2H-inde...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 600 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity of the compound against human thrombin was determined | Bioorg Med Chem Lett 9: 431-6 (1999) BindingDB Entry DOI: 10.7270/Q26T0KS3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine protease 1 (Homo sapiens (Human)) | BDBM50078557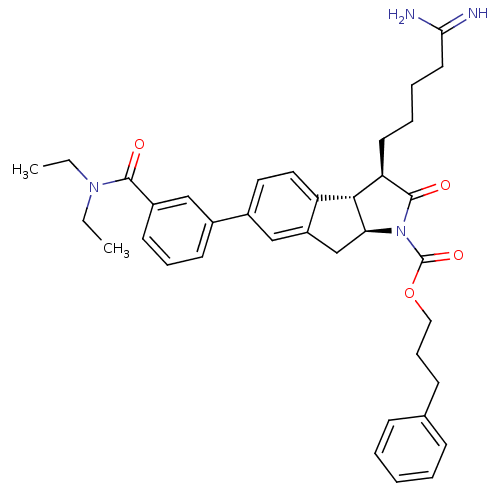 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-6-(3-diethy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 620 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of trypsin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50078557 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-6-(3-diethy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 660 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of thrombin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50075120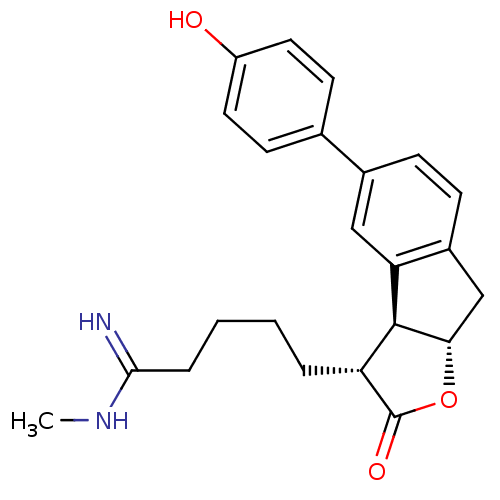 (5-[(3R,3aS,8aS)-5-(4-Hydroxy-phenyl)-2-oxo-3,3a,8,...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity of the compound against human thrombin was determined | Bioorg Med Chem Lett 9: 431-6 (1999) BindingDB Entry DOI: 10.7270/Q26T0KS3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine protease 1 (Homo sapiens (Human)) | BDBM50078549 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-6-(3-diethy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of trypsin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine protease 1 (Homo sapiens (Human)) | BDBM50078551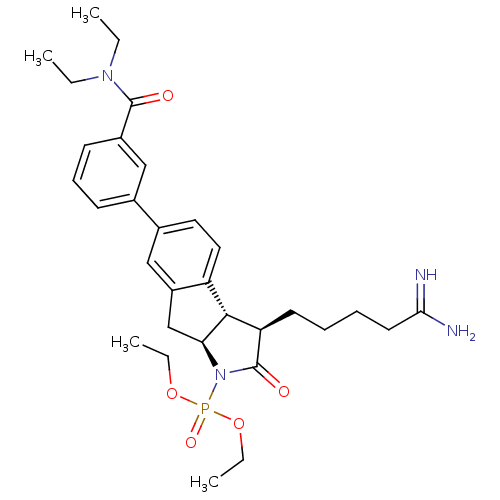 (CHEMBL46365 | [(3R,3aS,8aS)-3-(4-Carbamimidoyl-but...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of trypsin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50075119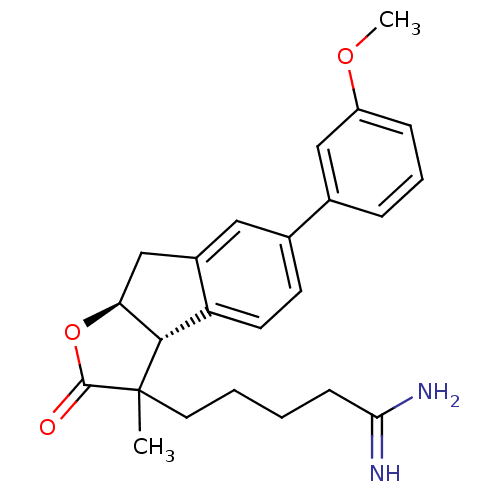 (5-[(3aR,8aS)-6-(3-Methoxy-phenyl)-3-methyl-2-oxo-3...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibitory activity of the compound against human thrombin was determined | Bioorg Med Chem Lett 9: 431-6 (1999) BindingDB Entry DOI: 10.7270/Q26T0KS3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine protease 1 (Homo sapiens (Human)) | BDBM50078553 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-5-(3-diethy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of trypsin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50078553 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-5-(3-diethy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of activated Coagulation factor X | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine protease 1 (Homo sapiens (Human)) | BDBM50078552 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-5-(3-diethy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of trypsin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50078546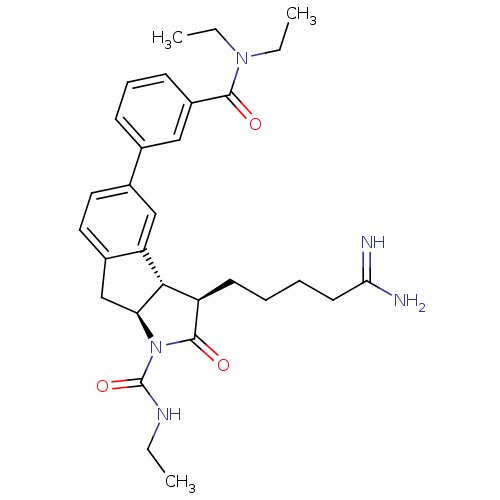 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-5-(3-diethy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of thrombin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50078551 (CHEMBL46365 | [(3R,3aS,8aS)-3-(4-Carbamimidoyl-but...