 Found 90 hits with Last Name = 'hurlburt' and Initial = 'w'
Found 90 hits with Last Name = 'hurlburt' and Initial = 'w' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50325424

((S)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES Fc1ccc(NC(=O)[C@@H]2CCCN2c2nc(Nc3cc([nH]n3)C3CC3)c3cccn3n2)cn1 |r| Show InChI InChI=1S/C22H22FN9O/c23-18-8-7-14(12-24-18)25-21(33)17-4-1-9-31(17)22-27-20(16-3-2-10-32(16)30-22)26-19-11-15(28-29-19)13-5-6-13/h2-3,7-8,10-13,17H,1,4-6,9H2,(H,25,33)(H2,26,27,28,29,30)/t17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R in IGF1R-SAL cells |
Bioorg Med Chem Lett 20: 5027-30 (2010)
Article DOI: 10.1016/j.bmcl.2010.07.045
BindingDB Entry DOI: 10.7270/Q2WH2Q5J |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50299148

((S)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES C[C@]1(CCCN1c1nc(Nc2cc([nH]n2)C2CC2)c2cccn2n1)C(=O)Nc1ccc(F)nc1 |r| Show InChI InChI=1S/C23H24FN9O/c1-23(21(34)26-15-7-8-18(24)25-13-15)9-3-10-32(23)22-28-20(17-4-2-11-33(17)31-22)27-19-12-16(29-30-19)14-5-6-14/h2,4,7-8,11-14H,3,5-6,9-10H2,1H3,(H,26,34)(H2,27,28,29,30,31)/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R after 60 mins by fluorescence electrophoresis |
J Med Chem 52: 7360-3 (2009)
Article DOI: 10.1021/jm900786r
BindingDB Entry DOI: 10.7270/Q2P27022 |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 2/G1/S-specific cyclin-E1/G1/S-specific cyclin-E2
(Homo sapiens (Human)) | BDBM50299138
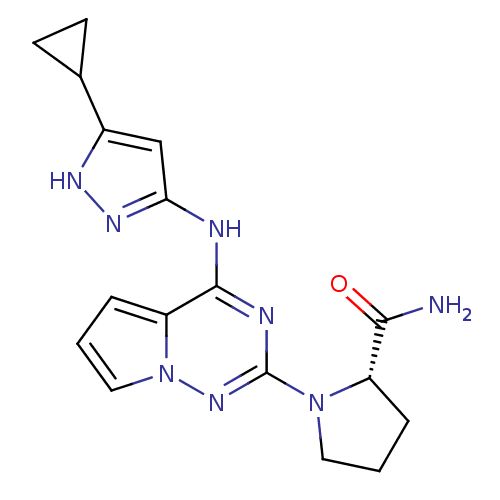
((S)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES NC(=O)[C@@H]1CCCN1c1nc(Nc2cc([nH]n2)C2CC2)c2cccn2n1 |r| Show InChI InChI=1S/C17H20N8O/c18-15(26)12-3-1-7-24(12)17-20-16(13-4-2-8-25(13)23-17)19-14-9-11(21-22-14)10-5-6-10/h2,4,8-10,12H,1,3,5-7H2,(H2,18,26)(H2,19,20,21,22,23)/t12-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E after 60 mins by fluorescence electrophoresis |
J Med Chem 52: 7360-3 (2009)
Article DOI: 10.1021/jm900786r
BindingDB Entry DOI: 10.7270/Q2P27022 |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50299143
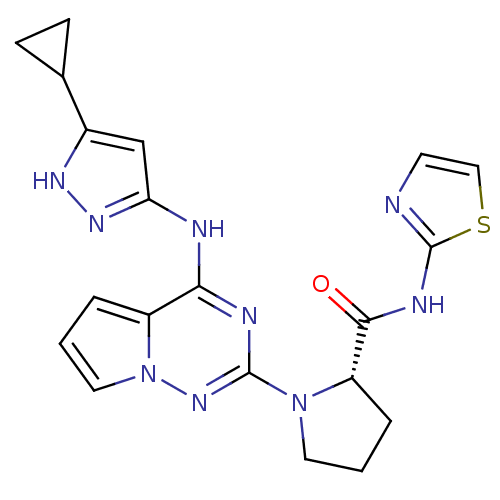
((S)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES O=C(Nc1nccs1)[C@@H]1CCCN1c1nc(Nc2cc([nH]n2)C2CC2)c2cccn2n1 |r| Show InChI InChI=1S/C20H21N9OS/c30-18(24-20-21-7-10-31-20)15-4-1-8-28(15)19-23-17(14-3-2-9-29(14)27-19)22-16-11-13(25-26-16)12-5-6-12/h2-3,7,9-12,15H,1,4-6,8H2,(H,21,24,30)(H2,22,23,25,26,27)/t15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R after 60 mins by fluorescence electrophoresis |
J Med Chem 52: 7360-3 (2009)
Article DOI: 10.1021/jm900786r
BindingDB Entry DOI: 10.7270/Q2P27022 |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50299148

((S)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES C[C@]1(CCCN1c1nc(Nc2cc([nH]n2)C2CC2)c2cccn2n1)C(=O)Nc1ccc(F)nc1 |r| Show InChI InChI=1S/C23H24FN9O/c1-23(21(34)26-15-7-8-18(24)25-13-15)9-3-10-32(23)22-28-20(17-4-2-11-33(17)31-22)27-19-12-16(29-30-19)14-5-6-14/h2,4,7-8,11-14H,3,5-6,9-10H2,1H3,(H,26,34)(H2,27,28,29,30,31)/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R in IGF1R-SAL cells |
Bioorg Med Chem Lett 20: 5027-30 (2010)
Article DOI: 10.1016/j.bmcl.2010.07.045
BindingDB Entry DOI: 10.7270/Q2WH2Q5J |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50325424

