| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Receptor tyrosine-protein kinase erbB-2 |
|---|
| Ligand | BDBM27879 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_321396 (CHEMBL880631) |
|---|
| IC50 | 17100±n/a nM |
|---|
| Citation |  Wittman, M; Carboni, J; Attar, R; Balasubramanian, B; Balimane, P; Brassil, P; Beaulieu, F; Chang, C; Clarke, W; Dell, J; Eummer, J; Frennesson, D; Gottardis, M; Greer, A; Hansel, S; Hurlburt, W; Jacobson, B; Krishnananthan, S; Lee, FY; Li, A; Lin, TA; Liu, P; Ouellet, C; Sang, X; Saulnier, MG; Stoffan, K; Sun, Y; Velaparthi, U; Wong, H; Yang, Z; Zimmermann, K; Zoeckler, M; Vyas, D Discovery of a (1H-benzoimidazol-2-yl)-1H-pyridin-2-one (BMS-536924) inhibitor of insulin-like growth factor I receptor kinase with in vivo antitumor activity. J Med Chem48:5639-43 (2005) [PubMed] Article Wittman, M; Carboni, J; Attar, R; Balasubramanian, B; Balimane, P; Brassil, P; Beaulieu, F; Chang, C; Clarke, W; Dell, J; Eummer, J; Frennesson, D; Gottardis, M; Greer, A; Hansel, S; Hurlburt, W; Jacobson, B; Krishnananthan, S; Lee, FY; Li, A; Lin, TA; Liu, P; Ouellet, C; Sang, X; Saulnier, MG; Stoffan, K; Sun, Y; Velaparthi, U; Wong, H; Yang, Z; Zimmermann, K; Zoeckler, M; Vyas, D Discovery of a (1H-benzoimidazol-2-yl)-1H-pyridin-2-one (BMS-536924) inhibitor of insulin-like growth factor I receptor kinase with in vivo antitumor activity. J Med Chem48:5639-43 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Receptor tyrosine-protein kinase erbB-2 |
|---|
| Name: | Receptor tyrosine-protein kinase erbB-2 |
|---|
| Synonyms: | 2.7.10.1 | C-erbB-2 | CD_antigen=CD340 | ERBB2 | ERBB2_HUMAN | ErbB-2/ErbB-3 heterodimer | FASN/HER2 | HER-2 Substrate | HER2 | MLN 19 | MLN19 | Metastatic lymph node gene 19 protein | NEU | NGL | Proto-oncogene Neu | Proto-oncogene c-ErbB-2 | Tyrosine kinase-type cell surface receptor HER2 | p185erbB2 |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 137894.47 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P04626 |
|---|
| Residue: | 1255 |
|---|
| Sequence: | MELAALCRWGLLLALLPPGAASTQVCTGTDMKLRLPASPETHLDMLRHLYQGCQVVQGNL
ELTYLPTNASLSFLQDIQEVQGYVLIAHNQVRQVPLQRLRIVRGTQLFEDNYALAVLDNG
DPLNNTTPVTGASPGGLRELQLRSLTEILKGGVLIQRNPQLCYQDTILWKDIFHKNNQLA
LTLIDTNRSRACHPCSPMCKGSRCWGESSEDCQSLTRTVCAGGCARCKGPLPTDCCHEQC
AAGCTGPKHSDCLACLHFNHSGICELHCPALVTYNTDTFESMPNPEGRYTFGASCVTACP
YNYLSTDVGSCTLVCPLHNQEVTAEDGTQRCEKCSKPCARVCYGLGMEHLREVRAVTSAN
IQEFAGCKKIFGSLAFLPESFDGDPASNTAPLQPEQLQVFETLEEITGYLYISAWPDSLP
DLSVFQNLQVIRGRILHNGAYSLTLQGLGISWLGLRSLRELGSGLALIHHNTHLCFVHTV
