 Found 166 hits with Last Name = 'wang' and Initial = 'yk'
Found 166 hits with Last Name = 'wang' and Initial = 'yk' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50239250

(CHEMBL4086080)Show SMILES CNC(=O)c1c(oc2ccc(cc12)-c1cc(ccc1C)C(=O)NC1(CC1)c1ccncc1)-c1ccc(F)cc1 Show InChI InChI=1S/C32H26FN3O3/c1-19-3-4-22(30(37)36-32(13-14-32)23-11-15-35-16-12-23)18-25(19)21-7-10-27-26(17-21)28(31(38)34-2)29(39-27)20-5-8-24(33)9-6-20/h3-12,15-18H,13-14H2,1-2H3,(H,34,38)(H,36,37) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP2C8 (unknown origin) |
J Med Chem 60: 4369-4385 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00328
BindingDB Entry DOI: 10.7270/Q20R9RKB |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50239251

(CHEMBL4096241)Show SMILES CNC(=O)c1c(oc2ccc(cc12)-c1cc(ccc1C)C(=O)NC1(CC1)c1cccnc1)-c1ccc(F)cc1 Show InChI InChI=1S/C32H26FN3O3/c1-19-5-6-22(30(37)36-32(13-14-32)23-4-3-15-35-18-23)17-25(19)21-9-12-27-26(16-21)28(31(38)34-2)29(39-27)20-7-10-24(33)11-8-20/h3-12,15-18H,13-14H2,1-2H3,(H,34,38)(H,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 63 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP3A4 (unknown origin) |
J Med Chem 60: 4369-4385 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00328
BindingDB Entry DOI: 10.7270/Q20R9RKB |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50239250

(CHEMBL4086080)Show SMILES CNC(=O)c1c(oc2ccc(cc12)-c1cc(ccc1C)C(=O)NC1(CC1)c1ccncc1)-c1ccc(F)cc1 Show InChI InChI=1S/C32H26FN3O3/c1-19-3-4-22(30(37)36-32(13-14-32)23-11-15-35-16-12-23)18-25(19)21-7-10-27-26(17-21)28(31(38)34-2)29(39-27)20-5-8-24(33)9-6-20/h3-12,15-18H,13-14H2,1-2H3,(H,34,38)(H,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 73 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP3A4 (unknown origin) |
J Med Chem 60: 4369-4385 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00328
BindingDB Entry DOI: 10.7270/Q20R9RKB |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50239251

(CHEMBL4096241)Show SMILES CNC(=O)c1c(oc2ccc(cc12)-c1cc(ccc1C)C(=O)NC1(CC1)c1cccnc1)-c1ccc(F)cc1 Show InChI InChI=1S/C32H26FN3O3/c1-19-5-6-22(30(37)36-32(13-14-32)23-4-3-15-35-18-23)17-25(19)21-9-12-27-26(16-21)28(31(38)34-2)29(39-27)20-7-10-24(33)11-8-20/h3-12,15-18H,13-14H2,1-2H3,(H,34,38)(H,36,37) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 350 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP2C8 (unknown origin) |
J Med Chem 60: 4369-4385 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00328
BindingDB Entry DOI: 10.7270/Q20R9RKB |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50254772
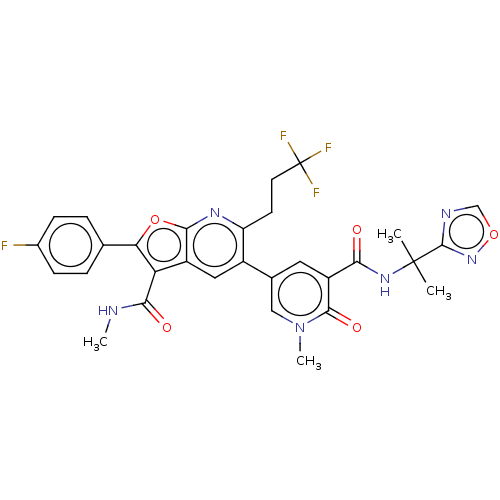
(CHEMBL4061621)Show SMILES CNC(=O)c1c(oc2nc(CCC(F)(F)F)c(cc12)-c1cc(C(=O)NC(C)(C)c2ncon2)c(=O)n(C)c1)-c1ccc(F)cc1 Show InChI InChI=1S/C30H26F4N6O5/c1-29(2,28-36-14-44-39-28)38-24(41)20-11-16(13-40(4)27(20)43)18-12-19-22(25(42)35-3)23(15-5-7-17(31)8-6-15)45-26(19)37-21(18)9-10-30(32,33)34/h5-8,11-14H,9-10H2,1-4H3,(H,35,42)(H,38,41) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 440 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development, 5 Research Parkway, Wallingford, Connecticut 06492, United States.
Curated by ChEMBL
| Assay Description
Competitive inhibition of CYP3A4 in human liver microsomes in presence of NADPH by LC-MS/MS analysis |
ACS Med Chem Lett 8: 771-774 (2017)
Article DOI: 10.1021/acsmedchemlett.7b00211
BindingDB Entry DOI: 10.7270/Q2KK9F7P |
More data for this
Ligand-Target Pair | |
RNA-directed RNA polymerase
(Hepacivirus C) | BDBM50484982

(CHEMBL2017865)Show SMILES OC(=O)\C=C\c1ccc(NC(=O)C2(CCCC2)NC(=O)c2ccc3c(C4CCCCC4)c4-c5ncccc5OCCn4c3c2)cc1 Show InChI InChI=1S/C37H38N4O5/c42-31(43)17-12-24-10-14-27(15-11-24)39-36(45)37(18-4-5-19-37)40-35(44)26-13-16-28-29(23-26)41-21-22-46-30-9-6-20-38-33(30)34(41)32(28)25-7-2-1-3-8-25/h6,9-17,20,23,25H,1-5,7-8,18-19,21-22H2,(H,39,45)(H,40,44)(H,42,43)/b17-12+ | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >600 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Inhibition of Hepatitis C virus NS5B RNA-dependent RNA polymerase P495L mutant |
Bioorg Med Chem Lett 22: 2866-71 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.063
BindingDB Entry DOI: 10.7270/Q2NK3HWC |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50239252

