 Found 15619 hits with Last Name = 'zhan' and Initial = 'c'
Found 15619 hits with Last Name = 'zhan' and Initial = 'c' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Prothrombin
(Homo sapiens (Human)) | BDBM14073
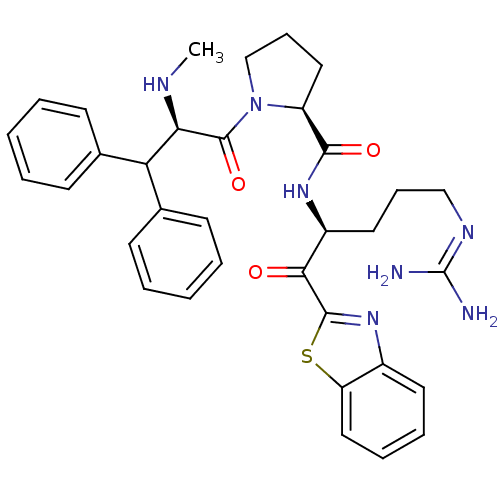
((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)Show SMILES [#6]-[#7]-[#6@H](-[#6](-c1ccccc1)-c1ccccc1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-c1nc2ccccc2s1 |r| Show InChI InChI=1S/C34H39N7O3S/c1-37-29(28(22-12-4-2-5-13-22)23-14-6-3-7-15-23)33(44)41-21-11-18-26(41)31(43)39-25(17-10-20-38-34(35)36)30(42)32-40-24-16-8-9-19-27(24)45-32/h2-9,12-16,19,25-26,28-29,37H,10-11,17-18,20-21H2,1H3,(H,39,43)(H4,35,36,38)/t25-,26-,29+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.000650 | -72.4 | 4.5 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Johnson & Johnson Pharmaceutical
| Assay Description
Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a... |
J Med Chem 48: 1984-2008 (2005)
Article DOI: 10.1021/jm0303857
BindingDB Entry DOI: 10.7270/Q2X0658X |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM14065
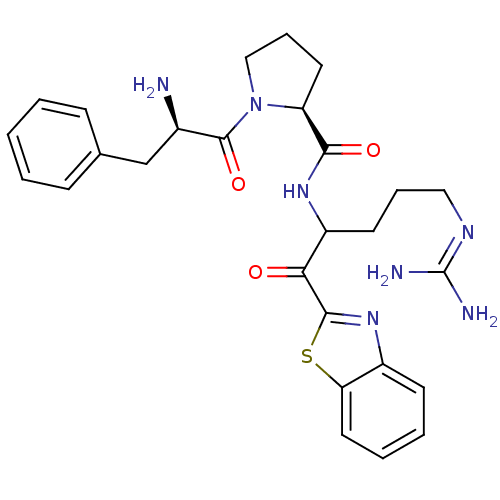
((2S)-1-[(2R)-2-amino-3-phenylpropanoyl]-N-[1-(1,3-...)Show SMILES [#7]-[#6@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-c1nc2ccccc2s1 |r| Show InChI InChI=1S/C27H33N7O3S/c28-18(16-17-8-2-1-3-9-17)26(37)34-15-7-12-21(34)24(36)32-20(11-6-14-31-27(29)30)23(35)25-33-19-10-4-5-13-22(19)38-25/h1-5,8-10,13,18,20-21H,6-7,11-12,14-16,28H2,(H,32,36)(H4,29,30,31)/t18-,20?,21+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00550 | -66.9 | 21 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Johnson & Johnson Pharmaceutical
| Assay Description
Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a... |
J Med Chem 48: 1984-2008 (2005)
Article DOI: 10.1021/jm0303857
BindingDB Entry DOI: 10.7270/Q2X0658X |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM14127
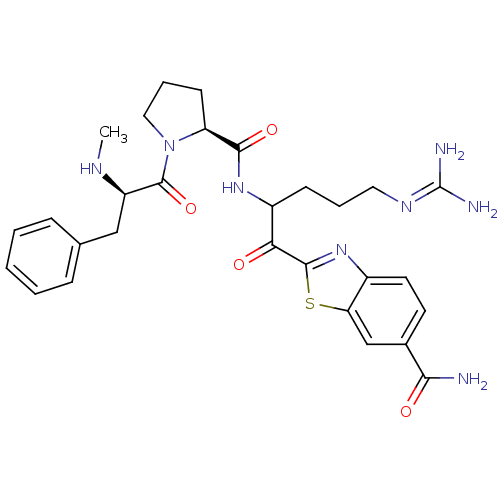
(2-(5-carbamimidamido-2-{[(2S)-1-[(2R)-2-(methylami...)Show SMILES [#6]-[#7]-[#6@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-c1nc2ccc(cc2s1)-[#6](-[#7])=O |r| Show InChI InChI=1S/C29H36N8O4S/c1-33-21(15-17-7-3-2-4-8-17)28(41)37-14-6-10-22(37)26(40)35-20(9-5-13-34-29(31)32)24(38)27-36-19-12-11-18(25(30)39)16-23(19)42-27/h2-4,7-8,11-12,16,20-22,33H,5-6,9-10,13-15H2,1H3,(H2,30,39)(H,35,40)(H4,31,32,34)/t20?,21-,22+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00700 | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical
| Assay Description
Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a... |
J Med Chem 48: 1984-2008 (2005)
Article DOI: 10.1021/jm0303857
BindingDB Entry DOI: 10.7270/Q2X0658X |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM14076
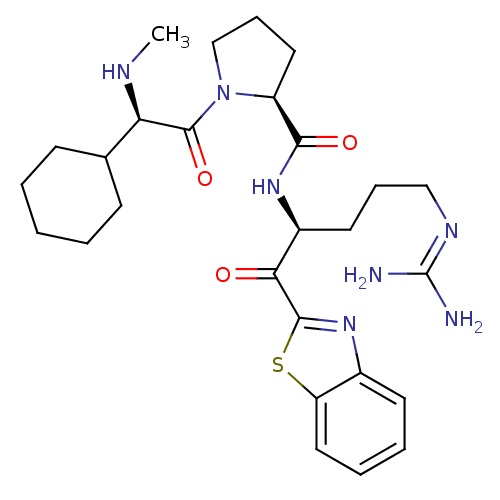
((2S)-N-[(2S)-1-(1,3-benzothiazol-2-yl)-5-carbamimi...)Show SMILES [#6]-[#7]-[#6@H](-[#6]-1-[#6]-[#6]-[#6]-[#6]-[#6]-1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-c1nc2ccccc2s1 |r| Show InChI InChI=1S/C27H39N7O3S/c1-30-22(17-9-3-2-4-10-17)26(37)34-16-8-13-20(34)24(36)32-19(12-7-15-31-27(28)29)23(35)25-33-18-11-5-6-14-21(18)38-25/h5-6,11,14,17,19-20,22,30H,2-4,7-10,12-13,15-16H2,1H3,(H,32,36)(H4,28,29,31)/t19-,20-,22+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0180 | -63.8 | 5.30 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Johnson & Johnson Pharmaceutical
| Assay Description
Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a... |
J Med Chem 48: 1984-2008 (2005)
Article DOI: 10.1021/jm0303857
BindingDB Entry DOI: 10.7270/Q2X0658X |
More data for this
Ligand-Target Pair | |
Nociceptin receptor
(Homo sapiens (Human)) | BDBM50121249