PWDQLFRNPHQALLHTANRPEDECVGEGLACHQLCARGHCWGPGPTQCVNCSQFLRGQEC
VEECRVLQGLPREYVNARHCLPCHPECQPQNGSVTCFGPEADQCVACAHYKDPPFCVARC
PSGVKPDLSYMPIWKFPDEEGACQPCPINCTHSCVDLDDKGCPAEQRASPLTSIISAVVG
ILLVVVLGVVFGILIKRRQQKIRKYTMRRLLQETELVEPLTPSGAMPNQAQMRILKETEL
RKVKVLGSGAFGTVYKGIWIPDGENVKIPVAIKVLRENTSPKANKEILDEAYVMAGVGSP
YVSRLLGICLTSTVQLVTQLMPYGCLLDHVRENRGRLGSQDLLNWCMQIAKGMSYLEDVR
LVHRDLAARNVLVKSPNHVKITDFGLARLLDIDETEYHADGGKVPIKWMALESILRRRFT
HQSDVWSYGVTVWELMTFGAKPYDGIPAREIPDLLEKGERLPQPPICTIDVYMIMVKCWM
IDSECRPRFRELVSEFSRMARDPQRFVVIQNEDLGPASPLDSTFYRSLLEDDDMGDLVDA
EEYLVPQQGFFCPDPAPGAGGMVHHRHRSSSTRSGGGDLTLGLEPSEEEAPRSPLAPSEG
AGSDVFDGDLGMGAAKGLQSLPTHDPSPLQRYSEDPTVPLPSETDGYVAPLTCSPQPEYV
NQPDVRPQPPSPREGPLPAARPAGATLERPKTLSPGKNGVVKDVFAFGGAVENPEYLTPQ
GGAAPQPHPPPAFSPAFDNLYYWDQDPPERGAPPSTFKGTPTAENPEYLGLDVPV
|
|
|
|---|
| BDBM27879 |
|---|
| n/a |
|---|
| Name | BDBM27879 |
|---|
| Synonyms: | 4-{[(2S)-2-(3-chlorophenyl)-2-hydroxyethyl]amino}-3-[4-methyl-6-(morpholin-4-yl)-1H-1,3-benzodiazol-2-yl]-1,2-dihydropyridin-2-one | BMS-536924 | CHEMBL401930 | US10710978, Compound BMS536924 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H26ClN5O3 |
|---|
| Mol. Mass. | 479.959 |
|---|
| SMILES | Cc1cc(cc2[nH]c(nc12)-c1c(NC[C@@H](O)c2cccc(Cl)c2)cc[nH]c1=O)N1CCOCC1 |r| |
|---|
| Structure | 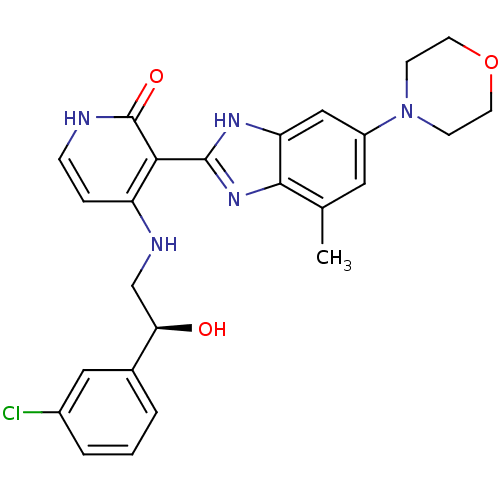
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wittman, M; Carboni, J; Attar, R; Balasubramanian, B; Balimane, P; Brassil, P; Beaulieu, F; Chang, C; Clarke, W; Dell, J; Eummer, J; Frennesson, D; Gottardis, M; Greer, A; Hansel, S; Hurlburt, W; Jacobson, B; Krishnananthan, S; Lee, FY; Li, A; Lin, TA; Liu, P; Ouellet, C; Sang, X; Saulnier, MG; Stoffan, K; Sun, Y; Velaparthi, U; Wong, H; Yang, Z; Zimmermann, K; Zoeckler, M; Vyas, D Discovery of a (1H-benzoimidazol-2-yl)-1H-pyridin-2-one (BMS-536924) inhibitor of insulin-like growth factor I receptor kinase with in vivo antitumor activity. J Med Chem48:5639-43 (2005) [PubMed] Article
Wittman, M; Carboni, J; Attar, R; Balasubramanian, B; Balimane, P; Brassil, P; Beaulieu, F; Chang, C; Clarke, W; Dell, J; Eummer, J; Frennesson, D; Gottardis, M; Greer, A; Hansel, S; Hurlburt, W; Jacobson, B; Krishnananthan, S; Lee, FY; Li, A; Lin, TA; Liu, P; Ouellet, C; Sang, X; Saulnier, MG; Stoffan, K; Sun, Y; Velaparthi, U; Wong, H; Yang, Z; Zimmermann, K; Zoeckler, M; Vyas, D Discovery of a (1H-benzoimidazol-2-yl)-1H-pyridin-2-one (BMS-536924) inhibitor of insulin-like growth factor I receptor kinase with in vivo antitumor activity. J Med Chem48:5639-43 (2005) [PubMed] Article