

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383315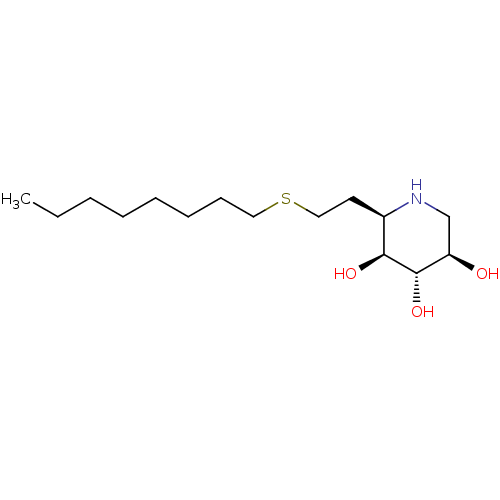 (CHEMBL2029773) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 4-methylumbelliferyl beta-D-glucopyranoside as substrate after 15 mins by Dixon an... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383327 (CHEMBL2029772) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 4-methylumbelliferyl beta-D-glucopyranoside as substrate after 15 mins by Dixon an... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383321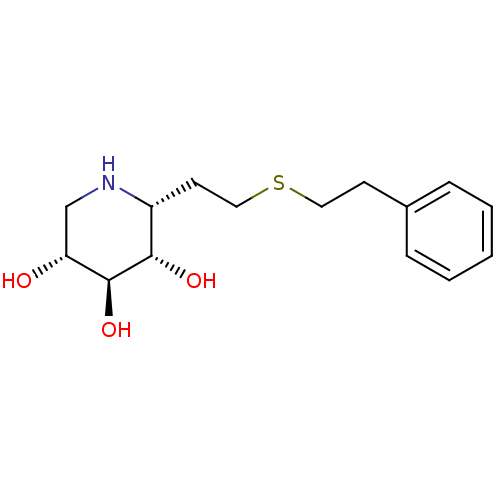 (CHEMBL2029780) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 3 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 4-methylumbelliferyl beta-D-glucopyranoside as substrate after 15 mins by Dixon an... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383323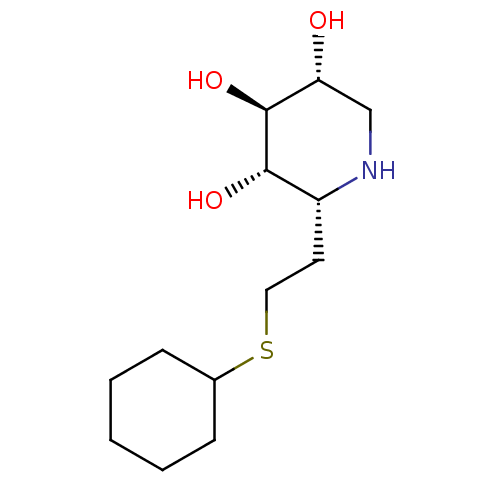 (CHEMBL2029778) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 4 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 4-methylumbelliferyl beta-D-glucopyranoside as substrate after 15 mins by Dixon an... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383325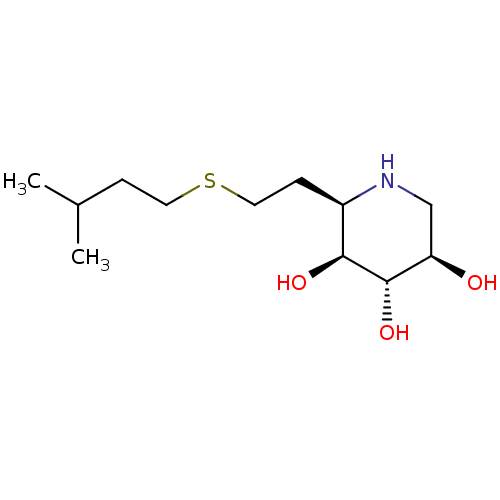 (CHEMBL2029776) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 4.40 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 4-methylumbelliferyl beta-D-glucopyranoside as substrate after 15 mins by Dixon an... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383324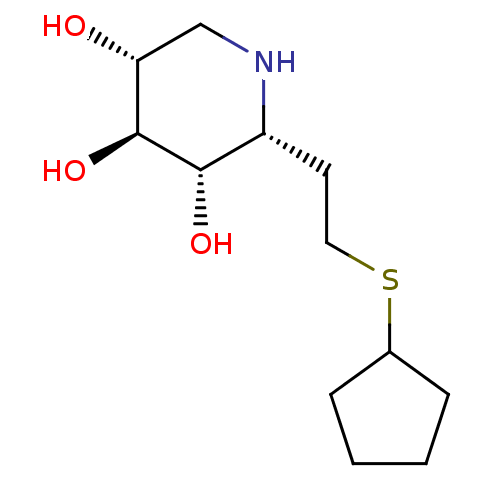 (CHEMBL2029777) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 4-methylumbelliferyl beta-D-glucopyranoside as substrate after 15 mins by Dixon an... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383319 (CHEMBL2029782) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 12 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 4-methylumbelliferyl beta-D-glucopyranoside as substrate after 15 mins by Dixon an... