 Found 893 hits with Last Name = 'watson' and Initial = 'd'
Found 893 hits with Last Name = 'watson' and Initial = 'd' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Aromatase
(Homo sapiens (Human)) | BDBM50044424
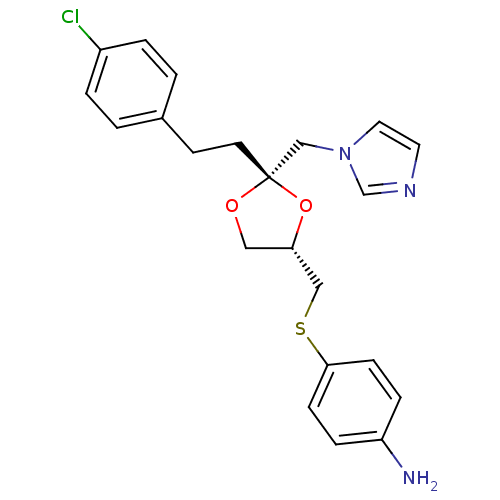
(4-(((2S,4S)-2-((1H-imidazol-1-yl)methyl)-2-(4-chlo...)Show SMILES Nc1ccc(SC[C@@H]2CO[C@](CCc3ccc(Cl)cc3)(Cn3ccnc3)O2)cc1 Show InChI InChI=1S/C22H24ClN3O2S/c23-18-3-1-17(2-4-18)9-10-22(15-26-12-11-25-16-26)27-13-20(28-22)14-29-21-7-5-19(24)6-8-21/h1-8,11-12,16,20H,9-10,13-15,24H2/t20-,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 7.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Binding affinity for Cytochrome P450 19A1 |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Lanosterol 14-alpha demethylase
(Rattus norvegicus) | BDBM50161586

(4-{(2S,4R)-2-[2-(4-Chloro-phenyl)-ethyl]-2-imidazo...)Show SMILES Nc1ccc(SC[C@H]2CO[C@](CCc3ccc(Cl)cc3)(Cn3ccnc3)O2)cc1 Show InChI InChI=1S/C22H24ClN3O2S/c23-18-3-1-17(2-4-18)9-10-22(15-26-12-11-25-16-26)27-13-20(28-22)14-29-21-7-5-19(24)6-8-21/h1-8,11-12,16,20H,9-10,13-15,24H2/t20-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Apparent Ki for rat Lanosterol 14-alpha demethylase |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM50044425
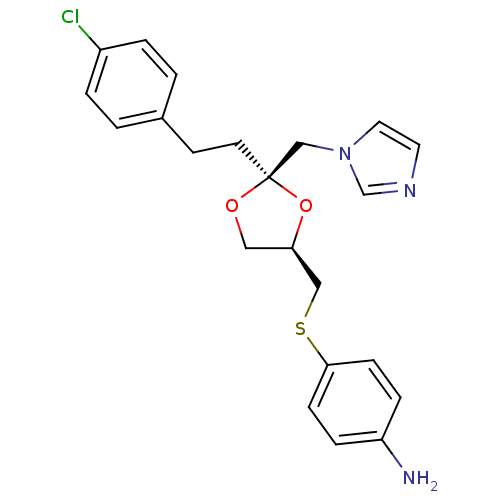
(4-{(2R,4R)-2-[2-(4-Chloro-phenyl)-ethyl]-2-imidazo...)Show SMILES Nc1ccc(SC[C@H]2CO[C@@](CCc3ccc(Cl)cc3)(Cn3ccnc3)O2)cc1 Show InChI InChI=1S/C22H24ClN3O2S/c23-18-3-1-17(2-4-18)9-10-22(15-26-12-11-25-16-26)27-13-20(28-22)14-29-21-7-5-19(24)6-8-21/h1-8,11-12,16,20H,9-10,13-15,24H2/t20-,22-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Binding affinity for Cytochrome P450 19A1 |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Cytochrome P450 11B1, mitochondrial
(Homo sapiens (Human)) | BDBM50044425

(4-{(2R,4R)-2-[2-(4-Chloro-phenyl)-ethyl]-2-imidazo...)Show SMILES Nc1ccc(SC[C@H]2CO[C@@](CCc3ccc(Cl)cc3)(Cn3ccnc3)O2)cc1 Show InChI InChI=1S/C22H24ClN3O2S/c23-18-3-1-17(2-4-18)9-10-22(15-26-12-11-25-16-26)27-13-20(28-22)14-29-21-7-5-19(24)6-8-21/h1-8,11-12,16,20H,9-10,13-15,24H2/t20-,22-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Binding affinity for corticoid 11-beta-hydroxylase |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Lanosterol 14-alpha demethylase
(Homo sapiens (Human)) | BDBM31768

(CHEMBL295698 | Ketoconazole | Nizoral | Panfungol)Show SMILES CC(=O)N1CCN(CC1)c1ccc(OC[C@@H]2CO[C@](Cn3ccnc3)(O2)c2ccc(Cl)cc2Cl)cc1 |r| Show InChI InChI=1S/C26H28Cl2N4O4/c1-19(33)31-10-12-32(13-11-31)21-3-5-22(6-4-21)34-15-23-16-35-26(36-23,17-30-9-8-29-18-30)24-7-2-20(27)14-25(24)28/h2-9,14,18,23H,10-13,15-17H2,1H3/t23-,26-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Inhibition of lanosterol 14-alpha-demethylase in hamster hepatic microsomes |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50044424

(4-(((2S,4S)-2-((1H-imidazol-1-yl)methyl)-2-(4-chlo...)Show SMILES Nc1ccc(SC[C@@H]2CO[C@](CCc3ccc(Cl)cc3)(Cn3ccnc3)O2)cc1 Show InChI InChI=1S/C22H24ClN3O2S/c23-18-3-1-17(2-4-18)9-10-22(15-26-12-11-25-16-26)27-13-20(28-22)14-29-21-7-5-19(24)6-8-21/h1-8,11-12,16,20H,9-10,13-15,24H2/t20-,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 28 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Binding affinity for progesterone 6-beta-hydroxylase of hepatic microsomes |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50044425

