 Found 301 hits with Last Name = 'lupisella' and Initial = 'j'
Found 301 hits with Last Name = 'lupisella' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Androgen receptor
(Homo sapiens (Human)) | BDBM29321
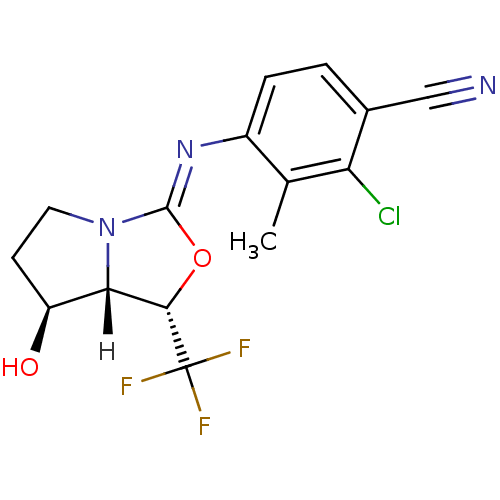
(oxazolidin-2-imine, 6d)Show SMILES [H][C@@]12[C@@H](O)CCN1\C(O[C@@H]2C(F)(F)F)=N\c1ccc(C#N)c(Cl)c1C |r| Show InChI InChI=1S/C15H13ClF3N3O2/c1-7-9(3-2-8(6-20)11(7)16)21-14-22-5-4-10(23)12(22)13(24-14)15(17,18)19/h2-3,10,12-13,23H,4-5H2,1H3/b21-14-/t10-,12-,13-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.200 | -54.8 | n/a | n/a | 19 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... |
J Med Chem 52: 2794-8 (2009)
Article DOI: 10.1021/jm801583j
BindingDB Entry DOI: 10.7270/Q2W66J30 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM29320

(BMS-665139 | oxazolidin-2-imine, 6c)Show SMILES [H][C@@]12[C@@H](O)CCN1\C(O[C@H]2C(F)(F)F)=N\c1ccc(C#N)c(Cl)c1C |r| Show InChI InChI=1S/C15H13ClF3N3O2/c1-7-9(3-2-8(6-20)11(7)16)21-14-22-5-4-10(23)12(22)13(24-14)15(17,18)19/h2-3,10,12-13,23H,4-5H2,1H3/b21-14-/t10-,12-,13+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.300 | -53.8 | n/a | n/a | 0.200 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... |
J Med Chem 52: 2794-8 (2009)
Article DOI: 10.1021/jm801583j
BindingDB Entry DOI: 10.7270/Q2W66J30 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Androgen receptor
(Homo sapiens (Human)) | BDBM18161

((1S,2S,7S,10R,11S,14S,15S)-14-hydroxy-2,15-dimethy...)Show SMILES [H][C@@]12CC[C@H](O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])CC[C@@]2([H])CC(=O)CC[C@]12C |r| Show InChI InChI=1S/C19H30O2/c1-18-9-7-13(20)11-12(18)3-4-14-15-5-6-17(21)19(15,2)10-8-16(14)18/h12,14-17,21H,3-11H2,1-2H3/t12-,14-,15-,16-,17-,18-,19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.300 | -53.8 | n/a | n/a | 2.80 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 49: 7596-9 (2006)
