 Found 897 hits with Last Name = 'price' and Initial = 'da'
Found 897 hits with Last Name = 'price' and Initial = 'da' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Muscarinic acetylcholine receptor M4
(Homo sapiens (Human)) | BDBM50296345

((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...)Show SMILES C[N+]1(C)[C@@H]2C[C@H](C[C@@H]1[C@@H]1O[C@H]21)OC(=O)C(O)(c1cccs1)c1cccs1 |TLB:11:5:1:8.10| Show InChI InChI=1S/C19H22NO4S2/c1-20(2)12-9-11(10-13(20)17-16(12)24-17)23-18(21)19(22,14-5-3-7-25-14)15-6-4-8-26-15/h3-8,11-13,16-17,22H,9-10H2,1-2H3/q+1/t11-,12-,13-,16-,17+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M4 receptor expressed in CHO cells after 24 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50296345

((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...)Show SMILES C[N+]1(C)[C@@H]2C[C@H](C[C@@H]1[C@@H]1O[C@H]21)OC(=O)C(O)(c1cccs1)c1cccs1 |TLB:11:5:1:8.10| Show InChI InChI=1S/C19H22NO4S2/c1-20(2)12-9-11(10-13(20)17-16(12)24-17)23-18(21)19(22,14-5-3-7-25-14)15-6-4-8-26-15/h3-8,11-13,16-17,22H,9-10H2,1-2H3/q+1/t11-,12-,13-,16-,17+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00980 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 24 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(Homo sapiens (Human)) | BDBM50296345

((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...)Show SMILES C[N+]1(C)[C@@H]2C[C@H](C[C@@H]1[C@@H]1O[C@H]21)OC(=O)C(O)(c1cccs1)c1cccs1 |TLB:11:5:1:8.10| Show InChI InChI=1S/C19H22NO4S2/c1-20(2)12-9-11(10-13(20)17-16(12)24-17)23-18(21)19(22,14-5-3-7-25-14)15-6-4-8-26-15/h3-8,11-13,16-17,22H,9-10H2,1-2H3/q+1/t11-,12-,13-,16-,17+/m1/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M1 receptor expressed in CHO cells after 24 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50296345

((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...)Show SMILES C[N+]1(C)[C@@H]2C[C@H](C[C@@H]1[C@@H]1O[C@H]21)OC(=O)C(O)(c1cccs1)c1cccs1 |TLB:11:5:1:8.10| Show InChI InChI=1S/C19H22NO4S2/c1-20(2)12-9-11(10-13(20)17-16(12)24-17)23-18(21)19(22,14-5-3-7-25-14)15-6-4-8-26-15/h3-8,11-13,16-17,22H,9-10H2,1-2H3/q+1/t11-,12-,13-,16-,17+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0215 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M2 receptor expressed in CHO cells after 24 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M5
(Homo sapiens (Human)) | BDBM50296345

((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...)Show SMILES C[N+]1(C)[C@@H]2C[C@H](C[C@@H]1[C@@H]1O[C@H]21)OC(=O)C(O)(c1cccs1)c1cccs1 |TLB:11:5:1:8.10| Show InChI InChI=1S/C19H22NO4S2/c1-20(2)12-9-11(10-13(20)17-16(12)24-17)23-18(21)19(22,14-5-3-7-25-14)15-6-4-8-26-15/h3-8,11-13,16-17,22H,9-10H2,1-2H3/q+1/t11-,12-,13-,16-,17+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0266 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M5 receptor expressed in CHO cells after 24 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355612
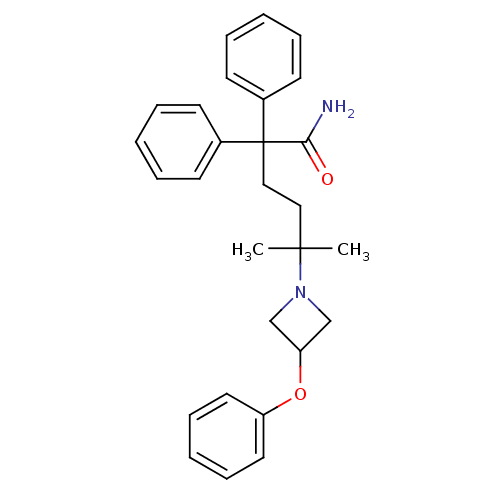
(CHEMBL1910848)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC(C1)Oc1ccccc1 Show InChI InChI=1S/C28H32N2O2/c1-27(2,30-20-25(21-30)32-24-16-10-5-11-17-24)18-19-28(26(29)31,22-12-6-3-7-13-22)23-14-8-4-9-15-23/h3-17,25H,18-21H2,1-2H3,(H2,29,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50296345

((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...)Show SMILES C[N+]1(C)[C@@H]2C[C@H](C[C@@H]1[C@@H]1O[C@H]21)OC(=O)C(O)(c1cccs1)c1cccs1 |TLB:11:5:1:8.10| Show InChI InChI=1S/C19H22NO4S2/c1-20(2)12-9-11(10-13(20)17-16(12)24-17)23-18(21)19(22,14-5-3-7-25-14)15-6-4-8-26-15/h3-8,11-13,16-17,22H,9-10H2,1-2H3/q+1/t11-,12-,13-,16-,17+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Glucagon-like peptide 1 receptor
(Homo sapiens (Human)) | BDBM50241203
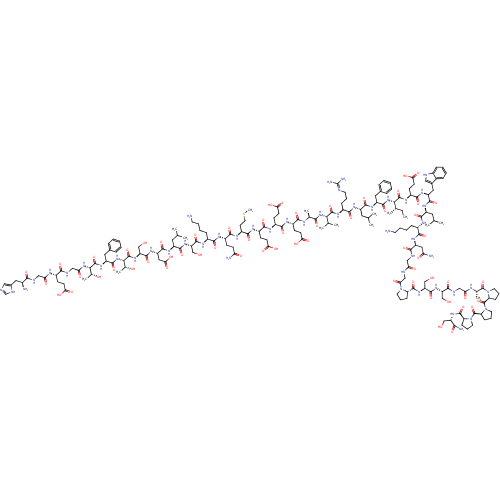
(CHEMBL414357 | HGEGTFTSDLSKQMEEEAVRLFIEWLKNGGPSSGS...)Show SMILES [H][C@](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H](N)Cc1cnc[nH]1)([C@@H](C)O)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@]([H])([C@@H](C)O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@]([H])([C@@H](C)CC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)NCC(=O)NCC(=O)N1CCC[C@@]1([H])C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](C)C(=O)N1CCC[C@@]1([H])C(=O)N1CCC[C@@]1([H])C(=O)N1CCC[C@@]1([H])C(=O)N[C@@H](CO)C(N)=O |r| Show InChI InChI=1S/C184H282N50O60S/c1-16-94(10)147(178(289)213-114(52-58-144(257)258)163(274)218-121(73-101-77-195-105-39-24-23-38-103(101)105)168(279)215-116(68-90(2)3)165(276)205-107(41-26-28-61-186)158(269)219-122(75-134(189)243)154(265)198-79-135(244)196-83-139(248)231-63-30-43-129(231)175(286)225-127(87-238)174(285)223-125(85-236)155(266)200-80-136(245)202-96(12)181(292)233-65-32-45-131(233)183(294)234-66-33-46-132(234)182(293)232-64-31-44-130(232)176(287)222-124(84-235)150(190)261)229-170(281)119(71-99-34-19-17-20-35-99)217-166(277)117(69-91(4)5)214-159(270)108(42-29-62-194-184(191)192)212-177(288)146(93(8)9)228-151(262)95(11)203-156(267)111(49-55-141(251)252)208-161(272)112(50-56-142(253)254)209-162(273)113(51-57-143(255)256)210-164(275)115(59-67-295-15)211-160(271)110(47-53-133(188)242)207-157(268)106(40-25-27-60-185)206-172(283)126(86-237)224-167(278)118(70-92(6)7)216-169(280)123(76-145(259)260)220-173(284)128(88-239)226-180(291)149(98(14)241)230-171(282)120(72-100-36-21-18-22-37-100)221-179(290)148(97(13)240)227-138(247)82-199-153(264)109(48-54-140(249)250)204-137(246)81-197-152(263)104(187)74-102-78-193-89-201-102/h17-24,34-39,77-78,89-98,104,106-132,146-149,195,235-241H,16,25-33,40-76,79-88,185-187H2,1-15H3,(H2,188,242)(H2,189,243)(H2,190,261)(H,193,201)(H,196,244)(H,197,263)(H,198,265)(H,199,264)(H,200,266)(H,202,245)(H,203,267)(H,204,246)(H,205,276)(H,206,283)(H,207,268)(H,208,272)(H,209,273)(H,210,275)(H,211,271)(H,212,288)(H,213,289)(H,214,270)(H,215,279)(H,216,280)(H,217,277)(H,218,274)(H,219,269)(H,220,284)(H,221,290)(H,222,287)(H,223,285)(H,224,278)(H,225,286)(H,226,291)(H,227,247)(H,228,262)(H,229,281)(H,230,282)(H,249,250)(H,251,252)(H,253,254)(H,255,256)(H,257,258)(H,259,260)(H4,191,192,194)/t94-,95-,96-,97+,98+,104-,106-,107-,108-,109-,110-,111-,112-,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,146-,147-,148-,149-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01856
BindingDB Entry DOI: 10.7270/Q2736W0V |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50355612