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383328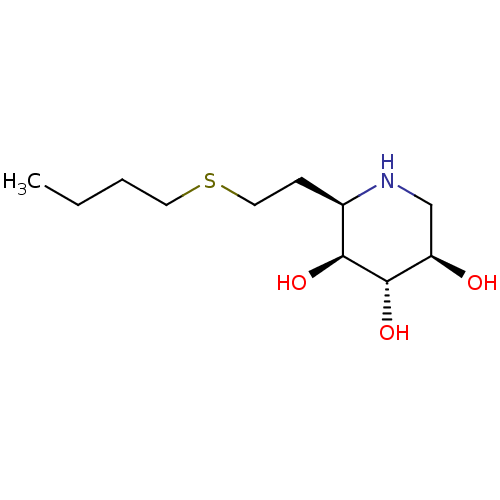 (CHEMBL2029771) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 18 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 4-methylumbelliferyl beta-D-glucopyranoside as substrate after 15 mins by Dixon an... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383322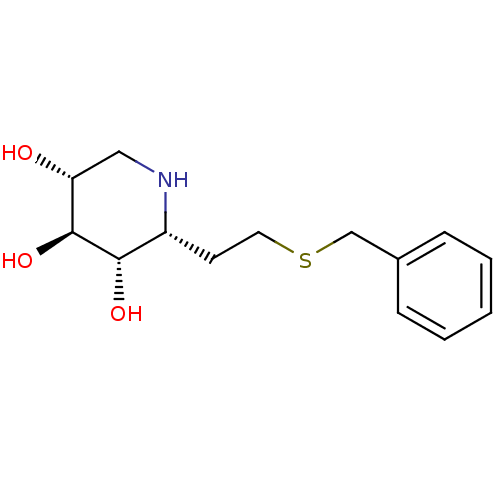 (CHEMBL2029779) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 19 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 4-methylumbelliferyl beta-D-glucopyranoside as substrate after 15 mins by Dixon an... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383326 (CHEMBL2029775) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 29 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 2,4-dinitrophenyl beta-D-glucopyranoside as substrate after 3 mins by Dixon and Li... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383318 (CHEMBL2029783) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 30 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 2,4-dinitrophenyl beta-D-glucopyranoside as substrate after 3 mins by Dixon and Li... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383314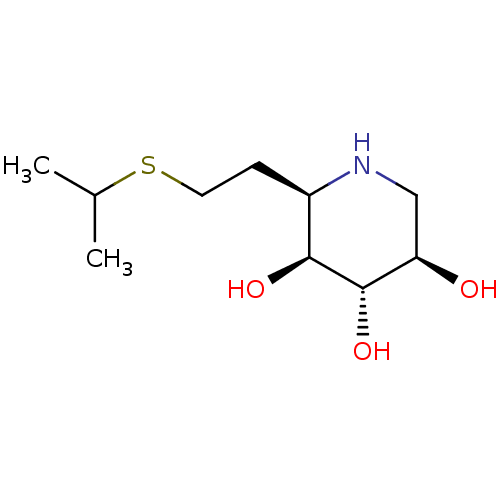 (CHEMBL2029774) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 76 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 2,4-dinitrophenyl beta-D-glucopyranoside as substrate after 3 mins by Dixon and Li... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383329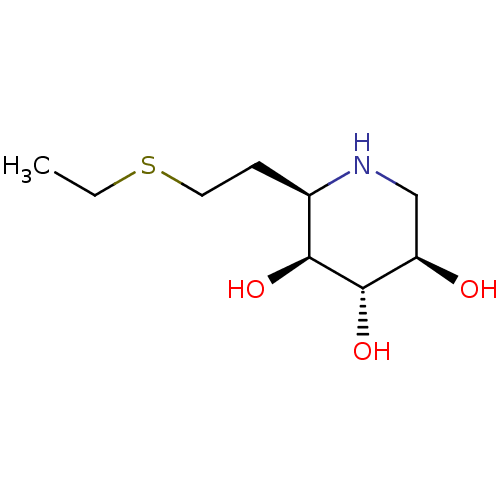 (CHEMBL2029770) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 180 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 2,4-dinitrophenyl beta-D-glucopyranoside as substrate after 3 mins by Dixon and Li... