 Found 87 hits with Last Name = 'leung' and Initial = 'a'
Found 87 hits with Last Name = 'leung' and Initial = 'a' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Tyrosinase
(Mus musculus (Mouse)) | BDBM50020713
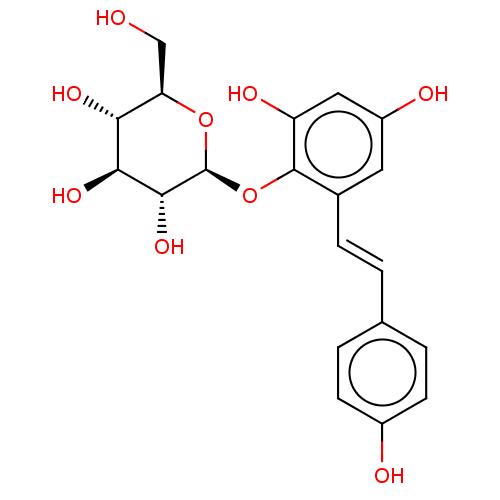
(CHEMBL460860)Show SMILES OC[C@H]1O[C@@H](Oc2c(O)cc(O)cc2\C=C\c2ccc(O)cc2)[C@H](O)[C@@H](O)[C@@H]1O |r| Show InChI InChI=1S/C20H22O9/c21-9-15-16(25)17(26)18(27)20(28-15)29-19-11(7-13(23)8-14(19)24)4-1-10-2-5-12(22)6-3-10/h1-8,15-18,20-27H,9H2/b4-1+/t15-,16-,17+,18-,20+/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.50E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Chinese University of Hong Kong
Curated by ChEMBL
| Assay Description
Inhibition of tyrosinase in mouse Melan-a cells assessed as decrease in L-DOPA Vmax at 240 uM by Lineweaver-Burk plot |
J Nat Prod 77: 1270-4 (2014)
Article DOI: 10.1021/np4008798
BindingDB Entry DOI: 10.7270/Q2MK6FFJ |
More data for this
Ligand-Target Pair | |
Tyrosinase
(Mus musculus (Mouse)) | BDBM50020713

(CHEMBL460860)Show SMILES OC[C@H]1O[C@@H](Oc2c(O)cc(O)cc2\C=C\c2ccc(O)cc2)[C@H](O)[C@@H](O)[C@@H]1O |r| Show InChI InChI=1S/C20H22O9/c21-9-15-16(25)17(26)18(27)20(28-15)29-19-11(7-13(23)8-14(19)24)4-1-10-2-5-12(22)6-3-10/h1-8,15-18,20-27H,9H2/b4-1+/t15-,16-,17+,18-,20+/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.80E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Chinese University of Hong Kong
Curated by ChEMBL
| Assay Description
Inhibition of tyrosinase in mouse Melan-a cells assessed as decrease in L-DOPA Vmax at 120 uM by Lineweaver-Burk plot |
J Nat Prod 77: 1270-4 (2014)
Article DOI: 10.1021/np4008798
BindingDB Entry DOI: 10.7270/Q2MK6FFJ |
More data for this
Ligand-Target Pair | |
Tyrosinase
(Mus musculus (Mouse)) | BDBM23926

((E)-resveratrol | 5-[(E)-2-(4-hydroxyphenyl)etheny...)Show InChI InChI=1S/C14H12O3/c15-12-5-3-10(4-6-12)1-2-11-7-13(16)9-14(17)8-11/h1-9,15-17H/b2-1+ | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 2.90E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Chinese University of Hong Kong
Curated by ChEMBL
| Assay Description
Inhibition of tyrosinase in mouse Melan-a cells assessed as decrease in L-DOPA Vmax at 120 uM by Lineweaver-Burk plot |
J Nat Prod 77: 1270-4 (2014)
Article DOI: 10.1021/np4008798
BindingDB Entry DOI: 10.7270/Q2MK6FFJ |
More data for this
Ligand-Target Pair | |
Tyrosinase
(Mus musculus (Mouse)) | BDBM50020713

(CHEMBL460860)Show SMILES OC[C@H]1O[C@@H](Oc2c(O)cc(O)cc2\C=C\c2ccc(O)cc2)[C@H](O)[C@@H](O)[C@@H]1O |r| Show InChI InChI=1S/C20H22O9/c21-9-15-16(25)17(26)18(27)20(28-15)29-19-11(7-13(23)8-14(19)24)4-1-10-2-5-12(22)6-3-10/h1-8,15-18,20-27H,9H2/b4-1+/t15-,16-,17+,18-,20+/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.10E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Chinese University of Hong Kong
Curated by ChEMBL
| Assay Description
Inhibition of tyrosinase in mouse Melan-a cells assessed as decrease in L-DOPA Vmax at 60 uM by Lineweaver-Burk plot |
J Nat Prod 77: 1270-4 (2014)
Article DOI: 10.1021/np4008798
BindingDB Entry DOI: 10.7270/Q2MK6FFJ |
More data for this
Ligand-Target Pair | |
Tyrosinase
(Mus musculus (Mouse)) | BDBM50031467
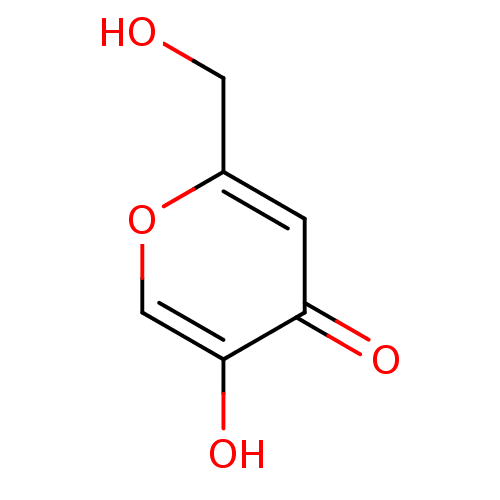
(5-HYDROXY-2-(HYDROXYMETHYL)-4H-PYRAN-4-ONE | 5-Hyd...)Show InChI InChI=1S/C6H6O4/c7-2-4-1-5(8)6(9)3-10-4/h1,3,7,9H,2H2 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 5.50E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Chinese University of Hong Kong
Curated by ChEMBL
| Assay Description
Inhibition of tyrosinase in mouse Melan-a cells assessed as decrease in L-DOPA Vmax at 120 uM by Lineweaver-Burk plot |
J Nat Prod 77: 1270-4 (2014)
Article DOI: 10.1021/np4008798
BindingDB Entry DOI: 10.7270/Q2MK6FFJ |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM8798
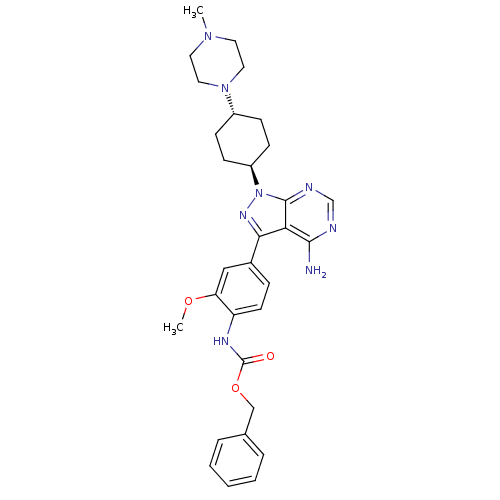
(CHEMBL47787 | Pyrazolo[3,4-d]pyrimidine 6 | benzyl...)Show SMILES COc1cc(ccc1NC(=O)OCc1ccccc1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |r,wU:22.23,wD:25.30,(.85,5.16,;-.29,6.2,;-1.75,5.73,;-2.08,4.23,;-3.55,3.76,;-4.69,4.8,;-4.36,6.3,;-2.89,6.77,;-2.56,8.27,;-1.1,8.74,;.04,7.7,;-.77,10.25,;.7,10.71,;1.03,12.22,;-.05,13.31,;.36,14.8,;1.85,15.19,;2.93,14.09,;2.52,12.61,;-4.03,2.3,;-3.12,1.05,;-4.03,-.2,;-3.55,-1.66,;-2.06,-2.04,;-1.64,-3.52,;-2.72,-4.63,;-4.21,-4.25,;-4.63,-2.76,;-2.3,-6.11,;-3.34,-7.24,;-2.87,-8.71,;-1.37,-9.04,;-.91,-10.51,;-.33,-7.91,;-.8,-6.44,;-5.49,.28,;-6.82,-.49,;-8.16,.28,;-8.16,1.82,;-6.82,2.59,;-6.82,4.13,;-5.49,1.82,)| Show InChI InChI=1S/C31H38N8O3/c1-37-14-16-38(17-15-37)23-9-11-24(12-10-23)39-30-27(29(32)33-20-34-30)28(36-39)22-8-13-25(26(18-22)41-2)35-31(40)42-19-21-6-4-3-5-7-21/h3-8,13,18,20,23-24H,9-12,14-17,19H2,1-2H3,(H,35,40)(H2,32,33,34)/t23-,24- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of p56 Lck tyrosine kinase activity with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Lactobacillus casei) | BDBM18050

