 Found 254 hits with Last Name = 'kobayashi' and Initial = 'a'
Found 254 hits with Last Name = 'kobayashi' and Initial = 'a' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.0680 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM21147

((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C49H62N10O16S3/c1-76-18-16-34(55-47(69)37(58-44(66)32(50)23-41(61)62)21-28-12-14-30(15-13-28)75-78(72,73)74)45(67)53-26-40(60)54-38(22-29-25-52-33-11-7-6-10-31(29)33)48(70)56-35(17-19-77-2)46(68)59-39(24-42(63)64)49(71)57-36(43(51)65)20-27-8-4-3-5-9-27/h3-15,25,32,34-39,52H,16-24,26,50H2,1-2H3,(H2,51,65)(H,53,67)(H,54,60)(H,55,69)(H,56,70)(H,57,71)(H,58,66)(H,59,68)(H,61,62)(H,63,64)(H,72,73,74)/t32-,34-,35-,36-,37-,38-,39-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0970 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50005463
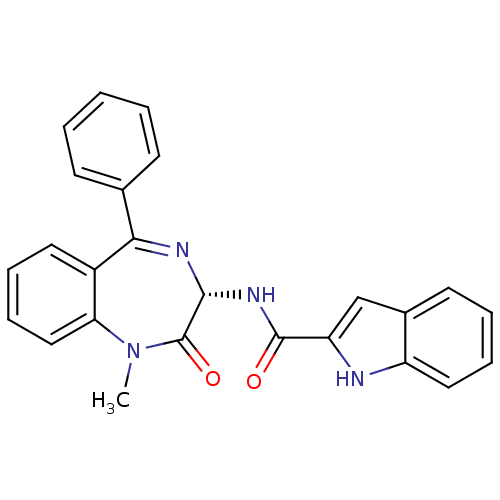
((R)-1H-Indole-2-carboxylic acid (1-methyl-2-oxo-5-...)Show SMILES CN1c2ccccc2C(=N[C@H](NC(=O)c2cc3ccccc3[nH]2)C1=O)c1ccccc1 |r,c:9| Show InChI InChI=1S/C25H20N4O2/c1-29-21-14-8-6-12-18(21)22(16-9-3-2-4-10-16)27-23(25(29)31)28-24(30)20-15-17-11-5-7-13-19(17)26-20/h2-15,23,26H,1H3,(H,28,30)/t23-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.540 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM82235
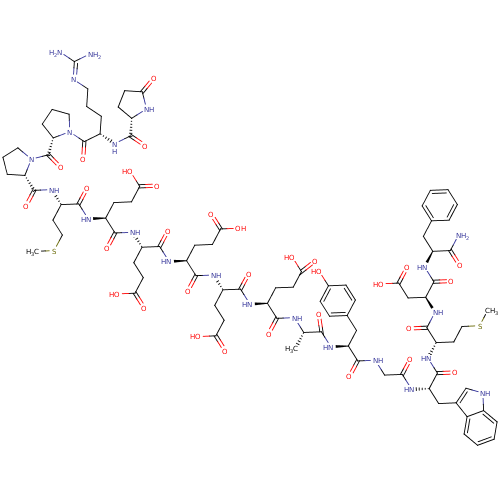
(Gastrin I | Gastrin-17 | Gastrin-I-(1-17))Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)CNC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](C)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCSC)NC(=O)[C@@H]1CCCN1C(=O)[C@@H]1CCCN1C(=O)[C@H](CCCN=C(N)N)NC(=O)[C@@H]1CCC(=O)N1)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,wU:4.4,138.143,38.41,52.60,70.78,88.95,96.98,wD:8.20,130.135,26.37,43.51,61.69,79.87,103.106,110.122,121.125,(26,6.14,;24.67,5.35,;24.67,3.8,;23.32,3.01,;23.32,1.49,;22,.73,;20.68,1.49,;20.68,3.04,;19.35,.73,;19.35,-.76,;20.68,-1.52,;21.94,-.7,;23.15,-1.69,;22.59,-3.1,;23.29,-4.48,;22.45,-5.77,;20.9,-5.71,;20.2,-4.31,;21.04,-2.98,;18,1.49,;16.68,.7,;16.68,-.76,;15.36,1.49,;14.03,.7,;12.68,1.46,;12.68,3.01,;11.36,.68,;11.36,-.79,;12.71,-1.55,;12.71,-3.1,;14.03,-3.86,;15.36,-3.07,;16.71,-3.86,;15.36,-1.55,;14.03,-.79,;10.01,1.44,;8.66,.68,;8.66,-.82,;7.33,1.44,;7.3,2.98,;6.01,.65,;4.66,1.44,;4.66,2.98,;3.34,.65,;3.34,-.84,;4.66,-1.6,;4.66,-3.15,;6.01,-3.91,;3.34,-3.91,;2.01,1.44,;.66,.65,;.66,-.84,;-.61,1.44,;-.61,2.96,;.66,3.74,;.66,5.32,;2.01,6.11,;-.61,6.11,;-1.93,.65,;-3.28,1.41,;-3.28,2.96,;-4.6,.65,;-4.6,-.84,;-3.28,-1.6,;-3.28,-3.15,;-1.93,-3.94,;-4.6,-3.94,;-5.93,1.41,;-7.28,.65,;-7.28,-.84,;-8.6,1.44,;-8.6,2.96,;-7.28,3.74,;-7.28,5.32,;-5.93,6.11,;-8.6,6.11,;-9.95,.65,;-11.33,1.44,;-11.33,2.98,;-12.65,.65,;-12.65,-.82,;-11.33,-1.6,;-11.33,-3.15,;-9.95,-3.94,;-12.65,-3.91,;-13.98,1.44,;-15.33,.68,;-15.33,-.79,;-16.65,1.44,;-16.65,2.96,;-15.3,3.77,;-15.3,5.29,;-13.98,6.08,;-17.97,.68,;-19.32,1.44,;-19.32,2.98,;-20.62,.68,;-19.69,1.83,;-20.39,3.26,;-21.91,2.96,;-21.91,1.44,;-23.24,.68,;-23.24,-.96,;-24.7,1.52,;-23.97,.34,;-24.56,-.98,;-26.16,-