(4-{(2R,4R)-2-[2-(4-Chloro-phenyl)-ethyl]-2-imidazo...)Show SMILES Nc1ccc(SC[C@H]2CO[C@@](CCc3ccc(Cl)cc3)(Cn3ccnc3)O2)cc1 Show InChI InChI=1S/C22H24ClN3O2S/c23-18-3-1-17(2-4-18)9-10-22(15-26-12-11-25-16-26)27-13-20(28-22)14-29-21-7-5-19(24)6-8-21/h1-8,11-12,16,20H,9-10,13-15,24H2/t20-,22-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 33 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Binding affinity for progesterone 6-beta-hydroxylase of hepatic microsomes |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Cytochrome P450 11B1, mitochondrial
(Homo sapiens (Human)) | BDBM50044424

(4-(((2S,4S)-2-((1H-imidazol-1-yl)methyl)-2-(4-chlo...)Show SMILES Nc1ccc(SC[C@@H]2CO[C@](CCc3ccc(Cl)cc3)(Cn3ccnc3)O2)cc1 Show InChI InChI=1S/C22H24ClN3O2S/c23-18-3-1-17(2-4-18)9-10-22(15-26-12-11-25-16-26)27-13-20(28-22)14-29-21-7-5-19(24)6-8-21/h1-8,11-12,16,20H,9-10,13-15,24H2/t20-,22-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 35 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Binding affinity for corticoid 11-beta-hydroxylase |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Lanosterol 14-alpha demethylase
(Rattus norvegicus) | BDBM50044425

(4-{(2R,4R)-2-[2-(4-Chloro-phenyl)-ethyl]-2-imidazo...)Show SMILES Nc1ccc(SC[C@H]2CO[C@@](CCc3ccc(Cl)cc3)(Cn3ccnc3)O2)cc1 Show InChI InChI=1S/C22H24ClN3O2S/c23-18-3-1-17(2-4-18)9-10-22(15-26-12-11-25-16-26)27-13-20(28-22)14-29-21-7-5-19(24)6-8-21/h1-8,11-12,16,20H,9-10,13-15,24H2/t20-,22-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Inhibition of lanosterol 14-alpha-demethylase of rat hepatic microsomes |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Lanosterol 14-alpha demethylase
(Homo sapiens (Human)) | BDBM50044425

(4-{(2R,4R)-2-[2-(4-Chloro-phenyl)-ethyl]-2-imidazo...)Show SMILES Nc1ccc(SC[C@H]2CO[C@@](CCc3ccc(Cl)cc3)(Cn3ccnc3)O2)cc1 Show InChI InChI=1S/C22H24ClN3O2S/c23-18-3-1-17(2-4-18)9-10-22(15-26-12-11-25-16-26)27-13-20(28-22)14-29-21-7-5-19(24)6-8-21/h1-8,11-12,16,20H,9-10,13-15,24H2/t20-,22-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Inhibition of lanosterol 14-alpha-demethylase in hamster hepatic microsomes |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Lanosterol 14-alpha demethylase
(Homo sapiens (Human)) | BDBM50044425

(4-{(2R,4R)-2-[2-(4-Chloro-phenyl)-ethyl]-2-imidazo...)Show SMILES Nc1ccc(SC[C@H]2CO[C@@](CCc3ccc(Cl)cc3)(Cn3ccnc3)O2)cc1 Show InChI InChI=1S/C22H24ClN3O2S/c23-18-3-1-17(2-4-18)9-10-22(15-26-12-11-25-16-26)27-13-20(28-22)14-29-21-7-5-19(24)6-8-21/h1-8,11-12,16,20H,9-10,13-15,24H2/t20-,22-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 40.4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Inhibition of lanosterol 14-alpha-demethylase in hamster hepatic microsomes |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50044425

(4-{(2R,4R)-2-[2-(4-Chloro-phenyl)-ethyl]-2-imidazo...)Show SMILES Nc1ccc(SC[C@H]2CO[C@@](CCc3ccc(Cl)cc3)(Cn3ccnc3)O2)cc1 Show InChI InChI=1S/C22H24ClN3O2S/c23-18-3-1-17(2-4-18)9-10-22(15-26-12-11-25-16-26)27-13-20(28-22)14-29-21-7-5-19(24)6-8-21/h1-8,11-12,16,20H,9-10,13-15,24H2/t20-,22-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 54 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Binding affinity for progesterone 17alpha,20-lyase |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Lanosterol 14-alpha demethylase
(Homo sapiens (Human)) | BDBM31768

(CHEMBL295698 | Ketoconazole | Nizoral | Panfungol)Show SMILES CC(=O)N1CCN(CC1)c1ccc(OC[C@@H]2CO[C@](Cn3ccnc3)(O2)c2ccc(Cl)cc2Cl)cc1 |r| Show InChI InChI=1S/C26H28Cl2N4O4/c1-19(33)31-10-12-32(13-11-31)21-3-5-22(6-4-21)34-15-23-16-35-26(36-23,17-30-9-8-29-18-30)24-7-2-20(27)14-25(24)28/h2-9,14,18,23H,10-13,15-17H2,1H3/t23-,26-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 64 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Inhibition of lanosterol 14-alpha-demethylase in hamster hepatic microsomes |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Lanosterol 14-alpha demethylase
(Rattus norvegicus) | BDBM31768

