

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50148259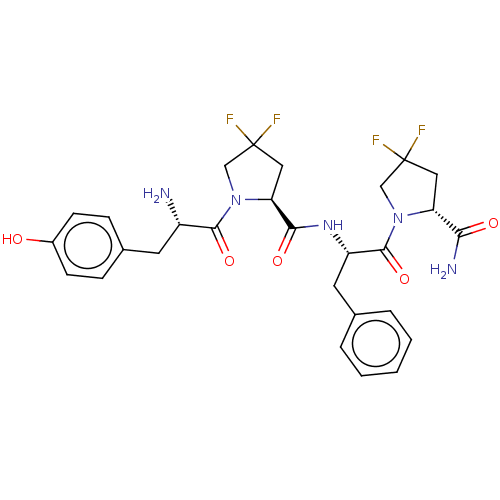 (CHEMBL3765406) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from MOR in Wistar rat brain homogenate by scintillation counting analysis | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50148257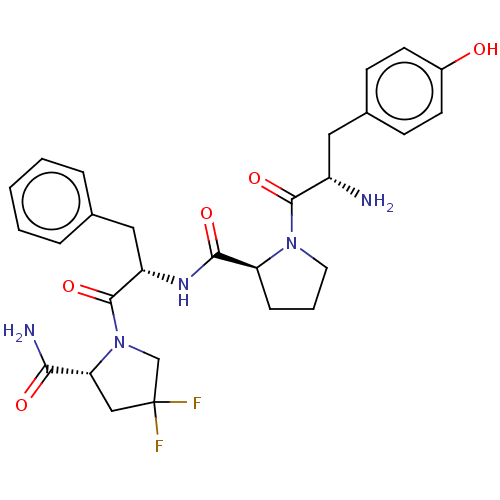 (CHEMBL3764219) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from MOR in Wistar rat brain homogenate by scintillation counting analysis | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50166066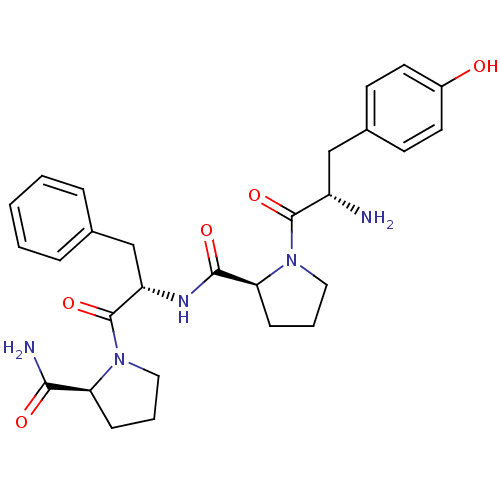 (CHEMBL362991 | H-Tyr-Pro-Phe-Pro-NH2 | MORPHICEPTI...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 36 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from MOR in Wistar rat brain homogenate by scintillation counting analysis | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50148258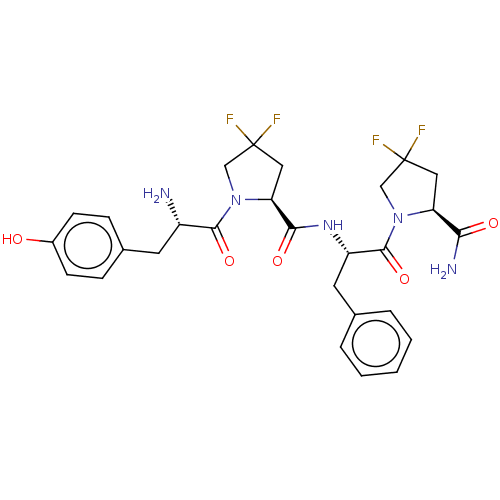 (CHEMBL3764033) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 106 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from MOR in Wistar rat brain homogenate by scintillation counting analysis | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Cavia porcellus (domestic guinea pig)) | BDBM50148259 (CHEMBL3765406) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 386 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Displacement of [3H]nor-BNI from KOR in Dunkin Hartley guinea pig brain homogenate by scintillation counting analysis | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Cavia porcellus (domestic guinea pig)) | BDBM50148257 (CHEMBL3764219) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Displacement of [3H]nor-BNI from KOR in Dunkin Hartley guinea pig brain homogenate by scintillation counting analysis | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50148259 (CHEMBL3765406) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Displacement of [3H][Ile5,6]deltorphin-2 from DOR in Wistar rat brain homogenate by scintillation counting analysis | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50148257 (CHEMBL3764219) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Displacement of [3H][Ile5,6]deltorphin-2 from DOR in Wistar rat brain homogenate by scintillation counting analysis | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Cavia porcellus (domestic guinea pig)) | BDBM50148256 (CHEMBL3765790) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Displacement of [3H]nor-BNI from KOR in Dunkin Hartley guinea pig brain homogenate by scintillation counting analysis | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50148256 (CHEMBL3765790) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from MOR in Wistar rat brain homogenate by scintillation counting analysis | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50148256 (CHEMBL3765790) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Displacement of [3H][Ile5,6]deltorphin-2 from DOR in Wistar rat brain homogenate by scintillation counting analysis | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Cavia porcellus (domestic guinea pig)) | BDBM50166066 (CHEMBL362991 | H-Tyr-Pro-Phe-Pro-NH2 | MORPHICEPTI...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Displacement of [3H]nor-BNI from KOR in Dunkin Hartley guinea pig brain homogenate by scintillation counting analysis | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Cavia porcellus (domestic guinea pig)) | BDBM50148258 (CHEMBL3764033) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Displacement of [3H]nor-BNI from KOR in Dunkin Hartley guinea pig brain homogenate by scintillation counting analysis | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50166066 (CHEMBL362991 | H-Tyr-Pro-Phe-Pro-NH2 | MORPHICEPTI...