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383320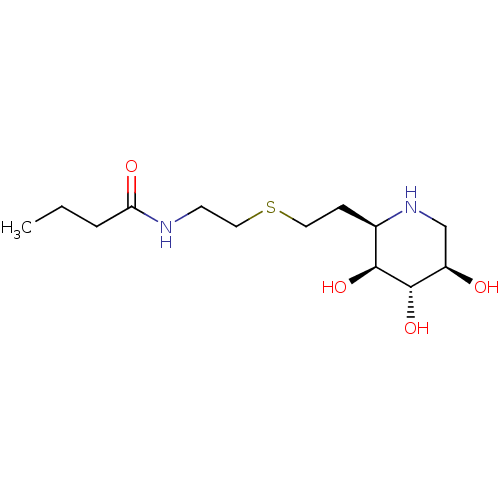 (CHEMBL2029781) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 200 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 2,4-dinitrophenyl beta-D-glucopyranoside as substrate after 3 mins by Dixon and Li... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383317 (CHEMBL2029784) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 250 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 2,4-dinitrophenyl beta-D-glucopyranoside as substrate after 3 mins by Dixon and Li... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383316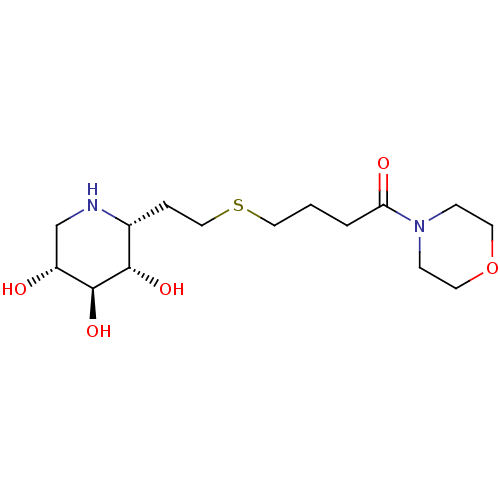 (CHEMBL2029785) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 450 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 2,4-dinitrophenyl beta-D-glucopyranoside as substrate after 3 mins by Dixon and Li... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383330 (CHEMBL2029769) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 2,4-dinitrophenyl beta-D-glucopyranoside as substrate after 3 mins by Dixon and Li... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50383331 (CHEMBL2029768) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 2,4-dinitrophenyl beta-D-glucopyranoside as substrate after 3 mins by Dixon and Li... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal acid glucosylceramidase (Homo sapiens (Human)) | BDBM50182798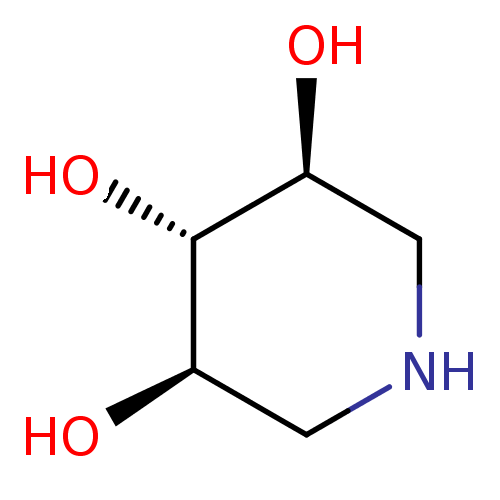 ((3R,4r,5S)-piperidine-3,4,5-triol | 1,5-Dideoxy-1,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 5.0 | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Competitive inhibition of wild type human glucocerebrosidase using 2,4-dinitrophenyl beta-D-glucopyranoside as substrate after 3 mins by Dixon and Li... | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytosolic beta-glucosidase (Homo sapiens (Human)) | BDBM50383315 (CHEMBL2029773) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of British Columbia Curated by ChEMBL | Assay Description Inhibition of human cytosolic beta-glucosidase using 4-methylumbelliferyl beta-D-glucopyranoside as substrate after 15 mins by fluorimetric analysis | J Med Chem 55: 2737-45 (2012) Article DOI: 10.1021/jm201633y BindingDB Entry DOI: 10.7270/Q2639QSX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM18512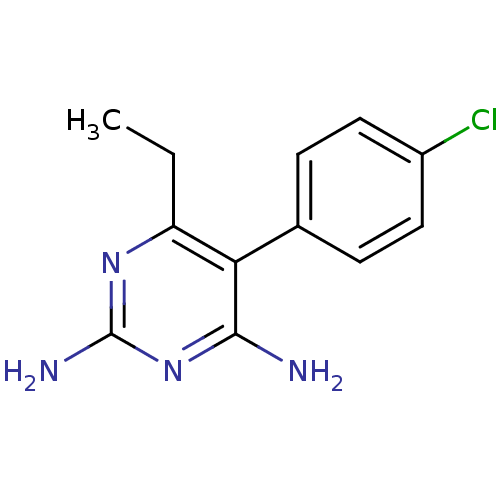 (5-(4-chlorophenyl)-6-ethylpyrimidine-2,4-diamine |...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 3.10E+3 | n/a | n/a | n/a | n/a | 7.0 | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using MUGS substrate incubated for 1 to 2 hrs at pH 7.0 by spectrofluorometry | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit beta (Homo sapiens (Human)) | BDBM18512 (5-(4-chlorophenyl)-6-ethylpyrimidine-2,4-diamine |...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 4.50E+3 | n/a | n/a | n/a | n/a | 7.0 | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexB using MUGS substrate incubated for 1 to 2 hrs at pH 7.0 by spectrofluorometry | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM18512 (5-(4-chlorophenyl)-6-ethylpyrimidine-2,4-diamine |...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 8.50E+3 | n/a | n/a | n/a | n/a | 4.5 | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of HexA alpha G269S mutant in ATSD patient fibroblasts using MUGS substrate incubated for 1 to 2 hrs at pH 4.5 by spectrofluorometry | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM18512 (5-(4-chlorophenyl)-6-ethylpyrimidine-2,4-diamine |...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 8.90E+3 | n/a | n/a | n/a | n/a | 4.5 | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using MUGS substrate incubated for 1 to 2 hrs at pH 4.5 by spectrofluorometry | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit beta (Homo sapiens (Human)) | BDBM18512 (5-(4-chlorophenyl)-6-ethylpyrimidine-2,4-diamine |...