(CHEMBL415845 | F-G-G-F-T-G-A-R-K-S-A-R-K-L-Aib-N-Q...)Show SMILES CC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)CNC(=O)[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccccc1)[C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(N)=O Show InChI InChI=1S/C80H132N28O21/c1-7-49(69(120)106-57(37-60(85)112)75(126)100-50(65(86)116)28-29-59(84)111)99-74(125)55(34-42(2)3)105-72(123)51(24-14-16-30-81)103-71(122)54(27-19-33-92-80(89)90)102-67(118)44(5)97-77(128)58(41-109)107-73(124)52(25-15-17-31-82)104-70(121)53(26-18-32-91-79(87)88)101-66(117)43(4)96-62(114)40-95-78(129)64(45(6)110)108-76(127)56(36-47-22-12-9-13-23-47)98-63(115)39-93-61(113)38-94-68(119)48(83)35-46-20-10-8-11-21-46/h8-13,20-23,42-45,48-58,64,109-110H,7,14-19,24-41,81-83H2,1-6H3,(H2,84,111)(H2,85,112)(H2,86,116)(H,93,113)(H,94,119)(H,95,129)(H,96,114)(H,97,128)(H,98,115)(H,99,125)(H,100,126)(H,101,117)(H,102,118)(H,103,122)(H,104,121)(H,105,123)(H,106,120)(H,107,124)(H,108,127)(H4,87,88,91)(H4,89,90,92)/t43-,44-,45+,48-,49-,50-,51-,52-,53-,54-,55-,56-,57-,58-,64-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue Pharma L. P.
Curated by ChEMBL
| Assay Description
Affinity for human Opioid receptor like 1 (ORL-1) expressed in HEK293 cells |
J Med Chem 45: 5280-6 (2002)
BindingDB Entry DOI: 10.7270/Q2474BKJ |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM370133
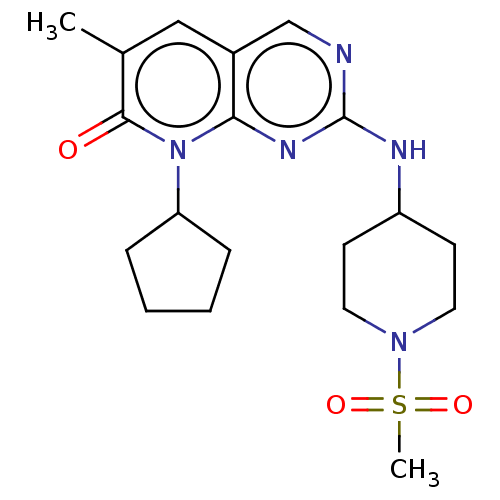
(US10233188, Example 22 | US10800783, Example 22 | ...)Show SMILES Cc1cc2cnc(NC3CCN(CC3)S(C)(=O)=O)nc2n(C2CCCC2)c1=O Show InChI InChI=1S/C19H27N5O3S/c1-13-11-14-12-20-19(21-15-7-9-23(10-8-15)28(2,26)27)22-17(14)24(18(13)25)16-5-3-4-6-16/h11-12,15-16H,3-10H2,1-2H3,(H,20,21,22) | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.0470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Nociceptin receptor
(Homo sapiens (Human)) | BDBM50121245
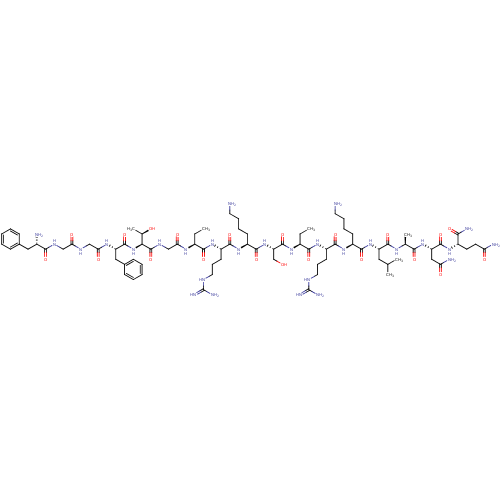
(CHEMBL266191 | F-G-G-F-T-G-Aib-R-K-S-Aib-R-K-L-A-N...)Show SMILES CC[C@H](NC(=O)CNC(=O)[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccccc1)[C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(N)=O Show InChI InChI=1S/C81H134N28O21/c1-7-49(98-64(116)41-96-79(130)65(45(6)111)109-77(128)57(37-47-23-13-10-14-24-47)99-63(115)40-94-62(114)39-95-68(119)48(84)36-46-21-11-9-12-22-46)69(120)102-54(27-19-33-92-80(88)89)71(122)105-53(26-16-18-32-83)74(125)108-59(42-110)78(129)100-50(8-2)70(121)103-55(28-20-34-93-81(90)91)72(123)104-52(25-15-17-31-82)73(124)107-56(35-43(3)4)75(126)97-44(5)67(118)106-58(38-61(86)113)76(127)101-51(66(87)117)29-30-60(85)112/h9-14,21-24,43-45,48-59,65,110-111H,7-8,15-20,25-42,82-84H2,1-6H3,(H2,85,112)(H2,86,113)(H2,87,117)(H,94,114)(H,95,119)(H,96,130)(H,97,126)(H,98,116)(H,99,115)(H,100,129)(H,101,127)(H,102,120)(H,103,121)(H,104,123)(H,105,122)(H,106,118)(H,107,124)(H,108,125)(H,109,128)(H4,88,89,92)(H4,90,91,93)/t44-,45+,48-,49-,50-,51-,52-,53-,54-,55-,56-,57-,58-,59-,65-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue Pharma L. P.
Curated by ChEMBL
| Assay Description
Affinity for human Opioid receptor like 1 (ORL-1) expressed in HEK293 cells |
J Med Chem 45: 5280-6 (2002)
BindingDB Entry DOI: 10.7270/Q2474BKJ |
More data for this
Ligand-Target Pair | |
Nociceptin receptor
(Homo sapiens (Human)) | BDBM50121247
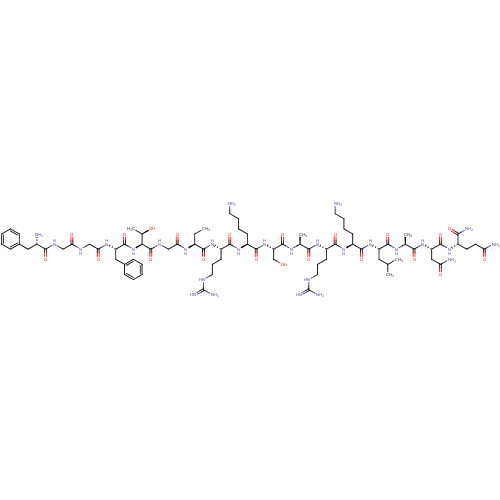
(CHEMBL414542 | F-G-G-F-T-G-Aib-R-K-S-A-R-K-L-A-N-Q...)Show SMILES CC[C@H](NC(=O)CNC(=O)[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccccc1)[C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(N)=O Show InChI InChI=1S/C80H132N28O21/c1-7-49(98-63(115)40-95-78(129)64(45(6)110)108-76(127)56(36-47-22-12-9-13-23-47)99-62(114)39-93-61(113)38-94-68(119)48(83)35-46-20-10-8-11-21-46)69(120)102-54(27-19-33-92-80(89)90)71(122)104-52(25-15-17-31-82)73(124)107-58(41-109)77(128)97-43(4)66(117)101-53(26-18-32-91-79(87)88)70(121)103-51(24-14-16-30-81)72(123)106-55(34-42(2)3)74(125)96-44(5)67(118)105-57(37-60(85)112)75(126)100-50(65(86)116)28-29-59(84)111/h8-13,20-23,42-45,48-58,64,109-110H,7,14-19,24-41,81-83H2,1-6H3,(H2,84,111)(H2,85,112)(H2,86,116)(H,93,113)(H,94,119)(H,95,129)(H,96,125)(H,97,128)(H,98,115)(H,99,114)(H,100,126)(H,101,117)(H,102,120)(H,103,121)(H,104,122)(H,105,118)(H,106,123)(H,107,124)(H,108,127)(H4,87,88,91)(H4,89,90,92)/t43-,44-,45+,48-,49-,50-,51-,52-,53-,54-,55-,56-,57-,58-,64-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue Pharma L. P.
Curated by ChEMBL
| Assay Description
Affinity for human Opioid receptor like 1 (ORL-1) expressed in HEK293 cells |
J Med Chem 45: 5280-6 (2002)
BindingDB Entry DOI: 10.7270/Q2474BKJ |
More data for this
Ligand-Target Pair | |
Nociceptin receptor
(Homo sapiens (Human)) | BDBM50121246
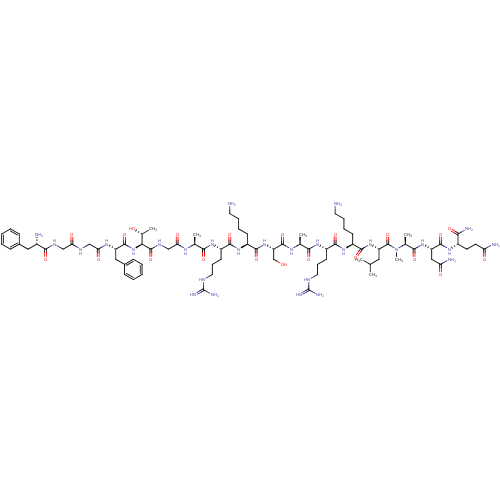
(CHEMBL438537 | F-G-G-F-T-G-A-R-K-S-A-R-K-L-MeA-N-Q...)Show SMILES CC(C)C[C@H](NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)CNC(=O)[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccccc1)[C@@H](C)O)C(=O)N(C)[C@@H](C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(N)=O Show InChI InChI=1S/C80H132N28O21/c1-42(2)34-57(78(129)108(7)45(5)68(119)104-56(37-60(85)112)74(125)99-50(65(86)116)28-29-59(84)111)105-72(123)51(24-14-16-30-81)102-71(122)54(27-19-33-92-80(89)90)101-67(118)44(4)97-76(127)58(41-109)106-73(124)52(25-15-17-31-82)103-70(121)53(26-18-32-91-79(87)88)100-66(117)43(3)96-62(114)40-95-77(128)64(46(6)110)107-75(126)55(36-48-22-12-9-13-23-48)98-63(115)39-93-61(113)38-94-69(120)49(83)35-47-20-10-8-11-21-47/h8-13,20-23,42-46,49-58,64,109-110H,14-19,24-41,81-83H2,1-7H3,(H2,84,111)(H2,85,112)(H2,86,116)(H,93,113)(H,94,120)(H,95,128)(H,96,114)(H,97,127)(H,98,115)(H,99,125)(H,100,117)(H,101,118)(H,102,122)(H,103,121)(H,104,119)(H,105,123)(H,106,124)(H,107,126)(H4,87,88,91)(H4,89,90,92)/t43-,44-,45-,46+,49-,50-,51-,52-,53-,54-,55-,56-,57-,58-,64-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue Pharma L. P.
Curated by ChEMBL
| Assay Description
Affinity for human Opioid receptor like 1 (ORL-1) expressed in HEK293 cells |
J Med Chem 45: 5280-6 (2002)
BindingDB Entry DOI: 10.7270/Q2474BKJ |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM467013
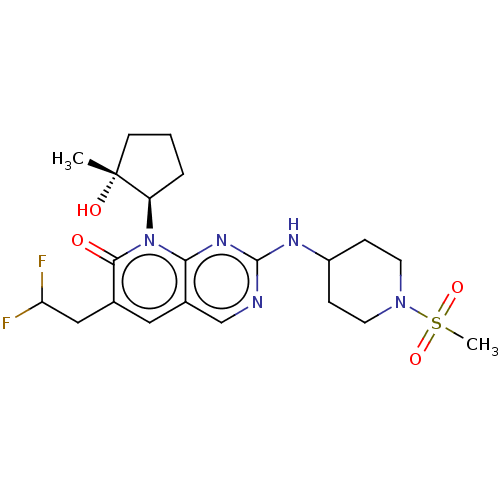
((-)-6-(2,2-difluoroethyl)-8-[(1R*,2R*)-2-hydroxy-2...)Show SMILES C[C@@]1(O)CCC[C@H]1n1c2nc(NC3CCN(CC3)S(C)(=O)=O)ncc2cc(CC(F)F)c1=O |r| Show InChI InChI=1S/C21H29F2N5O4S/c1-21(30)7-3-4-16(21)28-18-14(10-13(19(28)29)11-17(22)23)12-24-20(26-18)25-15-5-8-27(9-6-15)33(2,31)32/h10,12,15-17,30H,3-9,11H2,1-2H3,(H,24,25,26)/t16-,21-/m1/s1 | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.0640 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50049757