(CHEMBL295698 | Ketoconazole | Nizoral | Panfungol)Show SMILES CC(=O)N1CCN(CC1)c1ccc(OC[C@@H]2CO[C@](Cn3ccnc3)(O2)c2ccc(Cl)cc2Cl)cc1 |r| Show InChI InChI=1S/C26H28Cl2N4O4/c1-19(33)31-10-12-32(13-11-31)21-3-5-22(6-4-21)34-15-23-16-35-26(36-23,17-30-9-8-29-18-30)24-7-2-20(27)14-25(24)28/h2-9,14,18,23H,10-13,15-17H2,1H3/t23-,26-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 65 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Inhibition of lanosterol 14-alpha-demethylase of rat hepatic microsomes |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50044425

(4-{(2R,4R)-2-[2-(4-Chloro-phenyl)-ethyl]-2-imidazo...)Show SMILES Nc1ccc(SC[C@H]2CO[C@@](CCc3ccc(Cl)cc3)(Cn3ccnc3)O2)cc1 Show InChI InChI=1S/C22H24ClN3O2S/c23-18-3-1-17(2-4-18)9-10-22(15-26-12-11-25-16-26)27-13-20(28-22)14-29-21-7-5-19(24)6-8-21/h1-8,11-12,16,20H,9-10,13-15,24H2/t20-,22-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 109 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Binding affinity for cholesterol 17-alpha-hydroxylase |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Lanosterol 14-alpha demethylase
(Rattus norvegicus) | BDBM31653

(CHEMBL421109 | imidazole-dioxolane, 5)Show SMILES Nc1ccc(SC[C@@H]2CO[C@@](CCc3ccc(Cl)cc3)(Cn3ccnc3)O2)cc1 |r| Show InChI InChI=1S/C22H24ClN3O2S/c23-18-3-1-17(2-4-18)9-10-22(15-26-12-11-25-16-26)27-13-20(28-22)14-29-21-7-5-19(24)6-8-21/h1-8,11-12,16,20H,9-10,13-15,24H2/t20-,22+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 117 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Apparent Ki for rat Lanosterol 14-alpha demethylase |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50044424

(4-(((2S,4S)-2-((1H-imidazol-1-yl)methyl)-2-(4-chlo...)Show SMILES Nc1ccc(SC[C@@H]2CO[C@](CCc3ccc(Cl)cc3)(Cn3ccnc3)O2)cc1 Show InChI InChI=1S/C22H24ClN3O2S/c23-18-3-1-17(2-4-18)9-10-22(15-26-12-11-25-16-26)27-13-20(28-22)14-29-21-7-5-19(24)6-8-21/h1-8,11-12,16,20H,9-10,13-15,24H2/t20-,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 447 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Binding affinity for progesterone 17-alpha,20-lyase |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Steroid 17-alpha-hydroxylase/17,20 lyase
(Homo sapiens (Human)) | BDBM50044424

(4-(((2S,4S)-2-((1H-imidazol-1-yl)methyl)-2-(4-chlo...)Show SMILES Nc1ccc(SC[C@@H]2CO[C@](CCc3ccc(Cl)cc3)(Cn3ccnc3)O2)cc1 Show InChI InChI=1S/C22H24ClN3O2S/c23-18-3-1-17(2-4-18)9-10-22(15-26-12-11-25-16-26)27-13-20(28-22)14-29-21-7-5-19(24)6-8-21/h1-8,11-12,16,20H,9-10,13-15,24H2/t20-,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.63E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Syntex Discovery Research
Curated by ChEMBL
| Assay Description
Binding affinity for cholesterol 17-alpha-hydroxylase |
J Med Chem 36: 2235-7 (1993)
BindingDB Entry DOI: 10.7270/Q2ZW1K0P |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM138200
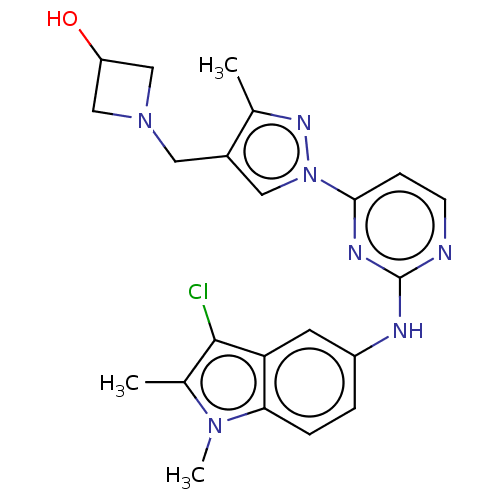
(US8871778, 141)Show SMILES Cc1nn(cc1CN1CC(O)C1)-c1ccnc(Nc2ccc3n(C)c(C)c(Cl)c3c2)n1 Show InChI InChI=1S/C22H24ClN7O/c1-13-15(9-29-11-17(31)12-29)10-30(27-13)20-6-7-24-22(26-20)25-16-4-5-19-18(8-16)21(23)14(2)28(19)3/h4-8,10,17,31H,9,11-12H2,1-3H3,(H,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant full length SYK (unknown origin) by biochemical Omnia assay |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127433
BindingDB Entry DOI: 10.7270/Q2C2511G |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50548361

(CHEMBL4744190)Show SMILES COC(=O)c1nn(C)c2c(C)cc(Nc3nccc(n3)-c3cc(CN(C)C)n(C)n3)cc12 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant full length SYK (unknown origin) by biochemical Omnia assay |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127433
BindingDB Entry DOI: 10.7270/Q2C2511G |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50548350
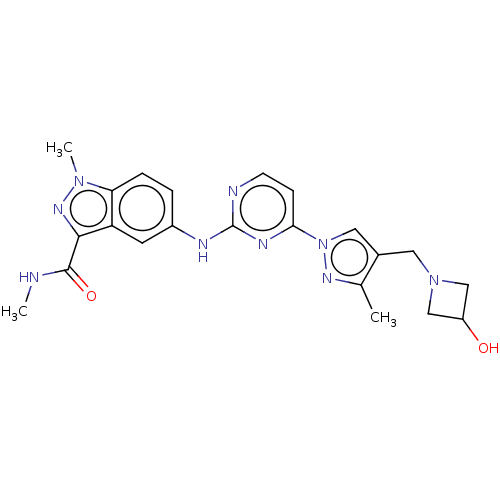
(CHEMBL4780257)Show SMILES CNC(=O)c1nn(C)c2ccc(Nc3nccc(n3)-n3cc(CN4CC(O)C4)c(C)n3)cc12 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant full length SYK (unknown origin) by biochemical Omnia assay |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127433
BindingDB Entry DOI: 10.7270/Q2C2511G |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50546397