(CHEMBL4078188)Show SMILES CNC(=O)c1c(oc2ccc(cc12)-c1cc(ccc1C)C(=O)NC1(CC1)c1ccccn1)-c1ccc(F)cc1 Show InChI InChI=1S/C32H26FN3O3/c1-19-6-7-22(30(37)36-32(14-15-32)27-5-3-4-16-35-27)18-24(19)21-10-13-26-25(17-21)28(31(38)34-2)29(39-26)20-8-11-23(33)12-9-20/h3-13,16-18H,14-15H2,1-2H3,(H,34,38)(H,36,37) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP2C8 (unknown origin) |
J Med Chem 60: 4369-4385 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00328
BindingDB Entry DOI: 10.7270/Q20R9RKB |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50239253
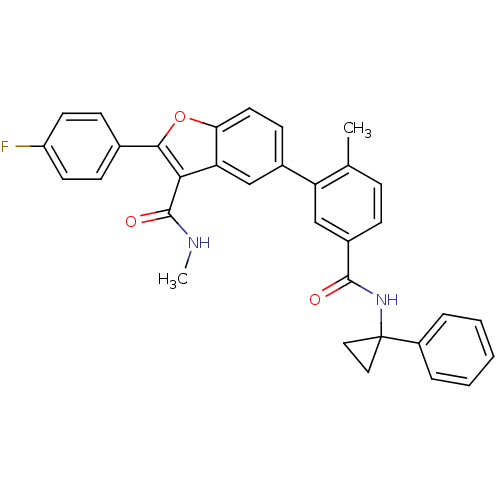
(CHEMBL4088517)Show SMILES CNC(=O)c1c(oc2ccc(cc12)-c1cc(ccc1C)C(=O)NC1(CC1)c1ccccc1)-c1ccc(F)cc1 Show InChI InChI=1S/C33H27FN2O3/c1-20-8-9-23(31(37)36-33(16-17-33)24-6-4-3-5-7-24)19-26(20)22-12-15-28-27(18-22)29(32(38)35-2)30(39-28)21-10-13-25(34)14-11-21/h3-15,18-19H,16-17H2,1-2H3,(H,35,38)(H,36,37) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP2C8 (unknown origin) |
J Med Chem 60: 4369-4385 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00328
BindingDB Entry DOI: 10.7270/Q20R9RKB |
More data for this
Ligand-Target Pair | |
RNA-directed RNA polymerase
(Hepacivirus C) | BDBM50484986
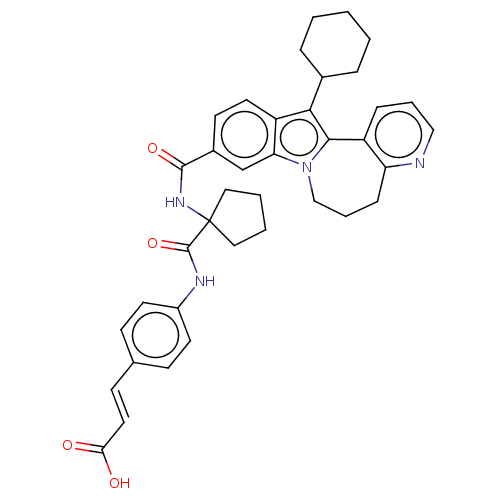
(CHEMBL2017863)Show SMILES OC(=O)\C=C\c1ccc(NC(=O)C2(CCCC2)NC(=O)c2ccc3c(C4CCCCC4)c4-c5cccnc5CCCn4c3c2)cc1 Show InChI InChI=1S/C38H40N4O4/c43-33(44)19-14-25-12-16-28(17-13-25)40-37(46)38(20-4-5-21-38)41-36(45)27-15-18-30-32(24-27)42-23-7-11-31-29(10-6-22-39-31)35(42)34(30)26-8-2-1-3-9-26/h6,10,12-19,22,24,26H,1-5,7-9,11,20-21,23H2,(H,40,46)(H,41,45)(H,43,44)/b19-14+ | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.73E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Inhibition of Hepatitis C virus NS5B RNA-dependent RNA polymerase P495L mutant |
Bioorg Med Chem Lett 22: 2866-71 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.063
BindingDB Entry DOI: 10.7270/Q2NK3HWC |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50239253

(CHEMBL4088517)Show SMILES CNC(=O)c1c(oc2ccc(cc12)-c1cc(ccc1C)C(=O)NC1(CC1)c1ccccc1)-c1ccc(F)cc1 Show InChI InChI=1S/C33H27FN2O3/c1-20-8-9-23(31(37)36-33(16-17-33)24-6-4-3-5-7-24)19-26(20)22-12-15-28-27(18-22)29(32(38)35-2)30(39-28)21-10-13-25(34)14-11-21/h3-15,18-19H,16-17H2,1-2H3,(H,35,38)(H,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP3A4 (unknown origin) |
J Med Chem 60: 4369-4385 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00328
BindingDB Entry DOI: 10.7270/Q20R9RKB |
More data for this
Ligand-Target Pair | |
RNA-directed RNA polymerase
(Hepacivirus C) | BDBM50484981
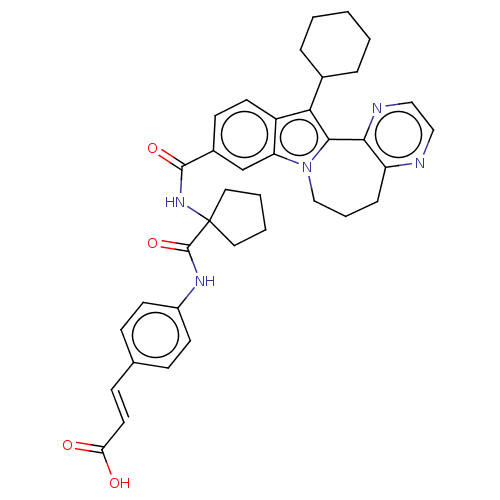
(CHEMBL2017862)Show SMILES OC(=O)\C=C\c1ccc(NC(=O)C2(CCCC2)NC(=O)c2ccc3c(C4CCCCC4)c4-c5nccnc5CCCn4c3c2)cc1 Show InChI InChI=1S/C37H39N5O4/c43-31(44)17-12-24-10-14-27(15-11-24)40-36(46)37(18-4-5-19-37)41-35(45)26-13-16-28-30(23-26)42-22-6-9-29-33(39-21-20-38-29)34(42)32(28)25-7-2-1-3-8-25/h10-17,20-21,23,25H,1-9,18-19,22H2,(H,40,46)(H,41,45)(H,43,44)/b17-12+ | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.28E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Inhibition of Hepatitis C virus NS5B RNA-dependent RNA polymerase P495L mutant |
Bioorg Med Chem Lett 22: 2866-71 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.063
BindingDB Entry DOI: 10.7270/Q2NK3HWC |
More data for this
Ligand-Target Pair | |
RNA-directed RNA polymerase
(Hepacivirus C) | BDBM50484983