.7,;-26.16,.68,;-27.52,1.44,;-27.52,2.98,;-28.84,.68,;-28.84,-.82,;-27.52,-1.55,;-27.52,-3.1,;-26.14,-3.86,;-26.11,-5.4,;-27.52,-6.16,;-24.78,-6.16,;-30.19,1.44,;-31.51,.68,;-31.51,-.82,;-32.84,1.41,;-31.85,.28,;-32.55,-1.1,;-34.07,-.79,;-35.37,-1.58,;-34.07,.68,;24.67,.73,;24.67,-.73,;26,1.49,;27.35,.7,;27.35,-.76,;28.7,-1.55,;30.02,-.76,;28.7,-3.07,;28.7,1.49,;28.7,3.04,;30.02,.7,;31.34,1.49,;31.34,3.01,;32.7,3.83,;32.7,5.38,;34.02,6.16,;35.37,5.38,;35.37,3.83,;34.02,3.01,;32.7,.7,;34.02,1.49,;32.7,-.76,)| Show InChI InChI=1S/C94H128N22O31S2/c1-48(79(133)113-65(43-50-19-21-52(117)22-20-50)80(134)100-47-71(119)103-66(44-51-46-99-54-15-8-7-14-53(51)54)89(143)109-61(35-40-148-2)88(142)114-67(45-77(130)131)90(144)112-64(78(95)132)42-49-12-5-4-6-13-49)101-81(135)56(24-30-72(120)121)104-83(137)57(25-31-73(122)123)105-84(138)58(26-32-74(124)125)106-85(139)59(27-33-75(126)127)107-86(140)60(28-34-76(128)129)108-87(141)62(36-41-149-3)110-91(145)68-17-10-38-115(68)93(147)69-18-11-39-116(69)92(146)63(16-9-37-98-94(96)97)111-82(136)55-23-29-70(118)102-55/h4-8,12-15,19-22,46,48,55-69,99,117H,9-11,16-18,23-45,47H2,1-3H3,(H2,95,132)(H,100,134)(H,101,135)(H,102,118)(H,103,119)(H,104,137)(H,105,138)(H,106,139)(H,107,140)(H,108,141)(H,109,143)(H,110,145)(H,111,136)(H,112,144)(H,113,133)(H,114,142)(H,120,121)(H,122,123)(H,124,125)(H,126,127)(H,128,129)(H,130,131)(H4,96,97,98)/t48-,55-,56-,57-,58-,59-,60-,61-,62-,63-,64-,65-,66-,67-,68-,69-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM82403
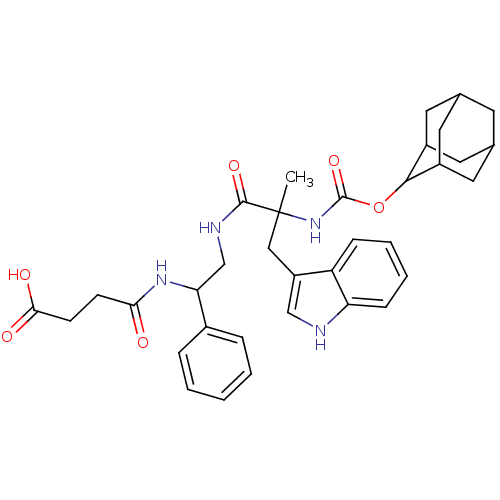
(CAS_108186 | CI-988 | NSC_108186)Show SMILES CC(Cc1c[nH]c2ccccc12)(NC(=O)OC1C2CC3CC(C2)CC1C3)C(=O)NCC(NC(=O)CCC(O)=O)c1ccccc1 |TLB:25:24:18.19.20:22,THB:15:16:18.19.20:22,20:19:16:21.22.23,20:21:16:18.19.25,25:19:22:16.23.24,(.76,2.29,;.32,.82,;-.11,-.66,;.95,-1.78,;.68,-3.29,;2.03,-4.02,;3.15,-2.96,;4.68,-3.07,;5.55,-1.8,;4.88,-.42,;3.35,-.3,;2.48,-1.57,;1.8,.38,;2.92,1.45,;2.55,2.94,;4.39,1.01,;5.51,2.08,;6.88,2.77,;6.88,4.17,;7.72,5.64,;9.09,4.95,;9.09,3.55,;8.25,2.08,;7.72,2.85,;6.35,3.55,;6.35,4.95,;-1.15,1.25,;-1.52,2.75,;-2.27,.19,;-3.75,.62,;-4.86,-.44,;-6.34,-.01,;-7.45,-1.07,;-7.09,-2.57,;-8.93,-.64,;-10.04,-1.7,;-11.52,-1.27,;-11.89,.23,;-12.64,-2.33,;-4.5,-1.94,;-3.02,-2.37,;-2.65,-3.87,;-3.77,-4.93,;-5.25,-4.5,;-5.61,-3,)| Show InChI InChI=1S/C35H42N4O6/c1-35(18-26-19-36-28-10-6-5-9-27(26)28,39-34(44)45-32-24-14-21-13-22(16-24)17-25(32)15-21)33(43)37-20-29(23-7-3-2-4-8-23)38-30(40)11-12-31(41)42/h2-10,19,21-22,24-25,29,32,36H,11-18,20H2,1H3,(H,37,43)(H,38,40)(H,39,44)(H,41,42) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 6.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50061220
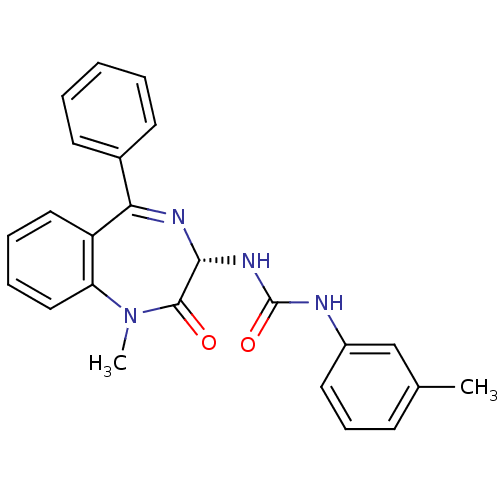
(1-((S)-1-Methyl-2-oxo-5-phenyl-2,3-dihydro-1H-benz...)Show SMILES CN1c2ccccc2C(=N[C@H](NC(=O)Nc2cccc(C)c2)C1=O)c1ccccc1 |c:9| Show InChI InChI=1S/C24H22N4O2/c1-16-9-8-12-18(15-16)25-24(30)27-22-23(29)28(2)20-14-7-6-13-19(20)21(26-22)17-10-4-3-5-11-17/h3-15,22H,1-2H3,(H2,25,27,30)/t22-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM21147

