 Found 76 hits of ic50 for UniProtKB: P0C1R8
Found 76 hits of ic50 for UniProtKB: P0C1R8 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50465326

(CHEMBL4283345)Show SMILES [H][C@](O[C@@H]1O[C@H](CN)[C@@H](O)[C@H]1OC)([C@H](NCCCNC(=O)[C@@H](NC(=O)[C@@H](NC(=O)N[C@@H](C(C)C)C(O)=O)[C@]1([H])CCNC(=N)N1)[C@@H](OC(=O)CCCCCCCCCCCCC(C)C)C(C)C)C(O)=O)[C@@]1([H])O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O |r| Show InChI InChI=1S/C54H93N11O18/c1-28(2)19-16-14-12-10-8-9-11-13-15-17-20-34(67)81-42(30(5)6)37(62-47(72)36(31-21-25-59-52(56)60-31)64-53(77)63-35(29(3)4)49(73)74)46(71)58-24-18-23-57-38(50(75)76)43(83-51-45(79-7)39(68)32(27-55)80-51)44-40(69)41(70)48(82-44)65-26-22-33(66)61-54(65)78/h22,26,28-32,35-45,48,51,57,68-70H,8-21,23-25,27,55H2,1-7H3,(H,58,71)(H,62,72)(H,73,74)(H,75,76)(H3,56,59,60)(H,61,66,78)(H2,63,64,77)/t31-,32+,35-,36-,37-,38-,39+,40-,41+,42-,43-,44-,45+,48+,51-/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.00400 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing University of Chinese Medicine
Curated by ChEMBL
| Assay Description
Inhibition of Staphylococcus aureus MraY expressed in Escherichia coli membrane using Park's nucleotide as substrate pretreated for 30 mins followed ... |
J Nat Prod 81: 942-948 (2018)
Article DOI: 10.1021/acs.jnatprod.7b01054
BindingDB Entry DOI: 10.7270/Q2XS5Z1V |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50465324
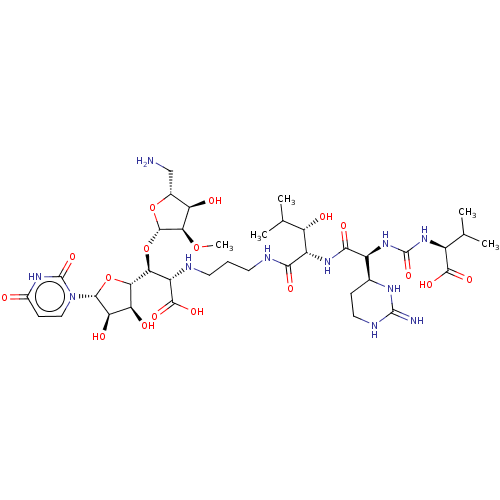
(CHEMBL4286766)Show SMILES [H][C@](O[C@@H]1O[C@H](CN)[C@@H](O)[C@H]1OC)([C@H](NCCCNC(=O)[C@@H](NC(=O)[C@@H](NC(=O)N[C@@H](C(C)C)C(O)=O)[C@]1([H])CCNC(=N)N1)[C@@H](O)C(C)C)C(O)=O)[C@@]1([H])O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O |r| Show InChI InChI=1S/C38H63N11O17/c1-14(2)19(33(57)58)47-37(61)48-20(16-7-11-43-36(40)44-16)31(56)46-21(23(51)15(3)4)30(55)42-10-6-9-41-22(34(59)60)27(66-35-29(63-5)24(52)17(13-39)64-35)28-25(53)26(54)32(65-28)49-12-8-18(50)45-38(49)62/h8,12,14-17,19-29,32,35,41,51-54H,6-7,9-11,13,39H2,1-5H3,(H,42,55)(H,46,56)(H,57,58)(H,59,60)(H3,40,43,44)(H,45,50,62)(H2,47,48,61)/t16-,17+,19-,20-,21-,22-,23-,24+,25-,26+,27-,28-,29+,32+,35-/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing University of Chinese Medicine
Curated by ChEMBL
| Assay Description
Inhibition of Staphylococcus aureus MraY expressed in Escherichia coli membrane using Park's nucleotide as substrate pretreated for 30 mins followed ... |
J Nat Prod 81: 942-948 (2018)
Article DOI: 10.1021/acs.jnatprod.7b01054
BindingDB Entry DOI: 10.7270/Q2XS5Z1V |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50465327
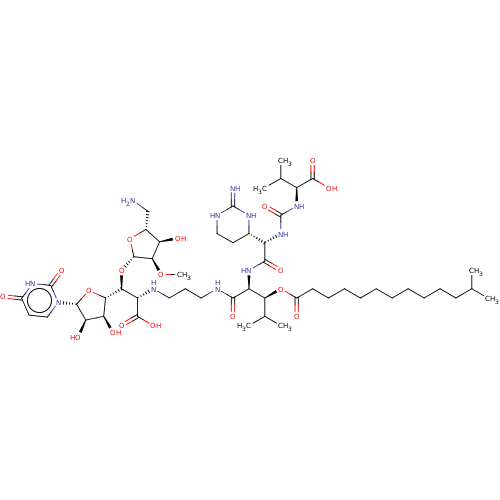
(CHEMBL4279378)Show SMILES [H][C@](O[C@@H]1O[C@H](CN)[C@@H](O)[C@H]1OC)([C@H](NCCCNC(=O)[C@@H](NC(=O)[C@@H](NC(=O)N[C@@H](C(C)C)C(O)=O)[C@]1([H])CCNC(=N)N1)[C@@H](OC(=O)CCCCCCCCCCC(C)C)C(C)C)C(O)=O)[C@@]1([H])O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O |r| Show InChI InChI=1S/C52H89N11O18/c1-26(2)17-14-12-10-8-9-11-13-15-18-32(65)79-40(28(5)6)35(60-45(70)34(29-19-23-57-50(54)58-29)62-51(75)61-33(27(3)4)47(71)72)44(69)56-22-16-21-55-36(48(73)74)41(81-49-43(77-7)37(66)30(25-53)78-49)42-38(67)39(68)46(80-42)63-24-20-31(64)59-52(63)76/h20,24,26-30,33-43,46,49,55,66-68H,8-19,21-23,25,53H2,1-7H3,(H,56,69)(H,60,70)(H,71,72)(H,73,74)(H3,54,57,58)(H,59,64,76)(H2,61,62,75)/t29-,30+,33-,34-,35-,36-,37+,38-,39+,40-,41-,42-,43+,46+,49-/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.0220 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing University of Chinese Medicine
Curated by ChEMBL
| Assay Description
Inhibition of Staphylococcus aureus MraY expressed in Escherichia coli membrane using Park's nucleotide as substrate pretreated for 30 mins followed ... |
J Nat Prod 81: 942-948 (2018)
Article DOI: 10.1021/acs.jnatprod.7b01054
BindingDB Entry DOI: 10.7270/Q2XS5Z1V |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50465325