(CHEMBL4776352)Show SMILES CN(C)Cc1cnn(c1)-c1ccnc2[nH]c(nc12)-c1cn(C)c2ccc(cc12)S(C)(=O)=O | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.0770 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of SYK (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127393
BindingDB Entry DOI: 10.7270/Q28W3HVM |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50548354
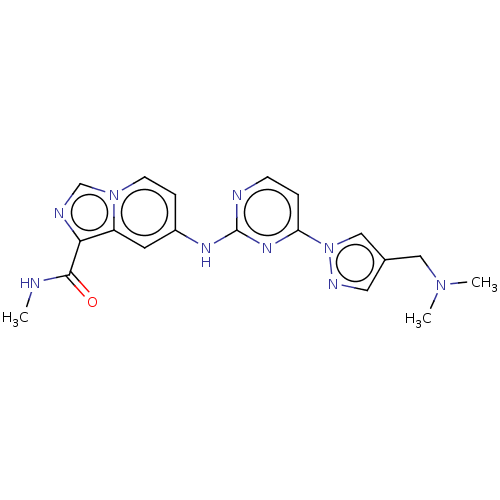
(CHEMBL4753469)Show SMILES CNC(=O)c1ncn2ccc(Nc3nccc(n3)-n3cc(CN(C)C)cn3)cc12 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant full length SYK (unknown origin) by biochemical Omnia assay |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127433
BindingDB Entry DOI: 10.7270/Q2C2511G |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50546402
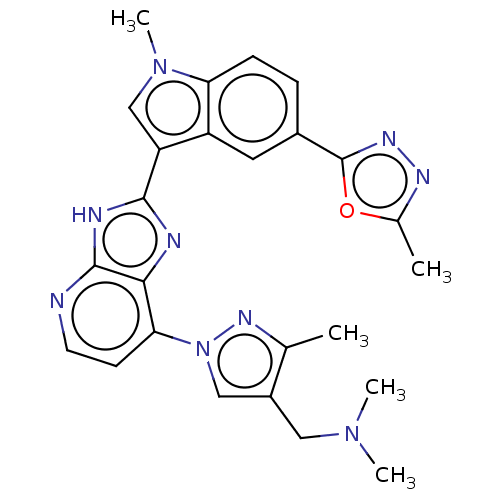
(CHEMBL4762995)Show SMILES CN(C)Cc1cn(nc1C)-c1ccnc2[nH]c(nc12)-c1cn(C)c2ccc(cc12)-c1nnc(C)o1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.0980 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of SYK (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127393
BindingDB Entry DOI: 10.7270/Q28W3HVM |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50548358

(CHEMBL4753141)Show SMILES CNC(=O)c1nn(C)c2ccc(Nc3nccc(n3)-n3cc(CN(C)C)c(OC)n3)cc12 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant full length SYK (unknown origin) by biochemical Omnia assay |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127433
BindingDB Entry DOI: 10.7270/Q2C2511G |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50548362
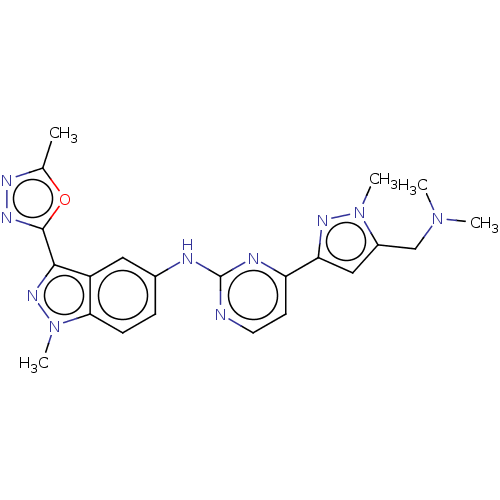
(CHEMBL4784088)Show SMILES CN(C)Cc1cc(nn1C)-c1ccnc(Nc2ccc3n(C)nc(-c4nnc(C)o4)c3c2)n1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant full length SYK (unknown origin) by biochemical Omnia assay |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127433
BindingDB Entry DOI: 10.7270/Q2C2511G |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50548357
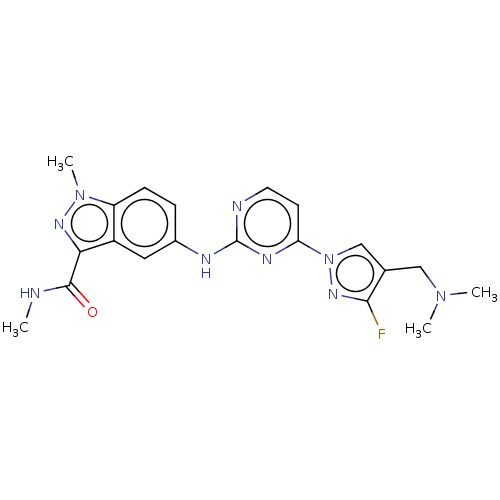
(CHEMBL4752008)Show SMILES CNC(=O)c1nn(C)c2ccc(Nc3nccc(n3)-n3cc(CN(C)C)c(F)n3)cc12 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant full length SYK (unknown origin) by biochemical Omnia assay |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127433
BindingDB Entry DOI: 10.7270/Q2C2511G |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50546398