((3S)-3-[(2S)-2-[(2S)-2-{2-[(2S)-2-[(2S)-2-[(3S)-3-...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C49H62N10O16S3/c1-76-18-16-34(55-47(69)37(58-44(66)32(50)23-41(61)62)21-28-12-14-30(15-13-28)75-78(72,73)74)45(67)53-26-40(60)54-38(22-29-25-52-33-11-7-6-10-31(29)33)48(70)56-35(17-19-77-2)46(68)59-39(24-42(63)64)49(71)57-36(43(51)65)20-27-8-4-3-5-9-27/h3-15,25,32,34-39,52H,16-24,26,50H2,1-2H3,(H2,51,65)(H,53,67)(H,54,60)(H,55,69)(H,56,70)(H,57,71)(H,58,66)(H,59,68)(H,61,62)(H,63,64)(H,72,73,74)/t32-,34-,35-,36-,37-,38-,39-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 31 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 63 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM82480

(YM022 S-isomer)Show SMILES Cc1cccc(NC(=O)N[C@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |r,t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| PubMed
| 140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM82480

(YM022 S-isomer)Show SMILES Cc1cccc(NC(=O)N[C@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |r,t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| PubMed
| 490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50005463

((R)-1H-Indole-2-carboxylic acid (1-methyl-2-oxo-5-...)Show SMILES CN1c2ccccc2C(=N[C@H](NC(=O)c2cc3ccccc3[nH]2)C1=O)c1ccccc1 |r,c:9| Show InChI InChI=1S/C25H20N4O2/c1-29-21-14-8-6-12-18(21)22(16-9-3-2-4-10-16)27-23(25(29)31)28-24(30)20-15-17-11-5-7-13-19(17)26-20/h2-15,23,26H,1H3,(H,28,30)/t23-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM50061220

(1-((S)-1-Methyl-2-oxo-5-phenyl-2,3-dihydro-1H-benz...)Show SMILES CN1c2ccccc2C(=N[C@H](NC(=O)Nc2cccc(C)c2)C1=O)c1ccccc1 |c:9| Show InChI InChI=1S/C24H22N4O2/c1-16-9-8-12-18(15-16)25-24(30)27-22-23(29)28(2)20-14-7-6-13-19(20)21(26-22)17-10-4-3-5-11-17/h3-15,22H,1-2H3,(H2,25,27,30)/t22-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 5.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM82403

(CAS_108186 | CI-988 | NSC_108186)Show SMILES CC(Cc1c[nH]c2ccccc12)(NC(=O)OC1C2CC3CC(C2)CC1C3)C(=O)NCC(NC(=O)CCC(O)=O)c1ccccc1 |TLB:25:24:18.19.20:22,THB:15:16:18.19.20:22,20:19:16:21.22.23,20:21:16:18.19.25,25:19:22:16.23.24,(.76,2.29,;.32,.82,;-.11,-.66,;.95,-1.78,;.68,-3.29,;2.03,-4.02,;3.15,-2.96,;4.68,-3.07,;5.55,-1.8,;4.88,-.42,;3.35,-.3,;2.48,-1.57,;1.8,.38,;2.92,1.45,;2.55,2.94,;4.39,1.01,;5.51,2.08,;6.88,2.77,;6.88,4.17,;7.72,5.64,;9.09,4.95,;9.09,3.55,;8.25,2.08,;7.72,2.85,;6.35,3.55,;6.35,4.95,;-1.15,1.25,;-1.52,2.75,;-2.27,.19,;-3.75,.62,;-4.86,-.44,;-6.34,-.01,;-7.45,-1.07,;-7.09,-2.57,;-8.93,-.64,;-10.04,-1.7,;-11.52,-1.27,;-11.89,.23,;-12.64,-2.33,;-4.5,-1.94,;-3.02,-2.37,;-2.65,-3.87,;-3.77,-4.93,;-5.25,-4.5,;-5.61,-3,)| Show InChI InChI=1S/C35H42N4O6/c1-35(18-26-19-36-28-10-6-5-9-27(26)28,39-34(44)45-32-24-14-21-13-22(16-24)17-25(32)15-21)33(43)37-20-29(23-7-3-2-4-8-23)38-30(40)11-12-31(41)42/h2-10,19,21-22,24-25,29,32,36H,11-18,20H2,1H3,(H,37,43)(H,38,40)(H,39,44)(H,41,42) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 6.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Sigma non-opioid intracellular receptor 1
(RAT) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Rattus norvegicus) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Cavia porcellus (domestic guinea pig)) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Gamma-aminobutyric acid receptor subunit alpha-1
(Rattus norvegicus (Rat)) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Gamma-aminobutyric acid receptor subunit alpha-1
(Rattus norvegicus (Rat)) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Rattus norvegicus (rat)) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
D(1A) dopamine receptor
(RAT) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(RAT) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(RAT) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Gastrin/cholecystokinin type B receptor
(RAT) | BDBM82235