(CHEMBL1910848)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC(C1)Oc1ccccc1 Show InChI InChI=1S/C28H32N2O2/c1-27(2,30-20-25(21-30)32-24-16-10-5-11-17-24)18-19-28(26(29)31,22-12-6-3-7-13-22)23-14-8-4-9-15-23/h3-17,25H,18-21H2,1-2H3,(H2,29,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0560 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M2 receptor expressed in CHO cells after 2 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50296345

((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...)Show SMILES C[N+]1(C)[C@@H]2C[C@H](C[C@@H]1[C@@H]1O[C@H]21)OC(=O)C(O)(c1cccs1)c1cccs1 |TLB:11:5:1:8.10| Show InChI InChI=1S/C19H22NO4S2/c1-20(2)12-9-11(10-13(20)17-16(12)24-17)23-18(21)19(22,14-5-3-7-25-14)15-6-4-8-26-15/h3-8,11-13,16-17,22H,9-10H2,1-2H3/q+1/t11-,12-,13-,16-,17+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0610 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M2 receptor expressed in CHO cells after 2 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50296345

((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...)Show SMILES C[N+]1(C)[C@@H]2C[C@H](C[C@@H]1[C@@H]1O[C@H]21)OC(=O)C(O)(c1cccs1)c1cccs1 |TLB:11:5:1:8.10| Show InChI InChI=1S/C19H22NO4S2/c1-20(2)12-9-11(10-13(20)17-16(12)24-17)23-18(21)19(22,14-5-3-7-25-14)15-6-4-8-26-15/h3-8,11-13,16-17,22H,9-10H2,1-2H3/q+1/t11-,12-,13-,16-,17+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0750 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 2 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50343152
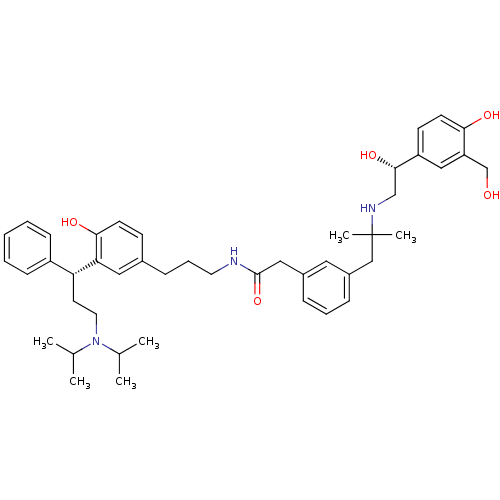
(CHEMBL1773196 | N-(3-(3-((R)-3-(diisopropylamino)-...)Show SMILES CC(C)N(CC[C@H](c1ccccc1)c1cc(CCCNC(=O)Cc2cccc(CC(C)(C)NC[C@H](O)c3ccc(O)c(CO)c3)c2)ccc1O)C(C)C |r| Show InChI InChI=1S/C45H61N3O5/c1-31(2)48(32(3)4)23-21-39(36-15-8-7-9-16-36)40-25-33(17-19-42(40)51)14-11-22-46-44(53)26-34-12-10-13-35(24-34)28-45(5,6)47-29-43(52)37-18-20-41(50)38(27-37)30-49/h7-10,12-13,15-20,24-25,27,31-32,39,43,47,49-52H,11,14,21-23,26,28-30H2,1-6H3,(H,46,53)/t39-,43+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0910 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methyl Scopolamine from human muscarinic M3 receptor expressed in CHO cells by scintillation proximity assay |
Bioorg Med Chem Lett 21: 2759-63 (2011)
Article DOI: 10.1016/j.bmcl.2010.10.132
BindingDB Entry DOI: 10.7270/Q2G73F1B |
More data for this
Ligand-Target Pair | |
Glucagon-like peptide 1 receptor
(Homo sapiens (Human)) | BDBM50241203

(CHEMBL414357 | HGEGTFTSDLSKQMEEEAVRLFIEWLKNGGPSSGS...)Show SMILES [H][C@](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H](N)Cc1cnc[nH]1)([C@@H](C)O)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@]([H])([C@@H](C)O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@]([H])([C@@H](C)CC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)NCC(=O)NCC(=O)N1CCC[C@@]1([H])C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](C)C(=O)N1CCC[C@@]1([H])C(=O)N1CCC[C@@]1([H])C(=O)N1CCC[C@@]1([H])C(=O)N[C@@H](CO)C(N)=O |r| Show InChI InChI=1S/C184H282N50O60S/c1-16-94(10)147(178(289)213-114(52-58-144(257)258)163(274)218-121(73-101-77-195-105-39-24-23-38-103(101)105)168(279)215-116(68-90(2)3)165(276)205-107(41-26-28-61-186)158(269)219-122(75-134(189)243)154(265)198-79-135(244)196-83-139(248)231-63-30-43-129(231)175(286)225-127(87-238)174(285)223-125(85-236)155(266)200-80-136(245)202-96(12)181(292)233-65-32-45-131(233)183(294)234-66-33-46-132(234)182(293)232-64-31-44-130(232)176(287)222-124(84-235)150(190)261)229-170(281)119(71-99-34-19-17-20-35-99)217-166(277)117(69-91(4)5)214-159(270)108(42-29-62-194-184(191)192)212-177(288)146(93(8)9)228-151(262)95(11)203-156(267)111(49-55-141(251)252)208-161(272)112(50-56-142(253)254)209-162(273)113(51-57-143(255)256)210-164(275)115(59-67-295-15)211-160(271)110(47-53-133(188)242)207-157(268)106(40-25-27-60-185)206-172(283)126(86-237)224-167(278)118(70-92(6)7)216-169(280)123(76-145(259)260)220-173(284)128(88-239)226-180(291)149(98(14)241)230-171(282)120(72-100-36-21-18-22-37-100)221-179(290)148(97(13)240)227-138(247)82-199-153(264)109(48-54-140(249)250)204-137(246)81-197-152(263)104(187)74-102-78-193-89-201-102/h17-24,34-39,77-78,89-98,104,106-132,146-149,195,235-241H,16,25-33,40-76,79-88,185-187H2,1-15H3,(H2,188,242)(H2,189,243)(H2,190,261)(H,193,201)(H,196,244)(H,197,263)(H,198,265)(H,199,264)(H,200,266)(H,202,245)(H,203,267)(H,204,246)(H,205,276)(H,206,283)(H,207,268)(H,208,272)(H,209,273)(H,210,275)(H,211,271)(H,212,288)(H,213,289)(H,214,270)(H,215,279)(H,216,280)(H,217,277)(H,218,274)(H,219,269)(H,220,284)(H,221,290)(H,222,287)(H,223,285)(H,224,278)(H,225,286)(H,226,291)(H,227,247)(H,228,262)(H,229,281)(H,230,282)(H,249,250)(H,251,252)(H,253,254)(H,255,256)(H,257,258)(H,259,260)(H4,191,192,194)/t94-,95-,96-,97+,98+,104-,106-,107-,108-,109-,110-,111-,112-,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,146-,147-,148-,149-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0920 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01856
BindingDB Entry DOI: 10.7270/Q2736W0V |
More data for this
Ligand-Target Pair | |
Glucagon-like peptide 1 receptor
(Homo sapiens (Human)) | BDBM50231952
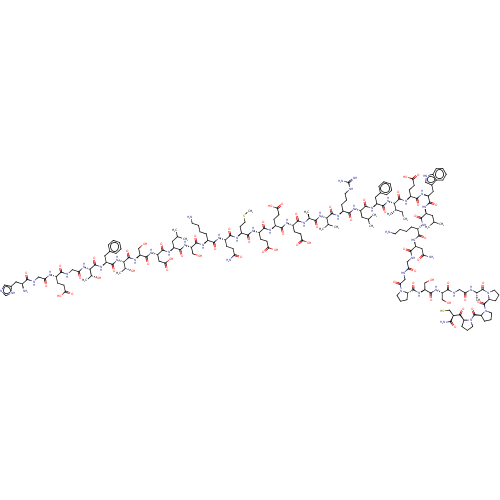
(CHEMBL4081554)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCSC)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H](N)Cc1cnc[nH]1)[C@@H](C)O)[C@@H](C)O)C(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)NCC(=O)NCC(=O)N1CCC[C@H]1C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N1CCC[C@H]1C(=O)N1CCC[C@H]1C(=O)[C@H](CS)C(N)=O |r| Show InChI InChI=1S/C184H281N49O59S2/c1-16-94(10)147(178(287)213-115(52-58-144(255)256)164(273)218-122(73-101-77-195-106-39-24-23-38-103(101)106)169(278)215-117(68-90(2)3)166(275)205-108(41-26-28-61-186)159(268)219-123(75-134(189)241)155(264)198-79-135(242)196-83-139(246)230-63-31-44-130(230)176(285)224-127(86-236)175(284)222-125(84-234)156(265)200-80-136(243)202-96(12)181(290)232-65-32-45-131(232)183(292)233-66-33-46-132(233)182(291)231-64-30-43-129(231)150(259)104(88-293)151(190)260)228-171(280)120(71-99-34-19-17-20-35-99)217-167(276)118(69-91(4)5)214-160(269)109(42-29-62-194-184(191)192)212-177(286)146(93(8)9)227-152(261)95(11)203-157(266)112(49-55-141(249)250)208-162(271)113(50-56-142(251)252)209-163(272)114(51-57-143(253)254)210-165(274)116(59-67-294-15)211-161(270)111(47-53-133(188)240)207-158(267)107(40-25-27-60-185)206-173(282)126(85-235)223-168(277)119(70-92(6)7)216-170(279)124(76-145(257)258)220-174(283)128(87-237)225-180(289)149(98(14)239)229-172(281)121(72-100-36-21-18-22-37-100)221-179(288)148(97(13)238)226-138(245)82-199-154(263)110(48-54-140(247)248)204-137(244)81-197-153(262)105(187)74-102-78-193-89-201-102/h17-24,34-39,77-78,89-98,104-105,107-132,146-149,195,234-239,293H,16,25-33,40-76,79-88,185-187H2,1-15H3,(H2,188,240)(H2,189,241)(H2,190,260)(H,193,201)(H,196,242)(H,197,262)(H,198,264)(H,199,263)(H,200,265)(H,202,243)(H,203,266)(H,204,244)(H,205,275)(H,206,282)(H,207,267)(H,208,271)(H,209,272)(H,210,274)(H,211,270)(H,212,286)(H,213,287)(H,214,269)(H,215,278)(H,216,279)(H,217,276)(H,218,273)(H,219,268)(H,220,283)(H,221,288)(H,222,284)(H,223,277)(H,224,285)(H,225,289)(H,226,245)(H,227,261)(H,228,280)(H,229,281)(H,247,248)(H,249,250)(H,251,252)(H,253,254)(H,255,256)(H,257,258)(H4,191,192,194)/t94-,95-,96-,97+,98+,104-,105-,107-,108-,109-,110-,111-,112-,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,146-,147-,148-,149-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Displacement of [125I]-GLP (7 to 36 residues) from human GLP1R expressed in CHO cell membranes incubated for 30 mins measured after 10 hrs by scintil... |
Eur J Med Chem 127: 703-714 (2017)
Article DOI: 10.1016/j.ejmech.2016.10.044
BindingDB Entry DOI: 10.7270/Q2T155W1 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355612