((S)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES Fc1ccc(NC(=O)[C@@H]2CCCN2c2nc(Nc3cc([nH]n3)C3CC3)c3cccn3n2)cn1 |r| Show InChI InChI=1S/C22H22FN9O/c23-18-8-7-14(12-24-18)25-21(33)17-4-1-9-31(17)22-27-20(16-3-2-10-32(16)30-22)26-19-11-15(28-29-19)13-5-6-13/h2-3,7-8,10-13,17H,1,4-6,9H2,(H,25,33)(H2,26,27,28,29,30)/t17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R |
Bioorg Med Chem Lett 20: 5027-30 (2010)
Article DOI: 10.1016/j.bmcl.2010.07.045
BindingDB Entry DOI: 10.7270/Q2WH2Q5J |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM107003
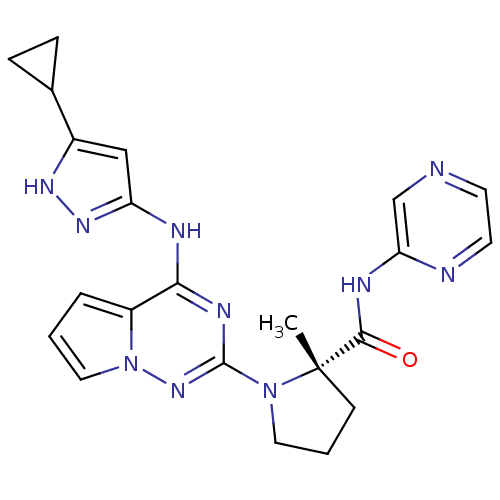
(CHEMBL575447 | US8592579, 110)Show SMILES C[C@]1(CCCN1c1nc(Nc2cc([nH]n2)C2CC2)c2cccn2n1)C(=O)Nc1cnccn1 |r| Show InChI InChI=1S/C22H24N10O/c1-22(20(33)26-18-13-23-8-9-24-18)7-3-10-31(22)21-27-19(16-4-2-11-32(16)30-21)25-17-12-15(28-29-17)14-5-6-14/h2,4,8-9,11-14H,3,5-7,10H2,1H3,(H,24,26,33)(H2,25,27,28,29,30)/t22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R after 60 mins by fluorescence electrophoresis |
J Med Chem 52: 7360-3 (2009)
Article DOI: 10.1021/jm900786r
BindingDB Entry DOI: 10.7270/Q2P27022 |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50299145
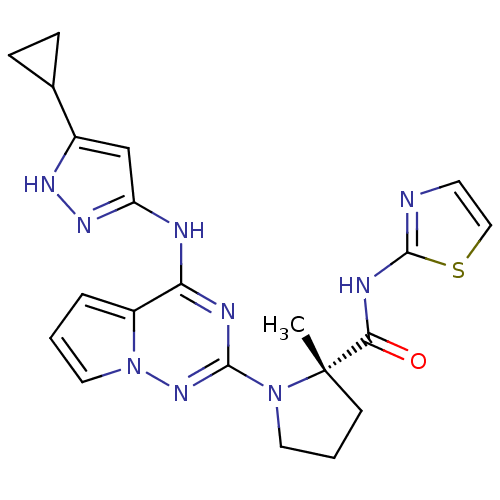
((S)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES C[C@]1(CCCN1c1nc(Nc2cc([nH]n2)C2CC2)c2cccn2n1)C(=O)Nc1nccs1 |r| Show InChI InChI=1S/C21H23N9OS/c1-21(18(31)25-20-22-8-11-32-20)7-3-9-29(21)19-24-17(15-4-2-10-30(15)28-19)23-16-12-14(26-27-16)13-5-6-13/h2,4,8,10-13H,3,5-7,9H2,1H3,(H,22,25,31)(H2,23,24,26,27,28)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R after 60 mins by fluorescence electrophoresis |
J Med Chem 52: 7360-3 (2009)
Article DOI: 10.1021/jm900786r
BindingDB Entry DOI: 10.7270/Q2P27022 |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50325430

(CHEMBL1222855 | N-((4-(5-cyclopropyl-1H-pyrazol-3-...)Show SMILES Fc1ccc(cn1)C(=O)NCc1nc(Nc2cc(n[nH]2)C2CC2)c2cccn2n1 Show InChI InChI=1S/C19H17FN8O/c20-15-6-5-12(9-21-15)19(29)22-10-17-24-18(14-2-1-7-28(14)27-17)23-16-8-13(25-26-16)11-3-4-11/h1-2,5-9,11H,3-4,10H2,(H,22,29)(H2,23,24,25,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R |
Bioorg Med Chem Lett 20: 5027-30 (2010)
Article DOI: 10.1016/j.bmcl.2010.07.045
BindingDB Entry DOI: 10.7270/Q2WH2Q5J |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50299146
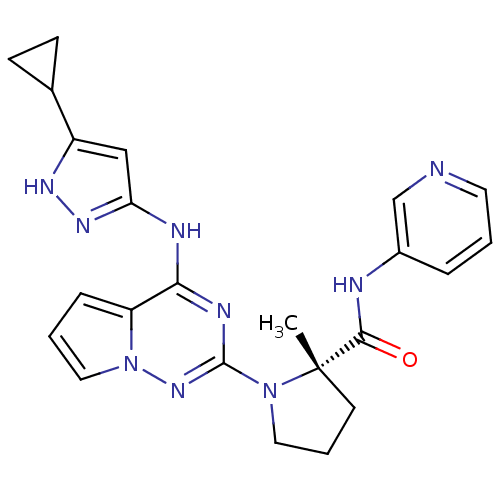
((S)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES C[C@]1(CCCN1c1nc(Nc2cc([nH]n2)C2CC2)c2cccn2n1)C(=O)Nc1cccnc1 |r| Show InChI InChI=1S/C23H25N9O/c1-23(21(33)25-16-5-2-10-24-14-16)9-4-11-31(23)22-27-20(18-6-3-12-32(18)30-22)26-19-13-17(28-29-19)15-7-8-15/h2-3,5-6,10,12-15H,4,7-9,11H2,1H3,(H,25,33)(H2,26,27,28,29,30)/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R after 60 mins by fluorescence electrophoresis |
J Med Chem 52: 7360-3 (2009)
Article DOI: 10.1021/jm900786r
BindingDB Entry DOI: 10.7270/Q2P27022 |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50325425

((S)-(2-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrr...)Show SMILES Fc1ccc(cn1)C(=O)N1CCC[C@H]1c1nc(Nc2cc(n[nH]2)C2CC2)c2cccn2n1 |r| Show InChI InChI=1S/C22H21FN8O/c23-18-8-7-14(12-24-18)22(32)30-9-1-3-16(30)21-26-20(17-4-2-10-31(17)29-21)25-19-11-15(27-28-19)13-5-6-13/h2,4,7-8,10-13,16H,1,3,5-6,9H2,(H2,25,26,27,28,29)/t16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R |
Bioorg Med Chem Lett 20: 5027-30 (2010)
Article DOI: 10.1016/j.bmcl.2010.07.045
BindingDB Entry DOI: 10.7270/Q2WH2Q5J |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50299148

((S)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES C[C@]1(CCCN1c1nc(Nc2cc([nH]n2)C2CC2)c2cccn2n1)C(=O)Nc1ccc(F)nc1 |r| Show InChI InChI=1S/C23H24FN9O/c1-23(21(34)26-15-7-8-18(24)25-13-15)9-3-10-32(23)22-28-20(17-4-2-11-33(17)31-22)27-19-12-16(29-30-19)14-5-6-14/h2,4,7-8,11-14H,3,5-6,9-10H2,1H3,(H,26,34)(H2,27,28,29,30,31)/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R |
Bioorg Med Chem Lett 20: 5027-30 (2010)
Article DOI: 10.1016/j.bmcl.2010.07.045
BindingDB Entry DOI: 10.7270/Q2WH2Q5J |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50299144