(Gastrin I | Gastrin-17 | Gastrin-I-(1-17))Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)CNC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](C)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCSC)NC(=O)[C@@H]1CCCN1C(=O)[C@@H]1CCCN1C(=O)[C@H](CCCN=C(N)N)NC(=O)[C@@H]1CCC(=O)N1)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |r,wU:4.4,138.143,38.41,52.60,70.78,88.95,96.98,wD:8.20,130.135,26.37,43.51,61.69,79.87,103.106,110.122,121.125,(26,6.14,;24.67,5.35,;24.67,3.8,;23.32,3.01,;23.32,1.49,;22,.73,;20.68,1.49,;20.68,3.04,;19.35,.73,;19.35,-.76,;20.68,-1.52,;21.94,-.7,;23.15,-1.69,;22.59,-3.1,;23.29,-4.48,;22.45,-5.77,;20.9,-5.71,;20.2,-4.31,;21.04,-2.98,;18,1.49,;16.68,.7,;16.68,-.76,;15.36,1.49,;14.03,.7,;12.68,1.46,;12.68,3.01,;11.36,.68,;11.36,-.79,;12.71,-1.55,;12.71,-3.1,;14.03,-3.86,;15.36,-3.07,;16.71,-3.86,;15.36,-1.55,;14.03,-.79,;10.01,1.44,;8.66,.68,;8.66,-.82,;7.33,1.44,;7.3,2.98,;6.01,.65,;4.66,1.44,;4.66,2.98,;3.34,.65,;3.34,-.84,;4.66,-1.6,;4.66,-3.15,;6.01,-3.91,;3.34,-3.91,;2.01,1.44,;.66,.65,;.66,-.84,;-.61,1.44,;-.61,2.96,;.66,3.74,;.66,5.32,;2.01,6.11,;-.61,6.11,;-1.93,.65,;-3.28,1.41,;-3.28,2.96,;-4.6,.65,;-4.6,-.84,;-3.28,-1.6,;-3.28,-3.15,;-1.93,-3.94,;-4.6,-3.94,;-5.93,1.41,;-7.28,.65,;-7.28,-.84,;-8.6,1.44,;-8.6,2.96,;-7.28,3.74,;-7.28,5.32,;-5.93,6.11,;-8.6,6.11,;-9.95,.65,;-11.33,1.44,;-11.33,2.98,;-12.65,.65,;-12.65,-.82,;-11.33,-1.6,;-11.33,-3.15,;-9.95,-3.94,;-12.65,-3.91,;-13.98,1.44,;-15.33,.68,;-15.33,-.79,;-16.65,1.44,;-16.65,2.96,;-15.3,3.77,;-15.3,5.29,;-13.98,6.08,;-17.97,.68,;-19.32,1.44,;-19.32,2.98,;-20.62,.68,;-19.69,1.83,;-20.39,3.26,;-21.91,2.96,;-21.91,1.44,;-23.24,.68,;-23.24,-.96,;-24.7,1.52,;-23.97,.34,;-24.56,-.98,;-26.16,-.7,;-26.16,.68,;-27.52,1.44,;-27.52,2.98,;-28.84,.68,;-28.84,-.82,;-27.52,-1.55,;-27.52,-3.1,;-26.14,-3.86,;-26.11,-5.4,;-27.52,-6.16,;-24.78,-6.16,;-30.19,1.44,;-31.51,.68,;-31.51,-.82,;-32.84,1.41,;-31.85,.28,;-32.55,-1.1,;-34.07,-.79,;-35.37,-1.58,;-34.07,.68,;24.67,.73,;24.67,-.73,;26,1.49,;27.35,.7,;27.35,-.76,;28.7,-1.55,;30.02,-.76,;28.7,-3.07,;28.7,1.49,;28.7,3.04,;30.02,.7,;31.34,1.49,;31.34,3.01,;32.7,3.83,;32.7,5.38,;34.02,6.16,;35.37,5.38,;35.37,3.83,;34.02,3.01,;32.7,.7,;34.02,1.49,;32.7,-.76,)| Show InChI InChI=1S/C94H128N22O31S2/c1-48(79(133)113-65(43-50-19-21-52(117)22-20-50)80(134)100-47-71(119)103-66(44-51-46-99-54-15-8-7-14-53(51)54)89(143)109-61(35-40-148-2)88(142)114-67(45-77(130)131)90(144)112-64(78(95)132)42-49-12-5-4-6-13-49)101-81(135)56(24-30-72(120)121)104-83(137)57(25-31-73(122)123)105-84(138)58(26-32-74(124)125)106-85(139)59(27-33-75(126)127)107-86(140)60(28-34-76(128)129)108-87(141)62(36-41-149-3)110-91(145)68-17-10-38-115(68)93(147)69-18-11-39-116(69)92(146)63(16-9-37-98-94(96)97)111-82(136)55-23-29-70(118)102-55/h4-8,12-15,19-22,46,48,55-69,99,117H,9-11,16-18,23-45,47H2,1-3H3,(H2,95,132)(H,100,134)(H,101,135)(H,102,118)(H,103,119)(H,104,137)(H,105,138)(H,106,139)(H,107,140)(H,108,141)(H,109,143)(H,110,145)(H,111,136)(H,112,144)(H,113,133)(H,114,142)(H,120,121)(H,122,123)(H,124,125)(H,126,127)(H,128,129)(H,130,131)(H4,96,97,98)/t48-,55-,56-,57-,58-,59-,60-,61-,62-,63-,64-,65-,66-,67-,68-,69-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Alpha-2C adrenergic receptor
(RAT) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Alpha-1A adrenergic receptor
(Rattus norvegicus (Rat)) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Rattus norvegicus (rat)) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Glutamate receptor 3
(RAT) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Rattus norvegicus (rat)) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Rattus norvegicus (rat)) | BDBM50056101

(1-[(R)-2-Oxo-1-(2-oxo-2-o-tolyl-ethyl)-5-phenyl-2,...)Show SMILES Cc1cccc(NC(=O)N[C@@H]2N=C(c3ccccc3)c3ccccc3N(CC(=O)c3ccccc3C)C2=O)c1 |t:11| Show InChI InChI=1S/C32H28N4O3/c1-21-11-10-15-24(19-21)33-32(39)35-30-31(38)36(20-28(37)25-16-7-6-12-22(25)2)27-18-9-8-17-26(27)29(34-30)23-13-4-3-5-14-23/h3-19,30H,20H2,1-2H3,(H2,33,35,39)/t30-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Co. Ltd.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 269: 725-31 (1994)
BindingDB Entry DOI: 10.7270/Q2WD3Z2F |
More data for this
Ligand-Target Pair | |
Chemerin-like receptor 1
(Homo sapiens (Human)) | BDBM50560538
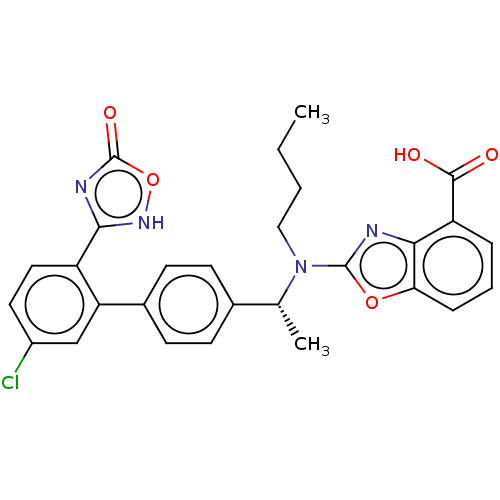
(CHEMBL4746987)Show SMILES CCCCN([C@H](C)c1ccc(cc1)-c1cc(Cl)ccc1-c1nc(=O)o[nH]1)c1nc2c(cccc2o1)C(O)=O |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of chemR23 in human CAL1 cells assessed as inhibition of chemerin-induced intracellular calcium ion concentration preincubated for 45 mins... |
Citation and Details
Article DOI: 10.1016/j.bmc.2020.115622
BindingDB Entry DOI: 10.7270/Q2NG4VC4 |
More data for this
Ligand-Target Pair | |
Chemerin-like receptor 1
(Homo sapiens (Human)) | BDBM50560541
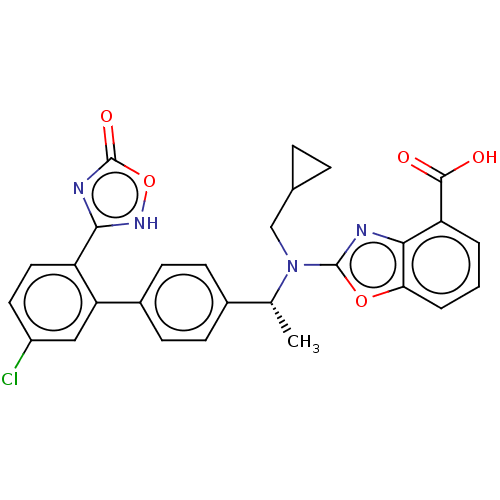
(CHEMBL4757632)Show SMILES C[C@@H](N(CC1CC1)c1nc2c(cccc2o1)C(O)=O)c1ccc(cc1)-c1cc(Cl)ccc1-c1nc(=O)o[nH]1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of chemR23 in human CAL1 cells assessed as inhibition of chemerin-induced intracellular calcium ion concentration preincubated for 45 mins... |
Citation and Details
Article DOI: 10.1016/j.bmc.2020.115622
BindingDB Entry DOI: 10.7270/Q2NG4VC4 |
More data for this
Ligand-Target Pair | |
Chemerin-like receptor 1
(Homo sapiens (Human)) | BDBM50560540