(CHEMBL1910848)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC(C1)Oc1ccccc1 Show InChI InChI=1S/C28H32N2O2/c1-27(2,30-20-25(21-30)32-24-16-10-5-11-17-24)18-19-28(26(29)31,22-12-6-3-7-13-22)23-14-8-4-9-15-23/h3-17,25H,18-21H2,1-2H3,(H2,29,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 2 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M4
(Homo sapiens (Human)) | BDBM50355622

(CHEMBL1910856)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC(C1)Oc1cccc(O)c1 Show InChI InChI=1S/C28H32N2O3/c1-27(2,30-19-25(20-30)33-24-15-9-14-23(31)18-24)16-17-28(26(29)32,21-10-5-3-6-11-21)22-12-7-4-8-13-22/h3-15,18,25,31H,16-17,19-20H2,1-2H3,(H2,29,32) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.115 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M4 receptor expressed in CHO cells after 24 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(Homo sapiens (Human)) | BDBM50355622

(CHEMBL1910856)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC(C1)Oc1cccc(O)c1 Show InChI InChI=1S/C28H32N2O3/c1-27(2,30-19-25(20-30)33-24-15-9-14-23(31)18-24)16-17-28(26(29)32,21-10-5-3-6-11-21)22-12-7-4-8-13-22/h3-15,18,25,31H,16-17,19-20H2,1-2H3,(H2,29,32) | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.115 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M1 receptor expressed in CHO cells after 24 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355622

(CHEMBL1910856)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC(C1)Oc1cccc(O)c1 Show InChI InChI=1S/C28H32N2O3/c1-27(2,30-19-25(20-30)33-24-15-9-14-23(31)18-24)16-17-28(26(29)32,21-10-5-3-6-11-21)22-12-7-4-8-13-22/h3-15,18,25,31H,16-17,19-20H2,1-2H3,(H2,29,32) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.128 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 24 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50355622

(CHEMBL1910856)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC(C1)Oc1cccc(O)c1 Show InChI InChI=1S/C28H32N2O3/c1-27(2,30-19-25(20-30)33-24-15-9-14-23(31)18-24)16-17-28(26(29)32,21-10-5-3-6-11-21)22-12-7-4-8-13-22/h3-15,18,25,31H,16-17,19-20H2,1-2H3,(H2,29,32) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.137 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M2 receptor expressed in CHO cells after 24 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355618
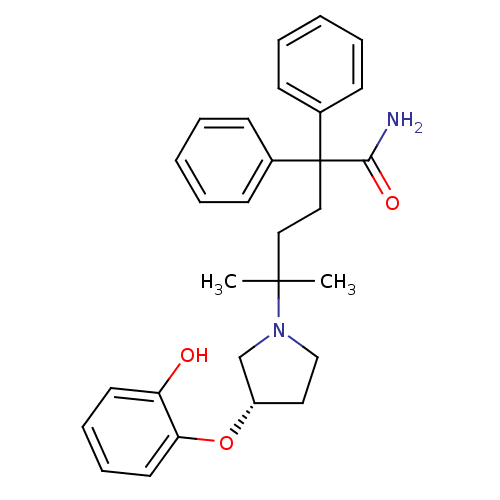
(CHEMBL1910852)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC[C@@H](C1)Oc1ccccc1O |r| Show InChI InChI=1S/C29H34N2O3/c1-28(2,31-20-17-24(21-31)34-26-16-10-9-15-25(26)32)18-19-29(27(30)33,22-11-5-3-6-12-22)23-13-7-4-8-14-23/h3-16,24,32H,17-21H2,1-2H3,(H2,30,33)/t24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355631
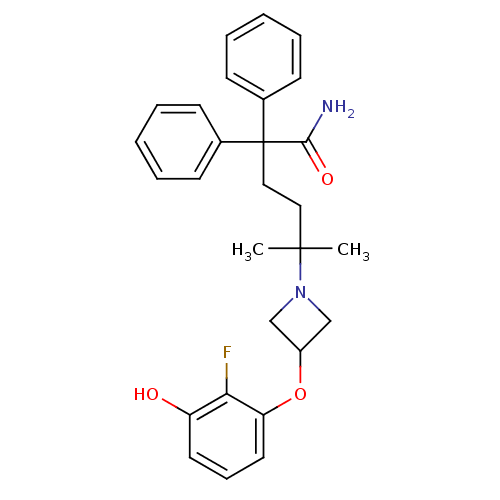
(CHEMBL1910865)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC(C1)Oc1cccc(O)c1F Show InChI InChI=1S/C28H31FN2O3/c1-27(2,31-18-22(19-31)34-24-15-9-14-23(32)25(24)29)16-17-28(26(30)33,20-10-5-3-6-11-20)21-12-7-4-8-13-21/h3-15,22,32H,16-19H2,1-2H3,(H2,30,33) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.163 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 24 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M5
(Homo sapiens (Human)) | BDBM50355622