(2-[(4-{[(2,4-diaminopteridin-6-yl)methyl](methyl)a...)Show SMILES CN(Cc1cnc2nc(N)nc(N)c2n1)c1ccc(cc1)C(=O)N[C@@H](CCC(O)=O)C(O)=O |r| Show InChI InChI=1S/C20H22N8O5/c1-28(9-11-8-23-17-15(24-11)16(21)26-20(22)27-17)12-4-2-10(3-5-12)18(31)25-13(19(32)33)6-7-14(29)30/h2-5,8,13H,6-7,9H2,1H3,(H,25,31)(H,29,30)(H,32,33)(H4,21,22,23,26,27)/t13-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Southern Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of Lactobacillus casei DHFR |
J Med Chem 50: 3283-9 (2007)
Article DOI: 10.1021/jm0701977
BindingDB Entry DOI: 10.7270/Q2TH8MCN |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM8798

(CHEMBL47787 | Pyrazolo[3,4-d]pyrimidine 6 | benzyl...)Show SMILES COc1cc(ccc1NC(=O)OCc1ccccc1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |r,wU:22.23,wD:25.30,(.85,5.16,;-.29,6.2,;-1.75,5.73,;-2.08,4.23,;-3.55,3.76,;-4.69,4.8,;-4.36,6.3,;-2.89,6.77,;-2.56,8.27,;-1.1,8.74,;.04,7.7,;-.77,10.25,;.7,10.71,;1.03,12.22,;-.05,13.31,;.36,14.8,;1.85,15.19,;2.93,14.09,;2.52,12.61,;-4.03,2.3,;-3.12,1.05,;-4.03,-.2,;-3.55,-1.66,;-2.06,-2.04,;-1.64,-3.52,;-2.72,-4.63,;-4.21,-4.25,;-4.63,-2.76,;-2.3,-6.11,;-3.34,-7.24,;-2.87,-8.71,;-1.37,-9.04,;-.91,-10.51,;-.33,-7.91,;-.8,-6.44,;-5.49,.28,;-6.82,-.49,;-8.16,.28,;-8.16,1.82,;-6.82,2.59,;-6.82,4.13,;-5.49,1.82,)| Show InChI InChI=1S/C31H38N8O3/c1-37-14-16-38(17-15-37)23-9-11-24(12-10-23)39-30-27(29(32)33-20-34-30)28(36-39)22-8-13-25(26(18-22)41-2)35-31(40)42-19-21-6-4-3-5-7-21/h3-8,13,18,20,23-24H,9-12,14-17,19H2,1-2H3,(H,35,40)(H2,32,33,34)/t23-,24- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of Src protein tryrosine kinase activity with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50145570

(1H-Indole-2-carboxylic acid (4-{4-amino-1-[4-(4-me...)Show SMILES COc1cc(ccc1NC(=O)c1cc2ccccc2[nH]1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |wU:23.25,wD:26.32,(1.45,-1.92,;1.52,-3.48,;2.9,-4.2,;2.97,-5.74,;4.32,-6.46,;5.63,-5.64,;5.57,-4.09,;4.21,-3.37,;4.2,-1.83,;5.51,-1.03,;6.86,-1.8,;5.49,.51,;6.63,1.55,;5.98,2.93,;6.61,4.35,;5.7,5.59,;4.18,5.43,;3.55,4.03,;4.46,2.77,;4.16,1.25,;4.39,-8,;5.62,-8.89,;5.14,-10.36,;6.09,-11.55,;7.63,-11.32,;8.58,-12.52,;8.02,-13.95,;6.49,-14.19,;5.53,-12.99,;8.99,-15.14,;10.5,-14.91,;11.47,-16.1,;10.91,-17.54,;11.88,-18.76,;9.38,-17.78,;8.43,-16.58,;3.61,-10.36,;2.57,-11.5,;1.08,-11.17,;.61,-9.71,;1.64,-8.57,;1.17,-7.1,;3.13,-8.89,)| Show InChI InChI=1S/C32H37N9O2/c1-39-13-15-40(16-14-39)22-8-10-23(11-9-22)41-31-28(30(33)34-19-35-31)29(38-41)21-7-12-25(27(18-21)43-2)37-32(42)26-17-20-5-3-4-6-24(20)36-26/h3-7,12,17-19,22-23,36H,8-11,13-16H2,1-2H3,(H,37,42)(H2,33,34,35)/t22-,23- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of p56 Lck tyrosine kinase activity with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Homo sapiens (Human)) | BDBM18050

(2-[(4-{[(2,4-diaminopteridin-6-yl)methyl](methyl)a...)Show SMILES CN(Cc1cnc2nc(N)nc(N)c2n1)c1ccc(cc1)C(=O)N[C@@H](CCC(O)=O)C(O)=O |r| Show InChI InChI=1S/C20H22N8O5/c1-28(9-11-8-23-17-15(24-11)16(21)26-20(22)27-17)12-4-2-10(3-5-12)18(31)25-13(19(32)33)6-7-14(29)30/h2-5,8,13H,6-7,9H2,1H3,(H,25,31)(H,29,30)(H,32,33)(H4,21,22,23,26,27)/t13-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Southern Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant DHFR |
J Med Chem 50: 3283-9 (2007)
Article DOI: 10.1021/jm0701977
BindingDB Entry DOI: 10.7270/Q2TH8MCN |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM8807
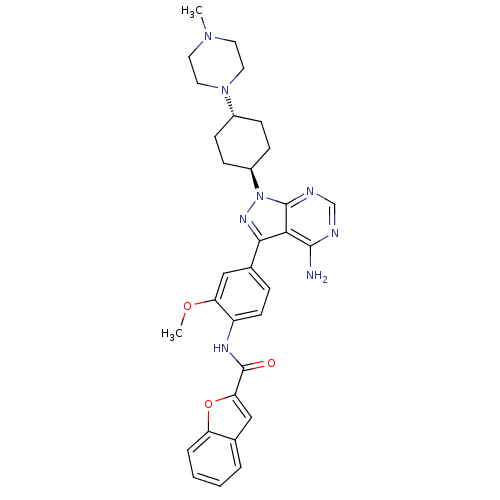
(CHEMBL43773 | N-(4-{4-amino-1-[4-(4-methylpiperazi...)Show SMILES COc1cc(ccc1NC(=O)c1cc2ccccc2o1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |r,wU:23.25,wD:26.32,(.85,5.16,;-.29,6.2,;-1.75,5.73,;-2.08,4.23,;-3.55,3.76,;-4.69,4.8,;-4.36,6.3,;-2.89,6.77,;-2.56,8.27,;-1.1,8.74,;.04,7.7,;-.77,10.25,;.65,10.85,;.52,12.38,;1.57,13.51,;1.12,14.98,;-.38,15.33,;-1.43,14.2,;-.98,12.73,;-1.78,11.41,;-4.03,2.3,;-3.12,1.05,;-4.03,-.2,;-3.55,-1.66,;-2.06,-2.04,;-1.64,-3.52,;-2.72,-4.63,;-4.21,-4.25,;-4.63,-2.76,;-2.3,-6.11,;-3.34,-7.24,;-2.87,-8.71,;-1.37,-9.04,;-.91,-10.51,;-.33,-7.91,;-.8,-6.44,;-5.49,.28,;-6.82,-.49,;-8.16,.28,;-8.16,1.82,;-6.82,2.59,;-6.82,4.13,;-5.49,1.82,)| Show InChI InChI=1S/C32H36N8O3/c1-38-13-15-39(16-14-38)22-8-10-23(11-9-22)40-31-28(30(33)34-19-35-31)29(37-40)21-7-12-24(26(18-21)42-2)36-32(41)27-17-20-5-3-4-6-25(20)43-27/h3-7,12,17-19,22-23H,8-11,13-16H2,1-2H3,(H,36,41)(H2,33,34,35)/t22-,23- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of p56 Lck tyrosine kinase activity with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM8803
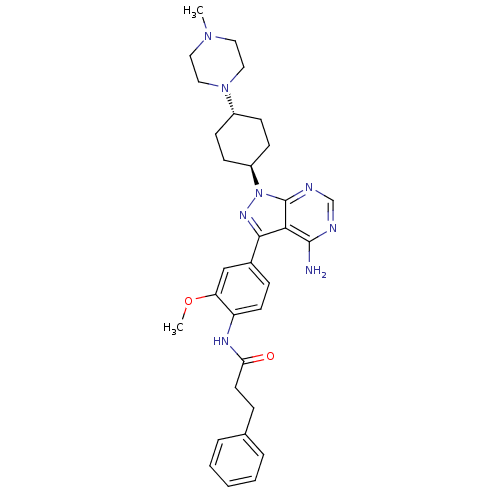
(CHEMBL295601 | N-(4-{4-amino-1-[4-(4-methylpiperaz...)Show SMILES COc1cc(ccc1NC(=O)CCc1ccccc1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |r,wU:22.23,wD:25.30,(.85,5.16,;-.29,6.2,;-1.75,5.73,;-2.08,4.23,;-3.55,3.76,;-4.69,4.8,;-4.36,6.3,;-2.89,6.77,;-2.56,8.27,;-1.1,8.74,;.04,7.7,;-.77,10.25,;.72,10.64,;1.13,12.12,;.1,13.28,;.59,14.74,;2.1,15.05,;3.12,13.9,;2.63,12.44,;-4.03,2.3,;-3.12,1.05,;-4.03,-.2,;-3.55,-1.66,;-2.06,-2.04,;-1.64,-3.52,;-2.72,-4.63,;-4.21,-4.25,;-4.63,-2.76,;-2.3,-6.11,;-3.34,-7.24,;-2.87,-8.71,;-1.37,-9.04,;-.91,-10.51,;-.33,-7.91,;-.8,-6.44,;-5.49,.28,;-6.82,-.49,;-8.16,.28,;-8.16,1.82,;-6.82,2.59,;-6.82,4.13,;-5.49,1.82,)| Show InChI InChI=1S/C32H40N8O2/c1-38-16-18-39(19-17-38)24-10-12-25(13-11-24)40-32-29(31(33)34-21-35-32)30(37-40)23-9-14-26(27(20-23)42-2)36-28(41)15-8-22-6-4-3-5-7-22/h3-7,9,14,20-21,24-25H,8,10-13,15-19H2,1-2H3,(H,36,41)(H2,33,34,35)/t24-,25- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of p56 Lck tyrosine kinase activity with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50145571
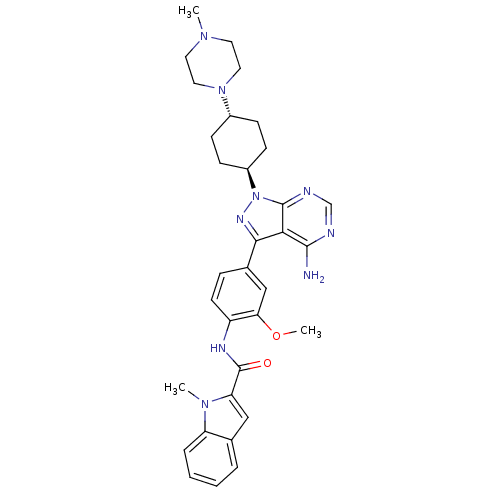
(1-Methyl-1H-indole-2-carboxylic acid (4-{4-amino-1...)Show SMILES COc1cc(ccc1NC(=O)c1cc2ccccc2n1C)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |wU:24.26,wD:27.33,(1.55,-1.69,;1.62,-3.24,;3.01,-3.96,;3.06,-5.49,;4.43,-6.21,;5.72,-5.39,;5.67,-3.86,;4.32,-3.13,;4.3,-1.59,;5.62,-.8,;6.97,-1.55,;5.6,.74,;6.72,1.79,;6.09,3.17,;6.72,4.59,;5.81,5.83,;4.29,5.68,;3.66,4.26,;4.56,3.01,;4.25,1.49,;2.85,.85,;4.49,-7.75,;5.72,-8.66,;5.25,-10.12,;6.2,-11.32,;7.73,-11.09,;8.68,-12.27,;8.12,-13.71,;6.6,-13.95,;5.63,-12.74,;9.08,-14.91,;10.61,-14.67,;11.57,-15.87,;11.02,-17.3,;11.97,-18.52,;9.49,-17.54,;8.52,-16.33,;3.72,-10.12,;2.68,-11.25,;1.19,-10.92,;.71,-9.48,;1.73,-8.33,;1.27,-6.86,;3.24,-8.66,)| Show InChI InChI=1S/C33H39N9O2/c1-39-14-16-41(17-15-39)23-9-11-24(12-10-23)42-32-29(31(34)35-20-36-32)30(38-42)22-8-13-25(28(19-22)44-3)37-33(43)27-18-21-6-4-5-7-26(21)40(27)2/h4-8,13,18-20,23-24H,9-12,14-17H2,1-3H3,(H,37,43)(H2,34,35,36)/t23-,24- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of p56 Lck tyrosine kinase activity with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50145570