(CHEMBL4793378)Show SMILES C[C@@H](N(CC1CC1)c1nc2c(cccc2o1)C(O)=O)c1ccc(cc1)-c1cc(F)ccc1-c1nc(=O)o[nH]1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of chemR23 in human CAL1 cells assessed as inhibition of chemerin-induced intracellular calcium ion concentration preincubated for 45 mins... |
Citation and Details
Article DOI: 10.1016/j.bmc.2020.115622
BindingDB Entry DOI: 10.7270/Q2NG4VC4 |
More data for this
Ligand-Target Pair | |
Chemerin-like receptor 1
(Homo sapiens (Human)) | BDBM50560537
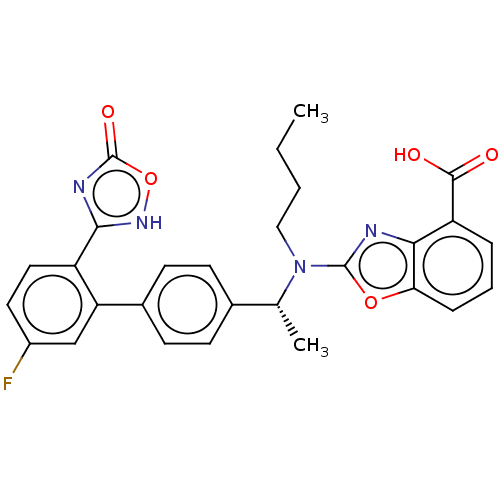
(CHEMBL4794289)Show SMILES CCCCN([C@H](C)c1ccc(cc1)-c1cc(F)ccc1-c1nc(=O)o[nH]1)c1nc2c(cccc2o1)C(O)=O |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of chemR23 in human CAL1 cells assessed as inhibition of chemerin-induced intracellular calcium ion concentration preincubated for 45 mins... |
Citation and Details
Article DOI: 10.1016/j.bmc.2020.115622
BindingDB Entry DOI: 10.7270/Q2NG4VC4 |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50003815
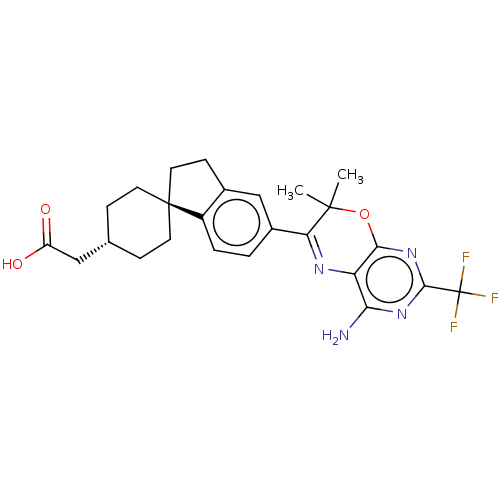
(CHEMBL3235321)Show SMILES CC1(C)Oc2nc(nc(N)c2N=C1c1ccc2c(CC[C@@]22CC[C@H](CC(O)=O)CC2)c1)C(F)(F)F |r,wU:20.22,wD:23.26,c:12,(27.23,-21.22,;25.69,-21.22,;26.46,-22.55,;24.36,-22,;23.03,-21.23,;21.69,-22.01,;20.36,-21.24,;20.36,-19.69,;21.69,-18.92,;21.68,-17.38,;23.02,-19.69,;24.35,-18.91,;25.69,-19.67,;27.02,-18.9,;27,-17.37,;28.32,-16.58,;29.67,-17.35,;29.68,-18.88,;31.15,-19.35,;32.04,-18.1,;31.12,-16.86,;30.02,-15.78,;30.42,-14.29,;31.91,-13.88,;32.3,-12.39,;33.79,-11.99,;34.89,-13.08,;34.19,-10.5,;33,-14.97,;32.61,-16.45,;28.35,-19.66,;19.02,-22.01,;17.69,-21.24,;19.02,-23.55,;17.68,-22.77,)| Show InChI InChI=1S/C25H27F3N4O3/c1-23(2)19(30-18-20(29)31-22(25(26,27)28)32-21(18)35-23)15-3-4-16-14(12-15)7-10-24(16)8-5-13(6-9-24)11-17(33)34/h3-4,12-13H,5-11H2,1-2H3,(H,33,34)(H2,29,31,32)/t13-,24- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of DGAT1 (unknown origin) by cell-based assay |
J Med Chem 57: 3464-83 (2014)
Article DOI: 10.1021/jm500135c
BindingDB Entry DOI: 10.7270/Q23X886W |
More data for this
Ligand-Target Pair | |
Chemerin-like receptor 1
(Homo sapiens (Human)) | BDBM50560539
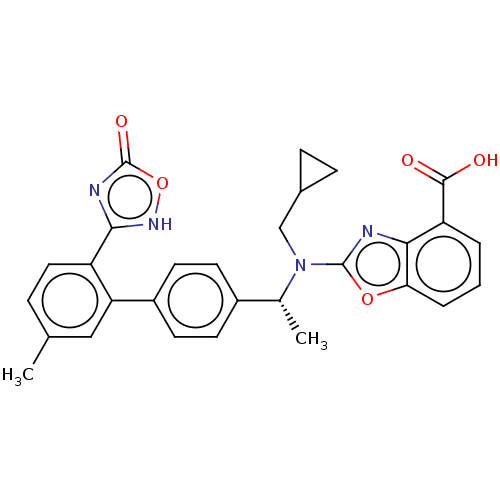
(CHEMBL4783675)Show SMILES C[C@@H](N(CC1CC1)c1nc2c(cccc2o1)C(O)=O)c1ccc(cc1)-c1cc(C)ccc1-c1nc(=O)o[nH]1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of chemR23 in human CAL1 cells assessed as inhibition of chemerin-induced intracellular calcium ion concentration preincubated for 45 mins... |
Citation and Details
Article DOI: 10.1016/j.bmc.2020.115622
BindingDB Entry DOI: 10.7270/Q2NG4VC4 |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
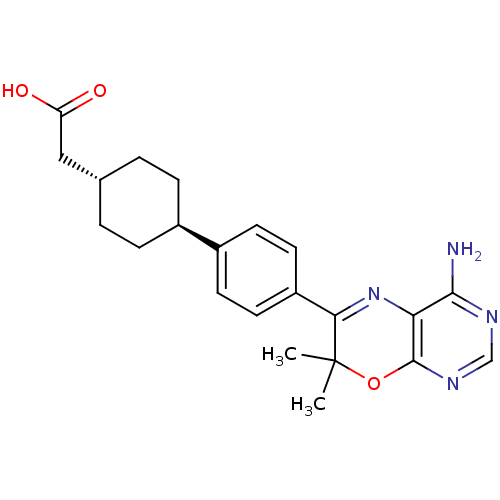
(Homo sapiens (Human)) | BDBM27947
(2-[4-(4-{4-amino-7,7-dimethyl-7H-pyrimido[4,5-b][1...)Show SMILES CC1(C)Oc2ncnc(N)c2N=C1c1ccc(cc1)[C@H]1CC[C@H](CC(O)=O)CC1 |r,wU:19.21,wD:22.25,c:12,(1.74,.99,;.97,2.32,;2.46,2.72,;-.37,1.55,;-1.7,2.32,;-3.03,1.55,;-4.37,2.32,;-4.37,3.86,;-3.03,4.63,;-3.03,6.17,;-1.7,3.86,;-.37,4.63,;.97,3.86,;2.3,4.63,;2.3,6.17,;3.63,6.94,;4.97,6.17,;4.97,4.63,;3.63,3.86,;6.19,6.75,;6.19,8.3,;7.53,9.07,;8.86,8.3,;10.19,9.07,;11.53,8.3,;12.86,9.07,;11.53,6.76,;8.86,6.75,;7.53,5.98,)| Show InChI InChI=1S/C22H26N4O3/c1-22(2)19(26-18-20(23)24-12-25-21(18)29-22)16-9-7-15(8-10-16)14-5-3-13(4-6-14)11-17(27)28/h7-10,12-14H,3-6,11H2,1-2H3,(H,27,28)(H2,23,24,25)/t13-,14- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of DGAT1 (unknown origin) by cell-based assay |
J Med Chem 57: 3464-83 (2014)
Article DOI: 10.1021/jm500135c
BindingDB Entry DOI: 10.7270/Q23X886W |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50003815