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 9.10E+3 | n/a | n/a | n/a | n/a | 4.5 | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexB using MUGS substrate incubated for 1 to 2 hrs at pH 4.5 by spectrofluorometry | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM18788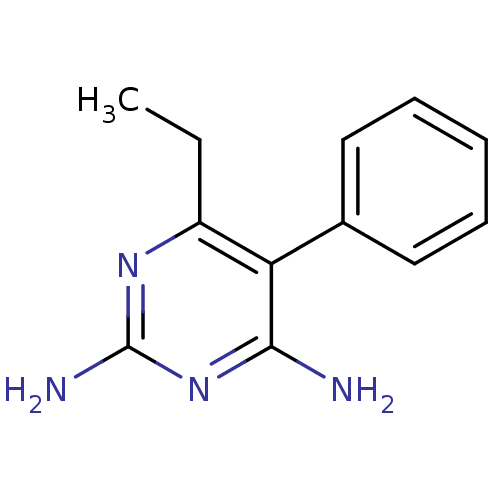 (6-ethyl-5-phenylpyrimidine-2,4-diamine | CHEMBL221...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | 7.0 | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using MUGS substrate incubated for 1 to 2 hrs at pH 7.0 by spectrofluorometry | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM18512 (5-(4-chlorophenyl)-6-ethylpyrimidine-2,4-diamine |...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.40E+4 | n/a | n/a | n/a | n/a | 4.5 | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of HexA in normal human fibroblasts using MUGS substrate incubated for 1 to 2 hrs at pH 4.5 by spectrofluorometry | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM50089680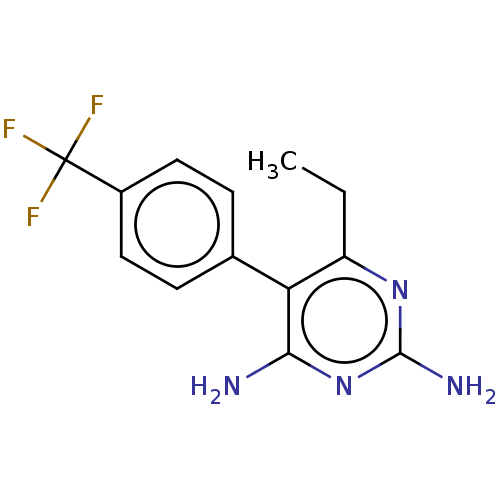 (CHEMBL3577321) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using pNPGlcNAc substrate | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha/beta (Homo sapiens (Human)) | BDBM18512 (5-(4-chlorophenyl)-6-ethylpyrimidine-2,4-diamine |...) | PDB UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of beta-N-acetylhexosaminidase (unknown origin) | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit beta (Homo sapiens (Human)) | BDBM18788 (6-ethyl-5-phenylpyrimidine-2,4-diamine | CHEMBL221...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.50E+4 | n/a | n/a | n/a | n/a | 7.0 | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexB using MUGS substrate incubated for 1 to 2 hrs at pH 7.0 by spectrofluorometry | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM18512 (5-(4-chlorophenyl)-6-ethylpyrimidine-2,4-diamine |...