(CHEMBL4754597)Show SMILES CNC(=O)c1ccc2n(C)cc(-c3nc4c(ccnc4[nH]3)-n3cc(CN(C)C)cn3)c2c1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.150 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of SYK (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127393
BindingDB Entry DOI: 10.7270/Q28W3HVM |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50546401
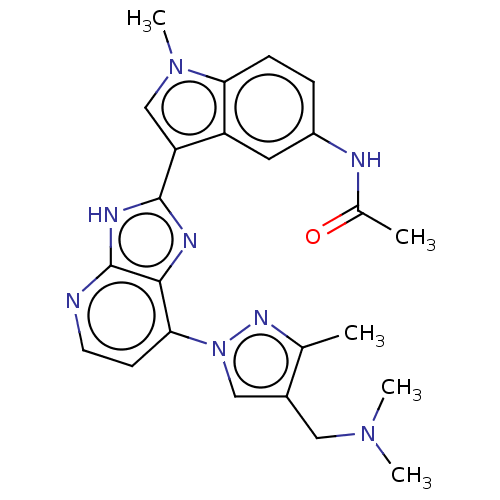
(CHEMBL4776842)Show SMILES CN(C)Cc1cn(nc1C)-c1ccnc2[nH]c(nc12)-c1cn(C)c2ccc(NC(C)=O)cc12 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.170 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of SYK (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127393
BindingDB Entry DOI: 10.7270/Q28W3HVM |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50546400
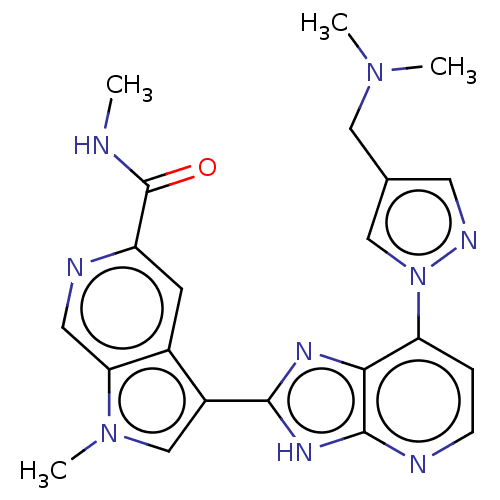
(CHEMBL4752127)Show SMILES CNC(=O)c1cc2c(cn(C)c2cn1)-c1nc2c(ccnc2[nH]1)-n1cc(CN(C)C)cn1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of SYK (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127393
BindingDB Entry DOI: 10.7270/Q28W3HVM |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50548363
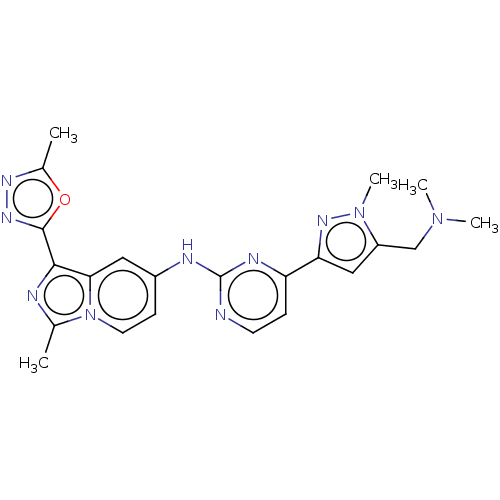
(CHEMBL4753164)Show SMILES CN(C)Cc1cc(nn1C)-c1ccnc(Nc2ccn3c(C)nc(-c4nnc(C)o4)c3c2)n1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant full length SYK (unknown origin) by biochemical Omnia assay |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127433
BindingDB Entry DOI: 10.7270/Q2C2511G |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50548356
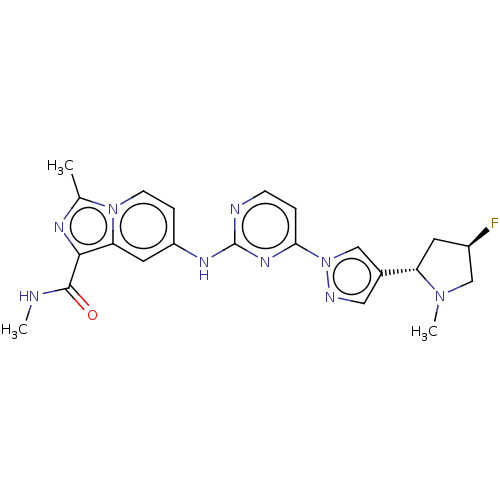
(CHEMBL4792651)Show SMILES CNC(=O)c1nc(C)n2ccc(Nc3nccc(n3)-n3cc(cn3)[C@@H]3C[C@@H](F)CN3C)cc12 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant full length SYK (unknown origin) by biochemical Omnia assay |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127433
BindingDB Entry DOI: 10.7270/Q2C2511G |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50548351

(CHEMBL4786316)Show SMILES CNC(=O)c1nn(C)c2ccc(Nc3nccc(n3)-n3cc(CN(C)C)cn3)cc12 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.210 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant full length SYK (unknown origin) by biochemical Omnia assay |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127433
BindingDB Entry DOI: 10.7270/Q2C2511G |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50546394

(CHEMBL4759636)Show SMILES CN(C)Cc1cc(nn1C)-c1ccnc(Nc2ccn3cnc(-c4nnc(C)o4)c3c2)n1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.210 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of SYK (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127393
BindingDB Entry DOI: 10.7270/Q28W3HVM |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50546407

(CHEMBL4740065)Show SMILES Cn1ccc2c(cccc12)-c1nc2c(ccnc2[nH]1)-n1cc(N[C@@H]2CCOC[C@@H]2N)cn1 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 0.210 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of SYK (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127393
BindingDB Entry DOI: 10.7270/Q28W3HVM |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50548353