(CHEMBL4284923)Show SMILES [H][C@](O[C@@H]1O[C@H](CNC(C)=O)[C@@H](O)[C@H]1OC)([C@H](NCCCNC(=O)[C@@H](NC(=O)[C@@H](NC(=O)N[C@@H](C(C)C)C(O)=O)[C@]1([H])CCNC(=N)N1)[C@@H](O)C(C)C)C(O)=O)[C@@]1([H])O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O |r| Show InChI InChI=1S/C40H65N11O18/c1-15(2)21(35(60)61)49-39(64)50-22(18-8-12-44-38(41)46-18)33(59)48-23(25(54)16(3)4)32(58)43-11-7-10-42-24(36(62)63)29(69-37-31(66-6)26(55)19(67-37)14-45-17(5)52)30-27(56)28(57)34(68-30)51-13-9-20(53)47-40(51)65/h9,13,15-16,18-19,21-31,34,37,42,54-57H,7-8,10-12,14H2,1-6H3,(H,43,58)(H,45,52)(H,48,59)(H,60,61)(H,62,63)(H3,41,44,46)(H,47,53,65)(H2,49,50,64)/t18-,19+,21-,22-,23-,24-,25-,26+,27-,28+,29-,30-,31+,34+,37-/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.0930 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing University of Chinese Medicine
Curated by ChEMBL
| Assay Description
Inhibition of Staphylococcus aureus MraY expressed in Escherichia coli membrane using Park's nucleotide as substrate pretreated for 30 mins followed ... |
J Nat Prod 81: 942-948 (2018)
Article DOI: 10.1021/acs.jnatprod.7b01054
BindingDB Entry DOI: 10.7270/Q2XS5Z1V |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50526643

(CHEMBL4475677)Show SMILES [H][C@@]1(O[C@H]([C@@]2([H])O[C@H]([C@H](O)[C@@H]2O)n2ccc(=O)[nH]c2=O)[C@]2([H])NC[C@@H](CCCCCCCCCCCCCCCCC)[C@H](N(C)C2=O)C(O)=O)O[C@H](CN)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C38H65N5O12/c1-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-23-22-40-26(34(49)42(2)27(23)36(50)51)32(55-37-31(48)28(45)24(21-39)53-37)33-29(46)30(47)35(54-33)43-20-19-25(44)41-38(43)52/h19-20,23-24,26-33,35,37,40,45-48H,3-18,21-22,39H2,1-2H3,(H,50,51)(H,41,44,52)/t23-,24-,26+,27+,28-,29+,30-,31-,32+,33+,35-,37+/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Griffith University
Curated by ChEMBL
| Assay Description
Inhibition of MraY in Staphylococcus aureus |
Eur J Med Chem 171: 462-474 (2019)
Article DOI: 10.1016/j.ejmech.2019.01.071
BindingDB Entry DOI: 10.7270/Q2K35Z3R |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50614429