Article DOI: 10.1021/jm061101w
BindingDB Entry DOI: 10.7270/Q2862DQ9 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Androgen receptor
(Homo sapiens (Human)) | BDBM29319
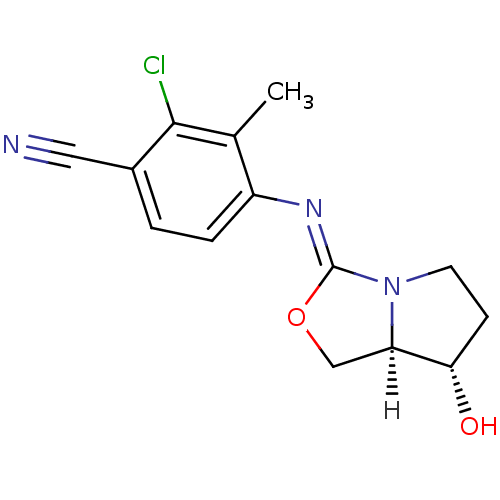
(oxazolidin-2-imine, 6b)Show SMILES [H][C@]12CO\C(=N/c3ccc(C#N)c(Cl)c3C)N1CC[C@@H]2O |r| Show InChI InChI=1S/C14H14ClN3O2/c1-8-10(3-2-9(6-16)13(8)15)17-14-18-5-4-12(19)11(18)7-20-14/h2-3,11-12,19H,4-5,7H2,1H3/b17-14-/t11-,12+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.300 | -53.8 | n/a | n/a | 14 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... |
J Med Chem 52: 2794-8 (2009)
Article DOI: 10.1021/jm801583j
BindingDB Entry DOI: 10.7270/Q2W66J30 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM29323
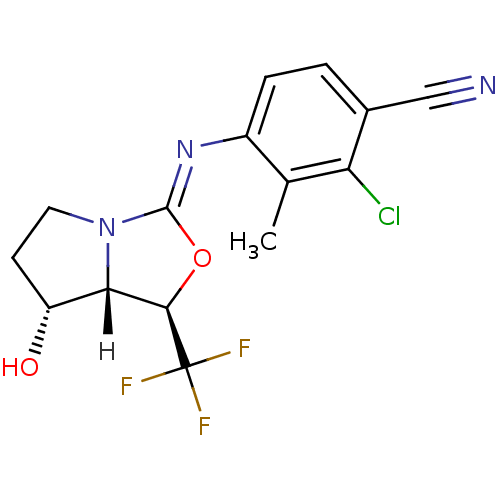
(oxazolidin-2-imine, 6f)Show SMILES [H][C@@]12[C@H](O)CCN1\C(O[C@H]2C(F)(F)F)=N\c1ccc(C#N)c(Cl)c1C |r| Show InChI InChI=1S/C15H13ClF3N3O2/c1-7-9(3-2-8(6-20)11(7)16)21-14-22-5-4-10(23)12(22)13(24-14)15(17,18)19/h2-3,10,12-13,23H,4-5H2,1H3/b21-14-/t10-,12+,13-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.300 | -53.8 | n/a | n/a | 1.40 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... |
J Med Chem 52: 2794-8 (2009)
Article DOI: 10.1021/jm801583j
BindingDB Entry DOI: 10.7270/Q2W66J30 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205111
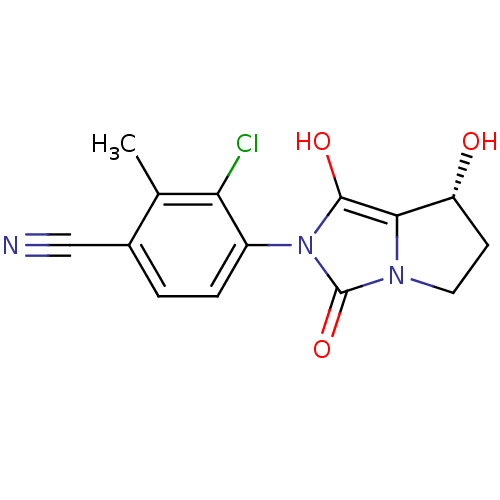