(()-2-(6-Chloro-pyridin-3-yl)-7-aza-bicyclo[2.2.1]h...)Show InChI InChI=1S/C11H13ClN2/c12-11-4-1-7(6-13-11)9-5-8-2-3-10(9)14-8/h1,4,6,8-10,14H,2-3,5H2 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.0790 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of New Orleans
Curated by ChEMBL
| Assay Description
In vitro binding affinity by inhibiting [3H]cytisine binding in rat brain tissue at Nicotinic acetylcholine receptor alpha4-beta2 |
J Med Chem 45: 3041-7 (2002)
Article DOI: 10.1021/jm0103561
BindingDB Entry DOI: 10.7270/Q2GQ71H0 |
More data for this
Ligand-Target Pair | |
Nociceptin receptor
(Homo sapiens (Human)) | BDBM50121253
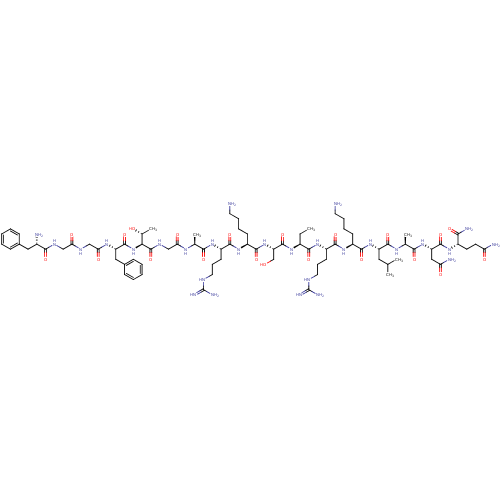
(CHEMBL408356 | F-G-G-F-T-G-A-R-K-S-Aib-R-K-L-A-N-Q...)Show SMILES CC[C@H](NC(=O)[C@H](CO)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)CNC(=O)[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccccc1)[C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(N)=O Show InChI InChI=1S/C80H132N28O21/c1-7-49(69(120)102-54(27-19-33-92-80(89)90)71(122)103-51(24-14-16-30-81)72(123)106-55(34-42(2)3)74(125)97-44(5)67(118)105-57(37-60(85)112)75(126)100-50(65(86)116)28-29-59(84)111)99-77(128)58(41-109)107-73(124)52(25-15-17-31-82)104-70(121)53(26-18-32-91-79(87)88)101-66(117)43(4)96-62(114)40-95-78(129)64(45(6)110)108-76(127)56(36-47-22-12-9-13-23-47)98-63(115)39-93-61(113)38-94-68(119)48(83)35-46-20-10-8-11-21-46/h8-13,20-23,42-45,48-58,64,109-110H,7,14-19,24-41,81-83H2,1-6H3,(H2,84,111)(H2,85,112)(H2,86,116)(H,93,113)(H,94,119)(H,95,129)(H,96,114)(H,97,125)(H,98,115)(H,99,128)(H,100,126)(H,101,117)(H,102,120)(H,103,122)(H,104,121)(H,105,118)(H,106,123)(H,107,124)(H,108,127)(H4,87,88,91)(H4,89,90,92)/t43-,44-,45+,48-,49-,50-,51-,52-,53-,54-,55-,56-,57-,58-,64-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue Pharma L. P.
Curated by ChEMBL
| Assay Description
Affinity for human Opioid receptor like 1 (ORL-1) expressed in HEK293 cells |
J Med Chem 45: 5280-6 (2002)
BindingDB Entry DOI: 10.7270/Q2474BKJ |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM370147
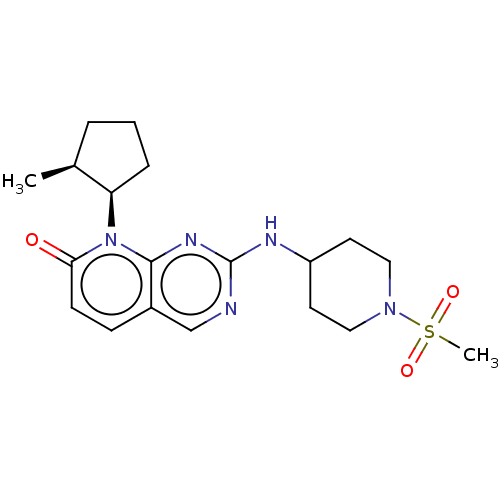
(US10233188, Example 36 | US10800783, Example 36 | ...)Show SMILES C[C@H]1CCC[C@H]1n1c2nc(NC3CCN(CC3)S(C)(=O)=O)ncc2ccc1=O |r| Show InChI InChI=1S/C19H27N5O3S/c1-13-4-3-5-16(13)24-17(25)7-6-14-12-20-19(22-18(14)24)21-15-8-10-23(11-9-15)28(2,26)27/h6-7,12-13,15-16H,3-5,8-11H2,1-2H3,(H,20,21,22)/t13-,16+/m0/s1 | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Nociceptin receptor
(Homo sapiens (Human)) | BDBM50121244
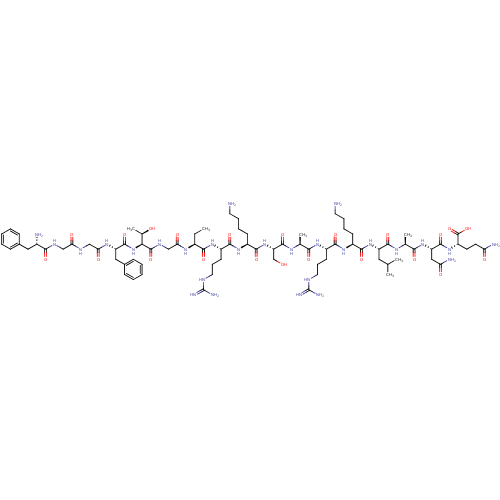
(CHEMBL264084 | F-G-G-F-T-G-Aib-R-K-S-A-R-K-L-A-N-Q...)Show SMILES CC[C@H](NC(=O)CNC(=O)[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccccc1)[C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(O)=O Show InChI InChI=1S/C80H131N27O22/c1-7-49(97-63(114)40-94-77(127)64(45(6)109)107-75(125)56(36-47-22-12-9-13-23-47)98-62(113)39-92-61(112)38-93-67(117)48(83)35-46-20-10-8-11-21-46)68(118)100-53(27-19-33-91-80(88)89)70(120)102-51(25-15-17-31-82)72(122)106-58(41-108)76(126)96-43(4)65(115)99-52(26-18-32-90-79(86)87)69(119)101-50(24-14-16-30-81)71(121)105-55(34-42(2)3)73(123)95-44(5)66(116)104-57(37-60(85)111)74(124)103-54(78(128)129)28-29-59(84)110/h8-13,20-23,42-45,48-58,64,108-109H,7,14-19,24-41,81-83H2,1-6H3,(H2,84,110)(H2,85,111)(H,92,112)(H,93,117)(H,94,127)(H,95,123)(H,96,126)(H,97,114)(H,98,113)(H,99,115)(H,100,118)(H,101,119)(H,102,120)(H,103,124)(H,104,116)(H,105,121)(H,106,122)(H,107,125)(H,128,129)(H4,86,87,90)(H4,88,89,91)/t43-,44-,45+,48-,49-,50-,51-,52-,53-,54-,55-,56-,57-,58-,64-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue Pharma L. P.
Curated by ChEMBL
| Assay Description
Affinity for human Opioid receptor like 1 (ORL-1) expressed in HEK293 cells |
J Med Chem 45: 5280-6 (2002)
BindingDB Entry DOI: 10.7270/Q2474BKJ |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM50609170