(CHEMBL1910856)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC(C1)Oc1cccc(O)c1 Show InChI InChI=1S/C28H32N2O3/c1-27(2,30-19-25(20-30)33-24-15-9-14-23(31)18-24)16-17-28(26(29)32,21-10-5-3-6-11-21)22-12-7-4-8-13-22/h3-15,18,25,31H,16-17,19-20H2,1-2H3,(H2,29,32) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.169 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M5 receptor expressed in CHO cells after 24 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355617
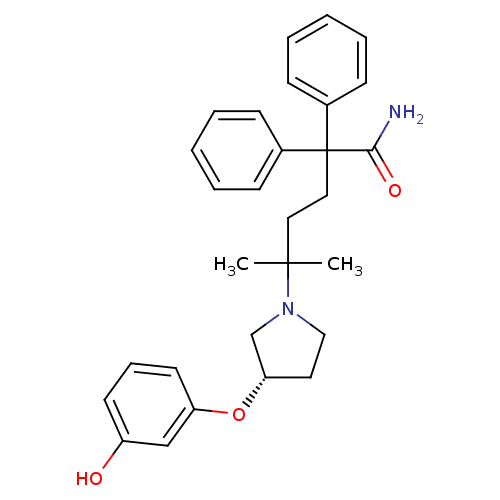
(CHEMBL1910851)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC[C@@H](C1)Oc1cccc(O)c1 |r| Show InChI InChI=1S/C29H34N2O3/c1-28(2,31-19-16-26(21-31)34-25-15-9-14-24(32)20-25)17-18-29(27(30)33,22-10-5-3-6-11-22)23-12-7-4-8-13-23/h3-15,20,26,32H,16-19,21H2,1-2H3,(H2,30,33)/t26-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 24 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355623
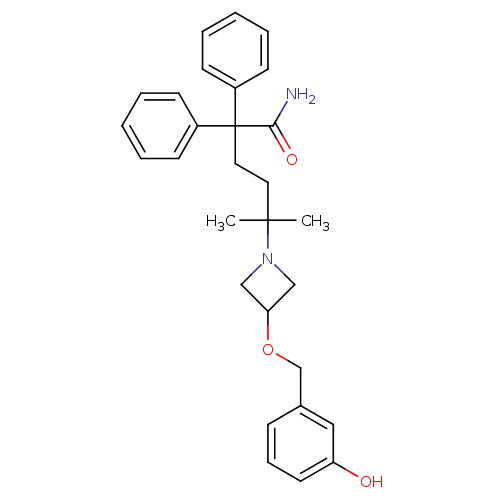
(CHEMBL1910857)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC(C1)OCc1cccc(O)c1 Show InChI InChI=1S/C29H34N2O3/c1-28(2,31-19-26(20-31)34-21-22-10-9-15-25(32)18-22)16-17-29(27(30)33,23-11-5-3-6-12-23)24-13-7-4-8-14-24/h3-15,18,26,32H,16-17,19-21H2,1-2H3,(H2,30,33) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355610

(CHEMBL1237108)Show SMILES CC(C)[N+]1(C)C2CCC1CC(C2)OC(=O)C(CO)c1ccccc1 |TLB:4:3:6.7:11.9.10,1:3:6.7:11.9.10,(10.09,.76,;9.35,2.05,;7.85,2.05,;10.09,3.35,;11.39,2.6,;10.09,4.89,;10.86,3.55,;10.3,2.58,;8.76,2.58,;7.43,3.35,;7.43,4.89,;8.76,5.66,;6.09,5.66,;6.09,7.2,;7.43,7.97,;4.76,7.97,;4.76,9.51,;3.43,10.28,;3.43,7.2,;3.43,5.66,;2.09,4.89,;.76,5.66,;.76,7.2,;2.09,7.97,)| Show InChI InChI=1S/C20H30NO3/c1-14(2)21(3)16-9-10-17(21)12-18(11-16)24-20(23)19(13-22)15-7-5-4-6-8-15/h4-8,14,16-19,22H,9-13H2,1-3H3/q+1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355622

(CHEMBL1910856)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC(C1)Oc1cccc(O)c1 Show InChI InChI=1S/C28H32N2O3/c1-27(2,30-19-25(20-30)33-24-15-9-14-23(31)18-24)16-17-28(26(29)32,21-10-5-3-6-11-21)22-12-7-4-8-13-22/h3-15,18,25,31H,16-17,19-20H2,1-2H3,(H2,29,32) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355616
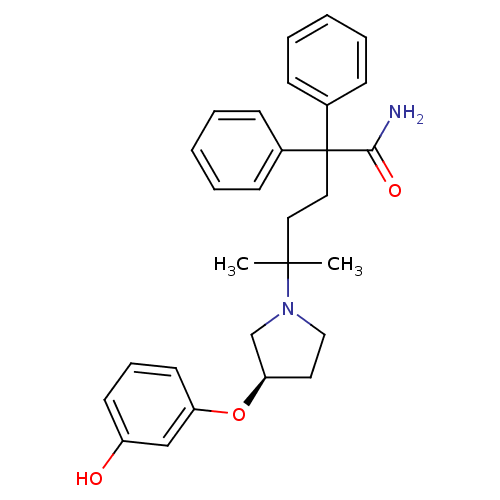
(CHEMBL1910850)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC[C@H](C1)Oc1cccc(O)c1 |r| Show InChI InChI=1S/C29H34N2O3/c1-28(2,31-19-16-26(21-31)34-25-15-9-14-24(32)20-25)17-18-29(27(30)33,22-10-5-3-6-11-22)23-12-7-4-8-13-23/h3-15,20,26,32H,16-19,21H2,1-2H3,(H2,30,33)/t26-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.201 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 24 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355617

(CHEMBL1910851)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC[C@@H](C1)Oc1cccc(O)c1 |r| Show InChI InChI=1S/C29H34N2O3/c1-28(2,31-19-16-26(21-31)34-25-15-9-14-24(32)20-25)17-18-29(27(30)33,22-10-5-3-6-11-22)23-12-7-4-8-13-23/h3-15,20,26,32H,16-19,21H2,1-2H3,(H2,30,33)/t26-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355610

(CHEMBL1237108)Show SMILES CC(C)[N+]1(C)C2CCC1CC(C2)OC(=O)C(CO)c1ccccc1 |TLB:4:3:6.7:11.9.10,1:3:6.7:11.9.10,(10.09,.76,;9.35,2.05,;7.85,2.05,;10.09,3.35,;11.39,2.6,;10.09,4.89,;10.86,3.55,;10.3,2.58,;8.76,2.58,;7.43,3.35,;7.43,4.89,;8.76,5.66,;6.09,5.66,;6.09,7.2,;7.43,7.97,;4.76,7.97,;4.76,9.51,;3.43,10.28,;3.43,7.2,;3.43,5.66,;2.09,4.89,;.76,5.66,;.76,7.2,;2.09,7.97,)| Show InChI InChI=1S/C20H30NO3/c1-14(2)21(3)16-9-10-17(21)12-18(11-16)24-20(23)19(13-22)15-7-5-4-6-8-15/h4-8,14,16-19,22H,9-13H2,1-3H3/q+1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.213 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 24 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355628
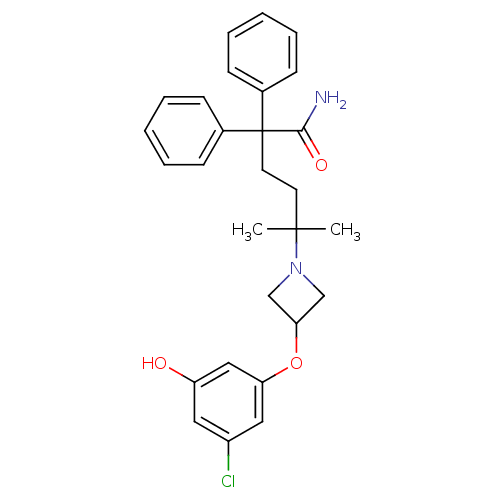
(CHEMBL1910862)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC(C1)Oc1cc(O)cc(Cl)c1 Show InChI InChI=1S/C28H31ClN2O3/c1-27(2,31-18-25(19-31)34-24-16-22(29)15-23(32)17-24)13-14-28(26(30)33,20-9-5-3-6-10-20)21-11-7-4-8-12-21/h3-12,15-17,25,32H,13-14,18-19H2,1-2H3,(H2,30,33) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Glucagon-like peptide 1 receptor
(Homo sapiens (Human)) | BDBM50231900
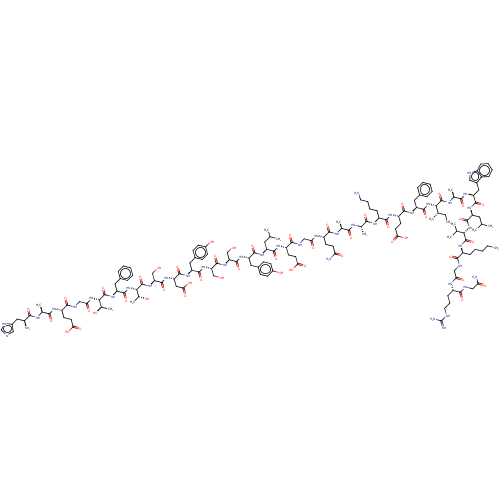
(CHEMBL4060480)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(N)=O)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)Cc1cnc[nH]1)[C@@H](C)O)[C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(N)=O |r| Show InChI InChI=1S/C155H229N41O47/c1-15-79(8)125(152(241)174-83(12)131(220)182-110(63-90-66-165-96-34-23-22-33-94(90)96)143(232)184-105(58-77(4)5)145(234)194-124(78(6)7)151(240)181-98(35-24-26-54-156)134(223)167-69-117(206)175-97(37-28-56-164-155(161)162)133(222)166-68-116(160)205)195-146(235)108(59-86-29-18-16-19-30-86)185-139(228)103(49-53-122(213)214)180-138(227)99(36-25-27-55-157)177-129(218)81(10)171-128(217)80(9)173-137(226)102(46-50-115(159)204)176-118(207)70-168-136(225)101(48-52-121(211)212)179-140(229)104(57-76(2)3)183-141(230)106(61-88-38-42-92(202)43-39-88)187-148(237)112(72-197)191-150(239)113(73-198)190-142(231)107(62-89-40-44-93(203)45-41-89)186-144(233)111(65-123(215)216)188-149(238)114(74-199)192-154(243)127(85(14)201)196-147(236)109(60-87-31-20-17-21-32-87)189-153(242)126(84(13)200)193-119(208)71-169-135(224)100(47-51-120(209)210)178-130(219)82(11)172-132(221)95(158)64-91-67-163-75-170-91/h16-23,29-34,38-45,66-67,75-85,95,97-114,124-127,165,197-203H,15,24-28,35-37,46-65,68-74,156-158H2,1-14H3,(H2,159,204)(H2,160,205)(H,163,170)(H,166,222)(H,167,223)(H,168,225)(H,169,224)(H,171,217)(H,172,221)(H,173,226)(H,174,241)(H,175,206)(H,176,207)(H,177,218)(H,178,219)(H,179,229)(H,180,227)(H,181,240)(H,182,220)(H,183,230)(H,184,232)(H,185,228)(H,186,233)(H,187,237)(H,188,238)(H,189,242)(H,190,231)(H,191,239)(H,192,243)(H,193,208)(H,194,234)(H,195,235)(H,196,236)(H,209,210)(H,211,212)(H,213,214)(H,215,216)(H4,161,162,164)/t79-,80-,81-,82-,83-,84+,85+,95-,97-,98-,99-,100-,101-,102-,103-,104-,105-,106-,107-,108-,109-,110-,111-,112-,113-,114-,124-,125-,126-,127-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Agonist activity at human GLP1R expressed in CHO cells assessed as cAMP accumulation incubated for 30 mins by LANCE assay |
Eur J Med Chem 127: 703-714 (2017)
Article DOI: 10.1016/j.ejmech.2016.10.044
BindingDB Entry DOI: 10.7270/Q2T155W1 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355614
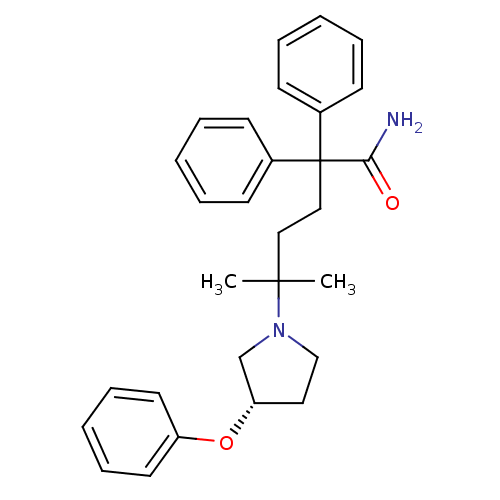
(CHEMBL1910846)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC[C@@H](C1)Oc1ccccc1 |r| Show InChI InChI=1S/C29H34N2O2/c1-28(2,31-21-18-26(22-31)33-25-16-10-5-11-17-25)19-20-29(27(30)32,23-12-6-3-7-13-23)24-14-8-4-9-15-24/h3-17,26H,18-22H2,1-2H3,(H2,30,32)/t26-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355631