(CHEMBL5265940) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50386961

(CHEMBL2048825)Show SMILES C[C@H](N)C(=O)N(C)[C@@H](C)[C@H](NC(=O)[C@H](C)NC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(O)=O)C(=O)N\C=C1\C[C@@H](O)[C@@H](O1)n1ccc(=O)[nH]c1=O |r| Show InChI InChI=1S/C32H41N9O10/c1-15(33)28(46)40(4)17(3)25(27(45)35-14-19-12-23(42)29(51-19)41-10-9-24(43)38-32(41)50)39-26(44)16(2)36-31(49)37-22(30(47)48)11-18-13-34-21-8-6-5-7-20(18)21/h5-10,13-17,22-23,25,29,34,42H,11-12,33H2,1-4H3,(H,35,45)(H,39,44)(H,47,48)(H2,36,37,49)(H,38,43,50)/b19-14-/t15-,16-,17-,22-,23+,25-,29+/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Staphylococcus aureus MraY using UDP-MurNAc-dansylpentapeptide substrate assessed as formation of dansylated lipid I incubated for 3 to... |
Bioorg Med Chem Lett 22: 4810-5 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.050
BindingDB Entry DOI: 10.7270/Q2J967FP |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50386963
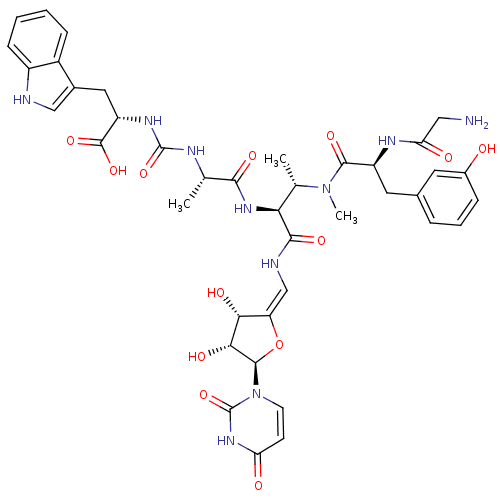
(CHEMBL2048828)Show SMILES C[C@H](NC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(O)=O)C(=O)N[C@@H]([C@H](C)N(C)C(=O)[C@H](Cc1cccc(O)c1)NC(=O)CN)C(=O)N\C=C1/O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O |r| Show InChI InChI=1S/C40H48N10O13/c1-19(44-39(61)46-27(38(59)60)15-22-17-42-25-10-5-4-9-24(22)25)34(56)48-31(20(2)49(3)36(58)26(45-30(53)16-41)14-21-7-6-8-23(51)13-21)35(57)43-18-28-32(54)33(55)37(63-28)50-12-11-29(52)47-40(50)62/h4-13,17-20,26-27,31-33,37,42,51,54-55H,14-16,41H2,1-3H3,(H,43,57)(H,45,53)(H,48,56)(H,59,60)(H2,44,46,61)(H,47,52,62)/b28-18-/t19-,20-,26-,27-,31-,32+,33+,37+/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Staphylococcus aureus MraY using UDP-MurNAc-dansylpentapeptide substrate assessed as formation of dansylated lipid I incubated for 3 to... |
Bioorg Med Chem Lett 22: 4810-5 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.050
BindingDB Entry DOI: 10.7270/Q2J967FP |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50526632

(CHEMBL4473600)Show SMILES [H][C@@]1(O[C@H]([C@@]2([H])O[C@H]([C@H](O)[C@@H]2O)n2ccc(=O)[nH]c2=O)[C@]2([H])C[C@H](CCN2)NC(=O)CCCCCCCCCCCCCCC)O[C@H](CN)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C35H61N5O10/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-25(41)38-22-16-18-37-23(20-22)31(50-34-30(46)27(43)24(21-36)48-34)32-28(44)29(45)33(49-32)40-19-17-26(42)39-35(40)47/h17,19,22-24,27-34,37,43-46H,2-16,18,20-21,36H2,1H3,(H,38,41)(H,39,42,47)/t22-,23-,24+,27+,28-,29+,30+,31-,32-,33+,34-/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Griffith University
Curated by ChEMBL
| Assay Description
Inhibition of MraY in Staphylococcus aureus |
Eur J Med Chem 171: 462-474 (2019)
Article DOI: 10.1016/j.ejmech.2019.01.071
BindingDB Entry DOI: 10.7270/Q2K35Z3R |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50526632

(CHEMBL4473600)Show SMILES [H][C@@]1(O[C@H]([C@@]2([H])O[C@H]([C@H](O)[C@@H]2O)n2ccc(=O)[nH]c2=O)[C@]2([H])C[C@H](CCN2)NC(=O)CCCCCCCCCCCCCCC)O[C@H](CN)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C35H61N5O10/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-25(41)38-22-16-18-37-23(20-22)31(50-34-30(46)27(43)24(21-36)48-34)32-28(44)29(45)33(49-32)40-19-17-26(42)39-35(40)47/h17,19,22-24,27-34,37,43-46H,2-16,18,20-21,36H2,1H3,(H,38,41)(H,39,42,47)/t22-,23-,24+,27+,28-,29+,30+,31-,32-,33+,34-/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Griffith University
Curated by ChEMBL
| Assay Description
Inhibition of MraY in Staphylococcus aureus |
Eur J Med Chem 171: 462-474 (2019)
Article DOI: 10.1016/j.ejmech.2019.01.071
BindingDB Entry DOI: 10.7270/Q2K35Z3R |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50614430
(CHEMBL5277590) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50386960

(CHEMBL2048826)Show SMILES C[C@H](N)C(=O)N(C)[C@@H](C)[C@H](NC(=O)[C@H](C)NC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(O)=O)C(=O)N\C=C1/O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O |r| Show InChI InChI=1S/C32H41N9O11/c1-14(33)28(47)40(4)16(3)23(27(46)35-13-21-24(43)25(44)29(52-21)41-10-9-22(42)38-32(41)51)39-26(45)15(2)36-31(50)37-20(30(48)49)11-17-12-34-19-8-6-5-7-18(17)19/h5-10,12-16,20,23-25,29,34,43-44H,11,33H2,1-4H3,(H,35,46)(H,39,45)(H,48,49)(H2,36,37,50)(H,38,42,51)/b21-13-/t14-,15-,16-,20-,23-,24+,25+,29+/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 42 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Staphylococcus aureus MraY using UDP-MurNAc-dansylpentapeptide substrate assessed as formation of dansylated lipid I incubated for 3 to... |
Bioorg Med Chem Lett 22: 4810-5 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.050
BindingDB Entry DOI: 10.7270/Q2J967FP |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50386965
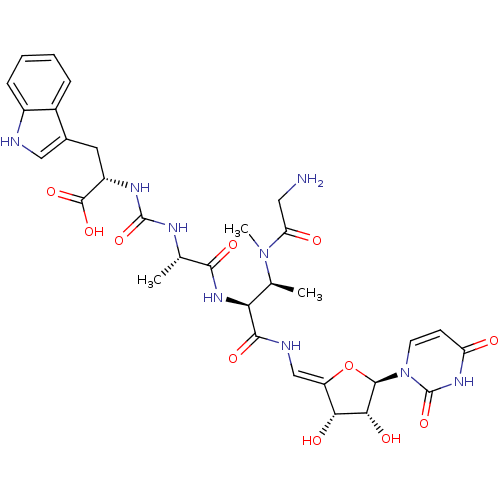
(CHEMBL2048830)Show SMILES C[C@H](NC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(O)=O)C(=O)N[C@@H]([C@H](C)N(C)C(=O)CN)C(=O)N\C=C1/O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O |r| Show InChI InChI=1S/C31H39N9O11/c1-14(35-30(49)36-19(29(47)48)10-16-12-33-18-7-5-4-6-17(16)18)26(45)38-23(15(2)39(3)22(42)11-32)27(46)34-13-20-24(43)25(44)28(51-20)40-9-8-21(41)37-31(40)50/h4-9,12-15,19,23-25,28,33,43-44H,10-11,32H2,1-3H3,(H,34,46)(H,38,45)(H,47,48)(H2,35,36,49)(H,37,41,50)/b20-13-/t14-,15-,19-,23-,24+,25+,28+/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 65 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Staphylococcus aureus MraY using UDP-MurNAc-dansylpentapeptide substrate assessed as formation of dansylated lipid I incubated for 3 to... |
Bioorg Med Chem Lett 22: 4810-5 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.050
BindingDB Entry DOI: 10.7270/Q2J967FP |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50526632