(CHEMBL5266837)Show SMILES CS(=O)(=O)N1CCC(CC1)Nc1ncc2cc(COC3CC3)c(=O)n(C3CCCC3)c2n1 | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| UniChem
| | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM467013

((-)-6-(2,2-difluoroethyl)-8-[(1R*,2R*)-2-hydroxy-2...)Show SMILES C[C@@]1(O)CCC[C@H]1n1c2nc(NC3CCN(CC3)S(C)(=O)=O)ncc2cc(CC(F)F)c1=O |r| Show InChI InChI=1S/C21H29F2N5O4S/c1-21(30)7-3-4-16(21)28-18-14(10-13(19(28)29)11-17(22)23)12-24-20(26-18)25-15-5-8-27(9-6-15)33(2,31)32/h10,12,15-17,30H,3-9,11H2,1-2H3,(H,24,25,26)/t16-,21-/m1/s1 | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM370121
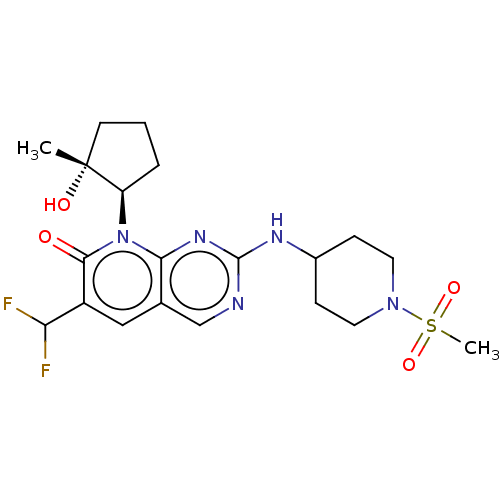
(6-(difluoromethyl)-8-[(1R,2R)-2-hydroxy-2-methylcy...)Show SMILES C[C@@]1(O)CCC[C@H]1n1c2nc(NC3CCN(CC3)S(C)(=O)=O)ncc2cc(C(F)F)c1=O |r| Show InChI InChI=1S/C20H27F2N5O4S/c1-20(29)7-3-4-15(20)27-17-12(10-14(16(21)22)18(27)28)11-23-19(25-17)24-13-5-8-26(9-6-13)32(2,30)31/h10-11,13,15-16,29H,3-9H2,1-2H3,(H,23,24,25)/t15-,20-/m1/s1 | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM14066
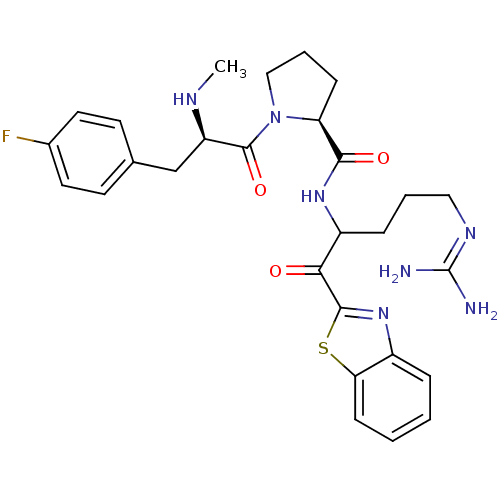
((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)Show SMILES [#6]-[#7]-[#6@H](-[#6]-c1ccc(F)cc1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-c1nc2ccccc2s1 |r| Show InChI InChI=1S/C28H34FN7O3S/c1-32-21(16-17-10-12-18(29)13-11-17)27(39)36-15-5-8-22(36)25(38)34-20(7-4-14-33-28(30)31)24(37)26-35-19-6-2-3-9-23(19)40-26/h2-3,6,9-13,20-22,32H,4-5,7-8,14-16H2,1H3,(H,34,38)(H4,30,31,33)/t20?,21-,22+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.120 | -58.9 | 15 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Johnson & Johnson Pharmaceutical
| Assay Description
Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a... |
J Med Chem 48: 1984-2008 (2005)
Article DOI: 10.1021/jm0303857
BindingDB Entry DOI: 10.7270/Q2X0658X |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM370205

(US10233188, Example 92 | US10800783, Example 92 | ...)Show SMILES Cc1cc2cnc(NC3CCN(CC3)S(C)(=O)=O)nc2n([C@@H]2CCC[C@@]2(C)O)c1=O |r| Show InChI InChI=1S/C20H29N5O4S/c1-13-11-14-12-21-19(22-15-6-9-24(10-7-15)30(3,28)29)23-17(14)25(18(13)26)16-5-4-8-20(16,2)27/h11-12,15-16,27H,4-10H2,1-3H3,(H,21,22,23)/t16-,20-/m1/s1 | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM370121

(6-(difluoromethyl)-8-[(1R,2R)-2-hydroxy-2-methylcy...)Show SMILES C[C@@]1(O)CCC[C@H]1n1c2nc(NC3CCN(CC3)S(C)(=O)=O)ncc2cc(C(F)F)c1=O |r| Show InChI InChI=1S/C20H27F2N5O4S/c1-20(29)7-3-4-15(20)27-17-12(10-14(16(21)22)18(27)28)11-23-19(25-17)24-13-5-8-26(9-6-13)32(2,30)31/h10-11,13,15-16,29H,3-9H2,1-2H3,(H,23,24,25)/t15-,20-/m1/s1 | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM14123
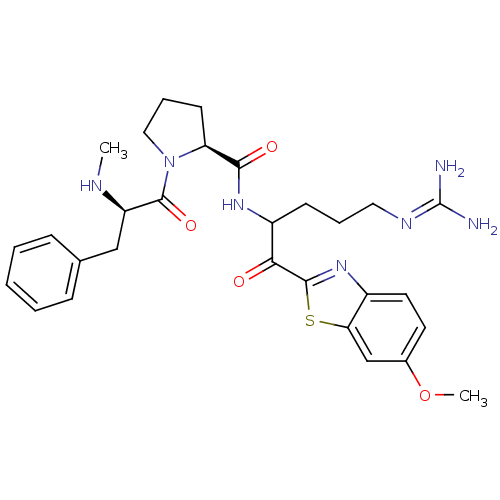
((2S)-N-[5-carbamimidamido-1-(6-methoxy-1,3-benzoth...)Show SMILES [#6]-[#7]-[#6@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-c1nc2ccc(-[#8]-[#6])cc2s1 |r| Show InChI InChI=1S/C29H37N7O4S/c1-32-22(16-18-8-4-3-5-9-18)28(39)36-15-7-11-23(36)26(38)34-21(10-6-14-33-29(30)31)25(37)27-35-20-13-12-19(40-2)17-24(20)41-27/h3-5,8-9,12-13,17,21-23,32H,6-7,10-11,14-16H2,1-2H3,(H,34,38)(H4,30,31,33)/t21?,22-,23+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.140 | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical
| Assay Description
Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a... |
J Med Chem 48: 1984-2008 (2005)
Article DOI: 10.1021/jm0303857
BindingDB Entry DOI: 10.7270/Q2X0658X |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50049757

(()-2-(6-Chloro-pyridin-3-yl)-7-aza-bicyclo[2.2.1]h...)Show InChI InChI=1S/C11H13ClN2/c12-11-4-1-7(6-13-11)9-5-8-2-3-10(9)14-8/h1,4,6,8-10,14H,2-3,5H2 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of New Orleans
Curated by ChEMBL
| Assay Description
in vitro binding affinity by inhibiting [3H]cytisine binding in rat brain tissue at Nicotinic acetylcholine receptor alpha4-beta2 |
J Med Chem 45: 3041-7 (2002)
Article DOI: 10.1021/jm0103561
BindingDB Entry DOI: 10.7270/Q2GQ71H0 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM14125