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of thrombin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50078557 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-6-(3-diethy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of activated Coagulation factor X | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50078552 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-5-(3-diethy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 3.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of activated Coagulation factor X | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50078560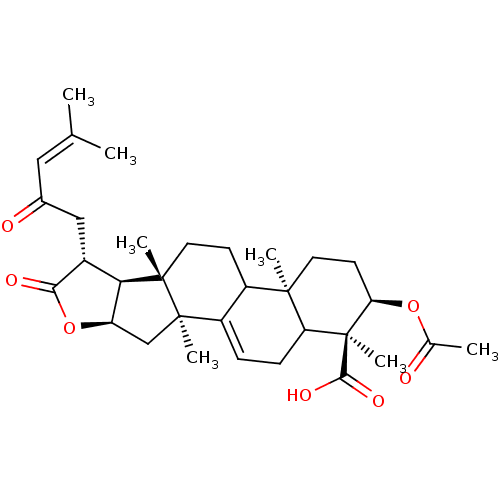 ((1S,2R,4aR,6aS,6bS,7R,9aR,10aS)-2-Acetoxy-1,4a,6a,...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of thrombin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50078548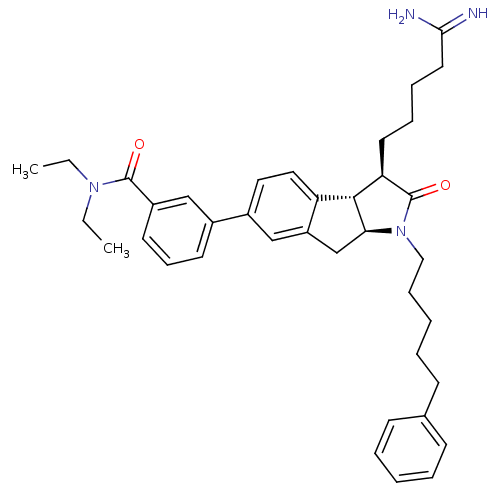 (3-[(3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-2-oxo-1-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of thrombin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50078547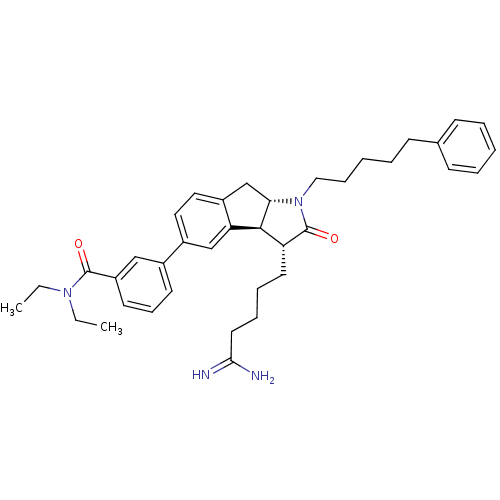 (3-[(3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-2-oxo-1-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of thrombin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50078549 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-6-(3-diethy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 5.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of thrombin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50078549 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-6-(3-diethy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 5.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of activated Coagulation factor X | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine protease 1 (Homo sapiens (Human)) | BDBM50078556 (3-[(3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-2-oxo-1-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 8.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of trypsin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine protease 1 (Homo sapiens (Human)) | BDBM50078548 (3-[(3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-2-oxo-1-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 8.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of trypsin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine protease 1 (Homo sapiens (Human)) | BDBM50078558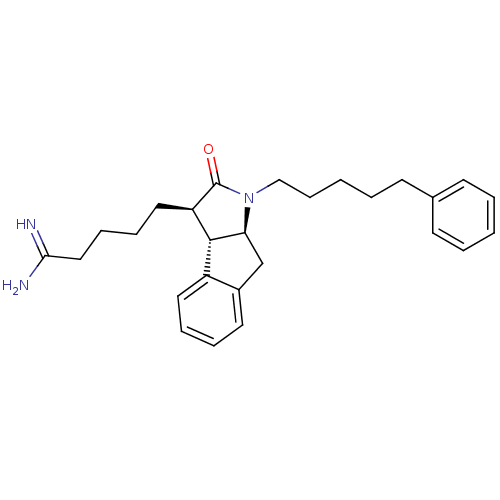 (5-[(3R,3aS,8aS)-2-Oxo-1-(5-phenyl-pentyl)-1,2,3,3a...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 9.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of trypsin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50078556 (3-[(3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-2-oxo-1-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 9.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of activated Coagulation factor X | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50078555 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-5-(3-diethy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of thrombin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50078545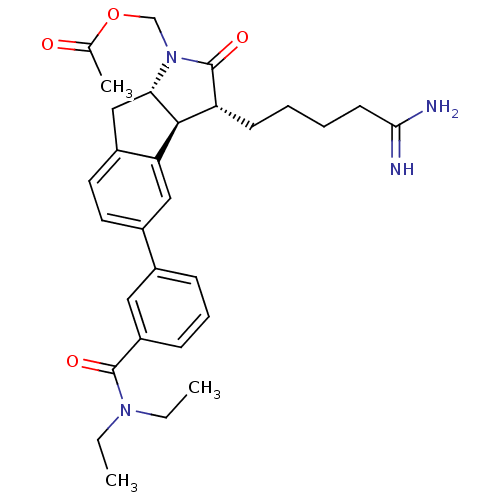 (Acetic acid (3R,3aS,8aS)-3-(4-carbamimidoyl-butyl)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of thrombin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50078558 (5-[(3R,3aS,8aS)-2-Oxo-1-(5-phenyl-pentyl)-1,2,3,3a...