(CHEMBL4473600)Show SMILES [H][C@@]1(O[C@H]([C@@]2([H])O[C@H]([C@H](O)[C@@H]2O)n2ccc(=O)[nH]c2=O)[C@]2([H])C[C@H](CCN2)NC(=O)CCCCCCCCCCCCCCC)O[C@H](CN)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C35H61N5O10/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-25(41)38-22-16-18-37-23(20-22)31(50-34-30(46)27(43)24(21-36)48-34)32-28(44)29(45)33(49-32)40-19-17-26(42)39-35(40)47/h17,19,22-24,27-34,37,43-46H,2-16,18,20-21,36H2,1H3,(H,38,41)(H,39,42,47)/t22-,23-,24+,27+,28-,29+,30+,31-,32-,33+,34-/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 206 | n/a | n/a | n/a | n/a | n/a | n/a |
Griffith University
Curated by ChEMBL
| Assay Description
Inhibition of MraY in Staphylococcus aureus |
Eur J Med Chem 171: 462-474 (2019)
Article DOI: 10.1016/j.ejmech.2019.01.071
BindingDB Entry DOI: 10.7270/Q2K35Z3R |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615884

(CHEMBL5279603) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50135599

((4S,5S,6S)-6-{(R)-Carbamoyl-[(2S,3S,4R,5R)-5-(2,4-...)Show SMILES CO[C@H]1[C@@H](O)[C@@H](O[C@@H]1[C@@H](O[C@H]1OC(=C[C@H](O)[C@@H]1O)C(=O)N[C@H]1CCCCNC1=O)C(N)=O)n1ccc(=O)[nH]c1=O |c:13| Show InChI InChI=1S/C23H31N5O12/c1-37-15-14(32)21(28-7-5-12(30)27-23(28)36)39-16(15)17(18(24)33)40-22-13(31)10(29)8-11(38-22)20(35)26-9-4-2-3-6-25-19(9)34/h5,7-10,13-17,21-22,29,31-32H,2-4,6H2,1H3,(H2,24,33)(H,25,34)(H,26,35)(H,27,30,36)/t9-,10-,13-,14+,15-,16-,17+,21+,22+/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 330 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmc.2022.117011
BindingDB Entry DOI: 10.7270/Q2GB285Q |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50537412

(CHEMBL4534172)Show SMILES [H][C@@]1(O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O)[C@H](O)C[C@H]1O[C@@]([H])(O[C@H]2O[C@H](CO)[C@@H](O)[C@H](O)[C@H]2NC(C)=O)[C@H](NC(=O)\C=C\CCCCCCCCCC(C)C)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C38H62N4O16/c1-19(2)13-11-9-7-5-4-6-8-10-12-14-24(46)40-27-31(51)28(48)22(55-37(27)58-36-26(39-20(3)44)30(50)29(49)23(18-43)56-36)17-21(45)34-32(52)33(53)35(57-34)42-16-15-25(47)41-38(42)54/h12,14-16,19,21-23,26-37,43,45,48-53H,4-11,13,17-18H2,1-3H3,(H,39,44)(H,40,46)(H,41,47,54)/b14-12+/t21-,22-,23-,26-,27-,28+,29-,30-,31-,32+,33-,34-,35-,36-,37+/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 350 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokkaido University
Curated by ChEMBL
| Assay Description
Inhibition of Staphylococcus aureus MraY assessed as reduction in dansylated lipid I formation using UDP-MurNAc-dansylpentapeptide incubated for 3 hr... |
Bioorg Med Chem 27: 1714-1719 (2019)
Article DOI: 10.1016/j.bmc.2019.02.035
BindingDB Entry DOI: 10.7270/Q2T43XMT |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50537412