(CHEMBL4762290)Show SMILES CNC(=O)c1nn(C)c2c(C)cc(Nc3nccc(n3)-n3cc(CN(C)C)cn3)cc12 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.220 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant full length SYK (unknown origin) by biochemical Omnia assay |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127433
BindingDB Entry DOI: 10.7270/Q2C2511G |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50548355
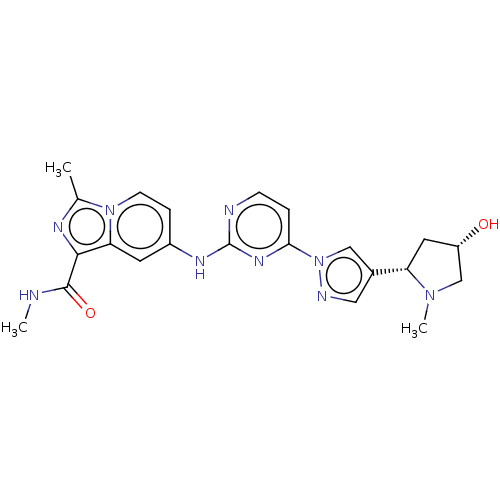
(CHEMBL4784783)Show SMILES CNC(=O)c1nc(C)n2ccc(Nc3nccc(n3)-n3cc(cn3)[C@@H]3C[C@H](O)CN3C)cc12 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.240 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant full length SYK (unknown origin) by biochemical Omnia assay |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127433
BindingDB Entry DOI: 10.7270/Q2C2511G |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50548360

(CHEMBL4790107)Show SMILES CNC(=O)c1nn(C)c2c(C)cc(Nc3nccc(n3)-c3cc(CN(C)C)n(C)n3)cc12 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant full length SYK (unknown origin) by biochemical Omnia assay |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127433
BindingDB Entry DOI: 10.7270/Q2C2511G |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50546393

(CHEMBL4751083)Show SMILES CNC(=O)c1nn(C)c2ccc(Nc3nccc(n3)-n3cc(CN(C)C)c(C)n3)cc12 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.310 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant full length SYK (unknown origin) by biochemical Omnia assay |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127433
BindingDB Entry DOI: 10.7270/Q2C2511G |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50546393

(CHEMBL4751083)Show SMILES CNC(=O)c1nn(C)c2ccc(Nc3nccc(n3)-n3cc(CN(C)C)c(C)n3)cc12 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.310 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of SYK (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127393
BindingDB Entry DOI: 10.7270/Q28W3HVM |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50546403

(CHEMBL4779316)Show SMILES Cn1ccc2c(cccc12)-c1nc2c(ccnc2[nH]1)-n1cc(CN2CC[C@@H](O)C2)cn1 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.480 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of SYK (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.bmcl.2020.127393
BindingDB Entry DOI: 10.7270/Q28W3HVM |
More data for this
Ligand-Target Pair | |
E3 ubiquitin-protein ligase Mdm2
(Homo sapiens (Human)) | BDBM427143

(1-({[(1R)-2-[(4-chloro-2-methanesulfonylphenyl)met...)Show SMILES C[C@@](O)(C1CCOCC1)c1cc2C(=O)N(Cc3ccc(Cl)cc3S(C)(=O)=O)[C@](OCC3(CC3)C(N)=O)(c2c(F)c1)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C34H35Cl2FN2O7S/c1-32(42,21-9-13-45-14-10-21)23-15-26-29(27(37)16-23)34(22-4-7-24(35)8-5-22,46-19-33(11-12-33)31(38)41)39(30(26)40)18-20-3-6-25(36)17-28(20)47(2,43)44/h3-8,15-17,21,42H,9-14,18-19H2,1-2H3,(H2,38,41)/t32-,34-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a |
ASTEX THERAPEUTICS LIMITED; CANCER RESEARCH TECHNOLOGY LIMITED
US Patent
| Assay Description
The ELISA assay was performed in streptavidin coated plates which were preincubated with 200 μl per well of 1 μg ml−1 biotinylated IP... |
US Patent US10544132 (2020)
BindingDB Entry DOI: 10.7270/Q27H1N06 |
More data for this
Ligand-Target Pair | |
Cellular tumor antigen p53 [1-83]/E3 ubiquitin-protein ligase Mdm2 [1-188]
(Homo sapiens (Human)) | BDBM427143

(1-({[(1R)-2-[(4-chloro-2-methanesulfonylphenyl)met...)Show SMILES C[C@@](O)(C1CCOCC1)c1cc2C(=O)N(Cc3ccc(Cl)cc3S(C)(=O)=O)[C@](OCC3(CC3)C(N)=O)(c2c(F)c1)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C34H35Cl2FN2O7S/c1-32(42,21-9-13-45-14-10-21)23-15-26-29(27(37)16-23)34(22-4-7-24(35)8-5-22,46-19-33(11-12-33)31(38)41)39(30(26)40)18-20-3-6-25(36)17-28(20)47(2,43)44/h3-8,15-17,21,42H,9-14,18-19H2,1-2H3,(H2,38,41)/t32-,34-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a |
ASTEX THERAPEUTICS LIMITED; CANCER RESEARCH TECHNOLOGY LIMITED
US Patent
| Assay Description
The ELISA assay was performed in streptavidin coated plates which were preincubated with 200 μl per well of 1 μg ml−1 biotinylated IP... |
US Patent US10981898 (2021)
BindingDB Entry DOI: 10.7270/Q2154M5V |
More data for this
Ligand-Target Pair | |
E3 ubiquitin-protein ligase Mdm2
(Homo sapiens (Human)) | BDBM427127