(CHEMBL2017866)Show SMILES OC(=O)\C=C\c1ccc(NC(=O)C2(CCCC2)NC(=O)c2ccc3c(C4CCCCC4)c4-c5cccnc5OCCn4c3c2)cc1 Show InChI InChI=1S/C37H38N4O5/c42-31(43)17-12-24-10-14-27(15-11-24)39-36(45)37(18-4-5-19-37)40-34(44)26-13-16-28-30(23-26)41-21-22-46-35-29(9-6-20-38-35)33(41)32(28)25-7-2-1-3-8-25/h6,9-17,20,23,25H,1-5,7-8,18-19,21-22H2,(H,39,45)(H,40,44)(H,42,43)/b17-12+ | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Inhibition of Hepatitis C virus NS5B RNA-dependent RNA polymerase P495L mutant |
Bioorg Med Chem Lett 22: 2866-71 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.063
BindingDB Entry DOI: 10.7270/Q2NK3HWC |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50565837
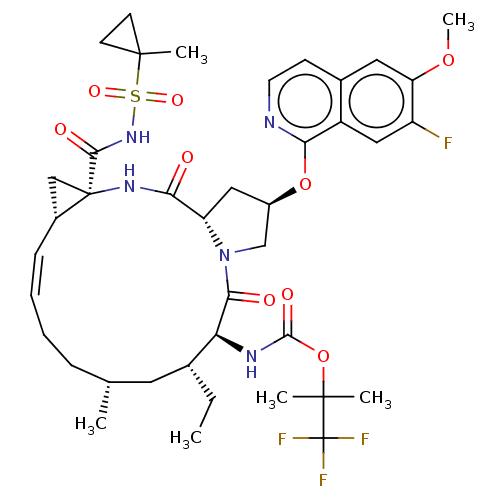
(CHEMBL4787795)Show SMILES [H][C@@]12C[C@]1(NC(=O)[C@]1([H])C[C@H](CN1C(=O)[C@@H](NC(=O)OC(C)(C)C(F)(F)F)[C@H](CC)C[C@H](C)CC\C=C/2)Oc1nccc2cc(OC)c(F)cc12)C(=O)NS(=O)(=O)C1(C)CC1 |r,c:37| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 2.85E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human CYP3A4 in human pooled liver microsomes in presence of midazolam as substrate preincubated for 30 mins in presence of NADPH follo... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01296
BindingDB Entry DOI: 10.7270/Q27D2ZWX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50565837

(CHEMBL4787795)Show SMILES [H][C@@]12C[C@]1(NC(=O)[C@]1([H])C[C@H](CN1C(=O)[C@@H](NC(=O)OC(C)(C)C(F)(F)F)[C@H](CC)C[C@H](C)CC\C=C/2)Oc1nccc2cc(OC)c(F)cc12)C(=O)NS(=O)(=O)C1(C)CC1 |r,c:37| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 3.02E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human CYP3A4 in human pooled liver microsomes in presence of testosterone as substrate preincubated for 30 mins in presence of NADPH fo... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01296
BindingDB Entry DOI: 10.7270/Q27D2ZWX |
More data for this
Ligand-Target Pair | |
RNA-directed RNA polymerase
(Hepacivirus C) | BDBM50484985

(CHEMBL2017864)Show SMILES OC(=O)\C=C\c1ccc(NC(=O)C2(CCCC2)NC(=O)c2ccc3c(C4CCCCC4)c4-c5ncccc5NC(=O)Cn4c3c2)cc1 Show InChI InChI=1S/C37H37N5O5/c43-30-22-42-29-21-25(13-16-27(29)32(24-7-2-1-3-8-24)34(42)33-28(40-30)9-6-20-38-33)35(46)41-37(18-4-5-19-37)36(47)39-26-14-10-23(11-15-26)12-17-31(44)45/h6,9-17,20-21,24H,1-5,7-8,18-19,22H2,(H,39,47)(H,40,43)(H,41,46)(H,44,45)/b17-12+ | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Inhibition of Hepatitis C virus NS5B RNA-dependent RNA polymerase P495L mutant |
Bioorg Med Chem Lett 22: 2866-71 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.063
BindingDB Entry DOI: 10.7270/Q2NK3HWC |
More data for this
Ligand-Target Pair | |
RNA-directed RNA polymerase
(Hepacivirus C) | BDBM50484977

(CHEMBL2017856)Show SMILES OC(=O)\C=C\c1ccc(NC(=O)C2(CCCC2)NC(=O)c2ccc3c(C4CCCCC4)c4-c5ccccc5CCCn4c3c2)cc1 Show InChI InChI=1S/C39H41N3O4/c43-34(44)21-16-26-14-18-30(19-15-26)40-38(46)39(22-6-7-23-39)41-37(45)29-17-20-32-33(25-29)42-24-8-12-27-9-4-5-13-31(27)36(42)35(32)28-10-2-1-3-11-28/h4-5,9,13-21,25,28H,1-3,6-8,10-12,22-24H2,(H,40,46)(H,41,45)(H,43,44)/b21-16+ | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.39E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Inhibition of Hepatitis C virus NS5B RNA-dependent RNA polymerase P495L mutant |
Bioorg Med Chem Lett 22: 2866-71 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.063
BindingDB Entry DOI: 10.7270/Q2NK3HWC |
More data for this
Ligand-Target Pair | |
RNA-directed RNA polymerase
(Hepacivirus C) | BDBM50484984

(CHEMBL2017858)Show SMILES OC(=O)\C=C\c1ccc(NC(=O)C2(CCCC2)NC(=O)c2ccc3c(C4CCCCC4)c4-c5ccoc5CCCn4c3c2)cc1 Show InChI InChI=1S/C37H39N3O5/c41-32(42)17-12-24-10-14-27(15-11-24)38-36(44)37(19-4-5-20-37)39-35(43)26-13-16-28-30(23-26)40-21-6-9-31-29(18-22-45-31)34(40)33(28)25-7-2-1-3-8-25/h10-18,22-23,25H,1-9,19-21H2,(H,38,44)(H,39,43)(H,41,42)/b17-12+ | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.72E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Inhibition of Hepatitis C virus NS5B RNA-dependent RNA polymerase P495L mutant |
Bioorg Med Chem Lett 22: 2866-71 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.063
BindingDB Entry DOI: 10.7270/Q2NK3HWC |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50239249

(CHEMBL4104000)Show SMILES CNC(=O)c1c(oc2ccc(cc12)-c1cc(ccc1C)C(=O)NC1(CC1)c1ncccn1)-c1ccc(F)cc1 Show InChI InChI=1S/C31H25FN4O3/c1-18-4-5-21(28(37)36-31(12-13-31)30-34-14-3-15-35-30)17-23(18)20-8-11-25-24(16-20)26(29(38)33-2)27(39-25)19-6-9-22(32)10-7-19/h3-11,14-17H,12-13H2,1-2H3,(H,33,38)(H,36,37) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP2C8 (unknown origin) |
J Med Chem 60: 4369-4385 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00328
BindingDB Entry DOI: 10.7270/Q20R9RKB |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50239252

(CHEMBL4078188)Show SMILES CNC(=O)c1c(oc2ccc(cc12)-c1cc(ccc1C)C(=O)NC1(CC1)c1ccccn1)-c1ccc(F)cc1 Show InChI InChI=1S/C32H26FN3O3/c1-19-6-7-22(30(37)36-32(14-15-32)27-5-3-4-16-35-27)18-24(19)21-10-13-26-25(17-21)28(31(38)34-2)29(39-26)20-8-11-23(33)12-9-20/h3-13,16-18H,14-15H2,1-2H3,(H,34,38)(H,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP3A4 (unknown origin) |
J Med Chem 60: 4369-4385 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00328
BindingDB Entry DOI: 10.7270/Q20R9RKB |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50239249