(CHEMBL4534172)Show SMILES [H][C@@]1(O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O)[C@H](O)C[C@H]1O[C@@]([H])(O[C@H]2O[C@H](CO)[C@@H](O)[C@H](O)[C@H]2NC(C)=O)[C@H](NC(=O)\C=C\CCCCCCCCCC(C)C)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C38H62N4O16/c1-19(2)13-11-9-7-5-4-6-8-10-12-14-24(46)40-27-31(51)28(48)22(55-37(27)58-36-26(39-20(3)44)30(50)29(49)23(18-43)56-36)17-21(45)34-32(52)33(53)35(57-34)42-16-15-25(47)41-38(42)54/h12,14-16,19,21-23,26-37,43,45,48-53H,4-11,13,17-18H2,1-3H3,(H,39,44)(H,40,46)(H,41,47,54)/b14-12+/t21-,22-,23-,26-,27-,28+,29-,30-,31-,32+,33-,34-,35-,36-,37+/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 350 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokkaido University
Curated by ChEMBL
| Assay Description
Inhibition of Staphylococcus aureus MraY assessed as reduction in dansylated lipid I formation using UDP-MurNAc-dansylpentapeptide incubated for 3 hr... |
Bioorg Med Chem 27: 1714-1719 (2019)
Article DOI: 10.1016/j.bmc.2019.02.035
BindingDB Entry DOI: 10.7270/Q2T43XMT |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615889
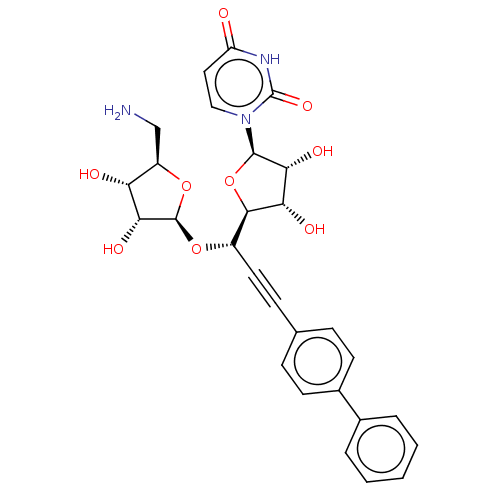
(CHEMBL5289885) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 590 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615888
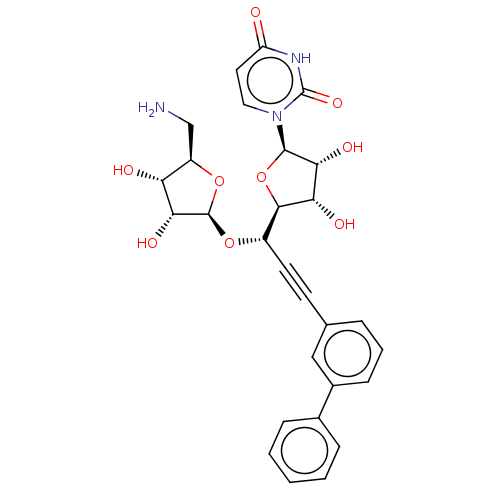
(CHEMBL5277496) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 650 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50386966
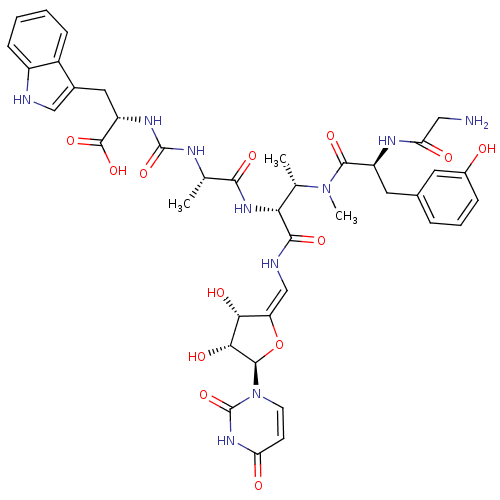
(CHEMBL2048831)Show SMILES C[C@H](NC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(O)=O)C(=O)N[C@H]([C@H](C)N(C)C(=O)[C@H](Cc1cccc(O)c1)NC(=O)CN)C(=O)N\C=C1/O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O |r| Show InChI InChI=1S/C40H48N10O13/c1-19(44-39(61)46-27(38(59)60)15-22-17-42-25-10-5-4-9-24(22)25)34(56)48-31(20(2)49(3)36(58)26(45-30(53)16-41)14-21-7-6-8-23(51)13-21)35(57)43-18-28-32(54)33(55)37(63-28)50-12-11-29(52)47-40(50)62/h4-13,17-20,26-27,31-33,37,42,51,54-55H,14-16,41H2,1-3H3,(H,43,57)(H,45,53)(H,48,56)(H,59,60)(H2,44,46,61)(H,47,52,62)/b28-18-/t19-,20-,26-,27-,31+,32+,33+,37+/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 650 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Staphylococcus aureus MraY using UDP-MurNAc-dansylpentapeptide substrate assessed as formation of dansylated lipid I incubated for 3 to... |
Bioorg Med Chem Lett 22: 4810-5 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.050
BindingDB Entry DOI: 10.7270/Q2J967FP |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615893

(CHEMBL5287014) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 900 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615880

