| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor VII |
|---|
| Ligand | BDBM13592 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_979046 (CHEMBL2421099) |
|---|
| IC50 | 5.0±n/a nM |
|---|
| Citation |  Bolton, SA; Sutton, JC; Anumula, R; Bisacchi, GS; Jacobson, B; Slusarchyk, WA; Treuner, UD; Wu, SC; Zhao, G; Pi, Z; Sheriff, S; Smirk, RA; Bisaha, S; Cheney, DL; Wei, A; Schumacher, WA; Hartl, KS; Liu, E; Zahler, R; Seiler, SM Discovery of nonbenzamidine factor VIIa inhibitors using a biaryl acid scaffold. Bioorg Med Chem Lett23:5239-43 (2013) [PubMed] Article Bolton, SA; Sutton, JC; Anumula, R; Bisacchi, GS; Jacobson, B; Slusarchyk, WA; Treuner, UD; Wu, SC; Zhao, G; Pi, Z; Sheriff, S; Smirk, RA; Bisaha, S; Cheney, DL; Wei, A; Schumacher, WA; Hartl, KS; Liu, E; Zahler, R; Seiler, SM Discovery of nonbenzamidine factor VIIa inhibitors using a biaryl acid scaffold. Bioorg Med Chem Lett23:5239-43 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Coagulation factor VII |
|---|
| Name: | Coagulation factor VII |
|---|
| Synonyms: | Eptacog alfa | F7 | FA7_HUMAN | Factor VIIa | Factor VIIa (fVIIa) | Proconvertin | SPCA | Thrombin and coagulation factor VII | serum prothrombin conversion accelerator |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 51599.89 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 466 |
|---|
| Sequence: | MVSQALRLLCLLLGLQGCLAAGGVAKASGGETRDMPWKPGPHRVFVTQEEAHGVLHRRRR
ANAFLEELRPGSLERECKEEQCSFEEAREIFKDAERTKLFWISYSDGDQCASSPCQNGGS
CKDQLQSYICFCLPAFEGRNCETHKDDQLICVNENGGCEQYCSDHTGTKRSCRCHEGYSL
LADGVSCTPTVEYPCGKIPILEKRNASKPQGRIVGGKVCPKGECPWQVLLLVNGAQLCGG
TLINTIWVVSAAHCFDKIKNWRNLIAVLGEHDLSEHDGDEQSRRVAQVIIPSTYVPGTTN
HDIALLRLHQPVVLTDHVVPLCLPERTFSERTLAFVRFSLVSGWGQLLDRGATALELMVL
NVPRLMTQDCLQQSRKVGDSPNITEYMFCAGYSDGSKDSCKGDSGGPHATHYRGTWYLTG
IVSWGQGCATVGHFGVYTRVSQYIEWLQKLMRSEPRPGVLLRAPFP
|
|
|
|---|
| BDBM13592 |
|---|
| n/a |
|---|
| Name | BDBM13592 |
|---|
| Synonyms: | 2-{2-[(4-carbamimidoylphenyl)carbamoyl]-6-methoxypyridin-3-yl}-5-{[(2S)-1-hydroxy-3,3-dimethylbutan-2-yl]carbamoyl}benzoic acid | Ono Compound 8 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H31N5O6 |
|---|
| Mol. Mass. | 533.5756 |
|---|
| SMILES | COc1ccc(-c2ccc(cc2C(O)=O)C(=O)N[C@H](CO)C(C)(C)C)c(n1)C(=O)Nc1ccc(cc1)C(N)=N |r| |
|---|
| Structure | 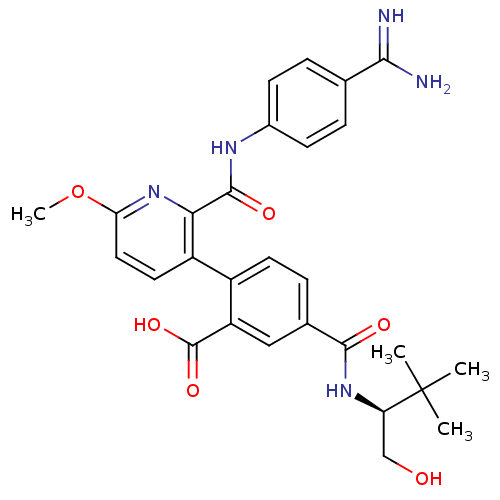
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bolton, SA; Sutton, JC; Anumula, R; Bisacchi, GS; Jacobson, B; Slusarchyk, WA; Treuner, UD; Wu, SC; Zhao, G; Pi, Z; Sheriff, S; Smirk, RA; Bisaha, S; Cheney, DL; Wei, A; Schumacher, WA; Hartl, KS; Liu, E; Zahler, R; Seiler, SM Discovery of nonbenzamidine factor VIIa inhibitors using a biaryl acid scaffold. Bioorg Med Chem Lett23:5239-43 (2013) [PubMed] Article
Bolton, SA; Sutton, JC; Anumula, R; Bisacchi, GS; Jacobson, B; Slusarchyk, WA; Treuner, UD; Wu, SC; Zhao, G; Pi, Z; Sheriff, S; Smirk, RA; Bisaha, S; Cheney, DL; Wei, A; Schumacher, WA; Hartl, KS; Liu, E; Zahler, R; Seiler, SM Discovery of nonbenzamidine factor VIIa inhibitors using a biaryl acid scaffold. Bioorg Med Chem Lett23:5239-43 (2013) [PubMed] Article