(CHEMBL3235321)Show SMILES CC1(C)Oc2nc(nc(N)c2N=C1c1ccc2c(CC[C@@]22CC[C@H](CC(O)=O)CC2)c1)C(F)(F)F |r,wU:20.22,wD:23.26,c:12,(27.23,-21.22,;25.69,-21.22,;26.46,-22.55,;24.36,-22,;23.03,-21.23,;21.69,-22.01,;20.36,-21.24,;20.36,-19.69,;21.69,-18.92,;21.68,-17.38,;23.02,-19.69,;24.35,-18.91,;25.69,-19.67,;27.02,-18.9,;27,-17.37,;28.32,-16.58,;29.67,-17.35,;29.68,-18.88,;31.15,-19.35,;32.04,-18.1,;31.12,-16.86,;30.02,-15.78,;30.42,-14.29,;31.91,-13.88,;32.3,-12.39,;33.79,-11.99,;34.89,-13.08,;34.19,-10.5,;33,-14.97,;32.61,-16.45,;28.35,-19.66,;19.02,-22.01,;17.69,-21.24,;19.02,-23.55,;17.68,-22.77,)| Show InChI InChI=1S/C25H27F3N4O3/c1-23(2)19(30-18-20(29)31-22(25(26,27)28)32-21(18)35-23)15-3-4-16-14(12-15)7-10-24(16)8-5-13(6-9-24)11-17(33)34/h3-4,12-13H,5-11H2,1-2H3,(H,33,34)(H2,29,31,32)/t13-,24- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human liver flag-tagged DGAT1 expressed in baculovirus infected insect cells after 2 hrs by SPA |
J Med Chem 57: 3464-83 (2014)
Article DOI: 10.1021/jm500135c
BindingDB Entry DOI: 10.7270/Q23X886W |
More data for this
Ligand-Target Pair | |
Chemerin-like receptor 1
(Homo sapiens (Human)) | BDBM50603313
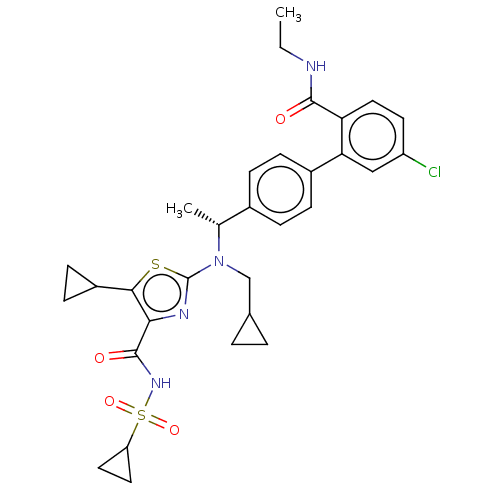
(CHEMBL5177967)Show SMILES CCNC(=O)c1ccc(Cl)cc1-c1ccc(cc1)[C@@H](C)N(CC1CC1)c1nc(C(=O)NS(=O)(=O)C2CC2)c(s1)C1CC1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmc.2021.116587
BindingDB Entry DOI: 10.7270/Q28W3JCR |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50003860