((2S)-N-[5-carbamimidamido-1-(6-fluoro-1,3-benzothi...)Show SMILES [#6]-[#7]-[#6@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-c1nc2ccc(F)cc2s1 |r| Show InChI InChI=1S/C28H34FN7O3S/c1-32-21(15-17-7-3-2-4-8-17)27(39)36-14-6-10-22(36)25(38)34-20(9-5-13-33-28(30)31)24(37)26-35-19-12-11-18(29)16-23(19)40-26/h2-4,7-8,11-12,16,20-22,32H,5-6,9-10,13-15H2,1H3,(H,34,38)(H4,30,31,33)/t20?,21-,22+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.150 | n/a | 7.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical
| Assay Description
Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a... |
J Med Chem 48: 1984-2008 (2005)
Article DOI: 10.1021/jm0303857
BindingDB Entry DOI: 10.7270/Q2X0658X |
More data for this
Ligand-Target Pair | |
Nociceptin receptor
(Homo sapiens (Human)) | BDBM50121256
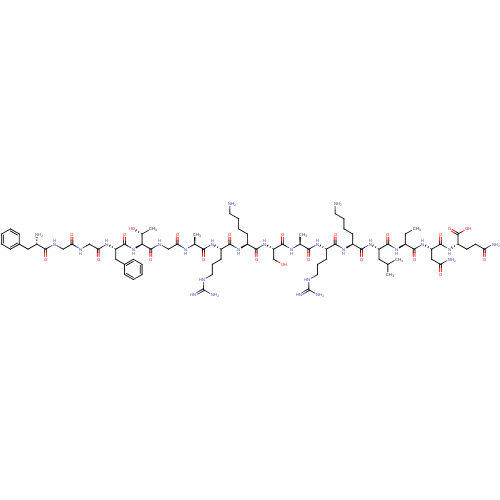
(CHEMBL410979 | F-G-G-F-T-G-A-R-K-S-A-R-K-L-Aib-N-Q...)Show SMILES CC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)CNC(=O)[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccccc1)[C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(O)=O Show InChI InChI=1S/C80H131N27O22/c1-7-49(68(118)105-57(37-60(85)111)74(124)103-54(78(128)129)28-29-59(84)110)98-73(123)55(34-42(2)3)104-71(121)50(24-14-16-30-81)101-70(120)53(27-19-33-91-80(88)89)100-66(116)44(5)96-76(126)58(41-108)106-72(122)51(25-15-17-31-82)102-69(119)52(26-18-32-90-79(86)87)99-65(115)43(4)95-62(113)40-94-77(127)64(45(6)109)107-75(125)56(36-47-22-12-9-13-23-47)97-63(114)39-92-61(112)38-93-67(117)48(83)35-46-20-10-8-11-21-46/h8-13,20-23,42-45,48-58,64,108-109H,7,14-19,24-41,81-83H2,1-6H3,(H2,84,110)(H2,85,111)(H,92,112)(H,93,117)(H,94,127)(H,95,113)(H,96,126)(H,97,114)(H,98,123)(H,99,115)(H,100,116)(H,101,120)(H,102,119)(H,103,124)(H,104,121)(H,105,118)(H,106,122)(H,107,125)(H,128,129)(H4,86,87,90)(H4,88,89,91)/t43-,44-,45+,48-,49-,50-,51-,52-,53-,54-,55-,56-,57-,58-,64-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue Pharma L. P.
Curated by ChEMBL
| Assay Description
Affinity for human Opioid receptor like 1 (ORL-1) expressed in HEK293 cells |
J Med Chem 45: 5280-6 (2002)
BindingDB Entry DOI: 10.7270/Q2474BKJ |
More data for this
Ligand-Target Pair | |
RAC-alpha serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM15131
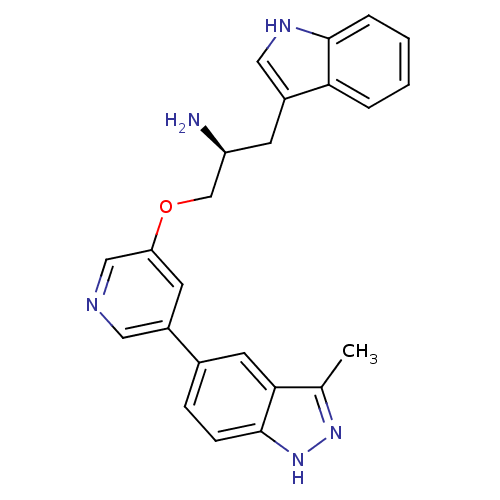
(5-indazolyl pyridine 3 | 5-{5-[(2S)-2-amino-3-(1H-...)Show SMILES Cc1n[nH]c2ccc(cc12)-c1cncc(OC[C@@H](N)Cc2c[nH]c3ccccc23)c1 |r| Show InChI InChI=1S/C24H23N5O/c1-15-22-10-16(6-7-24(22)29-28-15)17-9-20(13-26-11-17)30-14-19(25)8-18-12-27-23-5-3-2-4-21(18)23/h2-7,9-13,19,27H,8,14,25H2,1H3,(H,28,29)/t19-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of California
Curated by ChEMBL
| Assay Description
Inhibition of AKT1 |
Nat Chem Biol 5: 484-93 (2009)
Article DOI: 10.1038/nchembio.183
BindingDB Entry DOI: 10.7270/Q2D21XTB |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50143320
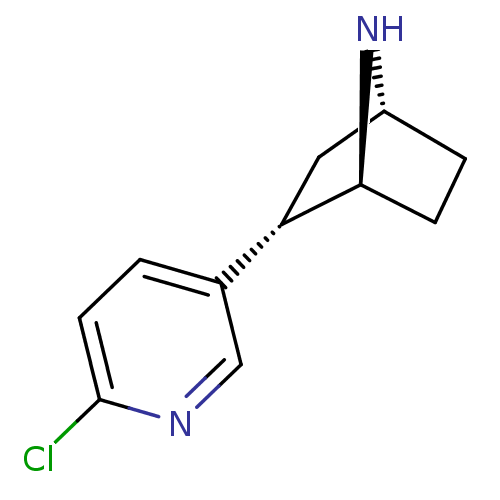
((+)-epibatidine | (-)-1-epidatidine | (1S,2S,4R)-2...)Show SMILES Clc1ccc(cn1)[C@@H]1C[C@H]2CC[C@@H]1N2 |THB:4:7:13:11.10| Show InChI InChI=1S/C11H13ClN2/c12-11-4-1-7(6-13-11)9-5-8-2-3-10(9)14-8/h1,4,6,8-10,14H,2-3,5H2/t8-,9+,10+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of New Orleans
Curated by ChEMBL
| Assay Description
in vitro binding affinity by inhibiting [3H]cytisine binding in rat brain tissue at Nicotinic acetylcholine receptor alpha4-beta2 |
J Med Chem 45: 3041-7 (2002)
Article DOI: 10.1021/jm0103561
BindingDB Entry DOI: 10.7270/Q2GQ71H0 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM14068
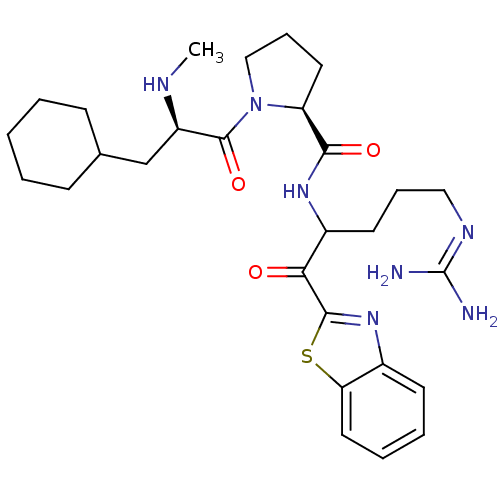
((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)Show SMILES [#6]-[#7]-[#6@H](-[#6]-[#6]-1-[#6]-[#6]-[#6]-[#6]-[#6]-1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-c1nc2ccccc2s1 |r| Show InChI InChI=1S/C28H41N7O3S/c1-31-21(17-18-9-3-2-4-10-18)27(38)35-16-8-13-22(35)25(37)33-20(12-7-15-32-28(29)30)24(36)26-34-19-11-5-6-14-23(19)39-26/h5-6,11,14,18,20-22,31H,2-4,7-10,12-13,15-17H2,1H3,(H,33,37)(H4,29,30,32)/t20?,21-,22+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.180 | -57.9 | 48 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Johnson & Johnson Pharmaceutical
| Assay Description
Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a... |
J Med Chem 48: 1984-2008 (2005)
Article DOI: 10.1021/jm0303857
BindingDB Entry DOI: 10.7270/Q2X0658X |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM467013