(3-chloro-4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydr...)Show SMILES Cc1c(Cl)c(ccc1C#N)-n1c(O)c2[C@H](O)CCn2c1=O |wU:14.15,(22.97,-37.01,;22.21,-35.67,;20.67,-35.65,;19.89,-36.98,;19.91,-34.31,;20.69,-32.99,;22.22,-32.99,;22.99,-34.34,;24.53,-34.34,;26.08,-34.34,;18.37,-34.31,;17.48,-33.06,;17.96,-31.6,;16.01,-33.53,;14.55,-33.04,;14.08,-31.57,;13.63,-34.28,;14.54,-35.53,;16,-35.06,;17.46,-35.55,;17.93,-37.02,)| Show InChI InChI=1S/C14H12ClN3O3/c1-7-8(6-16)2-3-9(11(7)15)18-13(20)12-10(19)4-5-17(12)14(18)21/h2-3,10,19-20H,4-5H2,1H3/t10-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM29324
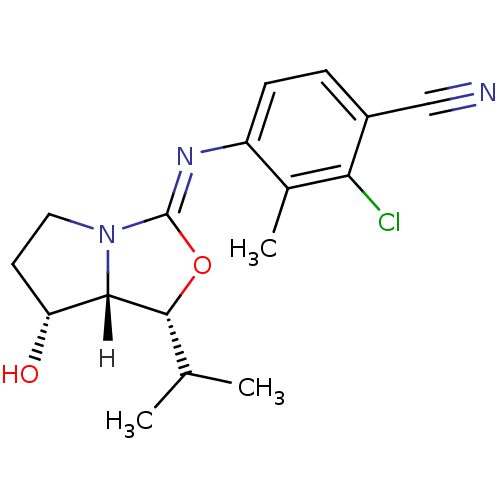
(oxazolidin-2-imine, 6g)Show SMILES [H][C@@]12[C@H](O)CCN1\C(O[C@@H]2C(C)C)=N\c1ccc(C#N)c(Cl)c1C |r| Show InChI InChI=1S/C17H20ClN3O2/c1-9(2)16-15-13(22)6-7-21(15)17(23-16)20-12-5-4-11(8-19)14(18)10(12)3/h4-5,9,13,15-16,22H,6-7H2,1-3H3/b20-17-/t13-,15+,16-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.700 | -51.7 | n/a | n/a | 3.70 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... |
J Med Chem 52: 2794-8 (2009)
Article DOI: 10.1021/jm801583j
BindingDB Entry DOI: 10.7270/Q2W66J30 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM29318

(oxazolidin-2-imine, 6a)Show SMILES [H][C@]12CO\C(=N/c3ccc(C#N)c(Cl)c3C)N1CC[C@H]2O |r| Show InChI InChI=1S/C14H14ClN3O2/c1-8-10(3-2-9(6-16)13(8)15)17-14-18-5-4-12(19)11(18)7-20-14/h2-3,11-12,19H,4-5,7H2,1H3/b17-14-/t11-,12-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.800 | -51.4 | n/a | n/a | 4.80 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... |
J Med Chem 52: 2794-8 (2009)
Article DOI: 10.1021/jm801583j
BindingDB Entry DOI: 10.7270/Q2W66J30 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18162
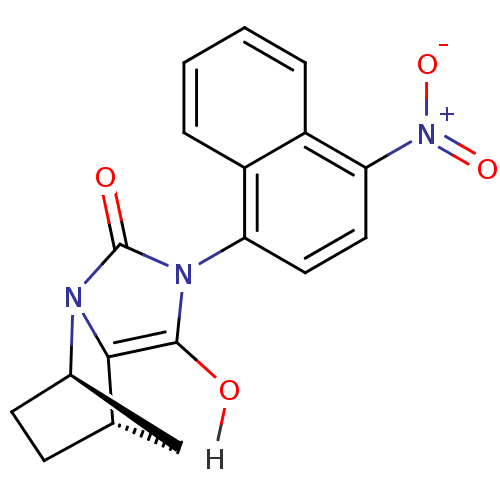