(1H-Indole-2-carboxylic acid (4-{4-amino-1-[4-(4-me...)Show SMILES COc1cc(ccc1NC(=O)c1cc2ccccc2[nH]1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |wU:23.25,wD:26.32,(1.45,-1.92,;1.52,-3.48,;2.9,-4.2,;2.97,-5.74,;4.32,-6.46,;5.63,-5.64,;5.57,-4.09,;4.21,-3.37,;4.2,-1.83,;5.51,-1.03,;6.86,-1.8,;5.49,.51,;6.63,1.55,;5.98,2.93,;6.61,4.35,;5.7,5.59,;4.18,5.43,;3.55,4.03,;4.46,2.77,;4.16,1.25,;4.39,-8,;5.62,-8.89,;5.14,-10.36,;6.09,-11.55,;7.63,-11.32,;8.58,-12.52,;8.02,-13.95,;6.49,-14.19,;5.53,-12.99,;8.99,-15.14,;10.5,-14.91,;11.47,-16.1,;10.91,-17.54,;11.88,-18.76,;9.38,-17.78,;8.43,-16.58,;3.61,-10.36,;2.57,-11.5,;1.08,-11.17,;.61,-9.71,;1.64,-8.57,;1.17,-7.1,;3.13,-8.89,)| Show InChI InChI=1S/C32H37N9O2/c1-39-13-15-40(16-14-39)22-8-10-23(11-9-22)41-31-28(30(33)34-19-35-31)29(38-41)21-7-12-25(27(18-21)43-2)37-32(42)26-17-20-5-3-4-6-24(20)36-26/h3-7,12,17-19,22-23,36H,8-11,13-16H2,1-2H3,(H,37,42)(H2,33,34,35)/t22-,23- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 56 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of Src protein tryrosine kinase activity with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM8807

(CHEMBL43773 | N-(4-{4-amino-1-[4-(4-methylpiperazi...)Show SMILES COc1cc(ccc1NC(=O)c1cc2ccccc2o1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |r,wU:23.25,wD:26.32,(.85,5.16,;-.29,6.2,;-1.75,5.73,;-2.08,4.23,;-3.55,3.76,;-4.69,4.8,;-4.36,6.3,;-2.89,6.77,;-2.56,8.27,;-1.1,8.74,;.04,7.7,;-.77,10.25,;.65,10.85,;.52,12.38,;1.57,13.51,;1.12,14.98,;-.38,15.33,;-1.43,14.2,;-.98,12.73,;-1.78,11.41,;-4.03,2.3,;-3.12,1.05,;-4.03,-.2,;-3.55,-1.66,;-2.06,-2.04,;-1.64,-3.52,;-2.72,-4.63,;-4.21,-4.25,;-4.63,-2.76,;-2.3,-6.11,;-3.34,-7.24,;-2.87,-8.71,;-1.37,-9.04,;-.91,-10.51,;-.33,-7.91,;-.8,-6.44,;-5.49,.28,;-6.82,-.49,;-8.16,.28,;-8.16,1.82,;-6.82,2.59,;-6.82,4.13,;-5.49,1.82,)| Show InChI InChI=1S/C32H36N8O3/c1-38-13-15-39(16-14-38)22-8-10-23(11-9-22)40-31-28(30(33)34-19-35-31)29(37-40)21-7-12-24(26(18-21)42-2)36-32(41)27-17-20-5-3-4-6-25(20)43-27/h3-7,12,17-19,22-23H,8-11,13-16H2,1-2H3,(H,36,41)(H2,33,34,35)/t22-,23- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 57 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of Src protein tryrosine kinase activity with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50145572
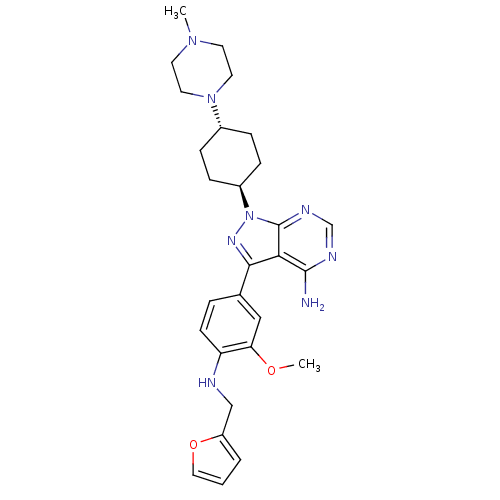
(3-{4-[(Furan-2-ylmethyl)-amino]-3-methoxy-phenyl}-...)Show SMILES COc1cc(ccc1NCc1ccco1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |wU:18.19,wD:21.26,(1.27,4.51,;1.34,2.96,;2.73,2.24,;2.78,.71,;4.15,-.01,;5.44,.81,;5.39,2.35,;4.04,3.08,;4.02,4.61,;5.34,5.4,;6.69,4.65,;8.02,5.45,;9.17,4.42,;8.56,3.02,;7.04,3.15,;4.2,-1.55,;5.44,-2.46,;4.96,-3.92,;5.92,-5.11,;7.44,-4.88,;8.4,-6.07,;7.84,-7.5,;6.32,-7.75,;5.35,-6.53,;8.79,-8.7,;8.24,-10.12,;9.21,-11.33,;10.73,-11.09,;11.68,-12.31,;11.29,-9.66,;10.33,-8.46,;3.43,-3.92,;2.4,-5.04,;.91,-4.72,;.43,-3.27,;1.45,-2.13,;.99,-.65,;2.96,-2.46,)| Show InChI InChI=1S/C28H36N8O2/c1-34-11-13-35(14-12-34)20-6-8-21(9-7-20)36-28-25(27(29)31-18-32-28)26(33-36)19-5-10-23(24(16-19)37-2)30-17-22-4-3-15-38-22/h3-5,10,15-16,18,20-21,30H,6-9,11-14,17H2,1-2H3,(H2,29,31,32)/t20-,21- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 64 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of p56 Lck tyrosine kinase activity with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50145571