(CHEMBL5268197) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 950 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615894
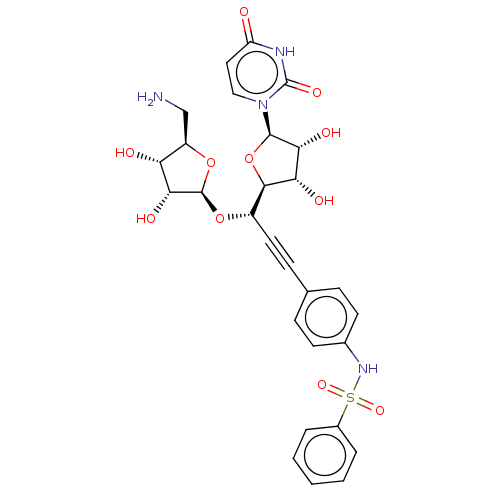
(CHEMBL5291217) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615892
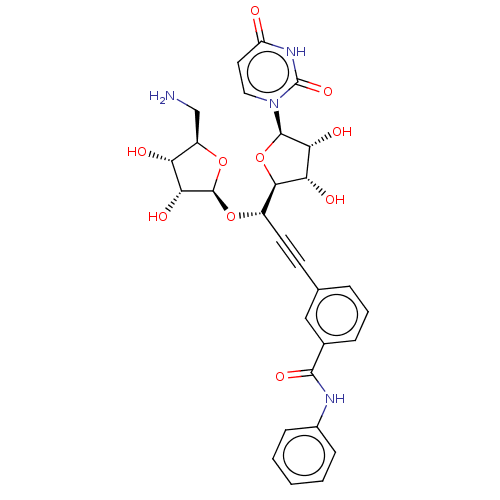
(CHEMBL5269635) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615883
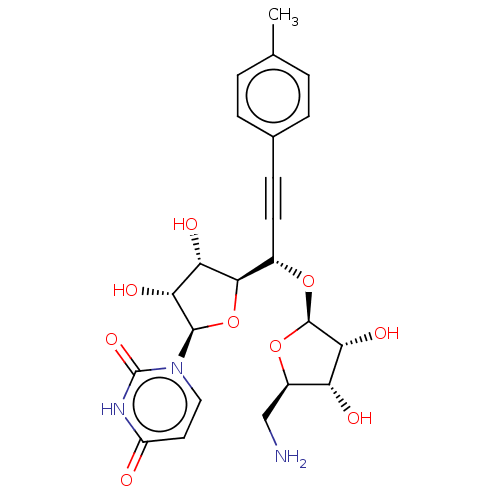
(CHEMBL5288794) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615879
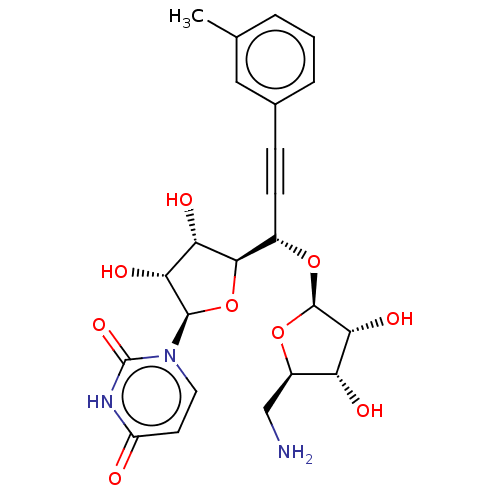
(CHEMBL5283961) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615891

(CHEMBL5278736) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615886

(CHEMBL5266213) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615881

(CHEMBL5290020) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50600491

(CHEMBL5209278)Show SMILES [H][C@@]1(O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O)[C@@H](O[C@@H]1O[C@H](CN)[C@@H](O)[C@H]1O)C#Cc1ccccc1 |r| | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615878

(CHEMBL5274543) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50553591

(CHEMBL4746444)Show SMILES [H][C@@]1(O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O)[C@@H](O[C@@]1([H])OC(=C[C@H](O)[C@@H]1O)C(=O)N[C@H]1CCCCNC1=O)C(N)=O |r,c:23| | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmc.2022.117011
BindingDB Entry DOI: 10.7270/Q2GB285Q |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615882

(CHEMBL5274523) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50600491

(CHEMBL5209278)Show SMILES [H][C@@]1(O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O)[C@@H](O[C@@H]1O[C@H](CN)[C@@H](O)[C@H]1O)C#Cc1ccccc1 |r| | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmc.2022.116744
BindingDB Entry DOI: 10.7270/Q2XG9W6P |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50526641

(CHEMBL4518281)Show SMILES [H][C@@]1(O[C@@H]([C@@H]2NC[C@H](CCCCCCCCCCCCCCCCC)[C@H](N(C)C2=O)C(O)=O)[C@@]2([H])O[C@H]([C@H](O)[C@@H]2O)n2ccc(=O)[nH]c2=O)O[C@H](CN)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C38H65N5O12/c1-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-23-22-40-26(34(49)42(2)27(23)36(50)51)32(55-37-31(48)28(45)24(21-39)53-37)33-29(46)30(47)35(54-33)43-20-19-25(44)41-38(43)52/h19-20,23-24,26-33,35,37,40,45-48H,3-18,21-22,39H2,1-2H3,(H,50,51)(H,41,44,52)/t23-,24+,26-,27-,28+,29-,30+,31+,32-,33-,35+,37-/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Griffith University
Curated by ChEMBL
| Assay Description
Inhibition of MraY in Staphylococcus aureus |
Eur J Med Chem 171: 462-474 (2019)
Article DOI: 10.1016/j.ejmech.2019.01.071
BindingDB Entry DOI: 10.7270/Q2K35Z3R |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50386962
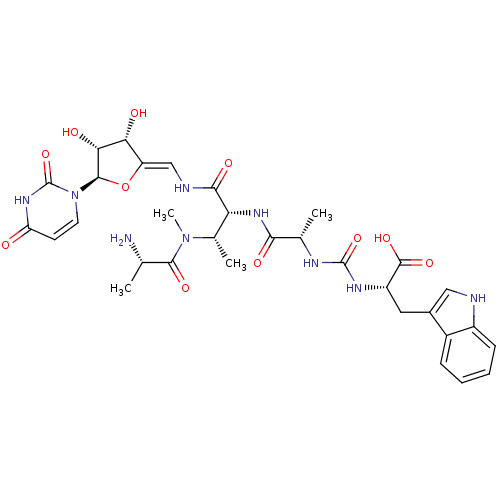
(CHEMBL2048827)Show SMILES C[C@H](N)C(=O)N(C)[C@@H](C)[C@@H](NC(=O)[C@H](C)NC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(O)=O)C(=O)N\C=C1/O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O |r| Show InChI InChI=1S/C32H41N9O11/c1-14(33)28(47)40(4)16(3)23(27(46)35-13-21-24(43)25(44)29(52-21)41-10-9-22(42)38-32(41)51)39-26(45)15(2)36-31(50)37-20(30(48)49)11-17-12-34-19-8-6-5-7-18(17)19/h5-10,12-16,20,23-25,29,34,43-44H,11,33H2,1-4H3,(H,35,46)(H,39,45)(H,48,49)(H2,36,37,50)(H,38,42,51)/b21-13-/t14-,15-,16-,20-,23+,24+,25+,29+/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Staphylococcus aureus MraY using UDP-MurNAc-dansylpentapeptide substrate assessed as formation of dansylated lipid I incubated for 3 to... |
Bioorg Med Chem Lett 22: 4810-5 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.050
BindingDB Entry DOI: 10.7270/Q2J967FP |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50386964