(CHEMBL1910865)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC(C1)Oc1cccc(O)c1F Show InChI InChI=1S/C28H31FN2O3/c1-27(2,31-18-22(19-31)34-24-15-9-14-23(32)25(24)29)16-17-28(26(30)33,20-10-5-3-6-11-20)21-12-7-4-8-13-21/h3-15,22,32H,16-19H2,1-2H3,(H2,30,33) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355611
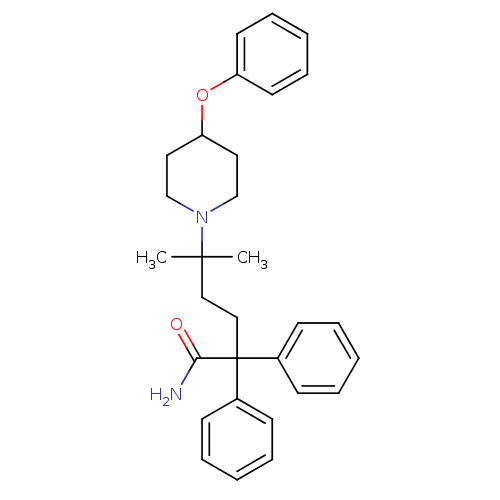
(CHEMBL1910849)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CCC(CC1)Oc1ccccc1 Show InChI InChI=1S/C30H36N2O2/c1-29(2,32-22-18-27(19-23-32)34-26-16-10-5-11-17-26)20-21-30(28(31)33,24-12-6-3-7-13-24)25-14-8-4-9-15-25/h3-17,27H,18-23H2,1-2H3,(H2,31,33) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50343153
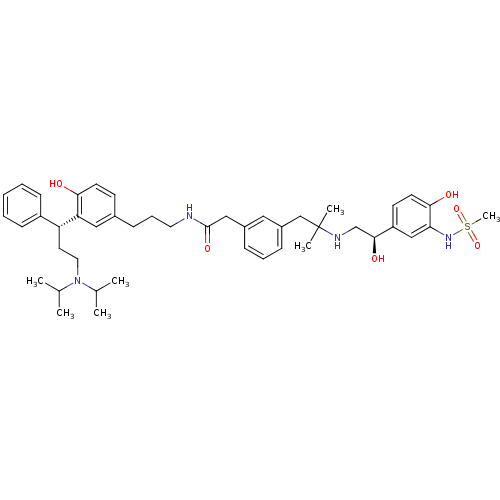
(CHEMBL1773197 | N-(3-(3-((R)-3-(diisopropylamino)-...)Show SMILES CC(C)N(CC[C@H](c1ccccc1)c1cc(CCCNC(=O)Cc2cccc(CC(C)(C)NC[C@H](O)c3ccc(O)c(NS(C)(=O)=O)c3)c2)ccc1O)C(C)C |r| Show InChI InChI=1S/C45H62N4O6S/c1-31(2)49(32(3)4)24-22-38(36-16-9-8-10-17-36)39-26-33(18-20-41(39)50)15-12-23-46-44(53)27-34-13-11-14-35(25-34)29-45(5,6)47-30-43(52)37-19-21-42(51)40(28-37)48-56(7,54)55/h8-11,13-14,16-21,25-26,28,31-32,38,43,47-48,50-52H,12,15,22-24,27,29-30H2,1-7H3,(H,46,53)/t38-,43+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.276 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methyl Scopolamine from human muscarinic M3 receptor expressed in CHO cells by scintillation proximity assay |
Bioorg Med Chem Lett 21: 2759-63 (2011)
Article DOI: 10.1016/j.bmcl.2010.10.132
BindingDB Entry DOI: 10.7270/Q2G73F1B |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355610