(1-({[(1R)-2-[(4-chloro-2-methanesulfonylphenyl)met...)Show SMILES Cn1cc(cn1)[C@](C)(O)c1cc2C(=O)N(Cc3ccc(Cl)cc3S(C)(=O)=O)[C@](OCC3(CC3)C(N)=O)(c2c(F)c1)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C33H31Cl2FN4O6S/c1-31(43,22-15-38-39(2)17-22)21-12-25-28(26(36)13-21)33(20-5-8-23(34)9-6-20,46-18-32(10-11-32)30(37)42)40(29(25)41)16-19-4-7-24(35)14-27(19)47(3,44)45/h4-9,12-15,17,43H,10-11,16,18H2,1-3H3,(H2,37,42)/t31-,33-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
ASTEX THERAPEUTICS LIMITED; CANCER RESEARCH TECHNOLOGY LIMITED
US Patent
| Assay Description
The ELISA assay was performed in streptavidin coated plates which were preincubated with 200 μl per well of 1 μg ml−1 biotinylated IP... |
US Patent US10544132 (2020)
BindingDB Entry DOI: 10.7270/Q27H1N06 |
More data for this
Ligand-Target Pair | |
Cellular tumor antigen p53 [1-83]/E3 ubiquitin-protein ligase Mdm2 [1-188]
(Homo sapiens (Human)) | BDBM427127

(1-({[(1R)-2-[(4-chloro-2-methanesulfonylphenyl)met...)Show SMILES Cn1cc(cn1)[C@](C)(O)c1cc2C(=O)N(Cc3ccc(Cl)cc3S(C)(=O)=O)[C@](OCC3(CC3)C(N)=O)(c2c(F)c1)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C33H31Cl2FN4O6S/c1-31(43,22-15-38-39(2)17-22)21-12-25-28(26(36)13-21)33(20-5-8-23(34)9-6-20,46-18-32(10-11-32)30(37)42)40(29(25)41)16-19-4-7-24(35)14-27(19)47(3,44)45/h4-9,12-15,17,43H,10-11,16,18H2,1-3H3,(H2,37,42)/t31-,33-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
ASTEX THERAPEUTICS LIMITED; CANCER RESEARCH TECHNOLOGY LIMITED
US Patent
| Assay Description
The ELISA assay was performed in streptavidin coated plates which were preincubated with 200 μl per well of 1 μg ml−1 biotinylated IP... |
US Patent US10981898 (2021)
BindingDB Entry DOI: 10.7270/Q2154M5V |
More data for this
Ligand-Target Pair | |
Cellular tumor antigen p53 [1-83]/E3 ubiquitin-protein ligase Mdm2 [1-188]
(Homo sapiens (Human)) | BDBM427195
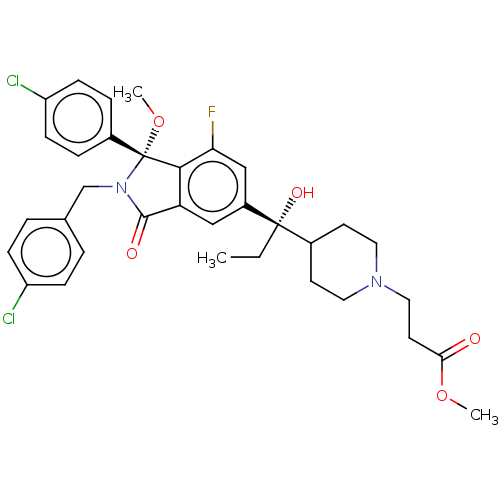
(US10544132, Example 136 | US10981898, Example 137 ...)Show SMILES CC[C@](O)(C1CCN(CCC(=O)OC)CC1)c1cc2C(=O)N(Cc3ccc(Cl)cc3)[C@](OC)(c2c(F)c1)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C34H37Cl2FN2O5/c1-4-33(42,23-13-16-38(17-14-23)18-15-30(40)43-2)25-19-28-31(29(37)20-25)34(44-3,24-7-11-27(36)12-8-24)39(32(28)41)21-22-5-9-26(35)10-6-22/h5-12,19-20,23,42H,4,13-18,21H2,1-3H3/t33-,34+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.570 | n/a | n/a | n/a | n/a | n/a | n/a |
ASTEX THERAPEUTICS LIMITED; CANCER RESEARCH TECHNOLOGY LIMITED
US Patent
| Assay Description
The ELISA assay was performed in streptavidin coated plates which were preincubated with 200 μl per well of 1 μg ml−1 biotinylated IP... |
US Patent US10981898 (2021)
BindingDB Entry DOI: 10.7270/Q2154M5V |
More data for this
Ligand-Target Pair | |
E3 ubiquitin-protein ligase Mdm2
(Homo sapiens (Human)) | BDBM427196
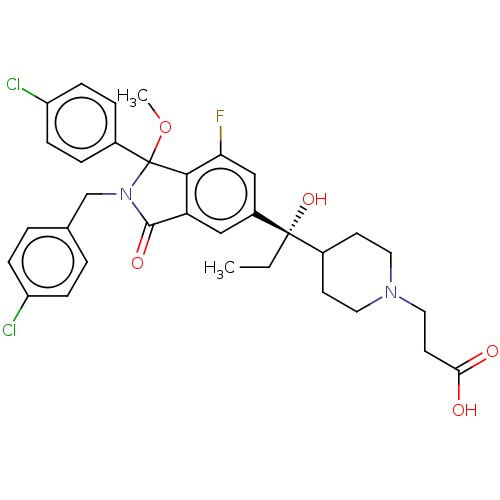
(US10544132, Example 137)Show SMILES CC[C@](O)(C1CCN(CCC(O)=O)CC1)c1cc2C(=O)N(Cc3ccc(Cl)cc3)C(OC)(c2c(F)c1)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C33H35Cl2FN2O5/c1-3-32(42,22-12-15-37(16-13-22)17-14-29(39)40)24-18-27-30(28(36)19-24)33(43-2,23-6-10-26(35)11-7-23)38(31(27)41)20-21-4-8-25(34)9-5-21/h4-11,18-19,22,42H,3,12-17,20H2,1-2H3,(H,39,40)/t32-,33?/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.570 | n/a | n/a | n/a | n/a | n/a | n/a |
ASTEX THERAPEUTICS LIMITED; CANCER RESEARCH TECHNOLOGY LIMITED
US Patent
| Assay Description
The ELISA assay was performed in streptavidin coated plates which were preincubated with 200 μl per well of 1 μg ml−1 biotinylated IP... |
US Patent US10544132 (2020)
BindingDB Entry DOI: 10.7270/Q27H1N06 |
More data for this
Ligand-Target Pair | |
E3 ubiquitin-protein ligase Mdm2
(Homo sapiens (Human)) | BDBM427156
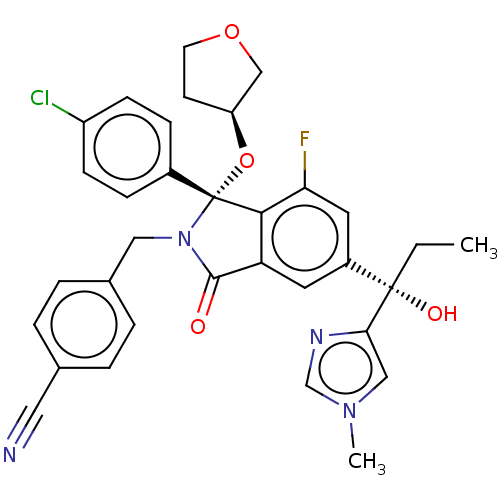
(4-{[(1R)-1-(4-chlorophenyl)-7-fluoro-5-[I-hydroxy-...)Show SMILES CC[C@@](O)(c1cn(C)cn1)c1cc2C(=O)N(Cc3ccc(cc3)C#N)[C@](O[C@H]3CCOC3)(c2c(F)c1)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C33H30ClFN4O4/c1-3-32(41,29-18-38(2)20-37-29)24-14-27-30(28(35)15-24)33(43-26-12-13-42-19-26,23-8-10-25(34)11-9-23)39(31(27)40)17-22-6-4-21(16-36)5-7-22/h4-11,14-15,18,20,26,41H,3,12-13,17,19H2,1-2H3/t26-,32-,33+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.640 | n/a | n/a | n/a | n/a | n/a | n/a |
ASTEX THERAPEUTICS LIMITED; CANCER RESEARCH TECHNOLOGY LIMITED
US Patent
| Assay Description
The ELISA assay was performed in streptavidin coated plates which were preincubated with 200 μl per well of 1 μg ml−1 biotinylated IP... |
US Patent US10544132 (2020)
BindingDB Entry DOI: 10.7270/Q27H1N06 |
More data for this
Ligand-Target Pair | |
Cellular tumor antigen p53 [1-83]/E3 ubiquitin-protein ligase Mdm2 [1-188]
(Homo sapiens (Human)) | BDBM427156