(CHEMBL3235317)Show SMILES CC1(C)Oc2nc(nc(N)c2N=C1c1ccc(cc1)[C@H]1CC[C@H](CC(O)=O)CC1)C(F)(F)F |r,wU:19.21,wD:22.25,c:12,(27.64,-8.52,;26.1,-8.52,;26.87,-9.85,;24.77,-9.3,;23.44,-8.53,;22.1,-9.31,;20.77,-8.54,;20.77,-6.99,;22.1,-6.22,;22.1,-4.68,;23.44,-6.99,;24.76,-6.21,;26.1,-6.97,;27.43,-6.19,;28.76,-6.96,;30.09,-6.19,;30.08,-4.64,;28.73,-3.88,;27.41,-4.66,;31.41,-3.86,;31.39,-2.32,;32.72,-1.55,;34.06,-2.31,;35.39,-1.54,;36.73,-2.3,;36.74,-3.84,;38.06,-1.53,;34.06,-3.85,;32.74,-4.63,;19.44,-9.31,;18.1,-8.53,;19.43,-10.85,;18.09,-10.06,)| Show InChI InChI=1S/C23H25F3N4O3/c1-22(2)18(28-17-19(27)29-21(23(24,25)26)30-20(17)33-22)15-9-7-14(8-10-15)13-5-3-12(4-6-13)11-16(31)32/h7-10,12-13H,3-6,11H2,1-2H3,(H,31,32)(H2,27,29,30)/t12-,13- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human liver flag-tagged DGAT1 expressed in baculovirus infected insect cells after 2 hrs by SPA |
J Med Chem 57: 3464-83 (2014)
Article DOI: 10.1021/jm500135c
BindingDB Entry DOI: 10.7270/Q23X886W |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM27947

(2-[4-(4-{4-amino-7,7-dimethyl-7H-pyrimido[4,5-b][1...)Show SMILES CC1(C)Oc2ncnc(N)c2N=C1c1ccc(cc1)[C@H]1CC[C@H](CC(O)=O)CC1 |r,wU:19.21,wD:22.25,c:12,(1.74,.99,;.97,2.32,;2.46,2.72,;-.37,1.55,;-1.7,2.32,;-3.03,1.55,;-4.37,2.32,;-4.37,3.86,;-3.03,4.63,;-3.03,6.17,;-1.7,3.86,;-.37,4.63,;.97,3.86,;2.3,4.63,;2.3,6.17,;3.63,6.94,;4.97,6.17,;4.97,4.63,;3.63,3.86,;6.19,6.75,;6.19,8.3,;7.53,9.07,;8.86,8.3,;10.19,9.07,;11.53,8.3,;12.86,9.07,;11.53,6.76,;8.86,6.75,;7.53,5.98,)| Show InChI InChI=1S/C22H26N4O3/c1-22(2)19(26-18-20(23)24-12-25-21(18)29-22)16-9-7-15(8-10-16)14-5-3-13(4-6-14)11-17(27)28/h7-10,12-14H,3-6,11H2,1-2H3,(H,27,28)(H2,23,24,25)/t13-,14- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human liver flag-tagged DGAT1 expressed in baculovirus infected insect cells after 2 hrs by SPA |
J Med Chem 57: 3464-83 (2014)
Article DOI: 10.1021/jm500135c
BindingDB Entry DOI: 10.7270/Q23X886W |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Diacylglycerol O-acyltransferase 1
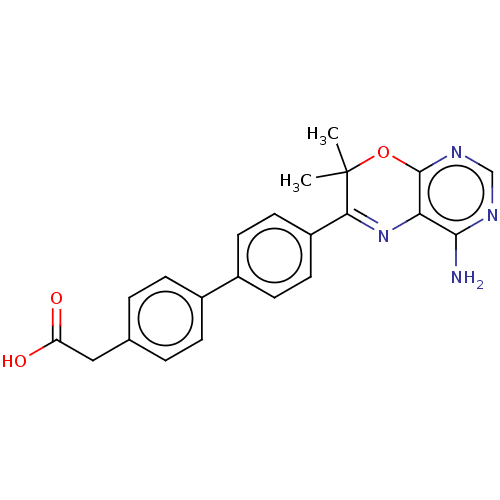
(Homo sapiens (Human)) | BDBM50003858
(CHEMBL3235315)Show SMILES CC1(C)Oc2ncnc(N)c2N=C1c1ccc(cc1)-c1ccc(CC(O)=O)cc1 |c:12| Show InChI InChI=1S/C22H20N4O3/c1-22(2)19(26-18-20(23)24-12-25-21(18)29-22)16-9-7-15(8-10-16)14-5-3-13(4-6-14)11-17(27)28/h3-10,12H,11H2,1-2H3,(H,27,28)(H2,23,24,25) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human liver flag-tagged DGAT1 expressed in baculovirus infected insect cells after 2 hrs by SPA |
J Med Chem 57: 3464-83 (2014)
Article DOI: 10.1021/jm500135c
BindingDB Entry DOI: 10.7270/Q23X886W |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50003857
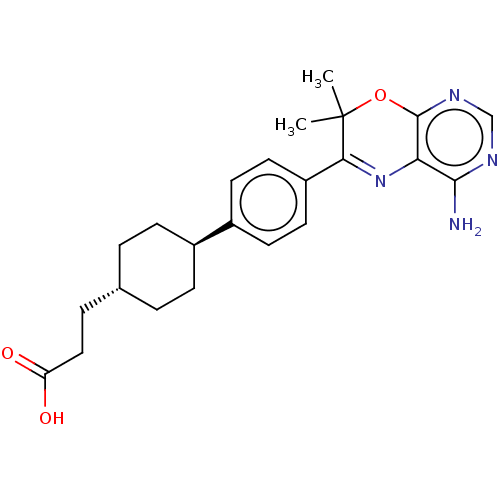
(CHEMBL3235314)Show SMILES CC1(C)Oc2ncnc(N)c2N=C1c1ccc(cc1)[C@H]1CC[C@H](CCC(O)=O)CC1 |r,wU:19.21,wD:22.25,c:12,(25.63,-58.39,;24.09,-58.39,;24.86,-59.72,;22.76,-59.17,;21.42,-58.4,;20.09,-59.18,;18.75,-58.41,;18.76,-56.86,;20.09,-56.09,;20.08,-54.55,;21.42,-56.86,;22.75,-56.08,;24.08,-56.84,;25.41,-56.06,;26.75,-56.83,;28.07,-56.06,;28.07,-54.51,;26.72,-53.75,;25.4,-54.53,;29.39,-53.73,;29.37,-52.2,;30.71,-51.42,;32.05,-52.18,;33.38,-51.41,;34.71,-52.17,;34.72,-53.71,;33.39,-54.49,;36.06,-54.48,;32.05,-53.72,;30.73,-54.5,)| Show InChI InChI=1S/C23H28N4O3/c1-23(2)20(27-19-21(24)25-13-26-22(19)30-23)17-10-8-16(9-11-17)15-6-3-14(4-7-15)5-12-18(28)29/h8-11,13-15H,3-7,12H2,1-2H3,(H,28,29)(H2,24,25,26)/t14-,15- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human liver flag-tagged DGAT1 expressed in baculovirus infected insect cells after 2 hrs by SPA |
J Med Chem 57: 3464-83 (2014)
Article DOI: 10.1021/jm500135c
BindingDB Entry DOI: 10.7270/Q23X886W |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50003862