((S)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES O=C(Nc1cccnc1)[C@@H]1CCCN1c1nc(Nc2cc([nH]n2)C2CC2)c2cccn2n1 |r| Show InChI InChI=1S/C22H23N9O/c32-21(24-15-4-1-9-23-13-15)18-6-2-10-30(18)22-26-20(17-5-3-11-31(17)29-22)25-19-12-16(27-28-19)14-7-8-14/h1,3-5,9,11-14,18H,2,6-8,10H2,(H,24,32)(H2,25,26,27,28,29)/t18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R after 60 mins by fluorescence electrophoresis |
J Med Chem 52: 7360-3 (2009)
Article DOI: 10.1021/jm900786r
BindingDB Entry DOI: 10.7270/Q2P27022 |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50325428
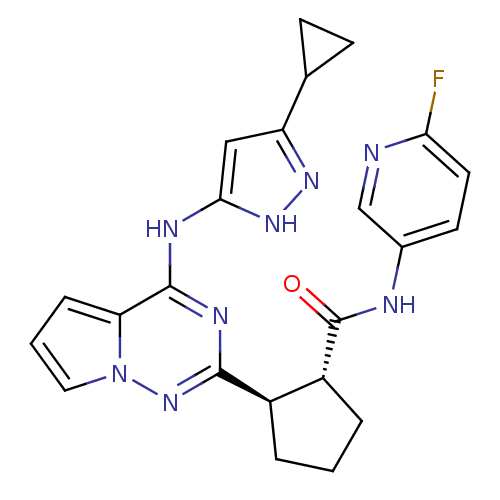
(CHEMBL1222791 | trans-2-(4-(5-cyclopropyl-1H-pyraz...)Show SMILES Fc1ccc(NC(=O)[C@@H]2CCC[C@H]2c2nc(Nc3cc(n[nH]3)C3CC3)c3cccn3n2)cn1 |r| Show InChI InChI=1S/C23H23FN8O/c24-19-9-8-14(12-25-19)26-23(33)16-4-1-3-15(16)21-28-22(18-5-2-10-32(18)31-21)27-20-11-17(29-30-20)13-6-7-13/h2,5,8-13,15-16H,1,3-4,6-7H2,(H,26,33)(H2,27,28,29,30,31)/t15-,16-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R |
Bioorg Med Chem Lett 20: 5027-30 (2010)
Article DOI: 10.1016/j.bmcl.2010.07.045
BindingDB Entry DOI: 10.7270/Q2WH2Q5J |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50325428

(CHEMBL1222791 | trans-2-(4-(5-cyclopropyl-1H-pyraz...)Show SMILES Fc1ccc(NC(=O)[C@@H]2CCC[C@H]2c2nc(Nc3cc(n[nH]3)C3CC3)c3cccn3n2)cn1 |r| Show InChI InChI=1S/C23H23FN8O/c24-19-9-8-14(12-25-19)26-23(33)16-4-1-3-15(16)21-28-22(18-5-2-10-32(18)31-21)27-20-11-17(29-30-20)13-6-7-13/h2,5,8-13,15-16H,1,3-4,6-7H2,(H,26,33)(H2,27,28,29,30,31)/t15-,16-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R |
Bioorg Med Chem Lett 20: 5027-30 (2010)
Article DOI: 10.1016/j.bmcl.2010.07.045
BindingDB Entry DOI: 10.7270/Q2WH2Q5J |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50325427
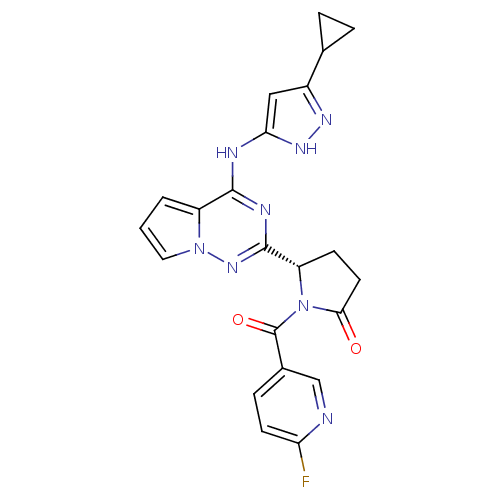
((S)-5-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES Fc1ccc(cn1)C(=O)N1[C@@H](CCC1=O)c1nc(Nc2cc(n[nH]2)C2CC2)c2cccn2n1 |r| Show InChI InChI=1S/C22H19FN8O2/c23-17-7-5-13(11-24-17)22(33)31-16(6-8-19(31)32)21-26-20(15-2-1-9-30(15)29-21)25-18-10-14(27-28-18)12-3-4-12/h1-2,5,7,9-12,16H,3-4,6,8H2,(H2,25,26,27,28,29)/t16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R |
Bioorg Med Chem Lett 20: 5027-30 (2010)
Article DOI: 10.1016/j.bmcl.2010.07.045
BindingDB Entry DOI: 10.7270/Q2WH2Q5J |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Mus musculus) | BDBM50299148

((S)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES C[C@]1(CCCN1c1nc(Nc2cc([nH]n2)C2CC2)c2cccn2n1)C(=O)Nc1ccc(F)nc1 |r| Show InChI InChI=1S/C23H24FN9O/c1-23(21(34)26-15-7-8-18(24)25-13-15)9-3-10-32(23)22-28-20(17-4-2-11-33(17)31-22)27-19-12-16(29-30-19)14-5-6-14/h2,4,7-8,11-14H,3,5-6,9-10H2,1H3,(H,26,34)(H2,27,28,29,30,31)/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| MMDB
Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R phosphorylation in mouse Sal cells by western blotting |
J Med Chem 52: 7360-3 (2009)
Article DOI: 10.1021/jm900786r
BindingDB Entry DOI: 10.7270/Q2P27022 |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50299140
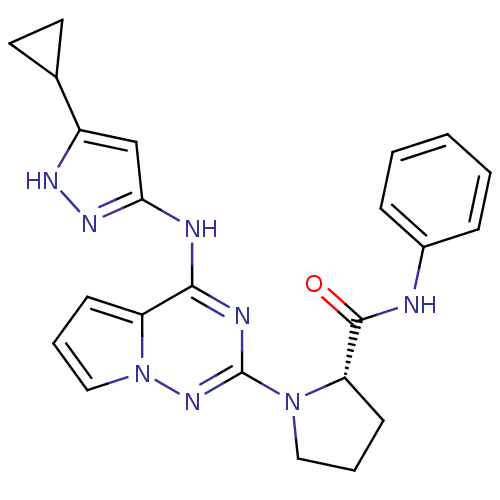
((S)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES O=C(Nc1ccccc1)[C@@H]1CCCN1c1nc(Nc2cc([nH]n2)C2CC2)c2cccn2n1 |r| Show InChI InChI=1S/C23H24N8O/c32-22(24-16-6-2-1-3-7-16)19-9-4-12-30(19)23-26-21(18-8-5-13-31(18)29-23)25-20-14-17(27-28-20)15-10-11-15/h1-3,5-8,13-15,19H,4,9-12H2,(H,24,32)(H2,25,26,27,28,29)/t19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R after 60 mins by fluorescence electrophoresis |
J Med Chem 52: 7360-3 (2009)
Article DOI: 10.1021/jm900786r
BindingDB Entry DOI: 10.7270/Q2P27022 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50201124
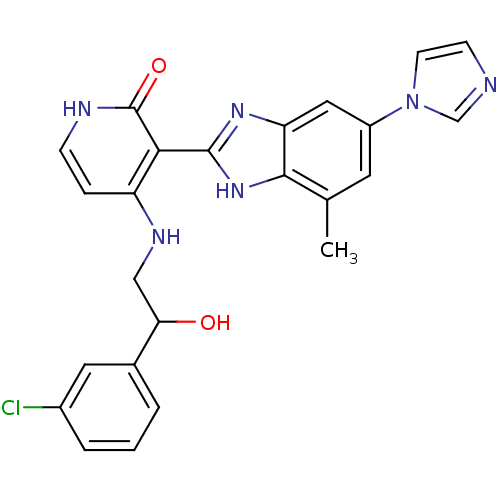
(3-(6-(1H-imidazol-1-yl)-4-methyl-1H-benzo[d]imidaz...)Show SMILES Cc1cc(cc2nc([nH]c12)-c1c(NCC(O)c2cccc(Cl)c2)cc[nH]c1=O)-n1ccnc1 Show InChI InChI=1S/C24H21ClN6O2/c1-14-9-17(31-8-7-26-13-31)11-19-22(14)30-23(29-19)21-18(5-6-27-24(21)33)28-12-20(32)15-3-2-4-16(25)10-15/h2-11,13,20,32H,12H2,1H3,(H,29,30)(H2,27,28,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | <18 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Concentration required to inhibit Cytochrome P450 1A2 in vitro by 50% |
J Med Chem 48: 5639-43 (2005)
Article DOI: 10.1021/jm050392q
BindingDB Entry DOI: 10.7270/Q2MW2GPP |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50201124