(CHEMBL2048829)Show SMILES C[C@H](NC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(O)=O)C(=O)N[C@@H]([C@H](C)N(C)C(=O)[C@H](Cc1ccc(O)cc1)NC(=O)CN)C(=O)N\C=C1/O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O |r| Show InChI InChI=1S/C40H48N10O13/c1-19(44-39(61)46-27(38(59)60)15-22-17-42-25-7-5-4-6-24(22)25)34(56)48-31(20(2)49(3)36(58)26(45-30(53)16-41)14-21-8-10-23(51)11-9-21)35(57)43-18-28-32(54)33(55)37(63-28)50-13-12-29(52)47-40(50)62/h4-13,17-20,26-27,31-33,37,42,51,54-55H,14-16,41H2,1-3H3,(H,43,57)(H,45,53)(H,48,56)(H,59,60)(H2,44,46,61)(H,47,52,62)/b28-18-/t19-,20-,26-,27-,31-,32+,33+,37+/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Staphylococcus aureus MraY using UDP-MurNAc-dansylpentapeptide substrate assessed as formation of dansylated lipid I incubated for 3 to... |
Bioorg Med Chem Lett 22: 4810-5 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.050
BindingDB Entry DOI: 10.7270/Q2J967FP |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615887

(CHEMBL5265879) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 4.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50600488

(CHEMBL5207643)Show SMILES [H][C@]12OC[C@@](O[C@H]1n1ccc(=O)[nH]c1=O)([C@H](CC)O[C@@H]1O[C@H](CN)[C@@H](O)[C@H]1O)[C@H]2O |r,TLB:7:6:3.2:28| | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmc.2022.116744
BindingDB Entry DOI: 10.7270/Q2XG9W6P |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615885

(CHEMBL5286726) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 5.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615890

(CHEMBL5277965) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 6.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50526640
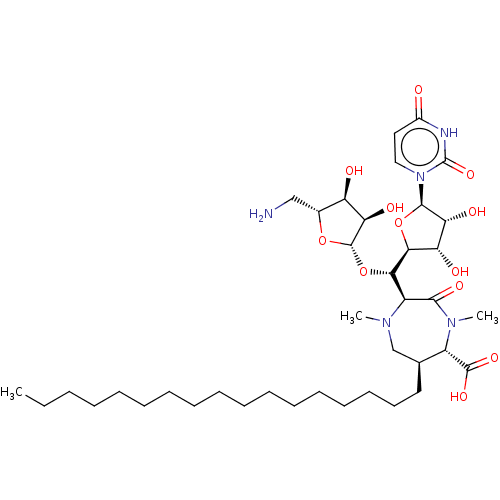
(CHEMBL4466734)Show SMILES [H][C@@]1(O[C@@H]([C@@H]2N(C)C[C@H](CCCCCCCCCCCCCCCCC)[C@H](N(C)C2=O)C(O)=O)[C@@]2([H])O[C@H]([C@H](O)[C@@H]2O)n2ccc(=O)[nH]c2=O)O[C@H](CN)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C39H67N5O12/c1-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-24-23-42(2)28(35(50)43(3)27(24)37(51)52)33(56-38-32(49)29(46)25(22-40)54-38)34-30(47)31(48)36(55-34)44-21-20-26(45)41-39(44)53/h20-21,24-25,27-34,36,38,46-49H,4-19,22-23,40H2,1-3H3,(H,51,52)(H,41,45,53)/t24-,25+,27-,28-,29+,30-,31+,32+,33-,34-,36+,38-/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 6.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Griffith University
Curated by ChEMBL
| Assay Description
Inhibition of MraY in Staphylococcus aureus |
Eur J Med Chem 171: 462-474 (2019)
Article DOI: 10.1016/j.ejmech.2019.01.071
BindingDB Entry DOI: 10.7270/Q2K35Z3R |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50386967
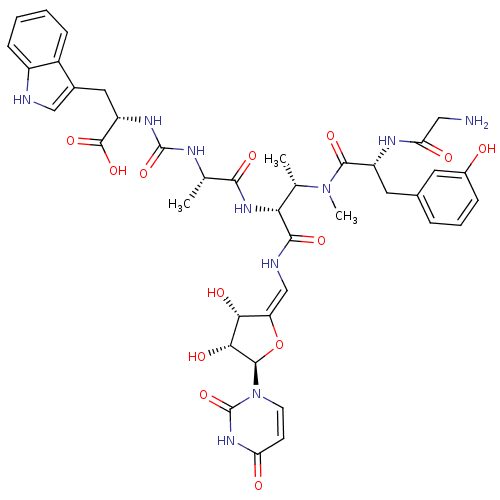
(CHEMBL2048832)Show SMILES C[C@H](NC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(O)=O)C(=O)N[C@H]([C@H](C)N(C)C(=O)[C@@H](Cc1cccc(O)c1)NC(=O)CN)C(=O)N\C=C1/O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O |r| Show InChI InChI=1S/C40H48N10O13/c1-19(44-39(61)46-27(38(59)60)15-22-17-42-25-10-5-4-9-24(22)25)34(56)48-31(20(2)49(3)36(58)26(45-30(53)16-41)14-21-7-6-8-23(51)13-21)35(57)43-18-28-32(54)33(55)37(63-28)50-12-11-29(52)47-40(50)62/h4-13,17-20,26-27,31-33,37,42,51,54-55H,14-16,41H2,1-3H3,(H,43,57)(H,45,53)(H,48,56)(H,59,60)(H2,44,46,61)(H,47,52,62)/b28-18-/t19-,20-,26+,27-,31+,32+,33+,37+/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of Staphylococcus aureus MraY using UDP-MurNAc-dansylpentapeptide substrate assessed as formation of dansylated lipid I incubated for 3 to... |
Bioorg Med Chem Lett 22: 4810-5 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.050
BindingDB Entry DOI: 10.7270/Q2J967FP |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50607107