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using pNPGlcNAc substrate | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM50089676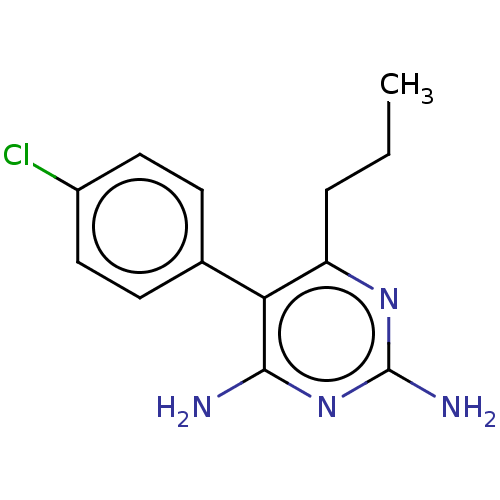 (CHEMBL3577317) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using pNPGlcNAc substrate | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM18780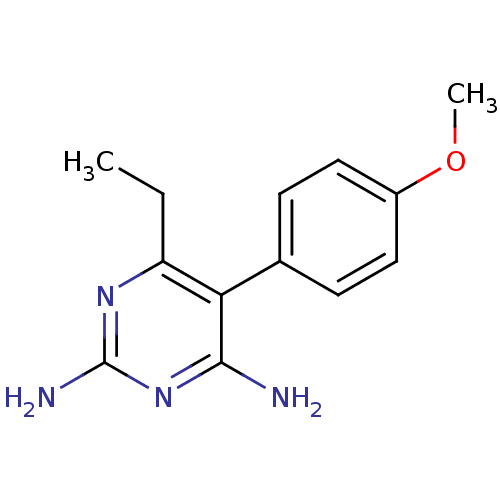 (6-ethyl-5-(4-methoxyphenyl)pyrimidine-2,4-diamine ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using pNPGlcNAc substrate | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM18788 (6-ethyl-5-phenylpyrimidine-2,4-diamine | CHEMBL221...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 2.70E+4 | n/a | n/a | n/a | n/a | 4.5 | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of HexA alpha G269S mutant in ATSD patient fibroblasts using MUGS substrate incubated for 1 to 2 hrs at pH 4.5 by spectrofluorometry | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM18788 (6-ethyl-5-phenylpyrimidine-2,4-diamine | CHEMBL221...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 2.70E+4 | n/a | n/a | n/a | n/a | 4.5 | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of HexA in normal human fibroblasts using MUGS substrate incubated for 1 to 2 hrs at pH 4.5 by spectrofluorometry | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM18788 (6-ethyl-5-phenylpyrimidine-2,4-diamine | CHEMBL221...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 3.00E+4 | n/a | n/a | n/a | n/a | 4.5 | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using MUGS substrate incubated for 1 to 2 hrs at pH 4.5 by spectrofluorometry | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM18788 (6-ethyl-5-phenylpyrimidine-2,4-diamine | CHEMBL221...