(3-(6-(1H-imidazol-1-yl)-4-methyl-1H-benzo[d]imidaz...)Show SMILES Cc1cc(cc2nc([nH]c12)-c1c(NCC(O)c2cccc(Cl)c2)cc[nH]c1=O)-n1ccnc1 Show InChI InChI=1S/C24H21ClN6O2/c1-14-9-17(31-8-7-26-13-31)11-19-22(14)30-23(29-19)21-18(5-6-27-24(21)33)28-12-20(32)15-3-2-4-16(25)10-15/h2-11,13,20,32H,12H2,1H3,(H,29,30)(H2,27,28,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | <18 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Concentration required to inhibit cytochrome P450 isozyme CYP3A4-BzRes in vitro by 50% |
J Med Chem 48: 5639-43 (2005)
Article DOI: 10.1021/jm050392q
BindingDB Entry DOI: 10.7270/Q2MW2GPP |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50201124

(3-(6-(1H-imidazol-1-yl)-4-methyl-1H-benzo[d]imidaz...)Show SMILES Cc1cc(cc2nc([nH]c12)-c1c(NCC(O)c2cccc(Cl)c2)cc[nH]c1=O)-n1ccnc1 Show InChI InChI=1S/C24H21ClN6O2/c1-14-9-17(31-8-7-26-13-31)11-19-22(14)30-23(29-19)21-18(5-6-27-24(21)33)28-12-20(32)15-3-2-4-16(25)10-15/h2-11,13,20,32H,12H2,1H3,(H,29,30)(H2,27,28,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | <18 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Concentration required to inhibit cytochrome P450 isozyme CYP3A4-BFC in vitro by 50% |
J Med Chem 48: 5639-43 (2005)
Article DOI: 10.1021/jm050392q
BindingDB Entry DOI: 10.7270/Q2MW2GPP |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM27887

(4-{[2-(4-chloro-1H-pyrazol-1-yl)ethyl]amino}-3-{6-...)Show SMILES COCCN1CCC(CC1)c1cc(C)c2nc([nH]c2c1)-c1c(NCCn2cc(Cl)cn2)cc[nH]c1=O Show InChI InChI=1S/C26H32ClN7O2/c1-17-13-19(18-4-8-33(9-5-18)11-12-36-2)14-22-24(17)32-25(31-22)23-21(3-6-29-26(23)35)28-7-10-34-16-20(27)15-30-34/h3,6,13-16,18H,4-5,7-12H2,1-2H3,(H,31,32)(H2,28,29,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | 7.0 | 30 |
Bristol-Myers Squibb Company
| Assay Description
The IGF-1 receptor tyrosine kinase was assayed using the synthetic polymer poly(Glu/Tyr) as a phosphoacceptor substrate. Incorporated radioactivity w... |
J Med Chem 51: 5897-900 (2008)
Article DOI: 10.1021/jm800832q
BindingDB Entry DOI: 10.7270/Q2513WJK |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM27881
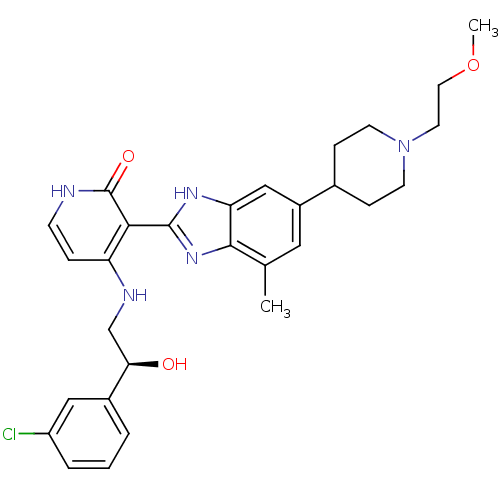
(4-{[(2S)-2-(3-chlorophenyl)-2-hydroxyethyl]amino}-...)Show SMILES COCCN1CCC(CC1)c1cc(C)c2nc([nH]c2c1)-c1c(NC[C@@H](O)c2cccc(Cl)c2)cc[nH]c1=O |r| Show InChI InChI=1S/C29H34ClN5O3/c1-18-14-21(19-7-10-35(11-8-19)12-13-38-2)16-24-27(18)34-28(33-24)26-23(6-9-31-29(26)37)32-17-25(36)20-4-3-5-22(30)15-20/h3-6,9,14-16,19,25,36H,7-8,10-13,17H2,1-2H3,(H,33,34)(H2,31,32,37)/t25-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | 7.0 | 30 |
Bristol-Myers Squibb Company
| Assay Description
The IGF-1 receptor tyrosine kinase was assayed using the synthetic polymer poly(Glu/Tyr) as a phosphoacceptor substrate. Incorporated radioactivity w... |
J Med Chem 51: 5897-900 (2008)
Article DOI: 10.1021/jm800832q
BindingDB Entry DOI: 10.7270/Q2513WJK |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50201124