(1-Methyl-1H-indole-2-carboxylic acid (4-{4-amino-1...)Show SMILES COc1cc(ccc1NC(=O)c1cc2ccccc2n1C)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |wU:24.26,wD:27.33,(1.55,-1.69,;1.62,-3.24,;3.01,-3.96,;3.06,-5.49,;4.43,-6.21,;5.72,-5.39,;5.67,-3.86,;4.32,-3.13,;4.3,-1.59,;5.62,-.8,;6.97,-1.55,;5.6,.74,;6.72,1.79,;6.09,3.17,;6.72,4.59,;5.81,5.83,;4.29,5.68,;3.66,4.26,;4.56,3.01,;4.25,1.49,;2.85,.85,;4.49,-7.75,;5.72,-8.66,;5.25,-10.12,;6.2,-11.32,;7.73,-11.09,;8.68,-12.27,;8.12,-13.71,;6.6,-13.95,;5.63,-12.74,;9.08,-14.91,;10.61,-14.67,;11.57,-15.87,;11.02,-17.3,;11.97,-18.52,;9.49,-17.54,;8.52,-16.33,;3.72,-10.12,;2.68,-11.25,;1.19,-10.92,;.71,-9.48,;1.73,-8.33,;1.27,-6.86,;3.24,-8.66,)| Show InChI InChI=1S/C33H39N9O2/c1-39-14-16-41(17-15-39)23-9-11-24(12-10-23)42-32-29(31(34)35-20-36-32)30(38-42)22-8-13-25(28(19-22)44-3)37-33(43)27-18-21-6-4-5-7-26(21)40(27)2/h4-8,13,18-20,23-24H,9-12,14-17H2,1-3H3,(H,37,43)(H2,34,35,36)/t23-,24- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of Src protein tryrosine kinase activity with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM8797
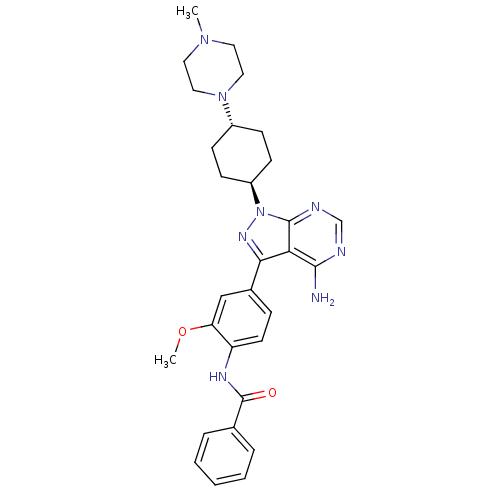
(CHEMBL297363 | N-(4-{4-amino-1-[4-(4-methylpiperaz...)Show SMILES COc1cc(ccc1NC(=O)c1ccccc1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |r,wU:20.21,wD:23.28,(.85,5.16,;-.29,6.2,;-1.75,5.73,;-2.08,4.23,;-3.55,3.76,;-4.69,4.8,;-4.36,6.3,;-2.89,6.77,;-2.56,8.27,;-1.1,8.74,;.04,7.7,;-.77,10.25,;-1.86,11.33,;-1.46,12.82,;.02,13.22,;1.12,12.14,;.72,10.65,;-4.03,2.3,;-3.12,1.05,;-4.03,-.2,;-3.55,-1.66,;-2.06,-2.04,;-1.64,-3.52,;-2.72,-4.63,;-4.21,-4.25,;-4.63,-2.76,;-2.3,-6.11,;-3.34,-7.24,;-2.87,-8.71,;-1.37,-9.04,;-.91,-10.51,;-.33,-7.91,;-.8,-6.44,;-5.49,.28,;-6.82,-.49,;-8.16,.28,;-8.16,1.82,;-6.82,2.59,;-6.82,4.13,;-5.49,1.82,)| Show InChI InChI=1S/C30H36N8O2/c1-36-14-16-37(17-15-36)22-9-11-23(12-10-22)38-29-26(28(31)32-19-33-29)27(35-38)21-8-13-24(25(18-21)40-2)34-30(39)20-6-4-3-5-7-20/h3-8,13,18-19,22-23H,9-12,14-17H2,1-2H3,(H,34,39)(H2,31,32,33)/t22-,23- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 93 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of p56 Lck tyrosine kinase activity with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50145572

(3-{4-[(Furan-2-ylmethyl)-amino]-3-methoxy-phenyl}-...)Show SMILES COc1cc(ccc1NCc1ccco1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |wU:18.19,wD:21.26,(1.27,4.51,;1.34,2.96,;2.73,2.24,;2.78,.71,;4.15,-.01,;5.44,.81,;5.39,2.35,;4.04,3.08,;4.02,4.61,;5.34,5.4,;6.69,4.65,;8.02,5.45,;9.17,4.42,;8.56,3.02,;7.04,3.15,;4.2,-1.55,;5.44,-2.46,;4.96,-3.92,;5.92,-5.11,;7.44,-4.88,;8.4,-6.07,;7.84,-7.5,;6.32,-7.75,;5.35,-6.53,;8.79,-8.7,;8.24,-10.12,;9.21,-11.33,;10.73,-11.09,;11.68,-12.31,;11.29,-9.66,;10.33,-8.46,;3.43,-3.92,;2.4,-5.04,;.91,-4.72,;.43,-3.27,;1.45,-2.13,;.99,-.65,;2.96,-2.46,)| Show InChI InChI=1S/C28H36N8O2/c1-34-11-13-35(14-12-34)20-6-8-21(9-7-20)36-28-25(27(29)31-18-32-28)26(33-36)19-5-10-23(24(16-19)37-2)30-17-22-4-3-15-38-22/h3-5,10,15-16,18,20-21,30H,6-9,11-14,17H2,1-2H3,(H2,29,31,32)/t20-,21- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 97 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of Src protein tryrosine kinase activity with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM8803

(CHEMBL295601 | N-(4-{4-amino-1-[4-(4-methylpiperaz...)Show SMILES COc1cc(ccc1NC(=O)CCc1ccccc1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |r,wU:22.23,wD:25.30,(.85,5.16,;-.29,6.2,;-1.75,5.73,;-2.08,4.23,;-3.55,3.76,;-4.69,4.8,;-4.36,6.3,;-2.89,6.77,;-2.56,8.27,;-1.1,8.74,;.04,7.7,;-.77,10.25,;.72,10.64,;1.13,12.12,;.1,13.28,;.59,14.74,;2.1,15.05,;3.12,13.9,;2.63,12.44,;-4.03,2.3,;-3.12,1.05,;-4.03,-.2,;-3.55,-1.66,;-2.06,-2.04,;-1.64,-3.52,;-2.72,-4.63,;-4.21,-4.25,;-4.63,-2.76,;-2.3,-6.11,;-3.34,-7.24,;-2.87,-8.71,;-1.37,-9.04,;-.91,-10.51,;-.33,-7.91,;-.8,-6.44,;-5.49,.28,;-6.82,-.49,;-8.16,.28,;-8.16,1.82,;-6.82,2.59,;-6.82,4.13,;-5.49,1.82,)| Show InChI InChI=1S/C32H40N8O2/c1-38-16-18-39(19-17-38)24-10-12-25(13-11-24)40-32-29(31(33)34-21-35-32)30(37-40)23-9-14-26(27(20-23)42-2)36-28(41)15-8-22-6-4-3-5-7-22/h3-7,9,14,20-21,24-25H,8,10-13,15-19H2,1-2H3,(H,36,41)(H2,33,34,35)/t24-,25- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 155 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of Src protein tryrosine kinase activity with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM8797