(CHEMBL1237108)Show SMILES CC(C)[N+]1(C)C2CCC1CC(C2)OC(=O)C(CO)c1ccccc1 |TLB:4:3:6.7:11.9.10,1:3:6.7:11.9.10,(10.09,.76,;9.35,2.05,;7.85,2.05,;10.09,3.35,;11.39,2.6,;10.09,4.89,;10.86,3.55,;10.3,2.58,;8.76,2.58,;7.43,3.35,;7.43,4.89,;8.76,5.66,;6.09,5.66,;6.09,7.2,;7.43,7.97,;4.76,7.97,;4.76,9.51,;3.43,10.28,;3.43,7.2,;3.43,5.66,;2.09,4.89,;.76,5.66,;.76,7.2,;2.09,7.97,)| Show InChI InChI=1S/C20H30NO3/c1-14(2)21(3)16-9-10-17(21)12-18(11-16)24-20(23)19(13-22)15-7-5-4-6-8-15/h4-8,14,16-19,22H,9-13H2,1-3H3/q+1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M3 receptor expressed in CHO cells after 2 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50343161
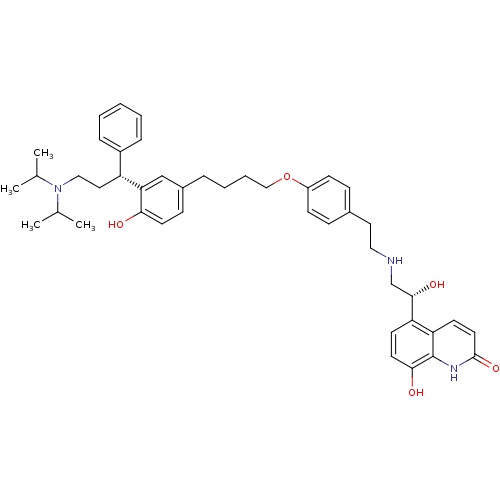
(5-((R)-2-(4-(4-(3-((R)-3-(diisopropylamino)-1-phen...)Show SMILES CC(C)N(CC[C@H](c1ccccc1)c1cc(CCCCOc2ccc(CCNC[C@H](O)c3ccc(O)c4[nH]c(=O)ccc34)cc2)ccc1O)C(C)C |r| Show InChI InChI=1S/C44H55N3O5/c1-30(2)47(31(3)4)26-24-36(34-11-6-5-7-12-34)39-28-33(15-20-40(39)48)10-8-9-27-52-35-16-13-32(14-17-35)23-25-45-29-42(50)37-18-21-41(49)44-38(37)19-22-43(51)46-44/h5-7,11-22,28,30-31,36,42,45,48-50H,8-10,23-27,29H2,1-4H3,(H,46,51)/t36-,42+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 0.305 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methyl Scopolamine from human muscarinic M3 receptor expressed in CHO cells by scintillation proximity assay |
Bioorg Med Chem Lett 21: 2759-63 (2011)
Article DOI: 10.1016/j.bmcl.2010.10.132
BindingDB Entry DOI: 10.7270/Q2G73F1B |
More data for this
Ligand-Target Pair | |
Glucagon-like peptide 1 receptor
(Homo sapiens (Human)) | BDBM50261506
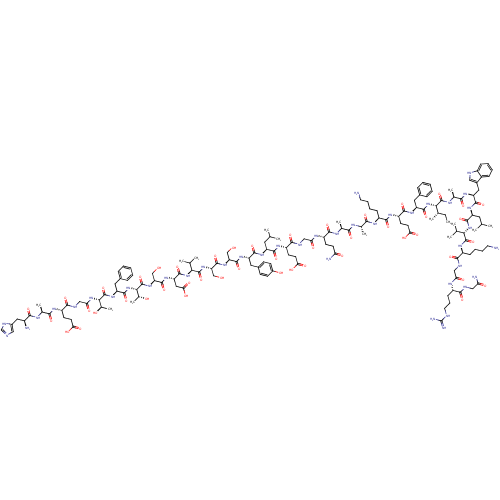
(CHEMBL499930 | HAEGTFTSDVSSYLEGQAAKEFIAWLVKGRG-NH2)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(N)=O)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)Cc1cnc[nH]1)[C@@H](C)O)[C@@H](C)O)C(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(N)=O |r| Show InChI InChI=1S/C151H229N41O46/c1-17-77(10)121(148(236)170-81(14)127(215)178-105(60-87-63-161-92-36-25-24-35-90(87)92)138(226)180-101(56-74(4)5)139(227)189-119(75(6)7)146(234)177-94(37-26-28-52-152)130(218)163-66-112(201)171-93(39-30-54-160-151(157)158)129(217)162-65-111(156)200)191-140(228)103(57-84-31-20-18-21-32-84)181-135(223)99(47-51-117(208)209)176-134(222)95(38-27-29-53-153)173-125(213)79(12)167-124(212)78(11)169-133(221)98(44-48-110(155)199)172-113(202)67-164-132(220)97(46-50-116(206)207)175-136(224)100(55-73(2)3)179-137(225)102(59-86-40-42-89(198)43-41-86)182-143(231)107(69-193)185-145(233)109(71-195)186-147(235)120(76(8)9)190-142(230)106(62-118(210)211)183-144(232)108(70-194)187-150(238)123(83(16)197)192-141(229)104(58-85-33-22-19-23-34-85)184-149(237)122(82(15)196)188-114(203)68-165-131(219)96(45-49-115(204)205)174-126(214)80(13)168-128(216)91(154)61-88-64-159-72-166-88/h18-25,31-36,40-43,63-64,72-83,91,93-109,119-123,161,193-198H,17,26-30,37-39,44-62,65-71,152-154H2,1-16H3,(H2,155,199)(H2,156,200)(H,159,166)(H,162,217)(H,163,218)(H,164,220)(H,165,219)(H,167,212)(H,168,216)(H,169,221)(H,170,236)(H,171,201)(H,172,202)(H,173,213)(H,174,214)(H,175,224)(H,176,222)(H,177,234)(H,178,215)(H,179,225)(H,180,226)(H,181,223)(H,182,231)(H,183,232)(H,184,237)(H,185,233)(H,186,235)(H,187,238)(H,188,203)(H,189,227)(H,190,230)(H,191,228)(H,192,229)(H,204,205)(H,206,207)(H,208,209)(H,210,211)(H4,157,158,160)/t77-,78-,79-,80-,81-,82+,83+,91-,93-,94-,95-,96-,97-,98-,99-,100-,101-,102-,103-,104-,105-,106-,107-,108-,109-,119-,120-,121-,122-,123-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Agonist activity at human GLP1R expressed in CHO cells assessed as cAMP accumulation incubated for 30 mins by LANCE assay |
Eur J Med Chem 127: 703-714 (2017)
Article DOI: 10.1016/j.ejmech.2016.10.044
BindingDB Entry DOI: 10.7270/Q2T155W1 |
More data for this
Ligand-Target Pair | |
Glucagon-like peptide 1 receptor
(Homo sapiens (Human)) | BDBM50231888
(CHEMBL4081357)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)Cc2cnc[nH]2)[C@@H](C)O)[C@@H](C)O)C(C)C)C(=O)N[C@@H](Cc2ccc(O)cc2)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N1)C(=O)N[C@@H](C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(N)=O |r| Show InChI InChI=1S/C156H236N42O46/c1-17-80(10)125(153(242)175-84(14)131(220)184-109(64-90-68-167-95-38-25-24-37-93(90)95)143(232)186-105(60-77(4)5)145(234)195-123(78(6)7)151(240)183-97(39-26-29-55-157)134(223)169-71-117(207)176-96(42-32-58-166-156(162)163)133(222)168-70-115(161)205)197-146(235)107(61-87-33-20-18-21-34-87)188-139(228)102(49-53-120(211)212)180-137(226)98(40-27-30-56-158)177-129(218)82(12)172-128(217)81(11)174-136(225)101(47-51-114(160)204)181-144(233)110-66-116(206)165-57-31-28-41-99(138(227)187-106(63-89-43-45-92(203)46-44-89)142(231)185-104(59-76(2)3)141(230)182-103(140(229)189-110)50-54-121(213)214)179-149(238)112(73-199)192-152(241)124(79(8)9)196-148(237)111(67-122(215)216)190-150(239)113(74-200)193-155(244)127(86(16)202)198-147(236)108(62-88-35-22-19-23-36-88)191-154(243)126(85(15)201)194-118(208)72-170-135(224)100(48-52-119(209)210)178-130(219)83(13)173-132(221)94(159)65-91-69-164-75-171-91/h18-25,33-38,43-46,68-69,75-86,94,96-113,123-127,167,199-203H,17,26-32,39-42,47-67,70-74,157-159H2,1-16H3,(H2,160,204)(H2,161,205)(H,164,171)(H,165,206)(H,168,222)(H,169,223)(H,170,224)(H,172,217)(H,173,221)(H,174,225)(H,175,242)(H,176,207)(H,177,218)(H,178,219)(H,179,238)(H,180,226)(H,181,233)(H,182,230)(H,183,240)(H,184,220)(H,185,231)(H,186,232)(H,187,227)(H,188,228)(H,189,229)(H,190,239)(H,191,243)(H,192,241)(H,193,244)(H,194,208)(H,195,234)(H,196,237)(H,197,235)(H,198,236)(H,209,210)(H,211,212)(H,213,214)(H,215,216)(H4,162,163,166)/t80-,81-,82-,83-,84-,85+,86+,94-,96-,97-,98-,99-,100-,101-,102-,103-,104-,105-,106-,107-,108-,109-,110-,111-,112-,113-,123-,124-,125-,126-,127-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Displacement of [125I]-GLP (7 to 36 residues) from human GLP1R expressed in CHO cell membranes incubated for 30 mins measured after 10 hrs by scintil... |
Eur J Med Chem 127: 703-714 (2017)
Article DOI: 10.1016/j.ejmech.2016.10.044
BindingDB Entry DOI: 10.7270/Q2T155W1 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M4
(Homo sapiens (Human)) | BDBM50355610

(CHEMBL1237108)Show SMILES CC(C)[N+]1(C)C2CCC1CC(C2)OC(=O)C(CO)c1ccccc1 |TLB:4:3:6.7:11.9.10,1:3:6.7:11.9.10,(10.09,.76,;9.35,2.05,;7.85,2.05,;10.09,3.35,;11.39,2.6,;10.09,4.89,;10.86,3.55,;10.3,2.58,;8.76,2.58,;7.43,3.35,;7.43,4.89,;8.76,5.66,;6.09,5.66,;6.09,7.2,;7.43,7.97,;4.76,7.97,;4.76,9.51,;3.43,10.28,;3.43,7.2,;3.43,5.66,;2.09,4.89,;.76,5.66,;.76,7.2,;2.09,7.97,)| Show InChI InChI=1S/C20H30NO3/c1-14(2)21(3)16-9-10-17(21)12-18(11-16)24-20(23)19(13-22)15-7-5-4-6-8-15/h4-8,14,16-19,22H,9-13H2,1-3H3/q+1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.382 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M4 receptor expressed in CHO cells after 24 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50343154
(2-(3-((R)-2-((R)-2-(3,5-dihydroxyphenyl)-2-hydroxy...)Show SMILES CC(C)N(CC[C@H](c1ccccc1)c1cc(CCCNC(=O)Cc2cccc(C[C@@H](C)NC[C@H](O)c3cc(O)cc(O)c3)c2)ccc1O)C(C)C |r| Show InChI InChI=1S/C43H57N3O5/c1-29(2)46(30(3)4)20-18-39(35-14-7-6-8-15-35)40-23-32(16-17-41(40)49)13-10-19-44-43(51)24-34-12-9-11-33(22-34)21-31(5)45-28-42(50)36-25-37(47)27-38(48)26-36/h6-9,11-12,14-17,22-23,25-27,29-31,39,42,45,47-50H,10,13,18-21,24,28H2,1-5H3,(H,44,51)/t31-,39-,42+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.397 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methyl Scopolamine from human muscarinic M3 receptor expressed in CHO cells by scintillation proximity assay |
Bioorg Med Chem Lett 21: 2759-63 (2011)
Article DOI: 10.1016/j.bmcl.2010.10.132
BindingDB Entry DOI: 10.7270/Q2G73F1B |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50355610

