| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM50536192 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1933085 (CHEMBL4478737) |
|---|
| IC50 | >100000±n/a nM |
|---|
| Citation |  Norcross, NR; Baraga�a, B; Wilson, C; Hallyburton, I; Osuna-Cabello, M; Norval, S; Riley, J; Stojanovski, L; Simeons, FR; Porzelle, A; Grimaldi, R; Wittlin, S; Duffy, S; Avery, VM; Meister, S; Sanz, L; Jim�nez-D�az, B; Angulo-Barturen, I; Ferrer, S; Mart�nez, MS; Gamo, FJ; Frearson, JA; Gray, DW; Fairlamb, AH; Winzeler, EA; Waterson, D; Campbell, SF; Willis, P; Read, KD; Gilbert, IH Trisubstituted Pyrimidines as Efficacious and Fast-Acting Antimalarials. J Med Chem59:6101-20 (2016) [PubMed] Article Norcross, NR; Baraga�a, B; Wilson, C; Hallyburton, I; Osuna-Cabello, M; Norval, S; Riley, J; Stojanovski, L; Simeons, FR; Porzelle, A; Grimaldi, R; Wittlin, S; Duffy, S; Avery, VM; Meister, S; Sanz, L; Jim�nez-D�az, B; Angulo-Barturen, I; Ferrer, S; Mart�nez, MS; Gamo, FJ; Frearson, JA; Gray, DW; Fairlamb, AH; Winzeler, EA; Waterson, D; Campbell, SF; Willis, P; Read, KD; Gilbert, IH Trisubstituted Pyrimidines as Efficacious and Fast-Acting Antimalarials. J Med Chem59:6101-20 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM50536192 |
|---|
| n/a |
|---|
| Name | BDBM50536192 |
|---|
| Synonyms: | CHEMBL4570567 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H30N6O |
|---|
| Mol. Mass. | 430.5453 |
|---|
| SMILES | Cc1cc(cc(C)n1)-c1nc(cc(n1)-c1cccnc1)N1CCC(CC1)N1CCOCC1 |
|---|
| Structure | 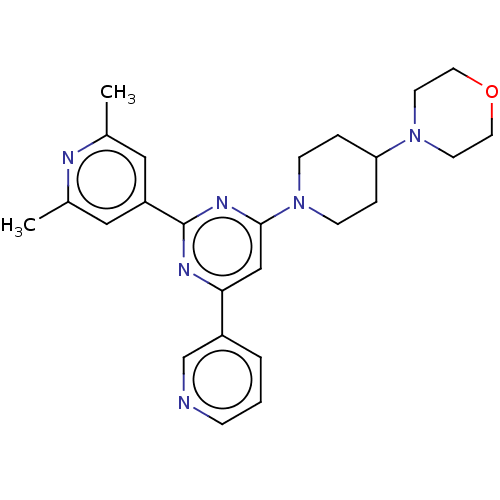
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Norcross, NR; Baraga�a, B; Wilson, C; Hallyburton, I; Osuna-Cabello, M; Norval, S; Riley, J; Stojanovski, L; Simeons, FR; Porzelle, A; Grimaldi, R; Wittlin, S; Duffy, S; Avery, VM; Meister, S; Sanz, L; Jim�nez-D�az, B; Angulo-Barturen, I; Ferrer, S; Mart�nez, MS; Gamo, FJ; Frearson, JA; Gray, DW; Fairlamb, AH; Winzeler, EA; Waterson, D; Campbell, SF; Willis, P; Read, KD; Gilbert, IH Trisubstituted Pyrimidines as Efficacious and Fast-Acting Antimalarials. J Med Chem59:6101-20 (2016) [PubMed] Article
Norcross, NR; Baraga�a, B; Wilson, C; Hallyburton, I; Osuna-Cabello, M; Norval, S; Riley, J; Stojanovski, L; Simeons, FR; Porzelle, A; Grimaldi, R; Wittlin, S; Duffy, S; Avery, VM; Meister, S; Sanz, L; Jim�nez-D�az, B; Angulo-Barturen, I; Ferrer, S; Mart�nez, MS; Gamo, FJ; Frearson, JA; Gray, DW; Fairlamb, AH; Winzeler, EA; Waterson, D; Campbell, SF; Willis, P; Read, KD; Gilbert, IH Trisubstituted Pyrimidines as Efficacious and Fast-Acting Antimalarials. J Med Chem59:6101-20 (2016) [PubMed] Article