(CHEMBL297363 | N-(4-{4-amino-1-[4-(4-methylpiperaz...)Show SMILES COc1cc(ccc1NC(=O)c1ccccc1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |r,wU:20.21,wD:23.28,(.85,5.16,;-.29,6.2,;-1.75,5.73,;-2.08,4.23,;-3.55,3.76,;-4.69,4.8,;-4.36,6.3,;-2.89,6.77,;-2.56,8.27,;-1.1,8.74,;.04,7.7,;-.77,10.25,;-1.86,11.33,;-1.46,12.82,;.02,13.22,;1.12,12.14,;.72,10.65,;-4.03,2.3,;-3.12,1.05,;-4.03,-.2,;-3.55,-1.66,;-2.06,-2.04,;-1.64,-3.52,;-2.72,-4.63,;-4.21,-4.25,;-4.63,-2.76,;-2.3,-6.11,;-3.34,-7.24,;-2.87,-8.71,;-1.37,-9.04,;-.91,-10.51,;-.33,-7.91,;-.8,-6.44,;-5.49,.28,;-6.82,-.49,;-8.16,.28,;-8.16,1.82,;-6.82,2.59,;-6.82,4.13,;-5.49,1.82,)| Show InChI InChI=1S/C30H36N8O2/c1-36-14-16-37(17-15-36)22-9-11-23(12-10-22)38-29-26(28(31)32-19-33-29)27(35-38)21-8-13-24(25(18-21)40-2)34-30(39)20-6-4-3-5-7-20/h3-8,13,18-19,22-23H,9-12,14-17H2,1-2H3,(H,34,39)(H2,31,32,33)/t22-,23- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 189 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of Src protein tryrosine kinase activity with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Mycobacterium avium) | BDBM18069
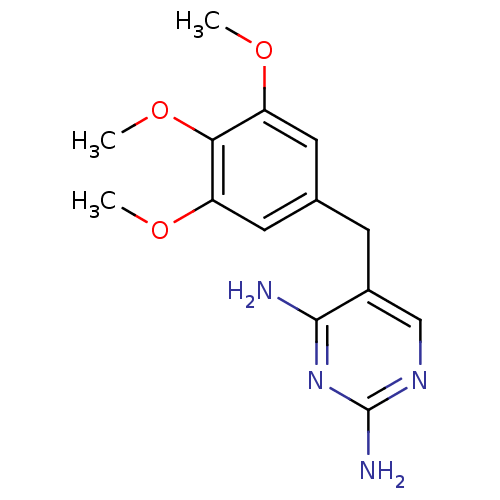
(5-[(3,4,5-trimethoxyphenyl)methyl]pyrimidine-2,4-d...)Show InChI InChI=1S/C14H18N4O3/c1-19-10-5-8(6-11(20-2)12(10)21-3)4-9-7-17-14(16)18-13(9)15/h5-7H,4H2,1-3H3,(H4,15,16,17,18) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
Southern Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium avium DHFR |
J Med Chem 50: 3283-9 (2007)
Article DOI: 10.1021/jm0701977
BindingDB Entry DOI: 10.7270/Q2TH8MCN |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Angiopoietin-1 receptor
(Homo sapiens (Human)) | BDBM50145570

(1H-Indole-2-carboxylic acid (4-{4-amino-1-[4-(4-me...)Show SMILES COc1cc(ccc1NC(=O)c1cc2ccccc2[nH]1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |wU:23.25,wD:26.32,(1.45,-1.92,;1.52,-3.48,;2.9,-4.2,;2.97,-5.74,;4.32,-6.46,;5.63,-5.64,;5.57,-4.09,;4.21,-3.37,;4.2,-1.83,;5.51,-1.03,;6.86,-1.8,;5.49,.51,;6.63,1.55,;5.98,2.93,;6.61,4.35,;5.7,5.59,;4.18,5.43,;3.55,4.03,;4.46,2.77,;4.16,1.25,;4.39,-8,;5.62,-8.89,;5.14,-10.36,;6.09,-11.55,;7.63,-11.32,;8.58,-12.52,;8.02,-13.95,;6.49,-14.19,;5.53,-12.99,;8.99,-15.14,;10.5,-14.91,;11.47,-16.1,;10.91,-17.54,;11.88,-18.76,;9.38,-17.78,;8.43,-16.58,;3.61,-10.36,;2.57,-11.5,;1.08,-11.17,;.61,-9.71,;1.64,-8.57,;1.17,-7.1,;3.13,-8.89,)| Show InChI InChI=1S/C32H37N9O2/c1-39-13-15-40(16-14-39)22-8-10-23(11-9-22)41-31-28(30(33)34-19-35-31)29(38-41)21-7-12-25(27(18-21)43-2)37-32(42)26-17-20-5-3-4-6-24(20)36-26/h3-7,12,17-19,22-23,36H,8-11,13-16H2,1-2H3,(H,37,42)(H2,33,34,35)/t22-,23- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 520 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of tyrosine protein kinase receptor TIE-2 with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Angiopoietin-1 receptor
(Homo sapiens (Human)) | BDBM8798

(CHEMBL47787 | Pyrazolo[3,4-d]pyrimidine 6 | benzyl...)Show SMILES COc1cc(ccc1NC(=O)OCc1ccccc1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |r,wU:22.23,wD:25.30,(.85,5.16,;-.29,6.2,;-1.75,5.73,;-2.08,4.23,;-3.55,3.76,;-4.69,4.8,;-4.36,6.3,;-2.89,6.77,;-2.56,8.27,;-1.1,8.74,;.04,7.7,;-.77,10.25,;.7,10.71,;1.03,12.22,;-.05,13.31,;.36,14.8,;1.85,15.19,;2.93,14.09,;2.52,12.61,;-4.03,2.3,;-3.12,1.05,;-4.03,-.2,;-3.55,-1.66,;-2.06,-2.04,;-1.64,-3.52,;-2.72,-4.63,;-4.21,-4.25,;-4.63,-2.76,;-2.3,-6.11,;-3.34,-7.24,;-2.87,-8.71,;-1.37,-9.04,;-.91,-10.51,;-.33,-7.91,;-.8,-6.44,;-5.49,.28,;-6.82,-.49,;-8.16,.28,;-8.16,1.82,;-6.82,2.59,;-6.82,4.13,;-5.49,1.82,)| Show InChI InChI=1S/C31H38N8O3/c1-37-14-16-38(17-15-37)23-9-11-24(12-10-23)39-30-27(29(32)33-20-34-30)28(36-39)22-8-13-25(26(18-22)41-2)35-31(40)42-19-21-6-4-3-5-7-21/h3-8,13,18,20,23-24H,9-12,14-17,19H2,1-2H3,(H,35,40)(H2,32,33,34)/t23-,24- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 521 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of tyrosine protein kinase receptor TIE-2 with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Lactobacillus casei) | BDBM18069

(5-[(3,4,5-trimethoxyphenyl)methyl]pyrimidine-2,4-d...)Show InChI InChI=1S/C14H18N4O3/c1-19-10-5-8(6-11(20-2)12(10)21-3)4-9-7-17-14(16)18-13(9)15/h5-7H,4H2,1-3H3,(H4,15,16,17,18) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 620 | n/a | n/a | n/a | n/a | n/a | n/a |
Southern Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of Lactobacillus casei DHFR |
J Med Chem 50: 3283-9 (2007)
Article DOI: 10.1021/jm0701977
BindingDB Entry DOI: 10.7270/Q2TH8MCN |
More data for this
Ligand-Target Pair | |
Angiopoietin-1 receptor
(Homo sapiens (Human)) | BDBM50145572

(3-{4-[(Furan-2-ylmethyl)-amino]-3-methoxy-phenyl}-...)Show SMILES COc1cc(ccc1NCc1ccco1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |wU:18.19,wD:21.26,(1.27,4.51,;1.34,2.96,;2.73,2.24,;2.78,.71,;4.15,-.01,;5.44,.81,;5.39,2.35,;4.04,3.08,;4.02,4.61,;5.34,5.4,;6.69,4.65,;8.02,5.45,;9.17,4.42,;8.56,3.02,;7.04,3.15,;4.2,-1.55,;5.44,-2.46,;4.96,-3.92,;5.92,-5.11,;7.44,-4.88,;8.4,-6.07,;7.84,-7.5,;6.32,-7.75,;5.35,-6.53,;8.79,-8.7,;8.24,-10.12,;9.21,-11.33,;10.73,-11.09,;11.68,-12.31,;11.29,-9.66,;10.33,-8.46,;3.43,-3.92,;2.4,-5.04,;.91,-4.72,;.43,-3.27,;1.45,-2.13,;.99,-.65,;2.96,-2.46,)| Show InChI InChI=1S/C28H36N8O2/c1-34-11-13-35(14-12-34)20-6-8-21(9-7-20)36-28-25(27(29)31-18-32-28)26(33-36)19-5-10-23(24(16-19)37-2)30-17-22-4-3-15-38-22/h3-5,10,15-16,18,20-21,30H,6-9,11-14,17H2,1-2H3,(H2,29,31,32)/t20-,21- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.02E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of tyrosine protein kinase receptor TIE-2 with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Angiopoietin-1 receptor
(Homo sapiens (Human)) | BDBM8807