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 3.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using pNPGlcNAc substrate | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM50089681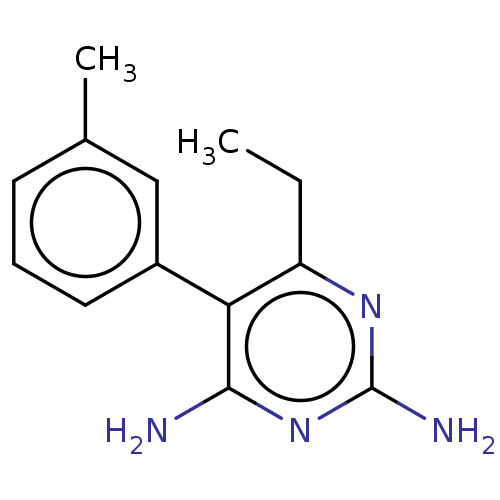 (CHEMBL3577322) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using pNPGlcNAc substrate | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit beta (Homo sapiens (Human)) | BDBM18788 (6-ethyl-5-phenylpyrimidine-2,4-diamine | CHEMBL221...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 3.80E+4 | n/a | n/a | n/a | n/a | 4.5 | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexB using MUGS substrate incubated for 1 to 2 hrs at pH 4.5 by spectrofluorometry | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM18779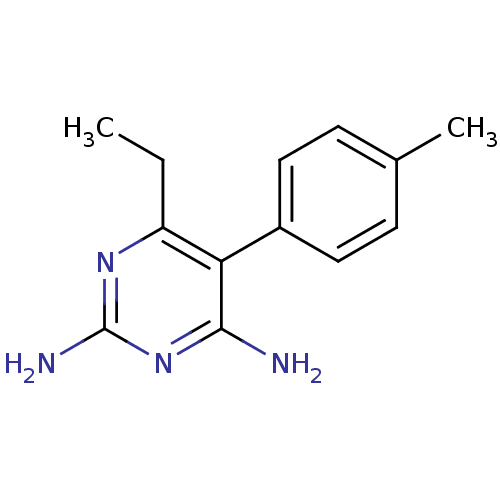 (6-ethyl-5-(4-methylphenyl)pyrimidine-2,4-diamine |...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using pNPGlcNAc substrate | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM50089682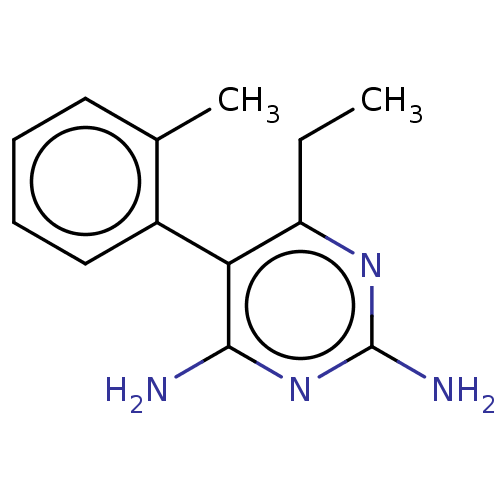 (CHEMBL3577323) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using pNPGlcNAc substrate | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM50089683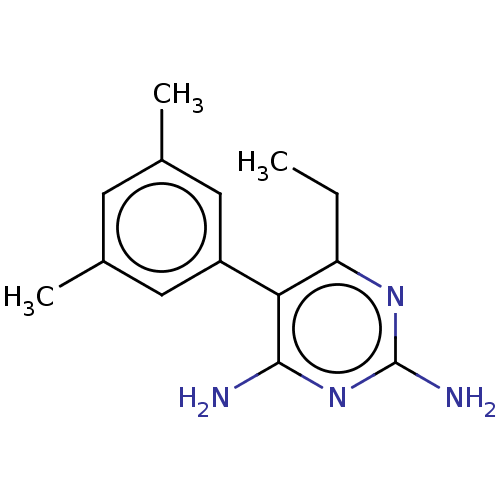 (CHEMBL3577324) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using pNPGlcNAc substrate | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM50089679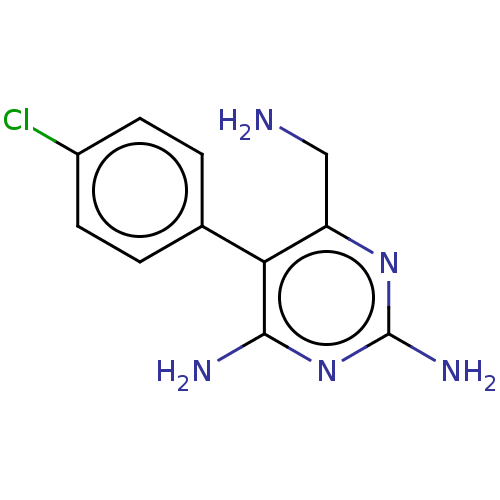 (CHEMBL3577320) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.80E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using pNPGlcNAc substrate | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM50089674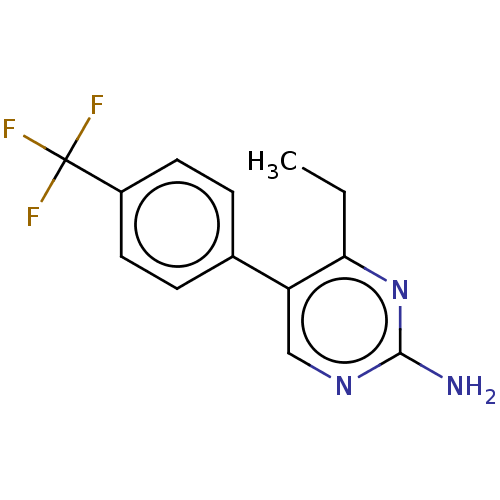 (CHEMBL3577315) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >4.