((-)-6-(2,2-difluoroethyl)-8-[(1R*,2R*)-2-hydroxy-2...)Show SMILES C[C@@]1(O)CCC[C@H]1n1c2nc(NC3CCN(CC3)S(C)(=O)=O)ncc2cc(CC(F)F)c1=O |r| Show InChI InChI=1S/C21H29F2N5O4S/c1-21(30)7-3-4-16(21)28-18-14(10-13(19(28)29)11-17(22)23)12-24-20(26-18)25-15-5-8-27(9-6-15)33(2,31)32/h10,12,15-17,30H,3-9,11H2,1-2H3,(H,24,25,26)/t16-,21-/m1/s1 | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50228853
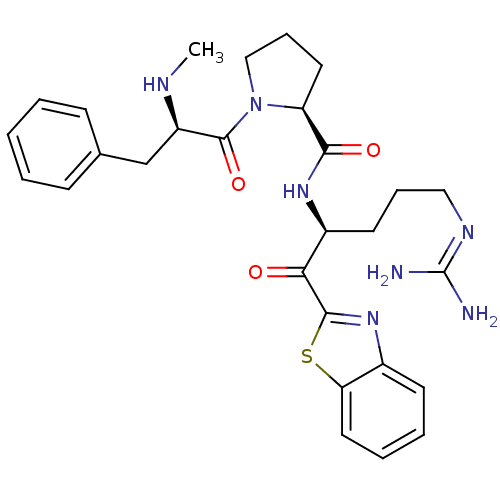
((S)-1-((R)-2-Methylamino-3-phenyl-propionyl)-pyrro...)Show SMILES [#6]-[#7]-[#6@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-c1nc2ccccc2s1 Show InChI InChI=1S/C28H35N7O3S/c1-31-21(17-18-9-3-2-4-10-18)27(38)35-16-8-13-22(35)25(37)33-20(12-7-15-32-28(29)30)24(36)26-34-19-11-5-6-14-23(19)39-26/h2-6,9-11,14,20-22,31H,7-8,12-13,15-17H2,1H3,(H,33,37)(H4,29,30,32)/t20-,21+,22-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
R. W. Johnson Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human alpha-thrombin. |
J Med Chem 39: 3039-43 (1996)
Article DOI: 10.1021/jm9603274
BindingDB Entry DOI: 10.7270/Q24B3208 |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM370256
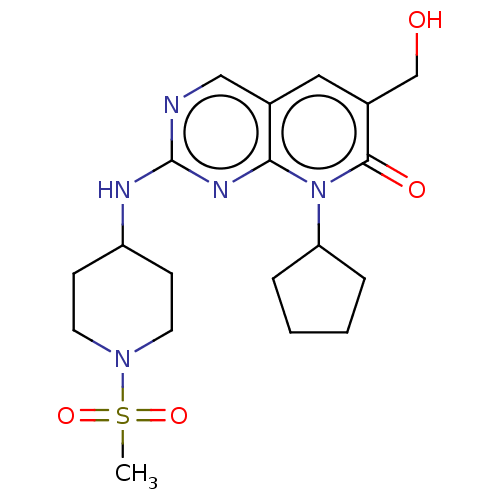
(BDBM467151 | US10233188, Example 143)Show SMILES CS(=O)(=O)N1CCC(CC1)Nc1ncc2cc(CO)c(=O)n(C3CCCC3)c2n1 | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| UniChem
| | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM370257
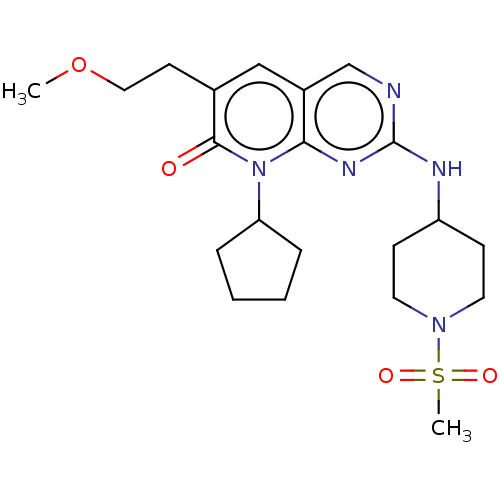
(BDBM467152 | US10233188, Example 144)Show SMILES COCCc1cc2cnc(NC3CCN(CC3)S(C)(=O)=O)nc2n(C2CCCC2)c1=O | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| UniChem
| | 0.196 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM14071
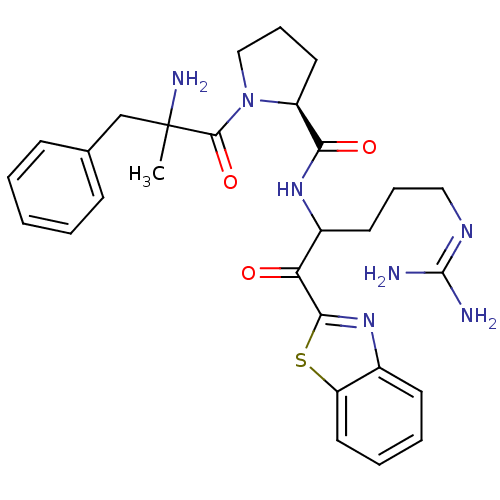
((2S)-1-(2-amino-2-benzylpropanoyl)-N-[1-(1,3-benzo...)Show SMILES [#6]C([#7])([#6]-c1ccccc1)[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-c1nc2ccccc2s1 |r| Show InChI InChI=1S/C28H35N7O3S/c1-28(31,17-18-9-3-2-4-10-18)26(38)35-16-8-13-21(35)24(37)33-20(12-7-15-32-27(29)30)23(36)25-34-19-11-5-6-14-22(19)39-25/h2-6,9-11,14,20-21H,7-8,12-13,15-17,31H2,1H3,(H,33,37)(H4,29,30,32)/t20?,21-,28?/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | -57.6 | 3.5 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Johnson & Johnson Pharmaceutical
| Assay Description
Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a... |
J Med Chem 48: 1984-2008 (2005)
Article DOI: 10.1021/jm0303857
BindingDB Entry DOI: 10.7270/Q2X0658X |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM14063
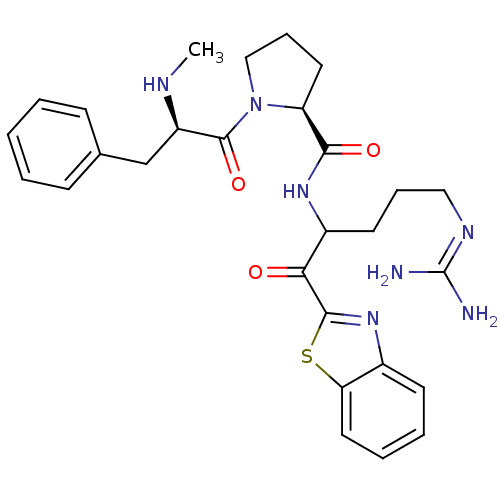
((2S)-N-[1-(1,3-benzothiazol-2-yl)-5-carbamimidamid...)Show SMILES [#6]-[#7]-[#6@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-c1nc2ccccc2s1 |r| Show InChI InChI=1S/C28H35N7O3S/c1-31-21(17-18-9-3-2-4-10-18)27(38)35-16-8-13-22(35)25(37)33-20(12-7-15-32-28(29)30)24(36)26-34-19-11-5-6-14-23(19)39-26/h2-6,9-11,14,20-22,31H,7-8,12-13,15-17H2,1H3,(H,33,37)(H4,29,30,32)/t20?,21-,22+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.200 | -57.6 | 29 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Johnson & Johnson Pharmaceutical
| Assay Description
Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a... |
J Med Chem 48: 1984-2008 (2005)
Article DOI: 10.1021/jm0303857
BindingDB Entry DOI: 10.7270/Q2X0658X |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM50609166

(CHEMBL5280928)Show SMILES CC(=O)c1c(C)c2cnc(NC3CCN(CC3)S(C)(=O)=O)nc2n(C2CCCC2)c1=O | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| UniChem
| | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM370120
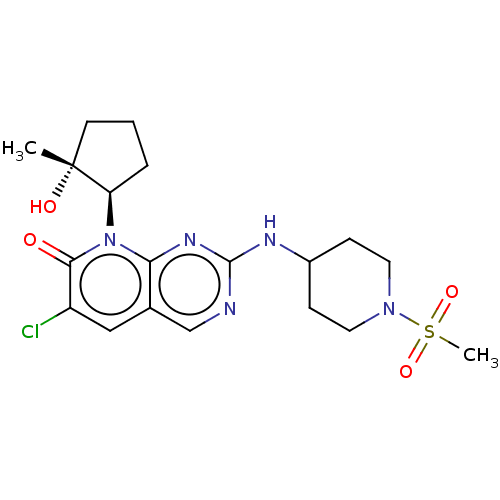
(6-chloro-8-[(1R,2R)-2-hydroxy-2-methylcyclopentyl]...)Show SMILES C[C@@]1(O)CCC[C@H]1n1c2nc(NC3CCN(CC3)S(C)(=O)=O)ncc2cc(Cl)c1=O |r| Show InChI InChI=1S/C19H26ClN5O4S/c1-19(27)7-3-4-15(19)25-16-12(10-14(20)17(25)26)11-21-18(23-16)22-13-5-8-24(9-6-13)30(2,28)29/h10-11,13,15,27H,3-9H2,1-2H3,(H,21,22,23)/t15-,19-/m1/s1 | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM370113
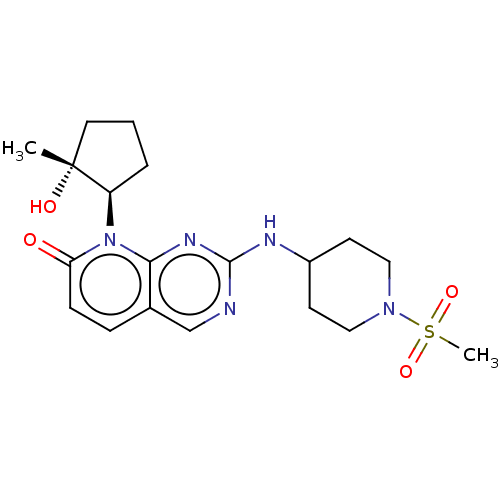
(8-[(1R,2R)-2-hydroxy-2-methylcyclopentyl]-2-{[1-(m...)Show SMILES C[C@@]1(O)CCC[C@H]1n1c2nc(NC3CCN(CC3)S(C)(=O)=O)ncc2ccc1=O |r| Show InChI InChI=1S/C19H27N5O4S/c1-19(26)9-3-4-15(19)24-16(25)6-5-13-12-20-18(22-17(13)24)21-14-7-10-23(11-8-14)29(2,27)28/h5-6,12,14-15,26H,3-4,7-11H2,1-2H3,(H,20,21,22)/t15-,19-/m1/s1 | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM50609166