(4-{[(1R)-1-(4-chlorophenyl)-7-fluoro-5-[I-hydroxy-...)Show SMILES CC[C@@](O)(c1cn(C)cn1)c1cc2C(=O)N(Cc3ccc(cc3)C#N)[C@](O[C@H]3CCOC3)(c2c(F)c1)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C33H30ClFN4O4/c1-3-32(41,29-18-38(2)20-37-29)24-14-27-30(28(35)15-24)33(43-26-12-13-42-19-26,23-8-10-25(34)11-9-23)39(31(27)40)17-22-6-4-21(16-36)5-7-22/h4-11,14-15,18,20,26,41H,3,12-13,17,19H2,1-2H3/t26-,32-,33+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.640 | n/a | n/a | n/a | n/a | n/a | n/a |
ASTEX THERAPEUTICS LIMITED; CANCER RESEARCH TECHNOLOGY LIMITED
US Patent
| Assay Description
The ELISA assay was performed in streptavidin coated plates which were preincubated with 200 μl per well of 1 μg ml−1 biotinylated IP... |
US Patent US10981898 (2021)
BindingDB Entry DOI: 10.7270/Q2154M5V |
More data for this
Ligand-Target Pair | |
E3 ubiquitin-protein ligase Mdm2
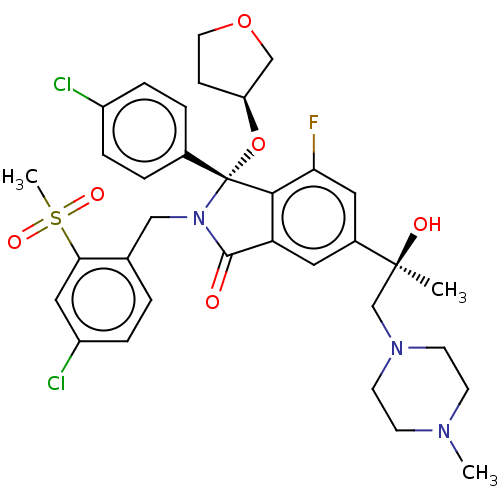
(Homo sapiens (Human)) | BDBM427122
(US10544132, Example 69 | US10981898, Example 70)Show SMILES CN1CCN(C[C@](C)(O)c2cc3C(=O)N(Cc4ccc(Cl)cc4S(C)(=O)=O)[C@](O[C@H]4CCOC4)(c3c(F)c2)c2ccc(Cl)cc2)CC1 |r| Show InChI InChI=1S/C34H38Cl2FN3O6S/c1-33(42,21-39-13-11-38(2)12-14-39)24-16-28-31(29(37)17-24)34(46-27-10-15-45-20-27,23-5-8-25(35)9-6-23)40(32(28)41)19-22-4-7-26(36)18-30(22)47(3,43)44/h4-9,16-18,27,42H,10-15,19-21H2,1-3H3/t27-,33-,34+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
ASTEX THERAPEUTICS LIMITED; CANCER RESEARCH TECHNOLOGY LIMITED
US Patent
| Assay Description
The ELISA assay was performed in streptavidin coated plates which were preincubated with 200 μl per well of 1 μg ml−1 biotinylated IP... |
US Patent US10544132 (2020)
BindingDB Entry DOI: 10.7270/Q27H1N06 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data