(3-(6-(1H-imidazol-1-yl)-4-methyl-1H-benzo[d]imidaz...)Show SMILES Cc1cc(cc2nc([nH]c12)-c1c(NCC(O)c2cccc(Cl)c2)cc[nH]c1=O)-n1ccnc1 Show InChI InChI=1S/C24H21ClN6O2/c1-14-9-17(31-8-7-26-13-31)11-19-22(14)30-23(29-19)21-18(5-6-27-24(21)33)28-12-20(32)15-3-2-4-16(25)10-15/h2-11,13,20,32H,12H2,1H3,(H,29,30)(H2,27,28,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | <18 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Concentration required to inhibit Cytochrome P450 2C9 in vitro by 50% |
J Med Chem 48: 5639-43 (2005)
Article DOI: 10.1021/jm050392q
BindingDB Entry DOI: 10.7270/Q2MW2GPP |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM27884
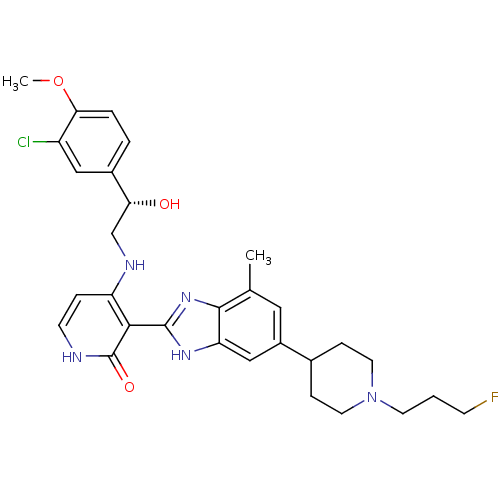
(4-{[(2S)-2-(3-chloro-4-methoxyphenyl)-2-hydroxyeth...)Show SMILES COc1ccc(cc1Cl)[C@H](O)CNc1cc[nH]c(=O)c1-c1nc2c(C)cc(cc2[nH]1)C1CCN(CCCF)CC1 |r| Show InChI InChI=1S/C30H35ClFN5O3/c1-18-14-21(19-7-12-37(13-8-19)11-3-9-32)16-24-28(18)36-29(35-24)27-23(6-10-33-30(27)39)34-17-25(38)20-4-5-26(40-2)22(31)15-20/h4-6,10,14-16,19,25,38H,3,7-9,11-13,17H2,1-2H3,(H,35,36)(H2,33,34,39)/t25-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | 7.0 | 30 |
Bristol-Myers Squibb Company
| Assay Description
The IGF-1 receptor tyrosine kinase was assayed using the synthetic polymer poly(Glu/Tyr) as a phosphoacceptor substrate. Incorporated radioactivity w... |
J Med Chem 51: 5897-900 (2008)
Article DOI: 10.1021/jm800832q
BindingDB Entry DOI: 10.7270/Q2513WJK |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM27880

(4-{[(2S)-2-(3-chlorophenyl)-2-hydroxyethyl]amino}-...)Show SMILES CN1CCC(CC1)c1cc(C)c2nc([nH]c2c1)-c1c(NC[C@@H](O)c2cccc(Cl)c2)cc[nH]c1=O |r| Show InChI InChI=1S/C27H30ClN5O2/c1-16-12-19(17-7-10-33(2)11-8-17)14-22-25(16)32-26(31-22)24-21(6-9-29-27(24)35)30-15-23(34)18-4-3-5-20(28)13-18/h3-6,9,12-14,17,23,34H,7-8,10-11,15H2,1-2H3,(H,31,32)(H2,29,30,35)/t23-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | 7.0 | 30 |
Bristol-Myers Squibb Company
| Assay Description
The IGF-1 receptor tyrosine kinase was assayed using the synthetic polymer poly(Glu/Tyr) as a phosphoacceptor substrate. Incorporated radioactivity w... |
J Med Chem 51: 5897-900 (2008)
Article DOI: 10.1021/jm800832q
BindingDB Entry DOI: 10.7270/Q2513WJK |
More data for this
Ligand-Target Pair | |
RAC-alpha serine/threonine-protein kinase
(Mus musculus) | BDBM50299148

((S)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES C[C@]1(CCCN1c1nc(Nc2cc([nH]n2)C2CC2)c2cccn2n1)C(=O)Nc1ccc(F)nc1 |r| Show InChI InChI=1S/C23H24FN9O/c1-23(21(34)26-15-7-8-18(24)25-13-15)9-3-10-32(23)22-28-20(17-4-2-11-33(17)31-22)27-19-12-16(29-30-19)14-5-6-14/h2,4,7-8,11-14H,3,5-6,9-10H2,1H3,(H,26,34)(H2,27,28,29,30,31)/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Inhibition of Akt phosphorylation in mouse Sal cells by western blotting |
J Med Chem 52: 7360-3 (2009)
Article DOI: 10.1021/jm900786r
BindingDB Entry DOI: 10.7270/Q2P27022 |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM27883
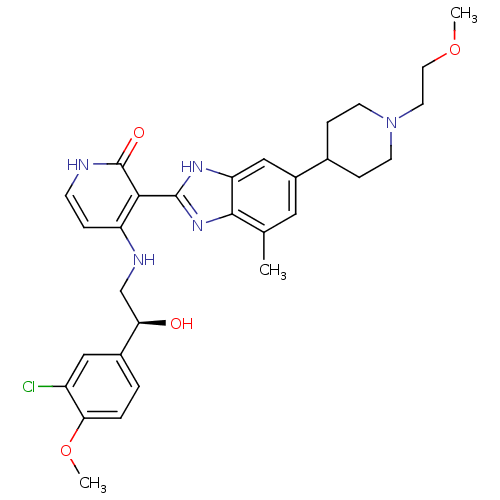
(4-{[(2S)-2-(3-chloro-4-methoxyphenyl)-2-hydroxyeth...)Show SMILES COCCN1CCC(CC1)c1cc(C)c2nc([nH]c2c1)-c1c(NC[C@@H](O)c2ccc(OC)c(Cl)c2)cc[nH]c1=O |r| Show InChI InChI=1S/C30H36ClN5O4/c1-18-14-21(19-7-10-36(11-8-19)12-13-39-2)16-24-28(18)35-29(34-24)27-23(6-9-32-30(27)38)33-17-25(37)20-4-5-26(40-3)22(31)15-20/h4-6,9,14-16,19,25,37H,7-8,10-13,17H2,1-3H3,(H,34,35)(H2,32,33,38)/t25-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | 7.0 | 30 |
Bristol-Myers Squibb Company
| Assay Description
The IGF-1 receptor tyrosine kinase was assayed using the synthetic polymer poly(Glu/Tyr) as a phosphoacceptor substrate. Incorporated radioactivity w... |
J Med Chem 51: 5897-900 (2008)
Article DOI: 10.1021/jm800832q
BindingDB Entry DOI: 10.7270/Q2513WJK |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 2/G1/S-specific cyclin-E1/G1/S-specific cyclin-E2
(Homo sapiens (Human)) | BDBM50299139

((R)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES NC(=O)[C@H]1CCCN1c1nc(Nc2cc([nH]n2)C2CC2)c2cccn2n1 |r| Show InChI InChI=1S/C17H20N8O/c18-15(26)12-3-1-7-24(12)17-20-16(13-4-2-8-25(13)23-17)19-14-9-11(21-22-14)10-5-6-10/h2,4,8-10,12H,1,3,5-7H2,(H2,18,26)(H2,19,20,21,22,23)/t12-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E after 60 mins by fluorescence electrophoresis |
J Med Chem 52: 7360-3 (2009)
Article DOI: 10.1021/jm900786r
BindingDB Entry DOI: 10.7270/Q2P27022 |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM27886

(4-{[2-(4-chloro-1H-pyrazol-1-yl)ethyl]amino}-3-{6-...)Show SMILES Cc1cc(cc2[nH]c(nc12)-c1c(NCCn2cc(Cl)cn2)cc[nH]c1=O)C1CCN(CCO)CC1 Show InChI InChI=1S/C25H30ClN7O2/c1-16-12-18(17-3-7-32(8-4-17)10-11-34)13-21-23(16)31-24(30-21)22-20(2-5-28-25(22)35)27-6-9-33-15-19(26)14-29-33/h2,5,12-15,17,34H,3-4,6-11H2,1H3,(H,30,31)(H2,27,28,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | 7.0 | 30 |
Bristol-Myers Squibb Company
| Assay Description
The IGF-1 receptor tyrosine kinase was assayed using the synthetic polymer poly(Glu/Tyr) as a phosphoacceptor substrate. Incorporated radioactivity w... |
J Med Chem 51: 5897-900 (2008)
Article DOI: 10.1021/jm800832q
BindingDB Entry DOI: 10.7270/Q2513WJK |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50201124