(CHEMBL4104000)Show SMILES CNC(=O)c1c(oc2ccc(cc12)-c1cc(ccc1C)C(=O)NC1(CC1)c1ncccn1)-c1ccc(F)cc1 Show InChI InChI=1S/C31H25FN4O3/c1-18-4-5-21(28(37)36-31(12-13-31)30-34-14-3-15-35-30)17-23(18)20-8-11-25-24(16-20)26(29(38)33-2)27(39-25)19-6-9-22(32)10-7-19/h3-11,14-17H,12-13H2,1-2H3,(H,33,38)(H,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by thallium flux assay |
J Med Chem 60: 4369-4385 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00328
BindingDB Entry DOI: 10.7270/Q20R9RKB |
More data for this
Ligand-Target Pair | |
RNA-directed RNA polymerase
(Hepacivirus C) | BDBM50484980
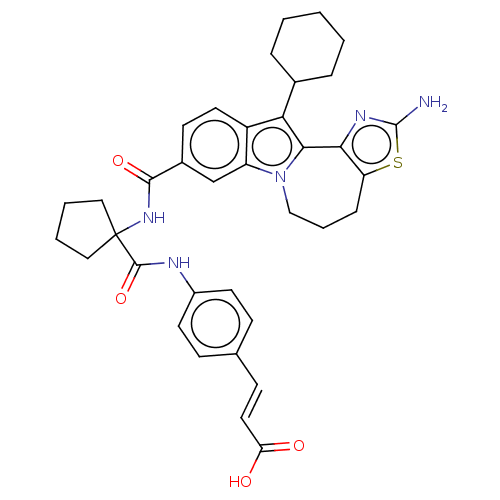
(CHEMBL2017861)Show SMILES Nc1nc-2c(CCCn3c-2c(C2CCCCC2)c2ccc(cc32)C(=O)NC2(CCCC2)C(=O)Nc2ccc(\C=C\C(O)=O)cc2)s1 Show InChI InChI=1S/C36H39N5O4S/c37-35-39-31-28(46-35)9-6-20-41-27-21-24(13-16-26(27)30(32(31)41)23-7-2-1-3-8-23)33(44)40-36(18-4-5-19-36)34(45)38-25-14-10-22(11-15-25)12-17-29(42)43/h10-17,21,23H,1-9,18-20H2,(H2,37,39)(H,38,45)(H,40,44)(H,42,43)/b17-12+ | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.84E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Inhibition of Hepatitis C virus NS5B RNA-dependent RNA polymerase P495L mutant |
Bioorg Med Chem Lett 22: 2866-71 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.063
BindingDB Entry DOI: 10.7270/Q2NK3HWC |
More data for this
Ligand-Target Pair | |
RNA-directed RNA polymerase
(Hepacivirus C) | BDBM50484979

(CHEMBL2017860)Show SMILES Cc1nc-2c(CCCn3c-2c(C2CCCCC2)c2ccc(cc32)C(=O)NC2(CCCC2)C(=O)Nc2ccc(\C=C\C(O)=O)cc2)s1 Show InChI InChI=1S/C37H40N4O4S/c1-23-38-33-30(46-23)10-7-21-41-29-22-26(14-17-28(29)32(34(33)41)25-8-3-2-4-9-25)35(44)40-37(19-5-6-20-37)36(45)39-27-15-11-24(12-16-27)13-18-31(42)43/h11-18,22,25H,2-10,19-21H2,1H3,(H,39,45)(H,40,44)(H,42,43)/b18-13+ | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Inhibition of Hepatitis C virus NS5B RNA-dependent RNA polymerase P495L mutant |
Bioorg Med Chem Lett 22: 2866-71 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.063
BindingDB Entry DOI: 10.7270/Q2NK3HWC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50239252

(CHEMBL4078188)Show SMILES CNC(=O)c1c(oc2ccc(cc12)-c1cc(ccc1C)C(=O)NC1(CC1)c1ccccn1)-c1ccc(F)cc1 Show InChI InChI=1S/C32H26FN3O3/c1-19-6-7-22(30(37)36-32(14-15-32)27-5-3-4-16-35-27)18-24(19)21-10-13-26-25(17-21)28(31(38)34-2)29(39-26)20-8-11-23(33)12-9-20/h3-13,16-18H,14-15H2,1-2H3,(H,34,38)(H,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by thallium flux assay |
J Med Chem 60: 4369-4385 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00328
BindingDB Entry DOI: 10.7270/Q20R9RKB |
More data for this
Ligand-Target Pair | |
RNA-directed RNA polymerase
(Hepacivirus C) | BDBM50484978

(CHEMBL2017857)Show SMILES OC(=O)\C=C\c1ccc(NC(=O)C2(CCCC2)NC(=O)c2ccc3c(C4CCCCC4)c4-c5ncoc5CCCn4c3c2)cc1 Show InChI InChI=1S/C36H38N4O5/c41-30(42)17-12-23-10-14-26(15-11-23)38-35(44)36(18-4-5-19-36)39-34(43)25-13-16-27-28(21-25)40-20-6-9-29-32(37-22-45-29)33(40)31(27)24-7-2-1-3-8-24/h10-17,21-22,24H,1-9,18-20H2,(H,38,44)(H,39,43)(H,41,42)/b17-12+ | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.85E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Inhibition of Hepatitis C virus NS5B RNA-dependent RNA polymerase P495L mutant |
Bioorg Med Chem Lett 22: 2866-71 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.063
BindingDB Entry DOI: 10.7270/Q2NK3HWC |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50239248
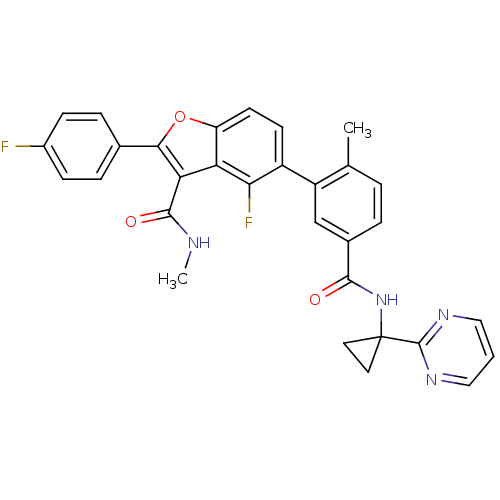
(CHEMBL4093031)Show SMILES CNC(=O)c1c(oc2ccc(c(F)c12)-c1cc(ccc1C)C(=O)NC1(CC1)c1ncccn1)-c1ccc(F)cc1 Show InChI InChI=1S/C31H24F2N4O3/c1-17-4-5-19(28(38)37-31(12-13-31)30-35-14-3-15-36-30)16-22(17)21-10-11-23-24(26(21)33)25(29(39)34-2)27(40-23)18-6-8-20(32)9-7-18/h3-11,14-16H,12-13H2,1-2H3,(H,34,39)(H,37,38) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP2C8 (unknown origin) |
J Med Chem 60: 4369-4385 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00328
BindingDB Entry DOI: 10.7270/Q20R9RKB |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50239249