((1R,6R,7S)-4-(4-nitronaphthalen-1-yl)-2,4-diazatri...)Show SMILES Oc1c2[C@H]3CC[C@H](C3)n2c(=O)n1-c1ccc([N+]([O-])=O)c2ccccc12 |r,wU:6.6,3.7,(12.2,-7.28,;12.6,-8.77,;11.56,-9.96,;10.02,-9.96,;9.25,-11.29,;10.02,-12.62,;11.56,-12.62,;10.02,-11.73,;12.33,-11.29,;13.87,-10.92,;15.03,-11.93,;14,-9.4,;15.49,-9,;16.07,-7.57,;17.59,-7.36,;18.54,-8.57,;20.06,-8.36,;20.83,-9.69,;20.64,-6.93,;17.96,-10,;18.91,-11.21,;18.34,-12.64,;16.81,-12.86,;15.86,-11.64,;16.44,-10.22,)| Show InChI InChI=1S/C18H15N3O4/c22-17-16-10-5-6-11(9-10)19(16)18(23)20(17)14-7-8-15(21(24)25)13-4-2-1-3-12(13)14/h1-4,7-8,10-11,22H,5-6,9H2/t10-,11+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | -50.9 | n/a | n/a | 385 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 49: 7596-9 (2006)
Article DOI: 10.1021/jm061101w
BindingDB Entry DOI: 10.7270/Q2862DQ9 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205116
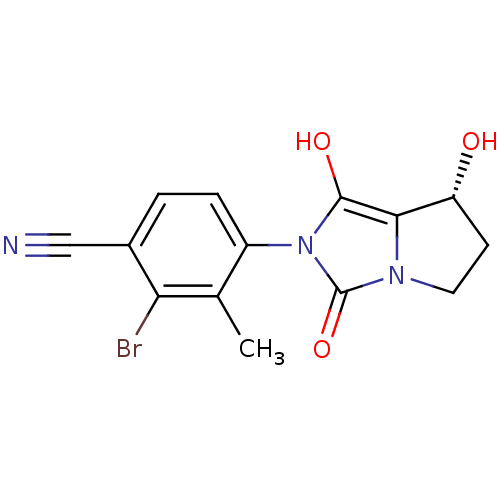
(2-bromo-4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydro...)Show SMILES Cc1c(Br)c(ccc1-n1c(O)c2[C@H](O)CCn2c1=O)C#N |wU:12.13,(21.54,-10.86,;22.32,-9.54,;23.86,-9.55,;24.62,-10.89,;24.64,-8.22,;23.87,-6.88,;22.33,-6.87,;21.56,-8.2,;20.02,-8.19,;19.13,-6.94,;19.61,-5.48,;17.66,-7.41,;16.2,-6.92,;15.73,-5.45,;15.28,-8.17,;16.19,-9.42,;17.65,-8.95,;19.11,-9.43,;19.58,-10.9,;26.18,-8.22,;27.72,-8.23,)| Show InChI InChI=1S/C14H12BrN3O3/c1-7-9(3-2-8(6-16)11(7)15)18-13(20)12-10(19)4-5-17(12)14(18)21/h2-3,10,19-20H,4-5H2,1H3/t10-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205114
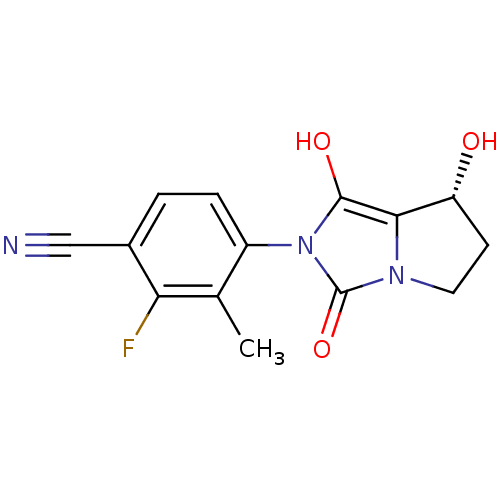
(2-fluoro-4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydr...)Show SMILES Cc1c(F)c(ccc1-n1c(O)c2[C@H](O)CCn2c1=O)C#N |wU:12.13,(21.37,-1.41,;22.15,-.08,;23.69,-.09,;24.45,-1.43,;24.47,1.24,;23.7,2.58,;22.16,2.59,;21.39,1.26,;19.85,1.27,;18.96,2.52,;19.44,3.98,;17.49,2.05,;16.03,2.54,;15.56,4.01,;15.11,1.29,;16.02,.04,;17.48,.51,;18.94,.03,;19.41,-1.44,;26.01,1.24,;27.55,1.23,)| Show InChI InChI=1S/C14H12FN3O3/c1-7-9(3-2-8(6-16)11(7)15)18-13(20)12-10(19)4-5-17(12)14(18)21/h2-3,10,19-20H,4-5H2,1H3/t10-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18165
