| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C9 |
|---|
| Ligand | BDBM50557806 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2060127 (CHEMBL4715128) |
|---|
| IC50 | 8500±n/a nM |
|---|
| Citation |  Liu, Q; Xiao, HY; Batt, DG; Xiao, Z; Zhu, Y; Yang, MG; Li, N; Yip, S; Li, P; Sun, D; Wu, DR; Ruzanov, M; Sack, JS; Weigelt, CA; Wang, J; Li, S; Shuster, DJ; Xie, JH; Song, Y; Sherry, T; Obermeier, MT; Fura, A; Stefanski, K; Cornelius, G; Chacko, S; Khandelwal, P; Dudhgaonkar, S; Rudra, A; Nagar, J; Murali, V; Govindarajan, A; Denton, R; Zhao, Q; Meanwell, NA; Borzilleri, R; Dhar, TGM Azatricyclic Inverse Agonists of ROR?t That Demonstrate Efficacy in Models of Rheumatoid Arthritis and Psoriasis. ACS Med Chem Lett12:827-835 (2021) [PubMed] Article Liu, Q; Xiao, HY; Batt, DG; Xiao, Z; Zhu, Y; Yang, MG; Li, N; Yip, S; Li, P; Sun, D; Wu, DR; Ruzanov, M; Sack, JS; Weigelt, CA; Wang, J; Li, S; Shuster, DJ; Xie, JH; Song, Y; Sherry, T; Obermeier, MT; Fura, A; Stefanski, K; Cornelius, G; Chacko, S; Khandelwal, P; Dudhgaonkar, S; Rudra, A; Nagar, J; Murali, V; Govindarajan, A; Denton, R; Zhao, Q; Meanwell, NA; Borzilleri, R; Dhar, TGM Azatricyclic Inverse Agonists of ROR?t That Demonstrate Efficacy in Models of Rheumatoid Arthritis and Psoriasis. ACS Med Chem Lett12:827-835 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C9 |
|---|
| Name: | Cytochrome P450 2C9 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55636.33 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11712 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKV
YGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFM
KSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYID
LLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFK
KSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM50557806 |
|---|
| n/a |
|---|
| Name | BDBM50557806 |
|---|
| Synonyms: | CHEMBL4800380 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H26F8N2O4S |
|---|
| Mol. Mass. | 614.548 |
|---|
| SMILES | [H][C@@]12CCc3nc(ccc3[C@@]1(CC[C@H]2NC(=O)CC(C)(C)O)S(=O)(=O)c1ccc(F)cc1)C(F)(C(F)(F)F)C(F)(F)F |r| |
|---|
| Structure | 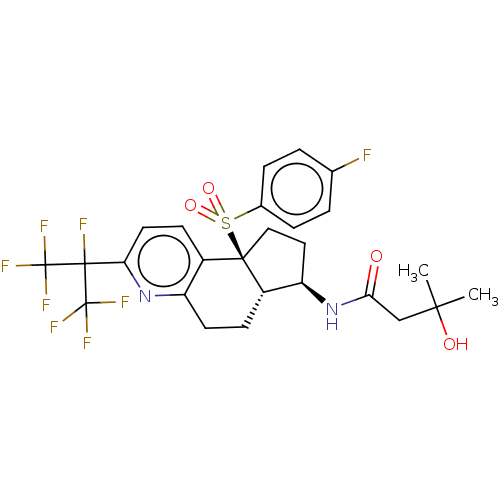
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Liu, Q; Xiao, HY; Batt, DG; Xiao, Z; Zhu, Y; Yang, MG; Li, N; Yip, S; Li, P; Sun, D; Wu, DR; Ruzanov, M; Sack, JS; Weigelt, CA; Wang, J; Li, S; Shuster, DJ; Xie, JH; Song, Y; Sherry, T; Obermeier, MT; Fura, A; Stefanski, K; Cornelius, G; Chacko, S; Khandelwal, P; Dudhgaonkar, S; Rudra, A; Nagar, J; Murali, V; Govindarajan, A; Denton, R; Zhao, Q; Meanwell, NA; Borzilleri, R; Dhar, TGM Azatricyclic Inverse Agonists of ROR?t That Demonstrate Efficacy in Models of Rheumatoid Arthritis and Psoriasis. ACS Med Chem Lett12:827-835 (2021) [PubMed] Article
Liu, Q; Xiao, HY; Batt, DG; Xiao, Z; Zhu, Y; Yang, MG; Li, N; Yip, S; Li, P; Sun, D; Wu, DR; Ruzanov, M; Sack, JS; Weigelt, CA; Wang, J; Li, S; Shuster, DJ; Xie, JH; Song, Y; Sherry, T; Obermeier, MT; Fura, A; Stefanski, K; Cornelius, G; Chacko, S; Khandelwal, P; Dudhgaonkar, S; Rudra, A; Nagar, J; Murali, V; Govindarajan, A; Denton, R; Zhao, Q; Meanwell, NA; Borzilleri, R; Dhar, TGM Azatricyclic Inverse Agonists of ROR?t That Demonstrate Efficacy in Models of Rheumatoid Arthritis and Psoriasis. ACS Med Chem Lett12:827-835 (2021) [PubMed] Article