(CHEMBL5220005)Show SMILES [H][C@@]1(O[C@H]([C@H](O)[C@@H]1OC)n1ccc(=O)[nH]c1=O)[C@H](O[C@H]1OC(=C[C@H](O)[C@@H]1O)C(=O)N[C@H]1CCCCNC1=O)C(N)=O |r,c:23| | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmc.2022.117011
BindingDB Entry DOI: 10.7270/Q2GB285Q |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50607108

(CHEMBL5218857)Show SMILES [H][C@@]1(O[C@H]([C@H](O)[C@@H]1OC)n1ccc(=O)[nH]c1=O)[C@@H](O[C@H]1OC(=C[C@H](O)[C@@H]1O)C(=O)N[C@H]1CCCCNC1=O)C#N |r,c:23| | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmc.2022.117011
BindingDB Entry DOI: 10.7270/Q2GB285Q |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50526649
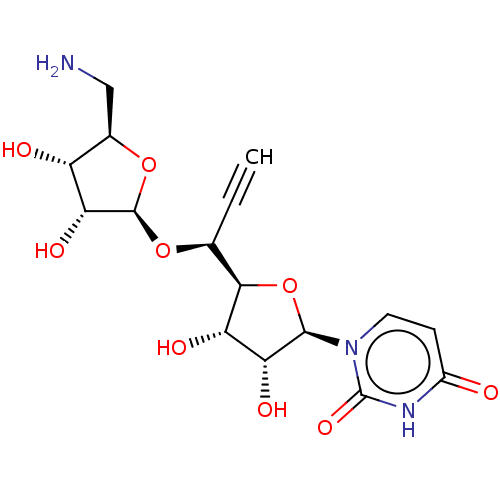
(CHEMBL4556639)Show SMILES [H][C@@]1(O[C@@H](C#C)[C@@]2([H])O[C@H]([C@H](O)[C@@H]2O)n2ccc(=O)[nH]c2=O)O[C@H](CN)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C16H21N3O9/c1-2-6(26-15-12(24)9(21)7(5-17)27-15)13-10(22)11(23)14(28-13)19-4-3-8(20)18-16(19)25/h1,3-4,6-7,9-15,21-24H,5,17H2,(H,18,20,25)/t6-,7+,9+,10-,11+,12+,13+,14+,15+/m0/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmc.2022.116744
BindingDB Entry DOI: 10.7270/Q2XG9W6P |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50526657
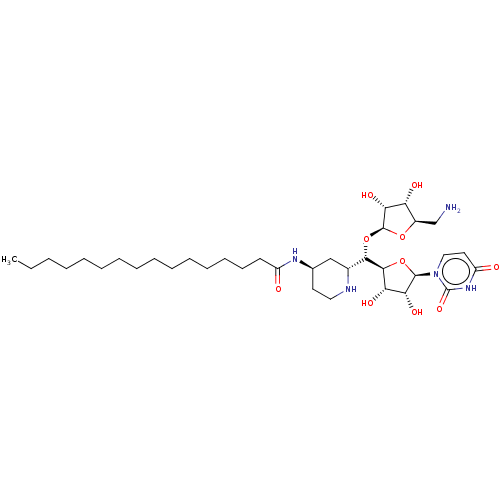
(CHEMBL4553977)Show SMILES [H][C@@]1(O[C@H]([C@@]2([H])O[C@H]([C@H](O)[C@@H]2O)n2ccc(=O)[nH]c2=O)[C@@]2([H])C[C@@H](CCN2)NC(=O)CCCCCCCCCCCCCCC)O[C@H](CN)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C35H61N5O10/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-25(41)38-22-16-18-37-23(20-22)31(50-34-30(46)27(43)24(21-36)48-34)32-28(44)29(45)33(49-32)40-19-17-26(42)39-35(40)47/h17,19,22-24,27-34,37,43-46H,2-16,18,20-21,36H2,1H3,(H,38,41)(H,39,42,47)/t22-,23-,24-,27-,28+,29-,30-,31+,32+,33-,34+/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Griffith University
Curated by ChEMBL
| Assay Description
Inhibition of MraY in Staphylococcus aureus |
Eur J Med Chem 171: 462-474 (2019)
Article DOI: 10.1016/j.ejmech.2019.01.071
BindingDB Entry DOI: 10.7270/Q2K35Z3R |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50600490

(CHEMBL5200223)Show SMILES [H][C@]12OC[C@@](O[C@H]1n1ccc(=O)[nH]c1=O)([C@@H](O[C@@H]1O[C@H](CN)[C@@H](O)[C@H]1O)C#C)[C@H]2O |r,TLB:7:6:3.2:28| | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmc.2022.116744
BindingDB Entry DOI: 10.7270/Q2XG9W6P |
More data for this
Ligand-Target Pair | |
Phospho-N-acetylmuramoyl-pentapeptide-transferase
(Staphylococcus aureus (strain MRSA252)) | BDBM50615877

(CHEMBL5273400) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 3.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data