(CHEMBL43773 | N-(4-{4-amino-1-[4-(4-methylpiperazi...)Show SMILES COc1cc(ccc1NC(=O)c1cc2ccccc2o1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |r,wU:23.25,wD:26.32,(.85,5.16,;-.29,6.2,;-1.75,5.73,;-2.08,4.23,;-3.55,3.76,;-4.69,4.8,;-4.36,6.3,;-2.89,6.77,;-2.56,8.27,;-1.1,8.74,;.04,7.7,;-.77,10.25,;.65,10.85,;.52,12.38,;1.57,13.51,;1.12,14.98,;-.38,15.33,;-1.43,14.2,;-.98,12.73,;-1.78,11.41,;-4.03,2.3,;-3.12,1.05,;-4.03,-.2,;-3.55,-1.66,;-2.06,-2.04,;-1.64,-3.52,;-2.72,-4.63,;-4.21,-4.25,;-4.63,-2.76,;-2.3,-6.11,;-3.34,-7.24,;-2.87,-8.71,;-1.37,-9.04,;-.91,-10.51,;-.33,-7.91,;-.8,-6.44,;-5.49,.28,;-6.82,-.49,;-8.16,.28,;-8.16,1.82,;-6.82,2.59,;-6.82,4.13,;-5.49,1.82,)| Show InChI InChI=1S/C32H36N8O3/c1-38-13-15-39(16-14-38)22-8-10-23(11-9-22)40-31-28(30(33)34-19-35-31)29(37-40)21-7-12-24(26(18-21)42-2)36-32(41)27-17-20-5-3-4-6-25(20)43-27/h3-7,12,17-19,22-23H,8-11,13-16H2,1-2H3,(H,36,41)(H2,33,34,35)/t22-,23- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.13E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of tyrosine protein kinase receptor TIE-2 with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Angiopoietin-1 receptor
(Homo sapiens (Human)) | BDBM50145571

(1-Methyl-1H-indole-2-carboxylic acid (4-{4-amino-1...)Show SMILES COc1cc(ccc1NC(=O)c1cc2ccccc2n1C)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |wU:24.26,wD:27.33,(1.55,-1.69,;1.62,-3.24,;3.01,-3.96,;3.06,-5.49,;4.43,-6.21,;5.72,-5.39,;5.67,-3.86,;4.32,-3.13,;4.3,-1.59,;5.62,-.8,;6.97,-1.55,;5.6,.74,;6.72,1.79,;6.09,3.17,;6.72,4.59,;5.81,5.83,;4.29,5.68,;3.66,4.26,;4.56,3.01,;4.25,1.49,;2.85,.85,;4.49,-7.75,;5.72,-8.66,;5.25,-10.12,;6.2,-11.32,;7.73,-11.09,;8.68,-12.27,;8.12,-13.71,;6.6,-13.95,;5.63,-12.74,;9.08,-14.91,;10.61,-14.67,;11.57,-15.87,;11.02,-17.3,;11.97,-18.52,;9.49,-17.54,;8.52,-16.33,;3.72,-10.12,;2.68,-11.25,;1.19,-10.92,;.71,-9.48,;1.73,-8.33,;1.27,-6.86,;3.24,-8.66,)| Show InChI InChI=1S/C33H39N9O2/c1-39-14-16-41(17-15-39)23-9-11-24(12-10-23)42-32-29(31(34)35-20-36-32)30(38-42)22-8-13-25(28(19-22)44-3)37-33(43)27-18-21-6-4-5-7-26(21)40(27)2/h4-8,13,18-20,23-24H,9-12,14-17H2,1-3H3,(H,37,43)(H2,34,35,36)/t23-,24- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.48E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of tyrosine protein kinase receptor TIE-2 with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-5
(Homo sapiens (Human)) | BDBM50600764
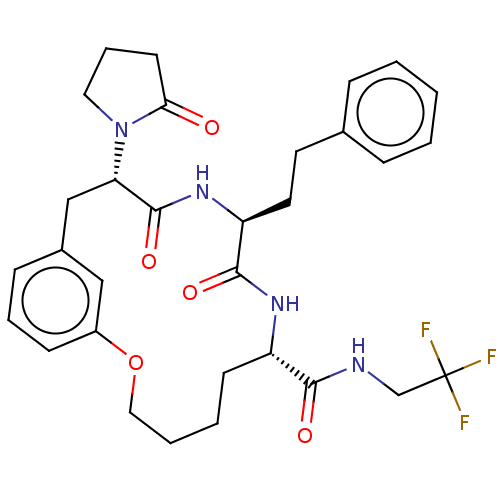
(CHEMBL5182017)Show SMILES FC(F)(F)CNC(=O)[C@@H]1CCCCOc2cccc(C[C@H](N3CCCC3=O)C(=O)N[C@@H](CCc3ccccc3)C(=O)N1)c2 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00611
BindingDB Entry DOI: 10.7270/Q20869BV |
More data for this
Ligand-Target Pair | |
Angiopoietin-1 receptor
(Homo sapiens (Human)) | BDBM8803

(CHEMBL295601 | N-(4-{4-amino-1-[4-(4-methylpiperaz...)Show SMILES COc1cc(ccc1NC(=O)CCc1ccccc1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |r,wU:22.23,wD:25.30,(.85,5.16,;-.29,6.2,;-1.75,5.73,;-2.08,4.23,;-3.55,3.76,;-4.69,4.8,;-4.36,6.3,;-2.89,6.77,;-2.56,8.27,;-1.1,8.74,;.04,7.7,;-.77,10.25,;.72,10.64,;1.13,12.12,;.1,13.28,;.59,14.74,;2.1,15.05,;3.12,13.9,;2.63,12.44,;-4.03,2.3,;-3.12,1.05,;-4.03,-.2,;-3.55,-1.66,;-2.06,-2.04,;-1.64,-3.52,;-2.72,-4.63,;-4.21,-4.25,;-4.63,-2.76,;-2.3,-6.11,;-3.34,-7.24,;-2.87,-8.71,;-1.37,-9.04,;-.91,-10.51,;-.33,-7.91,;-.8,-6.44,;-5.49,.28,;-6.82,-.49,;-8.16,.28,;-8.16,1.82,;-6.82,2.59,;-6.82,4.13,;-5.49,1.82,)| Show InChI InChI=1S/C32H40N8O2/c1-38-16-18-39(19-17-38)24-10-12-25(13-11-24)40-32-29(31(33)34-21-35-32)30(37-40)23-9-14-26(27(20-23)42-2)36-28(41)15-8-22-6-4-3-5-7-22/h3-7,9,14,20-21,24-25H,8,10-13,15-19H2,1-2H3,(H,36,41)(H2,33,34,35)/t24-,25- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.45E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of tyrosine protein kinase receptor TIE-2 with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-5
(Homo sapiens (Human)) | BDBM50600773
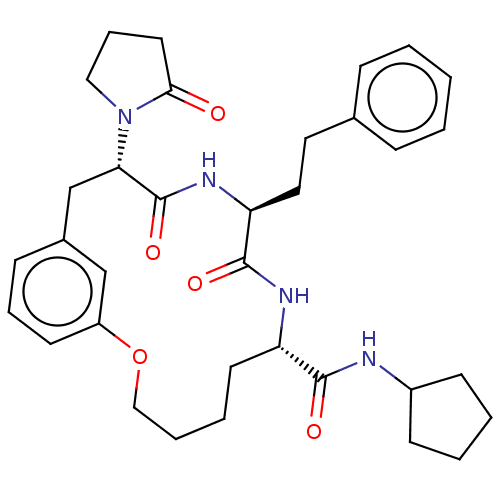
(CHEMBL5174759 | US20240043470, Compound 3-08)Show SMILES O=C(NC1CCCC1)[C@@H]1CCCCOc2cccc(C[C@H](N3CCCC3=O)C(=O)N[C@@H](CCc3ccccc3)C(=O)N1)c2 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00611
BindingDB Entry DOI: 10.7270/Q20869BV |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-5
(Homo sapiens (Human)) | BDBM50600775
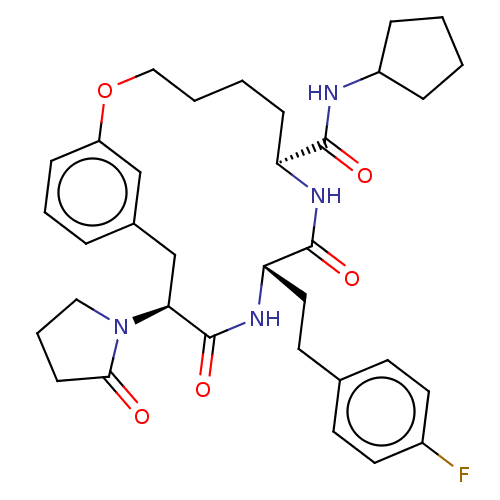
(CHEMBL5207942 | US20240043470, Compound 3-28)Show SMILES Fc1ccc(CC[C@@H]2NC(=O)[C@H](Cc3cccc(OCCCC[C@H](NC2=O)C(=O)NC2CCCC2)c3)N2CCCC2=O)cc1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00611
BindingDB Entry DOI: 10.7270/Q20869BV |
More data for this
Ligand-Target Pair | |
Bifunctional dihydrofolate reductase-thymidylate synthase
(Toxoplasma gondii) | BDBM18069