(3-(6-(1H-imidazol-1-yl)-4-methyl-1H-benzo[d]imidaz...)Show SMILES Cc1cc(cc2nc([nH]c12)-c1c(NCC(O)c2cccc(Cl)c2)cc[nH]c1=O)-n1ccnc1 Show InChI InChI=1S/C24H21ClN6O2/c1-14-9-17(31-8-7-26-13-31)11-19-22(14)30-23(29-19)21-18(5-6-27-24(21)33)28-12-20(32)15-3-2-4-16(25)10-15/h2-11,13,20,32H,12H2,1H3,(H,29,30)(H2,27,28,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Concentration required to inhibit cytochrome P450 isozyme CYP2C19 in vitro by 50% |
J Med Chem 48: 5639-43 (2005)
Article DOI: 10.1021/jm050392q
BindingDB Entry DOI: 10.7270/Q2MW2GPP |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM27885

(4-{[2-(4-chloro-1H-pyrazol-1-yl)ethyl]amino}-3-[4-...)Show SMILES CN1CCC(CC1)c1cc(C)c2nc([nH]c2c1)-c1c(NCCn2cc(Cl)cn2)cc[nH]c1=O Show InChI InChI=1S/C24H28ClN7O/c1-15-11-17(16-4-8-31(2)9-5-16)12-20-22(15)30-23(29-20)21-19(3-6-27-24(21)33)26-7-10-32-14-18(25)13-28-32/h3,6,11-14,16H,4-5,7-10H2,1-2H3,(H,29,30)(H2,26,27,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | 7.0 | 30 |
Bristol-Myers Squibb Company
| Assay Description
The IGF-1 receptor tyrosine kinase was assayed using the synthetic polymer poly(Glu/Tyr) as a phosphoacceptor substrate. Incorporated radioactivity w... |
J Med Chem 51: 5897-900 (2008)
Article DOI: 10.1021/jm800832q
BindingDB Entry DOI: 10.7270/Q2513WJK |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM27882

(4-{[(2S)-2-(3-chlorophenyl)-2-hydroxyethyl]amino}-...)Show SMILES Cc1cc(cc2[nH]c(nc12)-c1c(NC[C@@H](O)c2cccc(Cl)c2)cc[nH]c1=O)C1CCN(CCCF)CC1 |r| Show InChI InChI=1S/C29H33ClFN5O2/c1-18-14-21(19-7-12-36(13-8-19)11-3-9-31)16-24-27(18)35-28(34-24)26-23(6-10-32-29(26)38)33-17-25(37)20-4-2-5-22(30)15-20/h2,4-6,10,14-16,19,25,37H,3,7-9,11-13,17H2,1H3,(H,34,35)(H2,32,33,38)/t25-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | 7.0 | 30 |
Bristol-Myers Squibb Company
| Assay Description
The IGF-1 receptor tyrosine kinase was assayed using the synthetic polymer poly(Glu/Tyr) as a phosphoacceptor substrate. Incorporated radioactivity w... |
J Med Chem 51: 5897-900 (2008)
Article DOI: 10.1021/jm800832q
BindingDB Entry DOI: 10.7270/Q2513WJK |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50325429
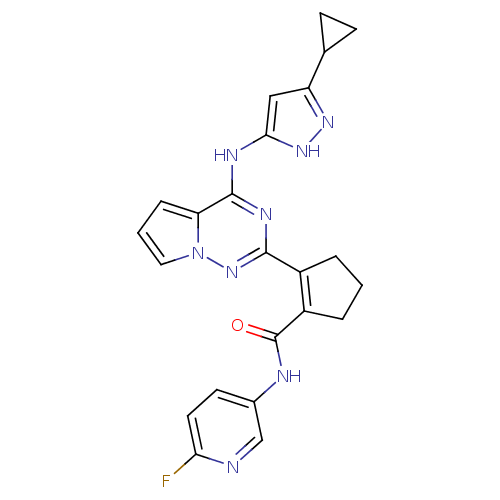
(2-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrrolo[1...)Show SMILES Fc1ccc(NC(=O)C2=C(CCC2)c2nc(Nc3cc(n[nH]3)C3CC3)c3cccn3n2)cn1 |t:8| Show InChI InChI=1S/C23H21FN8O/c24-19-9-8-14(12-25-19)26-23(33)16-4-1-3-15(16)21-28-22(18-5-2-10-32(18)31-21)27-20-11-17(29-30-20)13-6-7-13/h2,5,8-13H,1,3-4,6-7H2,(H,26,33)(H2,27,28,29,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R |
Bioorg Med Chem Lett 20: 5027-30 (2010)
Article DOI: 10.1016/j.bmcl.2010.07.045
BindingDB Entry DOI: 10.7270/Q2WH2Q5J |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM27888

(4-{[2-(4-chloro-1H-pyrazol-1-yl)ethyl]amino}-3-{6-...)Show SMILES Cc1cc(cc2[nH]c(nc12)-c1c(NCCn2cc(Cl)cn2)cc[nH]c1=O)C1CCN(CCCF)CC1 Show InChI InChI=1S/C26H31ClFN7O/c1-17-13-19(18-4-10-34(11-5-18)9-2-6-28)14-22-24(17)33-25(32-22)23-21(3-7-30-26(23)36)29-8-12-35-16-20(27)15-31-35/h3,7,13-16,18H,2,4-6,8-12H2,1H3,(H,32,33)(H2,29,30,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 34 | n/a | n/a | n/a | n/a | 7.0 | 30 |
Bristol-Myers Squibb Company
| Assay Description
The IGF-1 receptor tyrosine kinase was assayed using the synthetic polymer poly(Glu/Tyr) as a phosphoacceptor substrate. Incorporated radioactivity w... |
J Med Chem 51: 5897-900 (2008)
Article DOI: 10.1021/jm800832q
BindingDB Entry DOI: 10.7270/Q2513WJK |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 2/G1/S-specific cyclin-E1/G1/S-specific cyclin-E2
(Homo sapiens (Human)) | BDBM50299143