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Displacement of [3H][Ile5,6]deltorphin-2 from DOR in Wistar rat brain homogenate by scintillation counting analysis | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Rattus norvegicus (rat)) | BDBM50148258 (CHEMBL3764033) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Displacement of [3H][Ile5,6]deltorphin-2 from DOR in Wistar rat brain homogenate by scintillation counting analysis | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50148257 (CHEMBL3764219) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 20 | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Agonist activity at human recombinant MOR expressed in CHO cells by calcium mobilization assay | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM224024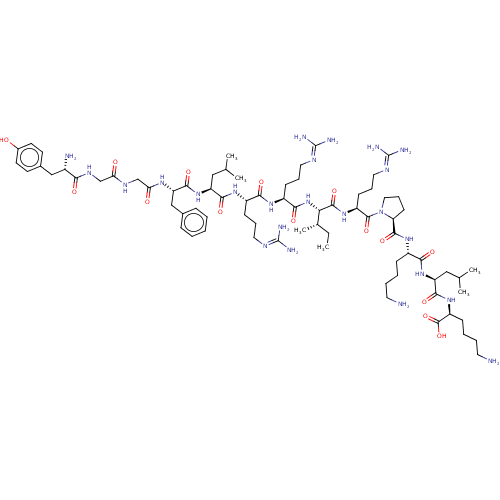 (BDBM50241435 | Dynorphin A (1-13) | YGGFLRRXRPKLK) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 19 | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Agonist activity at human recombinant DOR expressed in CHO cells by calcium mobilization assay | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM224024 (BDBM50241435 | Dynorphin A (1-13) | YGGFLRRXRPKLK) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Agonist activity at human recombinant KOR expressed in CHO cells by calcium mobilization assay | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50148258 (CHEMBL3764033) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 398 | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Agonist activity at human recombinant MOR expressed in CHO cells by calcium mobilization assay | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM224024 (BDBM50241435 | Dynorphin A (1-13) | YGGFLRRXRPKLK) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | n/a | n/a | n/a | n/a | 214 | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Agonist activity at human recombinant MOR expressed in CHO cells by calcium mobilization assay | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50142701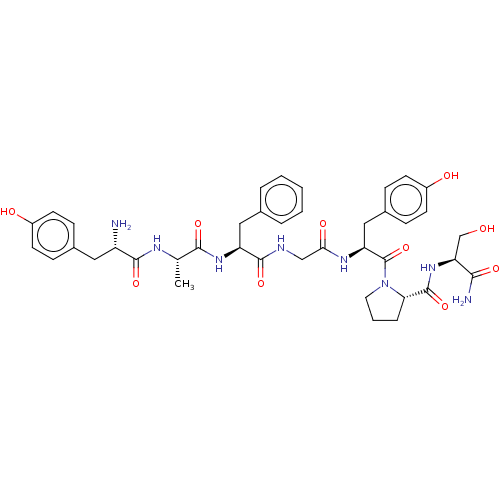 (CHEMBL285479) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 4 | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Agonist activity at human recombinant MOR expressed in CHO cells by calcium mobilization assay | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50166066 (CHEMBL362991 | H-Tyr-Pro-Phe-Pro-NH2 | MORPHICEPTI...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 741 | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Agonist activity at human recombinant MOR expressed in CHO cells by calcium mobilization assay | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50148259 (CHEMBL3765406) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 4.70 | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Agonist activity at human recombinant MOR expressed in CHO cells by calcium mobilization assay | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50148259 (CHEMBL3765406) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 813 | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Agonist activity at human recombinant KOR expressed in CHO cells by calcium mobilization assay | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM21008 ((4S,7S,13S)-13-[(2S)-2-amino-3-(4-hydroxyphenyl)pr...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 17 | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Agonist activity at human recombinant DOR expressed in CHO cells by calcium mobilization assay | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50142701 (CHEMBL285479) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 372 | n/a | n/a | n/a | n/a |
Medical University of Lodz Curated by ChEMBL | Assay Description Agonist activity at human recombinant DOR expressed in CHO cells by calcium mobilization assay | Bioorg Med Chem 24: 1582-8 (2016) Article DOI: 10.1016/j.bmc.2016.02.034 BindingDB Entry DOI: 10.7270/Q2Z321H7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||