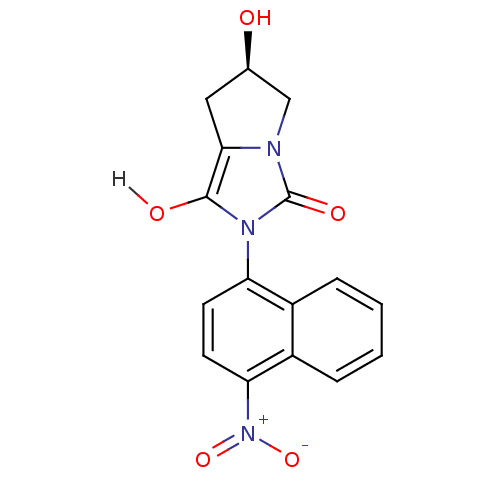
((6R)-6-hydroxy-2-(4-nitronaphthalen-1-yl)-hexahydr...)Show SMILES O[C@@H]1Cc2c(O)n(-c3ccc([N+]([O-])=O)c4ccccc34)c(=O)n2C1 |r,wD:1.0,(-8.34,1.15,;-6.88,1.62,;-6.4,3.09,;-4.86,3.09,;-3.62,3.99,;-3.62,5.53,;-2.37,3.09,;-.83,3.09,;.03,4.36,;1.57,4.25,;2.24,2.87,;3.78,2.76,;4.45,1.37,;4.64,4.03,;1.38,1.59,;2.05,.21,;1.19,-1.07,;-.35,-.96,;-1.02,.43,;-.16,1.7,;-2.85,1.62,;-1.94,.38,;-4.39,1.62,;-5.63,.72,)| Show InChI InChI=1S/C16H13N3O5/c20-9-7-14-15(21)18(16(22)17(14)8-9)12-5-6-13(19(23)24)11-4-2-1-3-10(11)12/h1-6,9,20-21H,7-8H2/t9-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.5 | -49.9 | n/a | n/a | 320 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 49: 7596-9 (2006)
Article DOI: 10.1021/jm061101w
BindingDB Entry DOI: 10.7270/Q2862DQ9 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205096
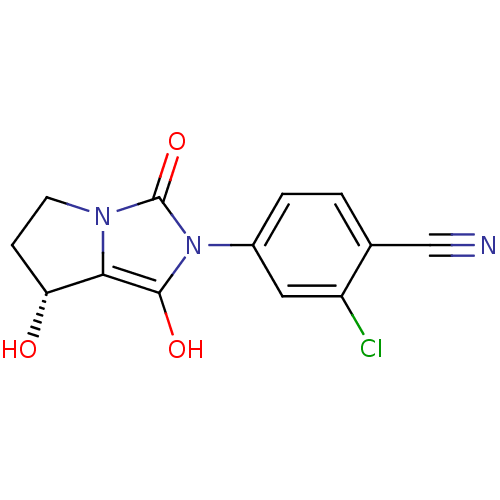
(2-chloro-4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydr...)Show SMILES O[C@@H]1CCn2c1c(O)n(-c1ccc(C#N)c(Cl)c1)c2=O Show InChI InChI=1S/C13H10ClN3O3/c14-9-5-8(2-1-7(9)6-15)17-12(19)11-10(18)3-4-16(11)13(17)20/h1-2,5,10,18-19H,3-4H2/t10-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18169
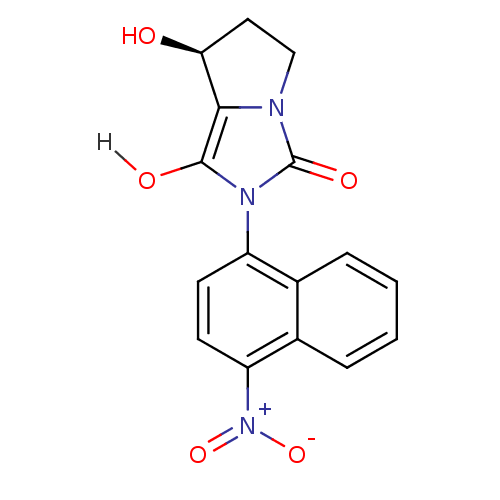
((7S,7aR)-7-hydroxy-2-(4-nitronaphthalen-1-yl)-hexa...)Show SMILES O[C@H]1CCn2c1c(O)n(-c1ccc([N+]([O-])=O)c3ccccc13)c2=O |r,wD:1.0,(-7.31,4.33,;-6.4,3.09,;-6.88,1.62,;-5.63,.72,;-4.39,1.62,;-4.86,3.09,;-3.62,3.99,;-3.62,5.53,;-2.37,3.09,;-.83,3.09,;.03,4.36,;1.57,4.25,;2.24,2.87,;3.78,2.76,;4.45,1.37,;4.64,4.03,;1.38,1.59,;2.05,.21,;1.19,-1.07,;-.35,-.96,;-1.02,.43,;-.16,1.7,;-2.85,1.62,;-1.94,.38,)| Show InChI InChI=1S/C16H13N3O5/c20-13-7-8-17-14(13)15(21)18(16(17)22)11-5-6-12(19(23)24)10-4-2-1-3-9(10)11/h1-6,13,20-21H,7-8H2/t13-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.5 | -49.9 | n/a | n/a | 281 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 49: 7596-9 (2006)
Article DOI: 10.1021/jm061101w
BindingDB Entry DOI: 10.7270/Q2862DQ9 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18176

(4-[(1S,7S,7aR)-7-hydroxy-1-methyl-3-oxo-hexahydro-...)Show SMILES [H][C@@]12[C@@H](O)CCN1C(=O)N([C@H]2C)c1ccc(C#N)c(Cl)c1C |r| Show InChI InChI=1S/C15H16ClN3O2/c1-8-11(4-3-10(7-17)13(8)16)19-9(2)14-12(20)5-6-18(14)15(19)21/h3-4,9,12,14,20H,5-6H2,1-2H3/t9-,12-,14+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.60 | -49.7 | n/a | n/a | 5.40 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... |
J Med Chem 52: 2794-8 (2009)
Article DOI: 10.1021/jm801583j
BindingDB Entry DOI: 10.7270/Q2W66J30 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18164
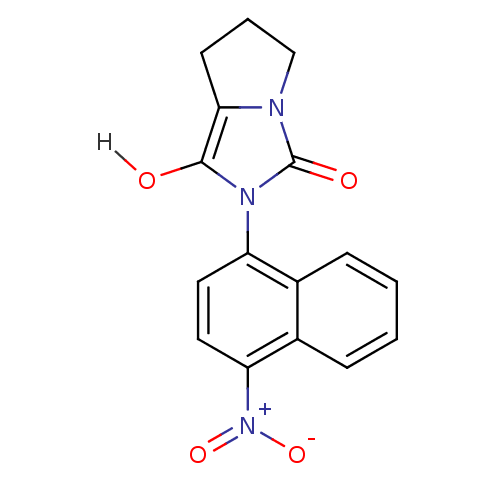
(2-(4-nitronaphthalen-1-yl)-hexahydro-1H-pyrrolo[1,...)Show SMILES Oc1c2CCCn2c(=O)n1-c1ccc([N+]([O-])=O)c2ccccc12 |(-3.62,5.53,;-3.62,3.99,;-4.86,3.09,;-6.4,3.09,;-6.88,1.62,;-5.63,.72,;-4.39,1.62,;-2.85,1.62,;-1.94,.38,;-2.37,3.09,;-.83,3.09,;.03,4.36,;1.57,4.25,;2.24,2.87,;3.78,2.76,;4.45,1.37,;4.64,4.03,;1.38,1.59,;2.05,.21,;1.19,-1.07,;-.35,-.96,;-1.02,.43,;-.16,1.7,)| Show InChI InChI=1S/C16H13N3O4/c20-15-14-6-3-9-17(14)16(21)18(15)12-7-8-13(19(22)23)11-5-2-1-4-10(11)12/h1-2,4-5,7-8,20H,3,6,9H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.70 | -49.6 | n/a | n/a | 270 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 49: 7596-9 (2006)