((S)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES O=C(Nc1nccs1)[C@@H]1CCCN1c1nc(Nc2cc([nH]n2)C2CC2)c2cccn2n1 |r| Show InChI InChI=1S/C20H21N9OS/c30-18(24-20-21-7-10-31-20)15-4-1-8-28(15)19-23-17(14-3-2-9-29(14)27-19)22-16-11-13(25-26-16)12-5-6-12/h2-3,7,9-12,15H,1,4-6,8H2,(H,21,24,30)(H2,22,23,25,26,27)/t15-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E after 60 mins by fluorescence electrophoresis |
J Med Chem 52: 7360-3 (2009)
Article DOI: 10.1021/jm900786r
BindingDB Entry DOI: 10.7270/Q2P27022 |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50299141
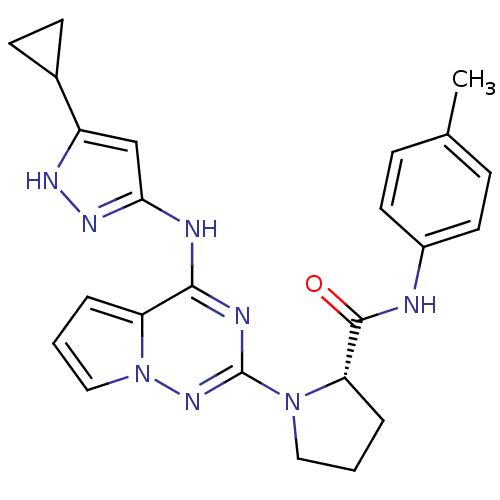
((S)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES Cc1ccc(NC(=O)[C@@H]2CCCN2c2nc(Nc3cc([nH]n3)C3CC3)c3cccn3n2)cc1 |r| Show InChI InChI=1S/C24H26N8O/c1-15-6-10-17(11-7-15)25-23(33)20-5-2-12-31(20)24-27-22(19-4-3-13-32(19)30-24)26-21-14-18(28-29-21)16-8-9-16/h3-4,6-7,10-11,13-14,16,20H,2,5,8-9,12H2,1H3,(H,25,33)(H2,26,27,28,29,30)/t20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R after 60 mins by fluorescence electrophoresis |
J Med Chem 52: 7360-3 (2009)
Article DOI: 10.1021/jm900786r
BindingDB Entry DOI: 10.7270/Q2P27022 |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50325430

(CHEMBL1222855 | N-((4-(5-cyclopropyl-1H-pyrazol-3-...)Show SMILES Fc1ccc(cn1)C(=O)NCc1nc(Nc2cc(n[nH]2)C2CC2)c2cccn2n1 Show InChI InChI=1S/C19H17FN8O/c20-15-6-5-12(9-21-15)19(29)22-10-17-24-18(14-2-1-7-28(14)27-17)23-16-8-13(25-26-16)11-3-4-11/h1-2,5-9,11H,3-4,10H2,(H,22,29)(H2,23,24,25,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R in IGF1R-SAL cells |
Bioorg Med Chem Lett 20: 5027-30 (2010)
Article DOI: 10.1016/j.bmcl.2010.07.045
BindingDB Entry DOI: 10.7270/Q2WH2Q5J |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50299138

((S)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES NC(=O)[C@@H]1CCCN1c1nc(Nc2cc([nH]n2)C2CC2)c2cccn2n1 |r| Show InChI InChI=1S/C17H20N8O/c18-15(26)12-3-1-7-24(12)17-20-16(13-4-2-8-25(13)23-17)19-14-9-11(21-22-14)10-5-6-10/h2,4,8-10,12H,1,3,5-7H2,(H2,18,26)(H2,19,20,21,22,23)/t12-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R after 60 mins by fluorescence electrophoresis |
J Med Chem 52: 7360-3 (2009)
Article DOI: 10.1021/jm900786r
BindingDB Entry DOI: 10.7270/Q2P27022 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM27879
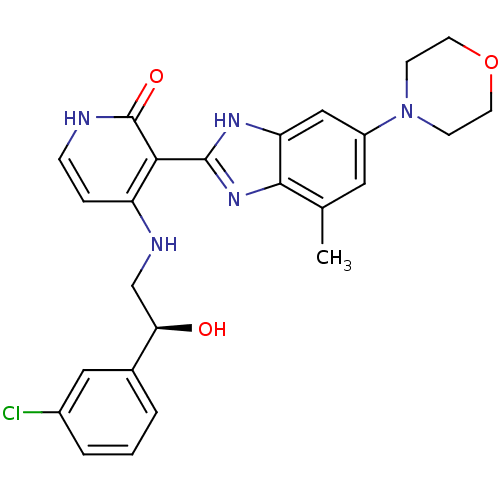
(4-{[(2S)-2-(3-chlorophenyl)-2-hydroxyethyl]amino}-...)Show SMILES Cc1cc(cc2[nH]c(nc12)-c1c(NC[C@@H](O)c2cccc(Cl)c2)cc[nH]c1=O)N1CCOCC1 |r| Show InChI InChI=1S/C25H26ClN5O3/c1-15-11-18(31-7-9-34-10-8-31)13-20-23(15)30-24(29-20)22-19(5-6-27-25(22)33)28-14-21(32)16-3-2-4-17(26)12-16/h2-6,11-13,21,32H,7-10,14H2,1H3,(H,29,30)(H2,27,28,33)/t21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Bristol-Myers Squibb Company
| Assay Description
The inhibition of recombinant human CYP3A4 was measured as the ability to perform a dealkylation of 7-benzyloxy-4-trifluoromethylcoumarin (BFC). Befo... |
J Med Chem 51: 5897-900 (2008)
Article DOI: 10.1021/jm800832q
BindingDB Entry DOI: 10.7270/Q2513WJK |
More data for this
Ligand-Target Pair | |
Insulin receptor
(Homo sapiens (Human)) | BDBM27879

(4-{[(2S)-2-(3-chlorophenyl)-2-hydroxyethyl]amino}-...)Show SMILES Cc1cc(cc2[nH]c(nc12)-c1c(NC[C@@H](O)c2cccc(Cl)c2)cc[nH]c1=O)N1CCOCC1 |r| Show InChI InChI=1S/C25H26ClN5O3/c1-15-11-18(31-7-9-34-10-8-31)13-20-23(15)30-24(29-20)22-19(5-6-27-25(22)33)28-14-21(32)16-3-2-4-17(26)12-16/h2-6,11-13,21,32H,7-10,14H2,1H3,(H,29,30)(H2,27,28,33)/t21-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 73 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration against IR kinase with ATP concentration at 1/2Km |
J Med Chem 48: 5639-43 (2005)
Article DOI: 10.1021/jm050392q
BindingDB Entry DOI: 10.7270/Q2MW2GPP |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM27879

(4-{[(2S)-2-(3-chlorophenyl)-2-hydroxyethyl]amino}-...)Show SMILES Cc1cc(cc2[nH]c(nc12)-c1c(NC[C@@H](O)c2cccc(Cl)c2)cc[nH]c1=O)N1CCOCC1 |r| Show InChI InChI=1S/C25H26ClN5O3/c1-15-11-18(31-7-9-34-10-8-31)13-20-23(15)30-24(29-20)22-19(5-6-27-25(22)33)28-14-21(32)16-3-2-4-17(26)12-16/h2-6,11-13,21,32H,7-10,14H2,1H3,(H,29,30)(H2,27,28,33)/t21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | 7.0 | 30 |
Bristol-Myers Squibb Company
| Assay Description
The IGF-1 receptor tyrosine kinase was assayed using the synthetic polymer poly(Glu/Tyr) as a phosphoacceptor substrate. Incorporated radioactivity w... |
J Med Chem 51: 5897-900 (2008)
Article DOI: 10.1021/jm800832q
BindingDB Entry DOI: 10.7270/Q2513WJK |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM27879