(CHEMBL4104000)Show SMILES CNC(=O)c1c(oc2ccc(cc12)-c1cc(ccc1C)C(=O)NC1(CC1)c1ncccn1)-c1ccc(F)cc1 Show InChI InChI=1S/C31H25FN4O3/c1-18-4-5-21(28(37)36-31(12-13-31)30-34-14-3-15-35-30)17-23(18)20-8-11-25-24(16-20)26(29(38)33-2)27(39-25)19-6-9-22(32)10-7-19/h3-11,14-17H,12-13H2,1-2H3,(H,33,38)(H,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.14E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP3A4 (unknown origin) |
J Med Chem 60: 4369-4385 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00328
BindingDB Entry DOI: 10.7270/Q20R9RKB |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50198324
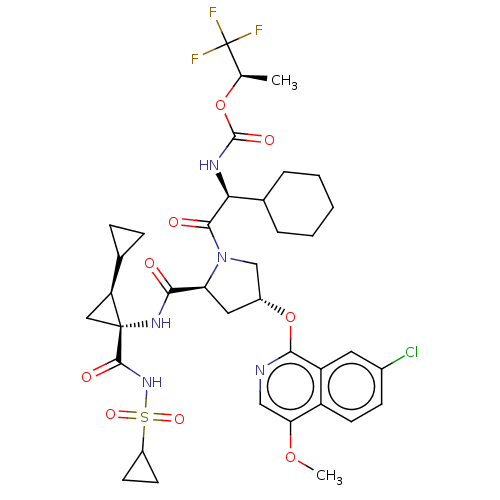
(CHEMBL3933212)Show SMILES COc1cnc(O[C@@H]2C[C@H](N(C2)C(=O)[C@@H](NC(=O)O[C@H](C)C(F)(F)F)C2CCCCC2)C(=O)N[C@@]2(C[C@H]2C2CC2)C(=O)NS(=O)(=O)C2CC2)c2cc(Cl)ccc12 |r| Show InChI InChI=1S/C37H45ClF3N5O9S/c1-19(37(39,40)41)54-35(50)43-30(21-6-4-3-5-7-21)33(48)46-18-23(55-32-26-14-22(38)10-13-25(26)29(53-2)17-42-32)15-28(46)31(47)44-36(16-27(36)20-8-9-20)34(49)45-56(51,52)24-11-12-24/h10,13-14,17,19-21,23-24,27-28,30H,3-9,11-12,15-16,18H2,1-2H3,(H,43,50)(H,44,47)(H,45,49)/t19-,23-,27+,28+,30+,36-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.25E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by patch clamp assay |
J Med Chem 59: 8042-60 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00821
BindingDB Entry DOI: 10.7270/Q21J9CR8 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50239248

(CHEMBL4093031)Show SMILES CNC(=O)c1c(oc2ccc(c(F)c12)-c1cc(ccc1C)C(=O)NC1(CC1)c1ncccn1)-c1ccc(F)cc1 Show InChI InChI=1S/C31H24F2N4O3/c1-17-4-5-19(28(38)37-31(12-13-31)30-35-14-3-15-36-30)16-22(17)21-10-11-23-24(26(21)33)25(29(39)34-2)27(40-23)18-6-8-20(32)9-7-18/h3-11,14-16H,12-13H2,1-2H3,(H,34,39)(H,37,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of microsomal CYP3A4 (unknown origin) |
J Med Chem 60: 4369-4385 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00328
BindingDB Entry DOI: 10.7270/Q20R9RKB |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50565837

(CHEMBL4787795)Show SMILES [H][C@@]12C[C@]1(NC(=O)[C@]1([H])C[C@H](CN1C(=O)[C@@H](NC(=O)OC(C)(C)C(F)(F)F)[C@H](CC)C[C@H](C)CC\C=C/2)Oc1nccc2cc(OC)c(F)cc12)C(=O)NS(=O)(=O)C1(C)CC1 |r,c:37| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 1.64E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human CYP3A4 in human pooled liver microsomes in presence of midazolam as substrate preincubated for 30 mins in presence of NADPH follo... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01296
BindingDB Entry DOI: 10.7270/Q27D2ZWX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50239248

(CHEMBL4093031)Show SMILES CNC(=O)c1c(oc2ccc(c(F)c12)-c1cc(ccc1C)C(=O)NC1(CC1)c1ncccn1)-c1ccc(F)cc1 Show InChI InChI=1S/C31H24F2N4O3/c1-17-4-5-19(28(38)37-31(12-13-31)30-35-14-3-15-36-30)16-22(17)21-10-11-23-24(26(21)33)25(29(39)34-2)27(40-23)18-6-8-20(32)9-7-18/h3-11,14-16H,12-13H2,1-2H3,(H,34,39)(H,37,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by patch clamp assay |
J Med Chem 60: 4369-4385 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00328
BindingDB Entry DOI: 10.7270/Q20R9RKB |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50198324

(CHEMBL3933212)Show SMILES COc1cnc(O[C@@H]2C[C@H](N(C2)C(=O)[C@@H](NC(=O)O[C@H](C)C(F)(F)F)C2CCCCC2)C(=O)N[C@@]2(C[C@H]2C2CC2)C(=O)NS(=O)(=O)C2CC2)c2cc(Cl)ccc12 |r| Show InChI InChI=1S/C37H45ClF3N5O9S/c1-19(37(39,40)41)54-35(50)43-30(21-6-4-3-5-7-21)33(48)46-18-23(55-32-26-14-22(38)10-13-25(26)29(53-2)17-42-32)15-28(46)31(47)44-36(16-27(36)20-8-9-20)34(49)45-56(51,52)24-11-12-24/h10,13-14,17,19-21,23-24,27-28,30H,3-9,11-12,15-16,18H2,1-2H3,(H,43,50)(H,44,47)(H,45,49)/t19-,23-,27+,28+,30+,36-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human leukocyte elastase |
J Med Chem 59: 8042-60 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00821
BindingDB Entry DOI: 10.7270/Q21J9CR8 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50239248

(CHEMBL4093031)Show SMILES CNC(=O)c1c(oc2ccc(c(F)c12)-c1cc(ccc1C)C(=O)NC1(CC1)c1ncccn1)-c1ccc(F)cc1 Show InChI InChI=1S/C31H24F2N4O3/c1-17-4-5-19(28(38)37-31(12-13-31)30-35-14-3-15-36-30)16-22(17)21-10-11-23-24(26(21)33)25(29(39)34-2)27(40-23)18-6-8-20(32)9-7-18/h3-11,14-16H,12-13H2,1-2H3,(H,34,39)(H,37,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human ERG by thallium flux assay |
J Med Chem 60: 4369-4385 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00328
BindingDB Entry DOI: 10.7270/Q20R9RKB |
More data for this
Ligand-Target Pair | |
DNA polymerase alpha catalytic subunit
(Homo sapiens (Human)) | BDBM50268397