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using pNPGlcNAc substrate | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM50089673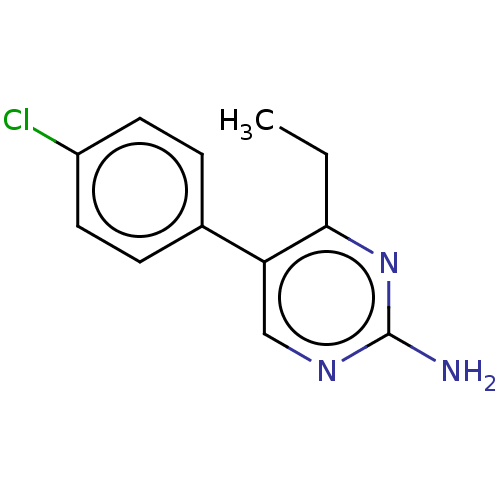 (CHEMBL3577314) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >4.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using pNPGlcNAc substrate | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM50089677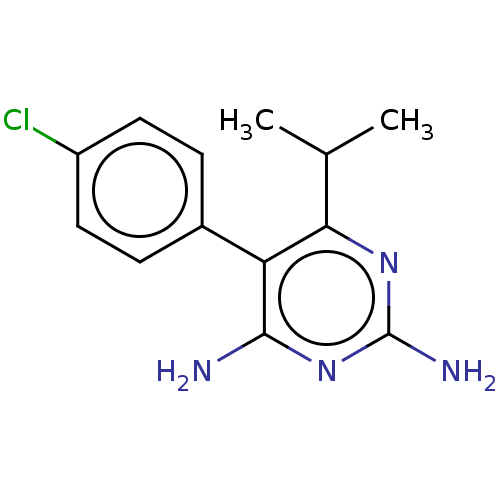 (CHEMBL3577318) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >4.10E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using pNPGlcNAc substrate | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM18775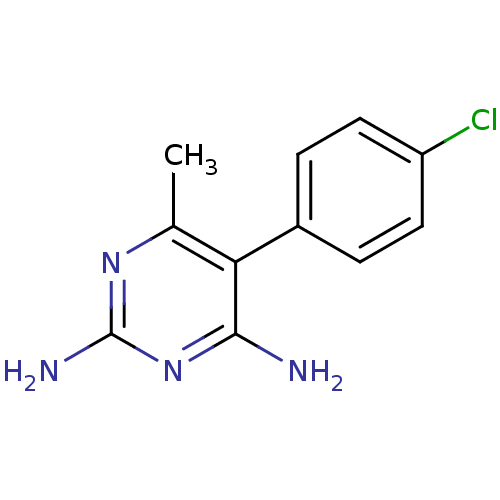 (5-(4-chlorophenyl)-6-methylpyrimidine-2,4-diamine ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >5.60E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using pNPGlcNAc substrate | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM50089675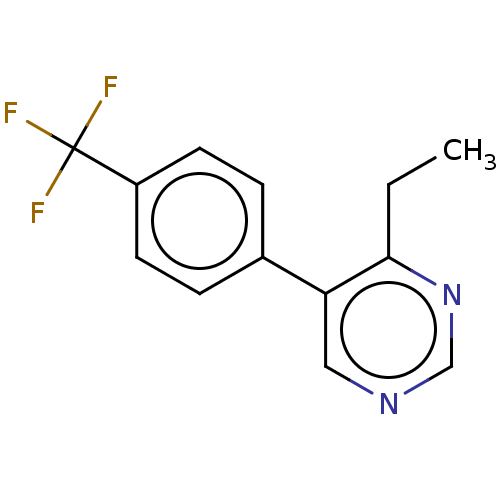 (CHEMBL3577316) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >9.40E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using pNPGlcNAc substrate | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM50089678 (CHEMBL3577319) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.40E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using pNPGlcNAc substrate | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-hexosaminidase subunit alpha (Homo sapiens (Human)) | BDBM50089672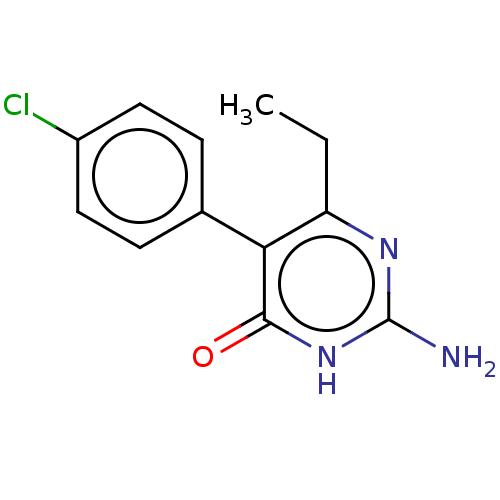 (CHEMBL3577313) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >2.10E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Genetics and Genome Biology, SickKids, PGCRL 686 Bay Street, Toronto, Ontario M5G 0A4, Canada. Curated by ChEMBL | Assay Description Inhibition of human placental HexA using pNPGlcNAc substrate | J Med Chem 58: 4483-93 (2015) Article DOI: 10.1021/jm5017895 BindingDB Entry DOI: 10.7270/Q28054CK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||