(CHEMBL1237108)Show SMILES CC(C)[N+]1(C)C2CCC1CC(C2)OC(=O)C(CO)c1ccccc1 |TLB:4:3:6.7:11.9.10,1:3:6.7:11.9.10,(10.09,.76,;9.35,2.05,;7.85,2.05,;10.09,3.35,;11.39,2.6,;10.09,4.89,;10.86,3.55,;10.3,2.58,;8.76,2.58,;7.43,3.35,;7.43,4.89,;8.76,5.66,;6.09,5.66,;6.09,7.2,;7.43,7.97,;4.76,7.97,;4.76,9.51,;3.43,10.28,;3.43,7.2,;3.43,5.66,;2.09,4.89,;.76,5.66,;.76,7.2,;2.09,7.97,)| Show InChI InChI=1S/C20H30NO3/c1-14(2)21(3)16-9-10-17(21)12-18(11-16)24-20(23)19(13-22)15-7-5-4-6-8-15/h4-8,14,16-19,22H,9-13H2,1-3H3/q+1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.413 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M2 receptor expressed in CHO cells after 24 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(Homo sapiens (Human)) | BDBM50355610

(CHEMBL1237108)Show SMILES CC(C)[N+]1(C)C2CCC1CC(C2)OC(=O)C(CO)c1ccccc1 |TLB:4:3:6.7:11.9.10,1:3:6.7:11.9.10,(10.09,.76,;9.35,2.05,;7.85,2.05,;10.09,3.35,;11.39,2.6,;10.09,4.89,;10.86,3.55,;10.3,2.58,;8.76,2.58,;7.43,3.35,;7.43,4.89,;8.76,5.66,;6.09,5.66,;6.09,7.2,;7.43,7.97,;4.76,7.97,;4.76,9.51,;3.43,10.28,;3.43,7.2,;3.43,5.66,;2.09,4.89,;.76,5.66,;.76,7.2,;2.09,7.97,)| Show InChI InChI=1S/C20H30NO3/c1-14(2)21(3)16-9-10-17(21)12-18(11-16)24-20(23)19(13-22)15-7-5-4-6-8-15/h4-8,14,16-19,22H,9-13H2,1-3H3/q+1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.433 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M1 receptor expressed in CHO cells after 24 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Glucagon-like peptide 1 receptor
(Homo sapiens (Human)) | BDBM50231942

(CHEMBL4065403)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](C)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)Cc1cnc[nH]1)[C@@H](C)O)[C@@H](C)O)C(=O)N[C@H]1CCCCNC(=O)C[C@H](NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC1=O)C(C)C)C(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(N)=O |r| Show InChI InChI=1S/C157H229N41O48/c1-15-79(8)126(154(244)182-99-36-25-27-55-165-118(208)65-111(136(226)170-70-119(209)176-97(37-28-56-166-157(162)163)134(224)168-69-117(161)207)191-153(243)125(78(6)7)196-147(237)105(58-77(4)5)184-145(235)110(188-140(99)230)63-90-67-167-96-34-23-22-33-94(90)96)197-148(238)108(59-86-29-18-16-19-30-86)185-141(231)103(49-53-123(215)216)180-139(229)98(35-24-26-54-158)177-130(220)81(10)172-129(219)80(9)174-137(227)101(46-50-116(160)206)179-132(222)83(12)175-138(228)102(48-52-122(213)214)181-142(232)104(57-76(2)3)183-143(233)106(61-88-38-42-92(204)43-39-88)187-150(240)113(72-199)193-152(242)114(73-200)192-144(234)107(62-89-40-44-93(205)45-41-89)186-146(236)112(66-124(217)218)189-151(241)115(74-201)194-156(246)128(85(14)203)198-149(239)109(60-87-31-20-17-21-32-87)190-155(245)127(84(13)202)195-120(210)71-169-135(225)100(47-51-121(211)212)178-131(221)82(11)173-133(223)95(159)64-91-68-164-75-171-91/h16-23,29-34,38-45,67-68,75-85,95,97-115,125-128,167,199-205H,15,24-28,35-37,46-66,69-74,158-159H2,1-14H3,(H2,160,206)(H2,161,207)(H,164,171)(H,165,208)(H,168,224)(H,169,225)(H,170,226)(H,172,219)(H,173,223)(H,174,227)(H,175,228)(H,176,209)(H,177,220)(H,178,221)(H,179,222)(H,180,229)(H,181,232)(H,182,244)(H,183,233)(H,184,235)(H,185,231)(H,186,236)(H,187,240)(H,188,230)(H,189,241)(H,190,245)(H,191,243)(H,192,234)(H,193,242)(H,194,246)(H,195,210)(H,196,237)(H,197,238)(H,198,239)(H,211,212)(H,213,214)(H,215,216)(H,217,218)(H4,162,163,166)/t79-,80-,81-,82-,83-,84+,85+,95-,97-,98-,99-,100-,101-,102-,103-,104-,105-,106-,107-,108-,109-,110-,111-,112-,113-,114-,115-,125-,126-,127-,128-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Displacement of [125I]-GLP (7 to 36 residues) from human GLP1R expressed in CHO cell membranes incubated for 30 mins measured after 10 hrs by scintil... |
Eur J Med Chem 127: 703-714 (2017)
Article DOI: 10.1016/j.ejmech.2016.10.044
BindingDB Entry DOI: 10.7270/Q2T155W1 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50355610