(CHEMBL4067852)Show SMILES [H][C@@]12C[C@@]1(Cn1c(c(C3CCCCC3)c3ccc(cc13)C(=O)NS(=O)(=O)C1(C)CC1)-c1ccc(F)cc21)C(=O)N1C2CCC1CN(C)C2 |r,TLB:37:39:47.45.44:41.42,THB:46:45:39:41.42| Show InChI InChI=1S/C37H43FN4O4S/c1-36(14-15-36)47(45,46)39-34(43)23-8-12-28-31(16-23)41-21-37(35(44)42-25-10-11-26(42)20-40(2)19-25)18-30(37)29-17-24(38)9-13-27(29)33(41)32(28)22-6-4-3-5-7-22/h8-9,12-13,16-17,22,25-26,30H,3-7,10-11,14-15,18-21H2,1-2H3,(H,39,43)/t25?,26?,30-,37-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Discovery Chemistry and Molecular Technologies, Bristol-Myers Squibb Research and Development, 5 Research Parkway, Wallingford, CT 06492, United States. Electronic address: zhizhenZheng
Curated by ChEMBL
| Assay Description
Inhibition of human DNA polymerase alpha |
Bioorg Med Chem Lett 27: 3294-3300 (2017)
Article DOI: 10.1016/j.bmcl.2017.06.024
BindingDB Entry DOI: 10.7270/Q20004KW |
More data for this
Ligand-Target Pair | |
DNA polymerase beta
(Homo sapiens (Human)) | BDBM50268397

(CHEMBL4067852)Show SMILES [H][C@@]12C[C@@]1(Cn1c(c(C3CCCCC3)c3ccc(cc13)C(=O)NS(=O)(=O)C1(C)CC1)-c1ccc(F)cc21)C(=O)N1C2CCC1CN(C)C2 |r,TLB:37:39:47.45.44:41.42,THB:46:45:39:41.42| Show InChI InChI=1S/C37H43FN4O4S/c1-36(14-15-36)47(45,46)39-34(43)23-8-12-28-31(16-23)41-21-37(35(44)42-25-10-11-26(42)20-40(2)19-25)18-30(37)29-17-24(38)9-13-27(29)33(41)32(28)22-6-4-3-5-7-22/h8-9,12-13,16-17,22,25-26,30H,3-7,10-11,14-15,18-21H2,1-2H3,(H,39,43)/t25?,26?,30-,37-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Discovery Chemistry and Molecular Technologies, Bristol-Myers Squibb Research and Development, 5 Research Parkway, Wallingford, CT 06492, United States. Electronic address: zhizhenZheng
Curated by ChEMBL
| Assay Description
Inhibition of human DNA polymerase beta |
Bioorg Med Chem Lett 27: 3294-3300 (2017)
Article DOI: 10.1016/j.bmcl.2017.06.024
BindingDB Entry DOI: 10.7270/Q20004KW |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50565837

(CHEMBL4787795)Show SMILES [H][C@@]12C[C@]1(NC(=O)[C@]1([H])C[C@H](CN1C(=O)[C@@H](NC(=O)OC(C)(C)C(F)(F)F)[C@H](CC)C[C@H](C)CC\C=C/2)Oc1nccc2cc(OC)c(F)cc12)C(=O)NS(=O)(=O)C1(C)CC1 |r,c:37| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >3.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human CYP3A4 in human pooled liver microsomes in presence of testosterone as substrate preincubated for 30 mins in presence of NADPH fo... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01296
BindingDB Entry DOI: 10.7270/Q27D2ZWX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50565837

(CHEMBL4787795)Show SMILES [H][C@@]12C[C@]1(NC(=O)[C@]1([H])C[C@H](CN1C(=O)[C@@H](NC(=O)OC(C)(C)C(F)(F)F)[C@H](CC)C[C@H](C)CC\C=C/2)Oc1nccc2cc(OC)c(F)cc12)C(=O)NS(=O)(=O)C1(C)CC1 |r,c:37| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human CYP3A4 in human pooled liver microsomes in presence of midazolam as substrate preincubated for 0 mins in presence of NADPH follow... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01296
BindingDB Entry DOI: 10.7270/Q27D2ZWX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50565837

(CHEMBL4787795)Show SMILES [H][C@@]12C[C@]1(NC(=O)[C@]1([H])C[C@H](CN1C(=O)[C@@H](NC(=O)OC(C)(C)C(F)(F)F)[C@H](CC)C[C@H](C)CC\C=C/2)Oc1nccc2cc(OC)c(F)cc12)C(=O)NS(=O)(=O)C1(C)CC1 |r,c:37| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human CYP3A4 in human pooled liver microsomes in presence of midazolam as substrate preincubated for 0 mins in presence of NADPH follow... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01296
BindingDB Entry DOI: 10.7270/Q27D2ZWX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50565837

(CHEMBL4787795)Show SMILES [H][C@@]12C[C@]1(NC(=O)[C@]1([H])C[C@H](CN1C(=O)[C@@H](NC(=O)OC(C)(C)C(F)(F)F)[C@H](CC)C[C@H](C)CC\C=C/2)Oc1nccc2cc(OC)c(F)cc12)C(=O)NS(=O)(=O)C1(C)CC1 |r,c:37| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human CYP3A4 in human pooled liver microsomes in presence of testosterone as substrate preincubated for 0 mins in presence of NADPH fol... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01296
BindingDB Entry DOI: 10.7270/Q27D2ZWX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50448498
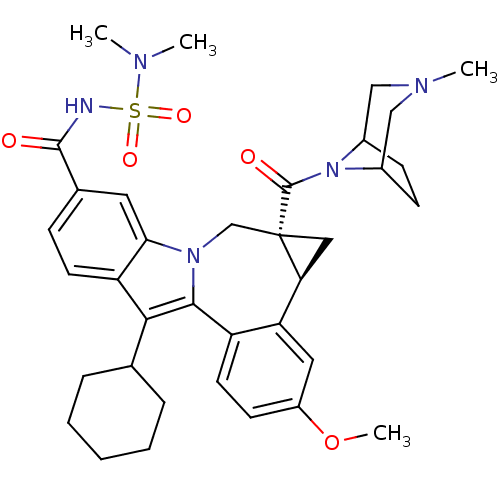
(BMS-791325 | Beclabuvir)Show SMILES COc1ccc2-c3c(C4CCCCC4)c4ccc(cc4n3C[C@]3(C[C@H]3c2c1)C(=O)N1C2CCC1CN(C)C2)C(=O)NS(=O)(=O)N(C)C |r,THB:36:35:29:31.32| Show InChI InChI=1S/C36H45N5O5S/c1-38(2)47(44,45)37-34(42)23-10-14-28-31(16-23)40-21-36(35(43)41-24-11-12-25(41)20-39(3)19-24)18-30(36)29-17-26(46-4)13-15-27(29)33(40)32(28)22-8-6-5-7-9-22/h10,13-17,22,24-25,30H,5-9,11-12,18-21H2,1-4H3,(H,37,42)/t24?,25?,30-,36-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
J Med Chem 57: 1855-79 (2014)
Article DOI: 10.1021/jm4016894
BindingDB Entry DOI: 10.7270/Q2V989JD |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50448498