(CHEMBL3235319)Show SMILES CC1(C)Oc2ncnc(N)c2N=C1c1ccc(cc1)[C@H]1CC[C@H](Cc2nnn[nH]2)CC1 |r,wU:19.21,wD:22.25,c:12,(58.66,-9.38,;57.12,-9.38,;57.89,-10.71,;55.79,-10.16,;54.46,-9.39,;53.12,-10.17,;51.79,-9.4,;51.79,-7.86,;53.12,-7.08,;53.11,-5.54,;54.45,-7.85,;55.78,-7.08,;57.12,-7.84,;58.45,-7.06,;59.78,-7.83,;61.11,-7.05,;61.1,-5.51,;59.75,-4.75,;58.43,-5.53,;62.43,-4.73,;62.4,-3.19,;63.74,-2.41,;65.08,-3.18,;66.41,-2.4,;67.75,-3.17,;69.14,-2.53,;70.18,-3.67,;69.41,-5.01,;67.9,-4.7,;65.08,-4.72,;63.76,-5.49,)| Show InChI InChI=1S/C22H26N8O/c1-22(2)19(26-18-20(23)24-12-25-21(18)31-22)16-9-7-15(8-10-16)14-5-3-13(4-6-14)11-17-27-29-30-28-17/h7-10,12-14H,3-6,11H2,1-2H3,(H2,23,24,25)(H,27,28,29,30)/t13-,14- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human liver flag-tagged DGAT1 expressed in baculovirus infected insect cells after 2 hrs by SPA |
J Med Chem 57: 3464-83 (2014)
Article DOI: 10.1021/jm500135c
BindingDB Entry DOI: 10.7270/Q23X886W |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50003864

(CHEMBL3235322)Show SMILES CC1(C)Oc2ncnc(N)c2C=C1c1ccc(cc1)[C@H]1CC[C@H](CC(O)=O)CC1 |r,wU:19.21,wD:22.25,c:12,(42.97,-20.87,;41.43,-20.87,;42.2,-22.2,;40.1,-21.65,;38.77,-20.88,;37.43,-21.66,;36.1,-20.89,;36.1,-19.34,;37.43,-18.57,;37.43,-17.03,;38.77,-19.34,;40.09,-18.56,;41.43,-19.32,;42.76,-18.55,;44.09,-19.31,;45.42,-18.54,;45.41,-17,;44.06,-16.23,;42.74,-17.02,;46.74,-16.22,;46.72,-14.68,;48.05,-13.9,;49.39,-14.66,;50.72,-13.89,;52.06,-14.65,;52.07,-16.19,;53.39,-13.88,;49.39,-16.2,;48.07,-16.98,)| Show InChI InChI=1S/C23H27N3O3/c1-23(2)19(12-18-21(24)25-13-26-22(18)29-23)17-9-7-16(8-10-17)15-5-3-14(4-6-15)11-20(27)28/h7-10,12-15H,3-6,11H2,1-2H3,(H,27,28)(H2,24,25,26)/t14-,15- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human liver flag-tagged DGAT1 expressed in baculovirus infected insect cells after 2 hrs by SPA |
J Med Chem 57: 3464-83 (2014)
Article DOI: 10.1021/jm500135c
BindingDB Entry DOI: 10.7270/Q23X886W |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50003859
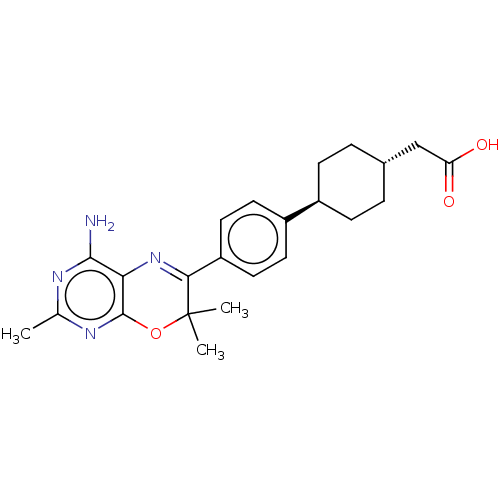
(CHEMBL3235316)Show SMILES Cc1nc(N)c2N=C(c3ccc(cc3)[C@H]3CC[C@H](CC(O)=O)CC3)C(C)(C)Oc2n1 |r,wU:14.14,wD:17.18,t:6,(.98,-9.8,;2.31,-9.03,;2.32,-7.49,;3.64,-6.72,;3.64,-5.18,;4.98,-7.48,;6.31,-6.71,;7.64,-7.47,;8.97,-6.69,;10.3,-7.46,;11.63,-6.68,;11.62,-5.14,;10.28,-4.38,;8.95,-5.16,;12.95,-4.36,;12.93,-2.82,;14.26,-2.05,;15.6,-2.81,;16.93,-2.04,;18.27,-2.8,;18.28,-4.34,;19.61,-2.02,;15.61,-4.35,;14.28,-5.12,;7.65,-9.01,;9.19,-9.01,;8.42,-10.35,;6.32,-9.79,;4.98,-9.03,;3.65,-9.8,)| Show InChI InChI=1S/C23H28N4O3/c1-13-25-21(24)19-22(26-13)30-23(2,3)20(27-19)17-10-8-16(9-11-17)15-6-4-14(5-7-15)12-18(28)29/h8-11,14-15H,4-7,12H2,1-3H3,(H,28,29)(H2,24,25,26)/t14-,15- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human liver flag-tagged DGAT1 expressed in baculovirus infected insect cells after 2 hrs by SPA |
J Med Chem 57: 3464-83 (2014)
Article DOI: 10.1021/jm500135c
BindingDB Entry DOI: 10.7270/Q23X886W |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data