(CHEMBL1237108)Show SMILES CC(C)[N+]1(C)C2CCC1CC(C2)OC(=O)C(CO)c1ccccc1 |TLB:4:3:6.7:11.9.10,1:3:6.7:11.9.10,(10.09,.76,;9.35,2.05,;7.85,2.05,;10.09,3.35,;11.39,2.6,;10.09,4.89,;10.86,3.55,;10.3,2.58,;8.76,2.58,;7.43,3.35,;7.43,4.89,;8.76,5.66,;6.09,5.66,;6.09,7.2,;7.43,7.97,;4.76,7.97,;4.76,9.51,;3.43,10.28,;3.43,7.2,;3.43,5.66,;2.09,4.89,;.76,5.66,;.76,7.2,;2.09,7.97,)| Show InChI InChI=1S/C20H30NO3/c1-14(2)21(3)16-9-10-17(21)12-18(11-16)24-20(23)19(13-22)15-7-5-4-6-8-15/h4-8,14,16-19,22H,9-13H2,1-3H3/q+1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from human recombinant M2 receptor expressed in CHO cells after 2 hrs by filter binding assay |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Glucagon-like peptide 1 receptor
(Homo sapiens (Human)) | BDBM50231943
(CHEMBL4093072)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCSC)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H](N)Cc1cnc[nH]1)[C@@H](C)O)[C@@H](C)O)C(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)NCC(=O)NCC(N)=O |r| Show InChI InChI=1S/C150H231N41O48S/c1-15-77(10)121(147(237)177-96(43-49-118(210)211)135(225)182-103(60-83-64-161-87-34-23-22-33-85(83)87)140(230)179-98(55-73(2)3)137(227)169-89(36-25-27-52-152)130(220)183-104(62-109(155)197)127(217)164-67-111(199)162-66-110(156)198)190-142(232)101(58-81-29-18-16-19-30-81)181-138(228)99(56-74(4)5)178-131(221)90(37-28-53-160-150(157)158)176-146(236)120(76(8)9)189-124(214)78(11)167-128(218)93(40-46-115(204)205)172-133(223)94(41-47-116(206)207)173-134(224)95(42-48-117(208)209)174-136(226)97(50-54-240-14)175-132(222)92(38-44-108(154)196)171-129(219)88(35-24-26-51-151)170-144(234)106(70-192)186-139(229)100(57-75(6)7)180-141(231)105(63-119(212)213)184-145(235)107(71-193)187-149(239)123(80(13)195)191-143(233)102(59-82-31-20-17-21-32-82)185-148(238)122(79(12)194)188-113(201)69-165-126(216)91(39-45-114(202)203)168-112(200)68-163-125(215)86(153)61-84-65-159-72-166-84/h16-23,29-34,64-65,72-80,86,88-107,120-123,161,192-195H,15,24-28,35-63,66-71,151-153H2,1-14H3,(H2,154,196)(H2,155,197)(H2,156,198)(H,159,166)(H,162,199)(H,163,215)(H,164,217)(H,165,216)(H,167,218)(H,168,200)(H,169,227)(H,170,234)(H,171,219)(H,172,223)(H,173,224)(H,174,226)(H,175,222)(H,176,236)(H,177,237)(H,178,221)(H,179,230)(H,180,231)(H,181,228)(H,182,225)(H,183,220)(H,184,235)(H,185,238)(H,186,229)(H,187,239)(H,188,201)(H,189,214)(H,190,232)(H,191,233)(H,202,203)(H,204,205)(H,206,207)(H,208,209)(H,210,211)(H,212,213)(H4,157,158,160)/t77-,78-,79+,80+,86-,88-,89-,90-,91-,92-,93-,94-,95-,96-,97-,98-,99-,100-,101-,102-,103-,104-,105-,106-,107-,120-,121-,122-,123-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Displacement of [125I]-GLP (7 to 36 residues) from human GLP1R expressed in CHO cell membranes incubated for 30 mins measured after 10 hrs by scintil... |
Eur J Med Chem 127: 703-714 (2017)
Article DOI: 10.1016/j.ejmech.2016.10.044
BindingDB Entry DOI: 10.7270/Q2T155W1 |
More data for this
Ligand-Target Pair | |
Glucagon-like peptide 1 receptor
(Homo sapiens (Human)) | BDBM50231949
(CHEMBL4069162)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(N)=O)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)Cc1cnc[nH]1)[C@@H](C)O)[C@@H](C)O)C(=O)N[C@H]1CCCCNC(=O)C[C@H](NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC1=O)C(C)C)C(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(N)=O |r| Show InChI InChI=1S/C156H227N41O48/c1-14-79(8)126(153(243)181-98-35-24-26-54-164-117(207)64-110(136(226)170-70-118(208)175-96(36-27-55-165-156(161)162)133(223)167-68-116(160)206)190-152(242)125(78(6)7)195-146(236)104(57-77(4)5)183-144(234)109(187-139(98)229)62-89-66-166-95-33-22-21-32-93(89)95)196-147(237)107(58-85-28-17-15-18-29-85)184-140(230)102(48-52-123(215)216)180-138(228)97(34-23-25-53-157)177-130(220)81(10)172-129(219)80(9)174-137(227)101(45-49-115(159)205)176-119(209)69-168-135(225)100(47-51-122(213)214)179-141(231)103(56-76(2)3)182-142(232)105(60-87-37-41-91(203)42-38-87)186-149(239)112(72-198)192-151(241)113(73-199)191-143(233)106(61-88-39-43-92(204)44-40-88)185-145(235)111(65-124(217)218)188-150(240)114(74-200)193-155(245)128(84(13)202)197-148(238)108(59-86-30-19-16-20-31-86)189-154(244)127(83(12)201)194-120(210)71-169-134(224)99(46-50-121(211)212)178-131(221)82(11)173-132(222)94(158)63-90-67-163-75-171-90/h15-22,28-33,37-44,66-67,75-84,94,96-114,125-128,166,198-204H,14,23-27,34-36,45-65,68-74,157-158H2,1-13H3,(H2,159,205)(H2,160,206)(H,163,171)(H,164,207)(H,167,223)(H,168,225)(H,169,224)(H,170,226)(H,172,219)(H,173,222)(H,174,227)(H,175,208)(H,176,209)(H,177,220)(H,178,221)(H,179,231)(H,180,228)(H,181,243)(H,182,232)(H,183,234)(H,184,230)(H,185,235)(H,186,239)(H,187,229)(H,188,240)(H,189,244)(H,190,242)(H,191,233)(H,192,241)(H,193,245)(H,194,210)(H,195,236)(H,196,237)(H,197,238)(H,211,212)(H,213,214)(H,215,216)(H,217,218)(H4,161,162,165)/t79-,80-,81-,82-,83+,84+,94-,96-,97-,98-,99-,100-,101-,102-,103-,104-,105-,106-,107-,108-,109-,110-,111-,112-,113-,114-,125-,126-,127-,128-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Displacement of [125I]-GLP (7 to 36 residues) from human GLP1R expressed in CHO cell membranes incubated for 30 mins measured after 10 hrs by scintil... |
Eur J Med Chem 127: 703-714 (2017)
Article DOI: 10.1016/j.ejmech.2016.10.044
BindingDB Entry DOI: 10.7270/Q2T155W1 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355616

(CHEMBL1910850)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC[C@H](C1)Oc1cccc(O)c1 |r| Show InChI InChI=1S/C29H34N2O3/c1-28(2,31-19-16-26(21-31)34-25-15-9-14-24(32)20-25)17-18-29(27(30)33,22-10-5-3-6-11-22)23-12-7-4-8-13-23/h3-15,20,26,32H,16-19,21H2,1-2H3,(H2,30,33)/t26-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |
Glucagon-like peptide 1 receptor
(Homo sapiens (Human)) | BDBM50231887
(CHEMBL4091638)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(N)=O)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H]1CC(=O)NCCCC[C@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)Cc2cnc[nH]2)[C@@H](C)O)[C@@H](C)O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H](Cc2ccc(O)cc2)C(=O)N1)C(=O)N[C@@H](C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(N)=O |r| Show InChI InChI=1S/C150H224N42O47/c1-13-75(6)120(147(237)170-79(10)126(216)179-103(59-85-63-161-90-34-21-20-33-88(85)90)137(227)180-99(55-73(2)3)140(230)190-119(74(4)5)146(236)178-92(35-22-25-51-151)129(219)163-66-112(202)171-91(38-28-54-160-150(156)157)128(218)162-65-110(155)200)191-141(231)101(56-82-29-16-14-17-30-82)181-135(225)98(46-50-117(209)210)177-133(223)93(36-23-26-52-152)173-124(214)77(8)167-123(213)76(7)169-132(222)97(43-47-109(154)199)172-113(203)67-164-131(221)96(45-49-116(207)208)176-138(228)104-61-111(201)159-53-27-24-37-94(134(224)186-107(70-194)145(235)187-106(69-193)143(233)182-100(136(226)183-104)58-84-39-41-87(198)42-40-84)175-139(229)105(62-118(211)212)184-144(234)108(71-195)188-149(239)122(81(12)197)192-142(232)102(57-83-31-18-15-19-32-83)185-148(238)121(80(11)196)189-114(204)68-165-130(220)95(44-48-115(205)206)174-125(215)78(9)168-127(217)89(153)60-86-64-158-72-166-86/h14-21,29-34,39-42,63-64,72-81,89,91-108,119-122,161,193-198H,13,22-28,35-38,43-62,65-71,151-153H2,1-12H3,(H2,154,199)(H2,155,200)(H,158,166)(H,159,201)(H,162,218)(H,163,219)(H,164,221)(H,165,220)(H,167,213)(H,168,217)(H,169,222)(H,170,237)(H,171,202)(H,172,203)(H,173,214)(H,174,215)(H,175,229)(H,176,228)(H,177,223)(H,178,236)(H,179,216)(H,180,227)(H,181,225)(H,182,233)(H,183,226)(H,184,234)(H,185,238)(H,186,224)(H,187,235)(H,188,239)(H,189,204)(H,190,230)(H,191,231)(H,192,232)(H,205,206)(H,207,208)(H,209,210)(H,211,212)(H4,156,157,160)/t75-,76-,77-,78-,79-,80+,81+,89-,91-,92-,93-,94-,95-,96-,97-,98-,99-,100-,101-,102-,103-,104-,105-,106-,107-,108-,119-,120-,121-,122-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.520 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Displacement of [125I]-GLP (7 to 36 residues) from human GLP1R expressed in CHO cell membranes incubated for 30 mins measured after 10 hrs by scintil... |
Eur J Med Chem 127: 703-714 (2017)
Article DOI: 10.1016/j.ejmech.2016.10.044
BindingDB Entry DOI: 10.7270/Q2T155W1 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50355626
(CHEMBL1910860)Show SMILES CC(C)(CCC(C(N)=O)(c1ccccc1)c1ccccc1)N1CC[C@H](C1)Oc1cc(O)cc(F)c1 |r| Show InChI InChI=1S/C29H33FN2O3/c1-28(2,32-16-13-25(20-32)35-26-18-23(30)17-24(33)19-26)14-15-29(27(31)34,21-9-5-3-6-10-21)22-11-7-4-8-12-22/h3-12,17-19,25,33H,13-16,20H2,1-2H3,(H2,31,34)/t25-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.560 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant M3 receptor expressed in CHO-K1 cells assessed as inhibition of carbamoyl choline-induced calcium currents a... |
J Med Chem 54: 6888-904 (2011)
Article DOI: 10.1021/jm200884j
BindingDB Entry DOI: 10.7270/Q2CZ37JW |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

















