(CHEMBL5280928)Show SMILES CC(=O)c1c(C)c2cnc(NC3CCN(CC3)S(C)(=O)=O)nc2n(C2CCCC2)c1=O | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| UniChem
| | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM6309
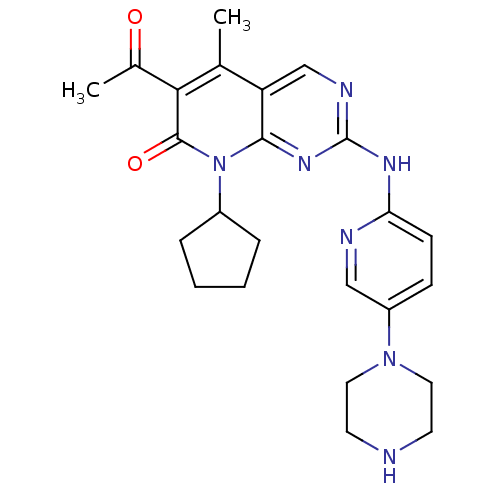
(6-Acetyl-8-cyclopentyl-5-methyl-2-(5-piperazin-1-y...)Show SMILES CC(=O)c1c(C)c2cnc(Nc3ccc(cn3)N3CCNCC3)nc2n(C2CCCC2)c1=O Show InChI InChI=1S/C24H29N7O2/c1-15-19-14-27-24(28-20-8-7-18(13-26-20)30-11-9-25-10-12-30)29-22(19)31(17-5-3-4-6-17)23(33)21(15)16(2)32/h7-8,13-14,17,25H,3-6,9-12H2,1-2H3,(H,26,27,28,29) | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM50609171
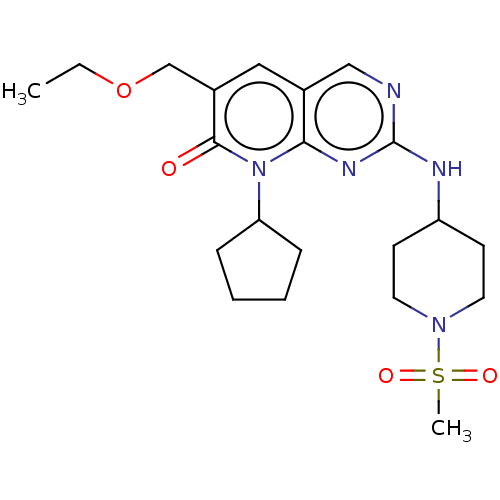
(CHEMBL5288078)Show SMILES CCOCc1cc2cnc(NC3CCN(CC3)S(C)(=O)=O)nc2n(C2CCCC2)c1=O | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| UniChem
| | 0.210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM14124
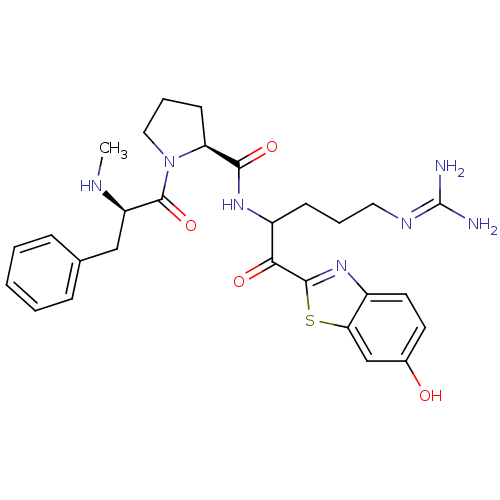
((2S)-N-[5-carbamimidamido-1-(6-hydroxy-1,3-benzoth...)Show SMILES [#6]-[#7]-[#6@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-c1nc2ccc(-[#8])cc2s1 |r| Show InChI InChI=1S/C28H35N7O4S/c1-31-21(15-17-7-3-2-4-8-17)27(39)35-14-6-10-22(35)25(38)33-20(9-5-13-32-28(29)30)24(37)26-34-19-12-11-18(36)16-23(19)40-26/h2-4,7-8,11-12,16,20-22,31,36H,5-6,9-10,13-15H2,1H3,(H,33,38)(H4,29,30,32)/t20?,21-,22+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.230 | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical
| Assay Description
Thrombin-catalyzed hydrolysis rates were measured spectrophotometrically using human alpha-thrombin, a chromogenic substrate in aqueous buffer, and a... |
J Med Chem 48: 1984-2008 (2005)
Article DOI: 10.1021/jm0303857
BindingDB Entry DOI: 10.7270/Q2X0658X |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM370205

(US10233188, Example 92 | US10800783, Example 92 | ...)Show SMILES Cc1cc2cnc(NC3CCN(CC3)S(C)(=O)=O)nc2n([C@@H]2CCC[C@@]2(C)O)c1=O |r| Show InChI InChI=1S/C20H29N5O4S/c1-13-11-14-12-21-19(22-15-6-9-24(10-7-15)30(3,28)29)23-17(14)25(18(13)26)16-5-4-8-20(16,2)27/h11-12,15-16,27H,4-10H2,1-3H3,(H,21,22,23)/t16-,20-/m1/s1 | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Nociceptin receptor
(Homo sapiens (Human)) | BDBM21842
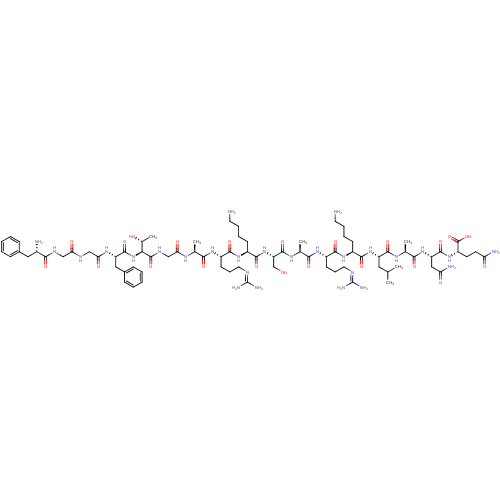
((2S)-2-[(2S)-2-[(2S)-2-[(2S)-2-[(2S)-6-amino-2-[(2...)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#8])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccccc1)-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@@H](-[#7])-[#6]-c1ccccc1)-[#6@@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6](-[#7])=O)-[#6](-[#8])=O Show InChI InChI=1S/C79H129N27O22/c1-41(2)33-54(72(122)95-44(5)66(116)103-56(36-59(84)110)73(123)102-53(77(127)128)27-28-58(83)109)104-70(120)49(23-13-15-29-80)100-69(119)52(26-18-32-90-79(87)88)99-65(115)43(4)96-75(125)57(40-107)105-71(121)50(24-14-16-30-81)101-68(118)51(25-17-31-89-78(85)86)98-64(114)42(3)94-61(112)39-93-76(126)63(45(6)108)106-74(124)55(35-47-21-11-8-12-22-47)97-62(113)38-91-60(111)37-92-67(117)48(82)34-46-19-9-7-10-20-46/h7-12,19-22,41-45,48-57,63,107-108H,13-18,23-40,80-82H2,1-6H3,(H2,83,109)(H2,84,110)(H,91,111)(H,92,117)(H,93,126)(H,94,112)(H,95,122)(H,96,125)(H,97,113)(H,98,114)(H,99,115)(H,100,119)(H,101,118)(H,102,123)(H,103,116)(H,104,120)(H,105,121)(H,106,124)(H,127,128)(H4,85,86,89)(H4,87,88,90)/t42-,43-,44-,45+,48-,49-,50-,51-,52-,53-,54-,55-,56-,57-,63-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue Pharma L. P.
Curated by ChEMBL
| Assay Description
Affinity for human Opioid receptor like 1 (ORL-1) expressed in HEK293 cells |
J Med Chem 45: 5280-6 (2002)
BindingDB Entry DOI: 10.7270/Q2474BKJ |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Bos taurus (bovine)) | BDBM14065