(BMS-791325 | Beclabuvir)Show SMILES COc1ccc2-c3c(C4CCCCC4)c4ccc(cc4n3C[C@]3(C[C@H]3c2c1)C(=O)N1C2CCC1CN(C)C2)C(=O)NS(=O)(=O)N(C)C |r,THB:36:35:29:31.32| Show InChI InChI=1S/C36H45N5O5S/c1-38(2)47(44,45)37-34(42)23-10-14-28-31(16-23)40-21-36(35(43)41-24-11-12-25(41)20-39(3)19-24)18-30(36)29-17-26(46-4)13-15-27(29)33(40)32(28)22-8-6-5-7-9-22/h10,13-17,22,24-25,30H,5-9,11-12,18-21H2,1-4H3,(H,37,42)/t24?,25?,30-,36-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
J Med Chem 57: 1855-79 (2014)
Article DOI: 10.1021/jm4016894
BindingDB Entry DOI: 10.7270/Q2V989JD |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50448498

(BMS-791325 | Beclabuvir)Show SMILES COc1ccc2-c3c(C4CCCCC4)c4ccc(cc4n3C[C@]3(C[C@H]3c2c1)C(=O)N1C2CCC1CN(C)C2)C(=O)NS(=O)(=O)N(C)C |r,THB:36:35:29:31.32| Show InChI InChI=1S/C36H45N5O5S/c1-38(2)47(44,45)37-34(42)23-10-14-28-31(16-23)40-21-36(35(43)41-24-11-12-25(41)20-39(3)19-24)18-30(36)29-17-26(46-4)13-15-27(29)33(40)32(28)22-8-6-5-7-9-22/h10,13-17,22,24-25,30H,5-9,11-12,18-21H2,1-4H3,(H,37,42)/t24?,25?,30-,36-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
J Med Chem 57: 1855-79 (2014)
Article DOI: 10.1021/jm4016894
BindingDB Entry DOI: 10.7270/Q2V989JD |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50448498

(BMS-791325 | Beclabuvir)Show SMILES COc1ccc2-c3c(C4CCCCC4)c4ccc(cc4n3C[C@]3(C[C@H]3c2c1)C(=O)N1C2CCC1CN(C)C2)C(=O)NS(=O)(=O)N(C)C |r,THB:36:35:29:31.32| Show InChI InChI=1S/C36H45N5O5S/c1-38(2)47(44,45)37-34(42)23-10-14-28-31(16-23)40-21-36(35(43)41-24-11-12-25(41)20-39(3)19-24)18-30(36)29-17-26(46-4)13-15-27(29)33(40)32(28)22-8-6-5-7-9-22/h10,13-17,22,24-25,30H,5-9,11-12,18-21H2,1-4H3,(H,37,42)/t24?,25?,30-,36-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
J Med Chem 57: 1855-79 (2014)
Article DOI: 10.1021/jm4016894
BindingDB Entry DOI: 10.7270/Q2V989JD |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50565837

(CHEMBL4787795)Show SMILES [H][C@@]12C[C@]1(NC(=O)[C@]1([H])C[C@H](CN1C(=O)[C@@H](NC(=O)OC(C)(C)C(F)(F)F)[C@H](CC)C[C@H](C)CC\C=C/2)Oc1nccc2cc(OC)c(F)cc12)C(=O)NS(=O)(=O)C1(C)CC1 |r,c:37| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human CYP3A4 in human pooled liver microsomes in presence of testosterone as substrate preincubated for 0 mins in presence of NADPH fol... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01296
BindingDB Entry DOI: 10.7270/Q27D2ZWX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50448494
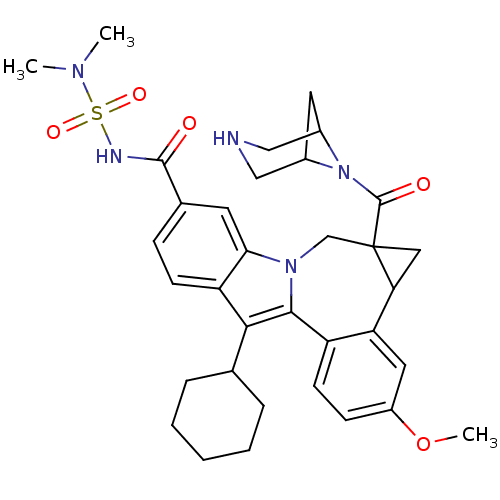
(CHEMBL3126657)Show SMILES COc1ccc2-c3c(C4CCCCC4)c4ccc(cc4n3CC3(CC3c2c1)C(=O)N1C2CC1CNC2)C(=O)NS(=O)(=O)N(C)C |TLB:27:29:35.34.33:31| Show InChI InChI=1S/C34H41N5O5S/c1-37(2)45(42,43)36-32(40)21-9-11-26-29(13-21)38-19-34(33(41)39-22-14-23(39)18-35-17-22)16-28(34)27-15-24(44-3)10-12-25(27)31(38)30(26)20-7-5-4-6-8-20/h9-13,15,20,22-23,28,35H,4-8,14,16-19H2,1-3H3,(H,36,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes after 30 mins in presence of NADPH |
J Med Chem 57: 1855-79 (2014)
Article DOI: 10.1021/jm4016894
BindingDB Entry DOI: 10.7270/Q2V989JD |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50448495
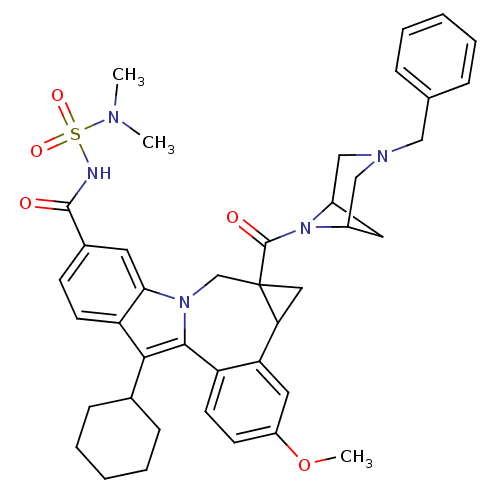
(CHEMBL3126836)Show SMILES COc1ccc2-c3c(C4CCCCC4)c4ccc(cc4n3CC3(CC3c2c1)C(=O)N1C2CC1CN(Cc1ccccc1)C2)C(=O)NS(=O)(=O)N(C)C |TLB:27:29:42.34.33:31,THB:35:34:29:31| Show InChI InChI=1S/C41H47N5O5S/c1-43(2)52(49,50)42-39(47)28-14-16-33-36(18-28)45-25-41(40(48)46-29-19-30(46)24-44(23-29)22-26-10-6-4-7-11-26)21-35(41)34-20-31(51-3)15-17-32(34)38(45)37(33)27-12-8-5-9-13-27/h4,6-7,10-11,14-18,20,27,29-30,35H,5,8-9,12-13,19,21-25H2,1-3H3,(H,42,47) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >1.30E+4 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 in human liver microsomes after 30 mins in presence of NADPH |
J Med Chem 57: 1855-79 (2014)
Article DOI: 10.1021/jm4016894
BindingDB Entry DOI: 10.7270/Q2V989JD |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50448496
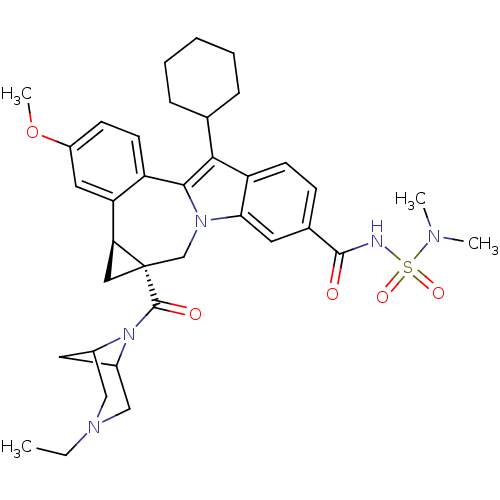
(CHEMBL3126835)Show SMILES CCN1CC2CC(C1)N2C(=O)[C@]12C[C@H]1c1cc(OC)ccc1-c1c(C3CCCCC3)c3ccc(cc3n1C2)C(=O)NS(=O)(=O)N(C)C |r,TLB:9:8:3.2.7:5,1:2:8:5| Show InChI InChI=1S/C36H45N5O5S/c1-5-39-19-24-16-25(20-39)41(24)35(43)36-18-30(36)29-17-26(46-4)12-14-27(29)33-32(22-9-7-6-8-10-22)28-13-11-23(15-31(28)40(33)21-36)34(42)37-47(44,45)38(2)3/h11-15,17,22,24-25,30H,5-10,16,18-21H2,1-4H3,(H,37,42)/t24?,25?,30-,36-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 in human liver microsomes after 30 mins in presence of NADPH |
J Med Chem 57: 1855-79 (2014)
Article DOI: 10.1021/jm4016894
BindingDB Entry DOI: 10.7270/Q2V989JD |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50448497
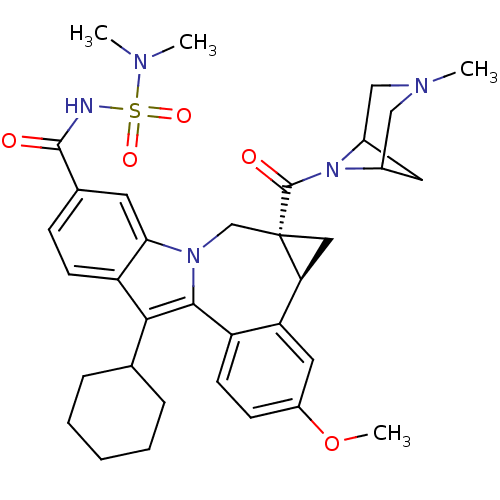
(CHEMBL3126834)Show SMILES COc1ccc2-c3c(C4CCCCC4)c4ccc(cc4n3C[C@]3(C[C@H]3c2c1)C(=O)N1C2CC1CN(C)C2)C(=O)NS(=O)(=O)N(C)C |r,TLB:27:29:36.34.33:31,THB:35:34:29:31| Show InChI InChI=1S/C35H43N5O5S/c1-37(2)46(43,44)36-33(41)22-10-12-27-30(14-22)39-20-35(34(42)40-23-15-24(40)19-38(3)18-23)17-29(35)28-16-25(45-4)11-13-26(28)32(39)31(27)21-8-6-5-7-9-21/h10-14,16,21,23-24,29H,5-9,15,17-20H2,1-4H3,(H,36,41)/t23?,24?,29-,35-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 in human liver microsomes after 30 mins in presence of NADPH |
J Med Chem 57: 1855-79 (2014)
Article DOI: 10.1021/jm4016894
BindingDB Entry DOI: 10.7270/Q2V989JD |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50448494