(5-[(3,4,5-trimethoxyphenyl)methyl]pyrimidine-2,4-d...)Show InChI InChI=1S/C14H18N4O3/c1-19-10-5-8(6-11(20-2)12(10)21-3)4-9-7-17-14(16)18-13(9)15/h5-7H,4H2,1-3H3,(H4,15,16,17,18) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.73E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Southern Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of Toxoplasma gondii DHFR |
J Med Chem 50: 3283-9 (2007)
Article DOI: 10.1021/jm0701977
BindingDB Entry DOI: 10.7270/Q2TH8MCN |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-5
(Homo sapiens (Human)) | BDBM50600767
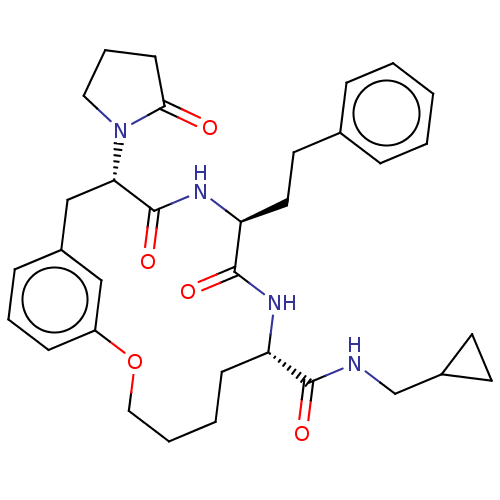
(CHEMBL5191353 | US20240043470, Compound 3-07)Show SMILES O=C(NCC1CC1)[C@@H]1CCCCOc2cccc(C[C@H](N3CCCC3=O)C(=O)N[C@@H](CCc3ccccc3)C(=O)N1)c2 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00611
BindingDB Entry DOI: 10.7270/Q20869BV |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-5
(Homo sapiens (Human)) | BDBM50600776

(CHEMBL5198676 | US20240043470, Compound 3-42)Show SMILES FC1(F)CCN(CC[C@@H]2NC(=O)[C@H](Cc3cccc(OCCCC[C@H](NC2=O)C(=O)NC2CCCC2)c3)N2CCCC2=O)CC1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00611
BindingDB Entry DOI: 10.7270/Q20869BV |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-5
(Homo sapiens (Human)) | BDBM50600762

(CHEMBL5180427 | US20240043470, Compound 1-53)Show SMILES CC(=O)N[C@H]1Cc2cccc(Oc3cccc(C[C@H](NC(=O)[C@H](CCc4ccccc4)NC1=O)C(=O)NCC(F)(F)F)c3)c2 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00611
BindingDB Entry DOI: 10.7270/Q20869BV |
More data for this
Ligand-Target Pair | |
Angiopoietin-1 receptor
(Homo sapiens (Human)) | BDBM8797

(CHEMBL297363 | N-(4-{4-amino-1-[4-(4-methylpiperaz...)Show SMILES COc1cc(ccc1NC(=O)c1ccccc1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |r,wU:20.21,wD:23.28,(.85,5.16,;-.29,6.2,;-1.75,5.73,;-2.08,4.23,;-3.55,3.76,;-4.69,4.8,;-4.36,6.3,;-2.89,6.77,;-2.56,8.27,;-1.1,8.74,;.04,7.7,;-.77,10.25,;-1.86,11.33,;-1.46,12.82,;.02,13.22,;1.12,12.14,;.72,10.65,;-4.03,2.3,;-3.12,1.05,;-4.03,-.2,;-3.55,-1.66,;-2.06,-2.04,;-1.64,-3.52,;-2.72,-4.63,;-4.21,-4.25,;-4.63,-2.76,;-2.3,-6.11,;-3.34,-7.24,;-2.87,-8.71,;-1.37,-9.04,;-.91,-10.51,;-.33,-7.91,;-.8,-6.44,;-5.49,.28,;-6.82,-.49,;-8.16,.28,;-8.16,1.82,;-6.82,2.59,;-6.82,4.13,;-5.49,1.82,)| Show InChI InChI=1S/C30H36N8O2/c1-36-14-16-37(17-15-36)22-9-11-23(12-10-22)38-29-26(28(31)32-19-33-29)27(35-38)21-8-13-24(25(18-21)40-2)34-30(39)20-6-4-3-5-7-20/h3-8,13,18-19,22-23H,9-12,14-17H2,1-2H3,(H,34,39)(H2,31,32,33)/t22-,23- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.68E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of tyrosine protein kinase receptor TIE-2 with 1 mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-5
(Homo sapiens (Human)) | BDBM50600765
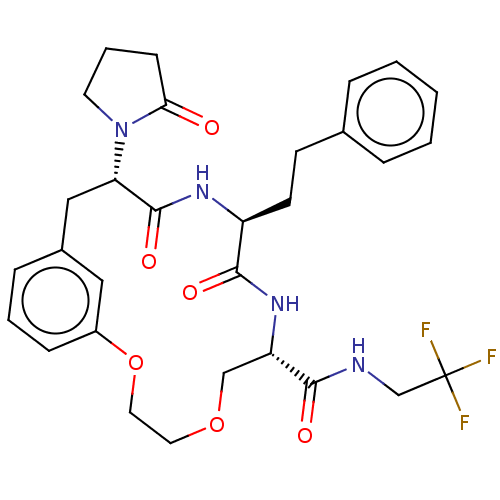
(CHEMBL5175549 | US20240043470, Compound 3-09)Show SMILES FC(F)(F)CNC(=O)[C@@H]1COCCOc2cccc(C[C@H](N3CCCC3=O)C(=O)N[C@@H](CCc3ccccc3)C(=O)N1)c2 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00611
BindingDB Entry DOI: 10.7270/Q20869BV |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-5
(Homo sapiens (Human)) | BDBM50600766

(CHEMBL5193963 | US20240043470, Compound 3-10)Show SMILES FC(F)CNC(=O)[C@@H]1CCCCOc2cccc(C[C@H](N3CCCC3=O)C(=O)N[C@@H](CCc3ccccc3)C(=O)N1)c2 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 8.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00611
BindingDB Entry DOI: 10.7270/Q20869BV |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-5
(Homo sapiens (Human)) | BDBM50600768
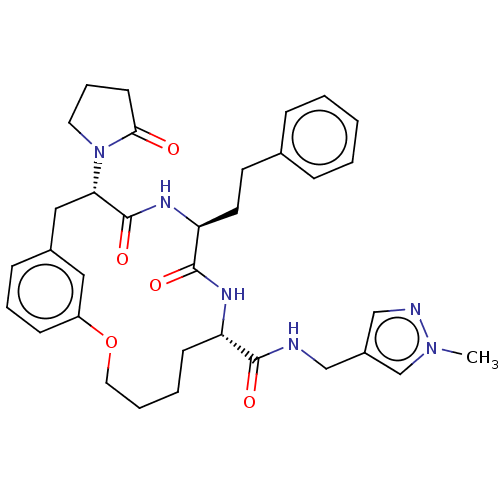
(CHEMBL5195365 | US20240043470, Compound 3-11)Show SMILES Cn1cc(CNC(=O)[C@@H]2CCCCOc3cccc(C[C@H](N4CCCC4=O)C(=O)N[C@@H](CCc4ccccc4)C(=O)N2)c3)cn1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 9.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00611
BindingDB Entry DOI: 10.7270/Q20869BV |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Pneumocystis carinii) | BDBM18069

(5-[(3,4,5-trimethoxyphenyl)methyl]pyrimidine-2,4-d...)Show InChI InChI=1S/C14H18N4O3/c1-19-10-5-8(6-11(20-2)12(10)21-3)4-9-7-17-14(16)18-13(9)15/h5-7H,4H2,1-3H3,(H4,15,16,17,18) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Southern Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of Pneumocystis carinii DHFR |
J Med Chem 50: 3283-9 (2007)
Article DOI: 10.1021/jm0701977
BindingDB Entry DOI: 10.7270/Q2TH8MCN |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Proteasome subunit beta type-5
(Homo sapiens (Human)) | BDBM50600774

(CHEMBL5200145 | US20240043470, Compound 3-35)Show SMILES O=C(NC1CCCC1)[C@@H]1CCCCOc2cccc(C[C@H](N3CCCC3=O)C(=O)N[C@@H](COc3ccccc3)C(=O)N1)c2 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.31E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00611
BindingDB Entry DOI: 10.7270/Q20869BV |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-8
(Homo sapiens (Human)) | BDBM50600782
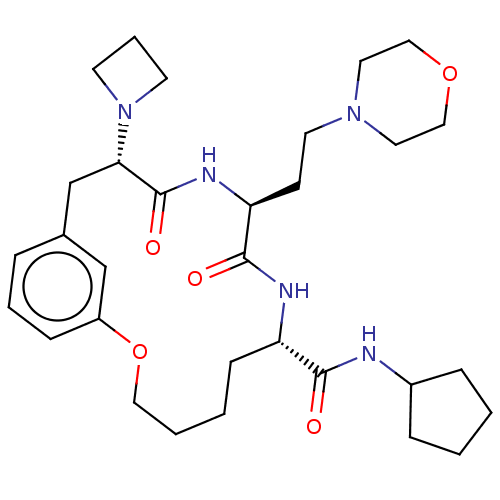
(CHEMBL5198080)Show SMILES O=C(NC1CCCC1)[C@@H]1CCCCOc2cccc(C[C@H](N3CCC3)C(=O)N[C@@H](CCN3CCOCC3)C(=O)N1)c2 |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.31E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00611
BindingDB Entry DOI: 10.7270/Q20869BV |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-5
(Homo sapiens (Human)) | BDBM50600782