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of thrombin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50078545 (Acetic acid (3R,3aS,8aS)-3-(4-carbamimidoyl-butyl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of activated Coagulation factor X | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine protease 1 (Homo sapiens (Human)) | BDBM50078555 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-5-(3-diethy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of trypsin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50078551 (CHEMBL46365 | [(3R,3aS,8aS)-3-(4-Carbamimidoyl-but...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | n/a | n/a | 2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of activated Coagulation factor X | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50078555 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-5-(3-diethy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of activated Coagulation factor X | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50078548 (3-[(3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-2-oxo-1-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of activated Coagulation factor X | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine protease 1 (Homo sapiens (Human)) | BDBM50078545 (Acetic acid (3R,3aS,8aS)-3-(4-carbamimidoyl-butyl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 3.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of trypsin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50078550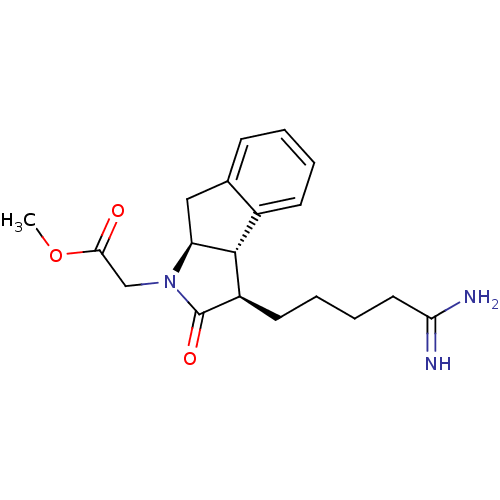 (CHEMBL295211 | [(3R,3aS,8aS)-3-(4-Carbamimidoyl-bu...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 4.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of thrombin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine protease 1 (Homo sapiens (Human)) | BDBM50078547 (3-[(3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-2-oxo-1-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of trypsin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50078547 (3-[(3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-2-oxo-1-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of activated Coagulation factor X | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50078546 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-5-(3-diethy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of activated Coagulation factor X | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine protease 1 (Homo sapiens (Human)) | BDBM50078546 ((3R,3aS,8aS)-3-(4-Carbamimidoyl-butyl)-5-(3-diethy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of trypsin | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50078558 (5-[(3R,3aS,8aS)-2-Oxo-1-(5-phenyl-pentyl)-1,2,3,3a...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Inhibition of activated Coagulation factor X | Bioorg Med Chem Lett 9: 1657-62 (1999) BindingDB Entry DOI: 10.7270/Q2VT1R8Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 55 total ) | Next | Last >> |