(CHEMBL3126657)Show SMILES COc1ccc2-c3c(C4CCCCC4)c4ccc(cc4n3CC3(CC3c2c1)C(=O)N1C2CC1CNC2)C(=O)NS(=O)(=O)N(C)C |TLB:27:29:35.34.33:31| Show InChI InChI=1S/C34H41N5O5S/c1-37(2)45(42,43)36-32(40)21-9-11-26-29(13-21)38-19-34(33(41)39-22-14-23(39)18-35-17-22)16-28(34)27-15-24(44-3)10-12-25(27)31(38)30(26)20-7-5-4-6-8-20/h9-13,15,20,22-23,28,35H,4-8,14,16-19H2,1-3H3,(H,36,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 in human liver microsomes after 30 mins in presence of NADPH |
J Med Chem 57: 1855-79 (2014)
Article DOI: 10.1021/jm4016894
BindingDB Entry DOI: 10.7270/Q2V989JD |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50448495

(CHEMBL3126836)Show SMILES COc1ccc2-c3c(C4CCCCC4)c4ccc(cc4n3CC3(CC3c2c1)C(=O)N1C2CC1CN(Cc1ccccc1)C2)C(=O)NS(=O)(=O)N(C)C |TLB:27:29:42.34.33:31,THB:35:34:29:31| Show InChI InChI=1S/C41H47N5O5S/c1-43(2)52(49,50)42-39(47)28-14-16-33-36(18-28)45-25-41(40(48)46-29-19-30(46)24-44(23-29)22-26-10-6-4-7-11-26)21-35(41)34-20-31(51-3)15-17-32(34)38(45)37(33)27-12-8-5-9-13-27/h4,6-7,10-11,14-18,20,27,29-30,35H,5,8-9,12-13,19,21-25H2,1-3H3,(H,42,47) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes after 30 mins in presence of NADPH |
J Med Chem 57: 1855-79 (2014)
Article DOI: 10.1021/jm4016894
BindingDB Entry DOI: 10.7270/Q2V989JD |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50448496

(CHEMBL3126835)Show SMILES CCN1CC2CC(C1)N2C(=O)[C@]12C[C@H]1c1cc(OC)ccc1-c1c(C3CCCCC3)c3ccc(cc3n1C2)C(=O)NS(=O)(=O)N(C)C |r,TLB:9:8:3.2.7:5,1:2:8:5| Show InChI InChI=1S/C36H45N5O5S/c1-5-39-19-24-16-25(20-39)41(24)35(43)36-18-30(36)29-17-26(46-4)12-14-27(29)33-32(22-9-7-6-8-10-22)28-13-11-23(15-31(28)40(33)21-36)34(42)37-47(44,45)38(2)3/h11-15,17,22,24-25,30H,5-10,16,18-21H2,1-4H3,(H,37,42)/t24?,25?,30-,36-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes after 30 mins in presence of NADPH |
J Med Chem 57: 1855-79 (2014)
Article DOI: 10.1021/jm4016894
BindingDB Entry DOI: 10.7270/Q2V989JD |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data