(CHEMBL5198080)Show SMILES O=C(NC1CCCC1)[C@@H]1CCCCOc2cccc(C[C@H](N3CCC3)C(=O)N[C@@H](CCN3CCOCC3)C(=O)N1)c2 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.31E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00611
BindingDB Entry DOI: 10.7270/Q20869BV |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-5
(Homo sapiens (Human)) | BDBM50600763
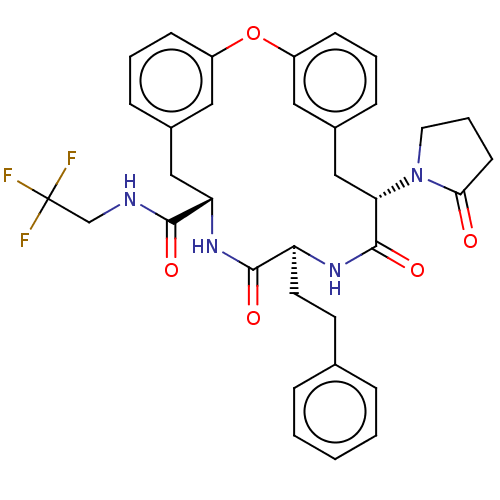
(CHEMBL5194112 | US20240043470, Compound 1-56)Show SMILES FC(F)(F)CNC(=O)[C@@H]1Cc2cccc(Oc3cccc(C[C@H](N4CCCC4=O)C(=O)N[C@@H](CCc4ccccc4)C(=O)N1)c3)c2 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.63E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00611
BindingDB Entry DOI: 10.7270/Q20869BV |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-5
(Homo sapiens (Human)) | BDBM50600779
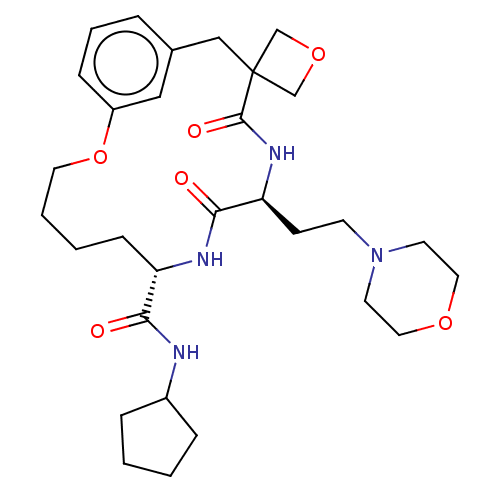
(CHEMBL5198924)Show SMILES O=C(NC1CCCC1)[C@@H]1CCCCOc2cccc(CC3(COC3)C(=O)N[C@@H](CCN3CCOCC3)C(=O)N1)c2 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.11E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00611
BindingDB Entry DOI: 10.7270/Q20869BV |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM8798

(CHEMBL47787 | Pyrazolo[3,4-d]pyrimidine 6 | benzyl...)Show SMILES COc1cc(ccc1NC(=O)OCc1ccccc1)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |r,wU:22.23,wD:25.30,(.85,5.16,;-.29,6.2,;-1.75,5.73,;-2.08,4.23,;-3.55,3.76,;-4.69,4.8,;-4.36,6.3,;-2.89,6.77,;-2.56,8.27,;-1.1,8.74,;.04,7.7,;-.77,10.25,;.7,10.71,;1.03,12.22,;-.05,13.31,;.36,14.8,;1.85,15.19,;2.93,14.09,;2.52,12.61,;-4.03,2.3,;-3.12,1.05,;-4.03,-.2,;-3.55,-1.66,;-2.06,-2.04,;-1.64,-3.52,;-2.72,-4.63,;-4.21,-4.25,;-4.63,-2.76,;-2.3,-6.11,;-3.34,-7.24,;-2.87,-8.71,;-1.37,-9.04,;-.91,-10.51,;-.33,-7.91,;-.8,-6.44,;-5.49,.28,;-6.82,-.49,;-8.16,.28,;-8.16,1.82,;-6.82,2.59,;-6.82,4.13,;-5.49,1.82,)| Show InChI InChI=1S/C31H38N8O3/c1-37-14-16-38(17-15-37)23-9-11-24(12-10-23)39-30-27(29(32)33-20-34-30)28(36-39)22-8-13-25(26(18-22)41-2)35-31(40)42-19-21-6-4-3-5-7-21/h3-8,13,18,20,23-24H,9-12,14-17,19H2,1-2H3,(H,35,40)(H2,32,33,34)/t23-,24- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of vascular endothelial growth factor receptor 2 activity with 1mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-5
(Homo sapiens (Human)) | BDBM50600778
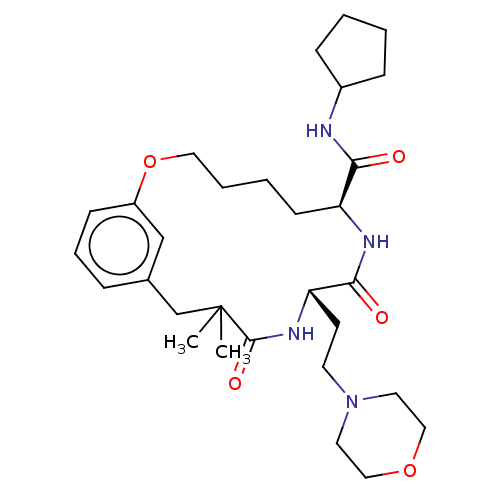
(CHEMBL5198102 | US20240043470, Compound 3-40)Show SMILES CC1(C)Cc2cccc(OCCCC[C@H](NC(=O)[C@H](CCN3CCOCC3)NC1=O)C(=O)NC1CCCC1)c2 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.52E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00611
BindingDB Entry DOI: 10.7270/Q20869BV |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-8
(Homo sapiens (Human)) | BDBM50600776

(CHEMBL5198676 | US20240043470, Compound 3-42)Show SMILES FC1(F)CCN(CC[C@@H]2NC(=O)[C@H](Cc3cccc(OCCCC[C@H](NC2=O)C(=O)NC2CCCC2)c3)N2CCCC2=O)CC1 |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.65E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00611
BindingDB Entry DOI: 10.7270/Q20869BV |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50145571

(1-Methyl-1H-indole-2-carboxylic acid (4-{4-amino-1...)Show SMILES COc1cc(ccc1NC(=O)c1cc2ccccc2n1C)-c1nn([C@H]2CC[C@@H](CC2)N2CCN(C)CC2)c2ncnc(N)c12 |wU:24.26,wD:27.33,(1.55,-1.69,;1.62,-3.24,;3.01,-3.96,;3.06,-5.49,;4.43,-6.21,;5.72,-5.39,;5.67,-3.86,;4.32,-3.13,;4.3,-1.59,;5.62,-.8,;6.97,-1.55,;5.6,.74,;6.72,1.79,;6.09,3.17,;6.72,4.59,;5.81,5.83,;4.29,5.68,;3.66,4.26,;4.56,3.01,;4.25,1.49,;2.85,.85,;4.49,-7.75,;5.72,-8.66,;5.25,-10.12,;6.2,-11.32,;7.73,-11.09,;8.68,-12.27,;8.12,-13.71,;6.6,-13.95,;5.63,-12.74,;9.08,-14.91,;10.61,-14.67,;11.57,-15.87,;11.02,-17.3,;11.97,-18.52,;9.49,-17.54,;8.52,-16.33,;3.72,-10.12,;2.68,-11.25,;1.19,-10.92,;.71,-9.48,;1.73,-8.33,;1.27,-6.86,;3.24,-8.66,)| Show InChI InChI=1S/C33H39N9O2/c1-39-14-16-41(17-15-39)23-9-11-24(12-10-23)42-32-29(31(34)35-20-36-32)30(38-42)22-8-13-25(28(19-22)44-3)37-33(43)27-18-21-6-4-5-7-26(21)40(27)2/h4-8,13,18-20,23-24H,9-12,14-17H2,1-3H3,(H,37,43)(H2,34,35,36)/t23-,24- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of vascular endothelial growth factor receptor 2 activity with 1mM ATP and biotinylated lck peptide |
Bioorg Med Chem Lett 14: 2613-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.101
BindingDB Entry DOI: 10.7270/Q2M61JQP |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data