((2S)-1-[(2R)-2-amino-3-phenylpropanoyl]-N-[1-(1,3-...)Show SMILES [#7]-[#6@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-c1nc2ccccc2s1 |r| Show InChI InChI=1S/C27H33N7O3S/c28-18(16-17-8-2-1-3-9-17)26(37)34-15-7-12-21(34)24(36)32-20(11-6-14-31-27(29)30)23(35)25-33-19-10-4-5-13-22(19)38-25/h1-5,8-10,13,18,20-21H,6-7,11-12,14-16,28H2,(H,32,36)(H4,29,30,31)/t18-,20?,21+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical
| Assay Description
Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp... |
J Med Chem 48: 1984-2008 (2005)
Article DOI: 10.1021/jm0303857
BindingDB Entry DOI: 10.7270/Q2X0658X |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Bos taurus (bovine)) | BDBM14127

(2-(5-carbamimidamido-2-{[(2S)-1-[(2R)-2-(methylami...)Show SMILES [#6]-[#7]-[#6@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-c1nc2ccc(cc2s1)-[#6](-[#7])=O |r| Show InChI InChI=1S/C29H36N8O4S/c1-33-21(15-17-7-3-2-4-8-17)28(41)37-14-6-10-22(37)26(40)35-20(9-5-13-34-29(31)32)24(38)27-36-19-12-11-18(25(30)39)16-23(19)42-27/h2-4,7-8,11-12,16,20-22,33H,5-6,9-10,13-15H2,1H3,(H2,30,39)(H,35,40)(H4,31,32,34)/t20?,21-,22+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical
| Assay Description
Trypsin-catalyzed hydrolysis rates were measured spectrophotometrically using bovine trypsin, a chromogenic substrate in aqueous buffer, and a microp... |
J Med Chem 48: 1984-2008 (2005)
Article DOI: 10.1021/jm0303857
BindingDB Entry DOI: 10.7270/Q2X0658X |
More data for this
Ligand-Target Pair | |
GABA-A receptor; alpha-5/beta-3/gamma-2
(Homo sapiens (Human)) | BDBM26267
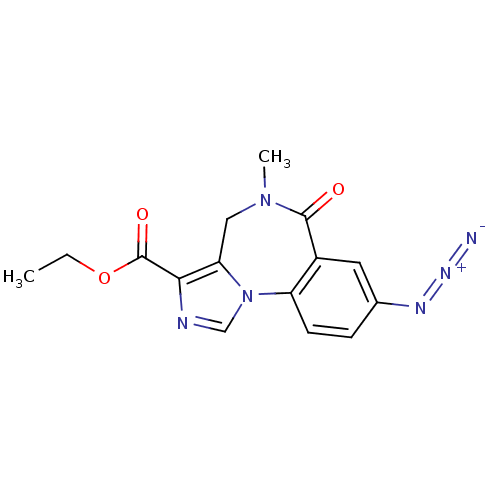
(RO-154513 | Ro15-4513 | [3H]Ro15-4513 | ethyl 12-a...)Show SMILES CCOC(=O)c1ncn-2c1CN(C)C(=O)c1cc(ccc-21)N=[N+]=[N-] Show InChI InChI=1S/C15H14N6O3/c1-3-24-15(23)13-12-7-20(2)14(22)10-6-9(18-19-16)4-5-11(10)21(12)8-17-13/h4-6,8H,3,7H2,1-2H3 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Milwaukee
Curated by ChEMBL
| Assay Description
In vitro binding affinity for human GABA-A receptor alpha5-beta3-gamma2 subunits expressed in L(tk-) cell membranes |
J Med Chem 46: 5567-70 (2003)
Article DOI: 10.1021/jm034164c
BindingDB Entry DOI: 10.7270/Q2R78DNC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM370140

(US10233188, Example 29 | US10800783, Example 29 | ...)Show SMILES CS(=O)(=O)N1CCC(CC1)Nc1ncc2cc(Cl)c(=O)n(C3CCCC3)c2n1 Show InChI InChI=1S/C18H24ClN5O3S/c1-28(26,27)23-8-6-13(7-9-23)21-18-20-11-12-10-15(19)17(25)24(16(12)22-18)14-4-2-3-5-14/h10-11,13-14H,2-9H2,1H3,(H,20,21,22) | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50194640
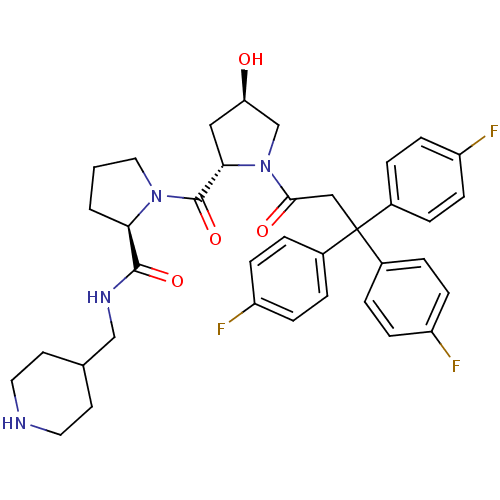
((2R)-1-((2S,4R)-4-hydroxy-1-[3,3,3-tris(4-fluoroph...)Show SMILES O[C@@H]1C[C@H](N(C1)C(=O)CC(c1ccc(F)cc1)(c1ccc(F)cc1)c1ccc(F)cc1)C(=O)N1CCC[C@@H]1C(=O)NCC1CCNCC1 Show InChI InChI=1S/C37H41F3N4O4/c38-28-9-3-25(4-10-28)37(26-5-11-29(39)12-6-26,27-7-13-30(40)14-8-27)21-34(46)44-23-31(45)20-33(44)36(48)43-19-1-2-32(43)35(47)42-22-24-15-17-41-18-16-24/h3-14,24,31-33,41,45H,1-2,15-23H2,(H,42,47)/t31-,32-,33+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arkansas for Medical Sciences
Curated by ChEMBL
| Assay Description
Binding affinity to human M3 muscarinic receptor |
J Med Chem 56: 1693-703 (2013)
Article DOI: 10.1021/jm301774u
BindingDB Entry DOI: 10.7270/Q2765GN2 |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM370248
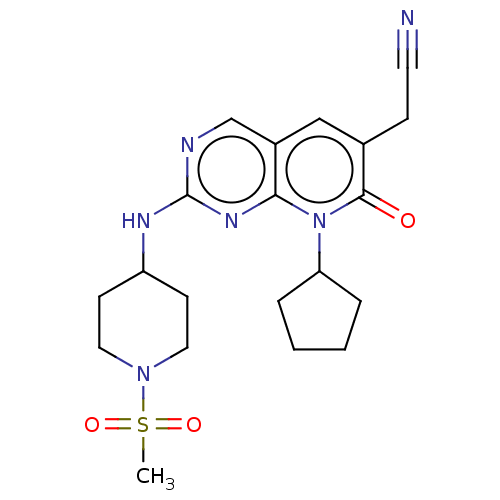
((8-cyclopentyl-2-{[1-(methylsulfonyl)piperidin-4-y...)Show SMILES CS(=O)(=O)N1CCC(CC1)Nc1ncc2cc(CC#N)c(=O)n(C3CCCC3)c2n1 Show InChI InChI=1S/C20H26N6O3S/c1-30(28,29)25-10-7-16(8-11-25)23-20-22-13-15-12-14(6-9-21)19(27)26(18(15)24-20)17-4-2-3-5-17/h12-13,16-17H,2-8,10-11H2,1H3,(H,22,23,24) | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM50293451

(1-Methyl-1-[2-(7-oxo-7H-1-aza-benzo[de]anthracen-9...)Show SMILES C[N+]1(CCC(=O)Nc2ccc-3c(c2)C(=O)c2cccc4ccnc-3c24)CCCCC1 Show InChI InChI=1S/C25H25N3O2/c1-28(13-3-2-4-14-28)15-11-22(29)27-18-8-9-19-21(16-18)25(30)20-7-5-6-17-10-12-26-24(19)23(17)20/h5-10,12,16H,2-4,11,13-15H2,1H3/p+1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sun Yat-sen University
Curated by ChEMBL
| Assay Description
Inhibition of electric eel AChE by LB plot |
Eur J Med Chem 44: 2523-32 (2009)
Article DOI: 10.1016/j.ejmech.2009.01.021
BindingDB Entry DOI: 10.7270/Q2Z89CFV |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha/beta
(Mus musculus (mouse)) | BDBM370133

(US10233188, Example 22 | US10800783, Example 22 | ...)Show SMILES Cc1cc2cnc(NC3CCN(CC3)S(C)(=O)=O)nc2n(C2CCCC2)c1=O Show InChI InChI=1S/C19H27N5O3S/c1-13-11-14-12-20-19(21-15-7-9-23(10-8-15)28(2,26)27)22-17(14)24(18(13)25)16-5-3-4-6-16/h11-12,15-16H,3-10H2,1-2H3,(H,20,21,22) | MMDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data