(4-{[(2S)-2-(3-chlorophenyl)-2-hydroxyethyl]amino}-...)Show SMILES Cc1cc(cc2[nH]c(nc12)-c1c(NC[C@@H](O)c2cccc(Cl)c2)cc[nH]c1=O)N1CCOCC1 |r| Show InChI InChI=1S/C25H26ClN5O3/c1-15-11-18(31-7-9-34-10-8-31)13-20-23(15)30-24(29-20)22-19(5-6-27-25(22)33)28-14-21(32)16-3-2-4-17(26)12-16/h2-6,11-13,21,32H,7-10,14H2,1H3,(H,29,30)(H2,27,28,33)/t21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration against IGF-1R kinase |
J Med Chem 48: 5639-43 (2005)
Article DOI: 10.1021/jm050392q
BindingDB Entry DOI: 10.7270/Q2MW2GPP |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM27879

(4-{[(2S)-2-(3-chlorophenyl)-2-hydroxyethyl]amino}-...)Show SMILES Cc1cc(cc2[nH]c(nc12)-c1c(NC[C@@H](O)c2cccc(Cl)c2)cc[nH]c1=O)N1CCOCC1 |r| Show InChI InChI=1S/C25H26ClN5O3/c1-15-11-18(31-7-9-34-10-8-31)13-20-23(15)30-24(29-20)22-19(5-6-27-25(22)33)28-14-21(32)16-3-2-4-17(26)12-16/h2-6,11-13,21,32H,7-10,14H2,1H3,(H,29,30)(H2,27,28,33)/t21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration against IGF-1R Sal kinase with ATP concentration at 1/2Km |
J Med Chem 48: 5639-43 (2005)
Article DOI: 10.1021/jm050392q
BindingDB Entry DOI: 10.7270/Q2MW2GPP |
More data for this
Ligand-Target Pair | |
Focal adhesion kinase 1
(Homo sapiens (Human)) | BDBM27879

(4-{[(2S)-2-(3-chlorophenyl)-2-hydroxyethyl]amino}-...)Show SMILES Cc1cc(cc2[nH]c(nc12)-c1c(NC[C@@H](O)c2cccc(Cl)c2)cc[nH]c1=O)N1CCOCC1 |r| Show InChI InChI=1S/C25H26ClN5O3/c1-15-11-18(31-7-9-34-10-8-31)13-20-23(15)30-24(29-20)22-19(5-6-27-25(22)33)28-14-21(32)16-3-2-4-17(26)12-16/h2-6,11-13,21,32H,7-10,14H2,1H3,(H,29,30)(H2,27,28,33)/t21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration against FAK with ATP concentration at 1/2Km |
J Med Chem 48: 5639-43 (2005)
Article DOI: 10.1021/jm050392q
BindingDB Entry DOI: 10.7270/Q2MW2GPP |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50325425

((S)-(2-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrr...)Show SMILES Fc1ccc(cn1)C(=O)N1CCC[C@H]1c1nc(Nc2cc(n[nH]2)C2CC2)c2cccn2n1 |r| Show InChI InChI=1S/C22H21FN8O/c23-18-8-7-14(12-24-18)22(32)30-9-1-3-16(30)21-26-20(17-4-2-10-31(17)29-21)25-19-11-15(27-28-19)13-5-6-13/h2,4,7-8,10-13,16H,1,3,5-6,9H2,(H2,25,26,27,28,29)/t16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 162 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R in IGF1R-SAL cells |
Bioorg Med Chem Lett 20: 5027-30 (2010)
Article DOI: 10.1016/j.bmcl.2010.07.045
BindingDB Entry DOI: 10.7270/Q2WH2Q5J |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50201124

(3-(6-(1H-imidazol-1-yl)-4-methyl-1H-benzo[d]imidaz...)Show SMILES Cc1cc(cc2nc([nH]c12)-c1c(NCC(O)c2cccc(Cl)c2)cc[nH]c1=O)-n1ccnc1 Show InChI InChI=1S/C24H21ClN6O2/c1-14-9-17(31-8-7-26-13-31)11-19-22(14)30-23(29-19)21-18(5-6-27-24(21)33)28-12-20(32)15-3-2-4-16(25)10-15/h2-11,13,20,32H,12H2,1H3,(H,29,30)(H2,27,28,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 180 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration against IGF-1R kinase |
J Med Chem 48: 5639-43 (2005)
Article DOI: 10.1021/jm050392q
BindingDB Entry DOI: 10.7270/Q2MW2GPP |
More data for this
Ligand-Target Pair | |
Dual specificity mitogen-activated protein kinase kinase 1
(Homo sapiens (Human)) | BDBM27879

(4-{[(2S)-2-(3-chlorophenyl)-2-hydroxyethyl]amino}-...)Show SMILES Cc1cc(cc2[nH]c(nc12)-c1c(NC[C@@H](O)c2cccc(Cl)c2)cc[nH]c1=O)N1CCOCC1 |r| Show InChI InChI=1S/C25H26ClN5O3/c1-15-11-18(31-7-9-34-10-8-31)13-20-23(15)30-24(29-20)22-19(5-6-27-25(22)33)28-14-21(32)16-3-2-4-17(26)12-16/h2-6,11-13,21,32H,7-10,14H2,1H3,(H,29,30)(H2,27,28,33)/t21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 182 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration against MEK with ATP concentration at 1/2Km |
J Med Chem 48: 5639-43 (2005)
Article DOI: 10.1021/jm050392q
BindingDB Entry DOI: 10.7270/Q2MW2GPP |
More data for this
Ligand-Target Pair | |
Insulin-like growth factor 1 receptor
(Homo sapiens (Human)) | BDBM50299139

((R)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES NC(=O)[C@H]1CCCN1c1nc(Nc2cc([nH]n2)C2CC2)c2cccn2n1 |r| Show InChI InChI=1S/C17H20N8O/c18-15(26)12-3-1-7-24(12)17-20-16(13-4-2-8-25(13)23-17)19-14-9-11(21-22-14)10-5-6-10/h2,4,8-10,12H,1,3,5-7H2,(H2,18,26)(H2,19,20,21,22,23)/t12-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 199 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Inhibition of IGF1R after 60 mins by fluorescence electrophoresis |
J Med Chem 52: 7360-3 (2009)
Article DOI: 10.1021/jm900786r
BindingDB Entry DOI: 10.7270/Q2P27022 |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 2/G1/S-specific cyclin-E1/G1/S-specific cyclin-E2
(Homo sapiens (Human)) | BDBM50299142

((S)-1-(4-(5-cyclopropyl-1H-pyrazol-3-ylamino)pyrro...)Show SMILES CN(C(=O)[C@@H]1CCCN1c1nc(Nc2cc([nH]n2)C2CC2)c2cccn2n1)c1ccccc1 |r| Show InChI InChI=1S/C24H26N8O/c1-30(17-7-3-2-4-8-17)23(33)20-10-5-13-31(20)24-26-22(19-9-6-14-32(19)29-24)25-21-15-18(27-28-21)16-11-12-16/h2-4,6-9,14-16,20H,5,10-13H2,1H3,(H2,25,26,27,28,29)/t20-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Co
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E after 60 mins by fluorescence electrophoresis |
J Med Chem 52: 7360-3 (2009)
Article DOI: 10.1021/jm900786r
BindingDB Entry DOI: 10.7270/Q2P27022 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data