Article DOI: 10.1021/jm061101w
BindingDB Entry DOI: 10.7270/Q2862DQ9 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM29326

(guanidine derivative, 12)Show SMILES [H][C@]12CN(C)\C(=N/c3ccc(C#N)c(Cl)c3C)N1CC[C@H]2O |r| Show InChI InChI=1S/C15H17ClN4O/c1-9-11(4-3-10(7-17)14(9)16)18-15-19(2)8-12-13(21)5-6-20(12)15/h3-4,12-13,21H,5-6,8H2,1-2H3/b18-15+/t12-,13-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.90 | -49.3 | n/a | n/a | 44 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... |
J Med Chem 52: 2794-8 (2009)
Article DOI: 10.1021/jm801583j
BindingDB Entry DOI: 10.7270/Q2W66J30 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18168
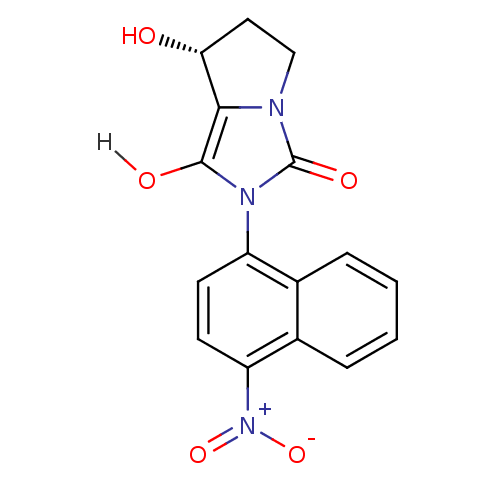
((7R,7aS)-7-hydroxy-2-(4-nitronaphthalen-1-yl)-hexa...)Show SMILES O[C@@H]1CCn2c1c(O)n(-c1ccc([N+]([O-])=O)c3ccccc13)c2=O |r,wU:1.0,(-7.31,4.33,;-6.4,3.09,;-6.88,1.62,;-5.63,.72,;-4.39,1.62,;-4.86,3.09,;-3.62,3.99,;-3.62,5.53,;-2.37,3.09,;-.83,3.09,;.03,4.36,;1.57,4.25,;2.24,2.87,;3.78,2.76,;4.45,1.37,;4.64,4.03,;1.38,1.59,;2.05,.21,;1.19,-1.07,;-.35,-.96,;-1.02,.43,;-.16,1.7,;-2.85,1.62,;-1.94,.38,)| Show InChI InChI=1S/C16H13N3O5/c20-13-7-8-17-14(13)15(21)18(16(17)22)11-5-6-12(19(23)24)10-4-2-1-3-9(10)11/h1-6,13,20-21H,7-8H2/t13-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.10 | -49.0 | n/a | n/a | 1.5 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 49: 7596-9 (2006)
Article DOI: 10.1021/jm061101w
BindingDB Entry DOI: 10.7270/Q2862DQ9 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18173
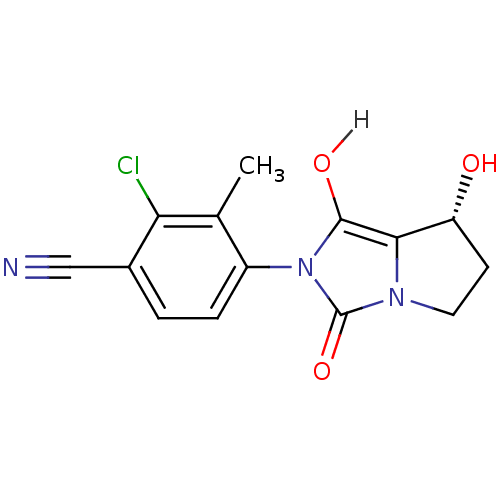
(4-[(7R,7aS)-7-hydroxy-1,3-dioxo-hexahydro-1H-pyrro...)Show SMILES Cc1c(Cl)c(ccc1-n1c(O)c2[C@H](O)CCn2c1=O)C#N |r,wU:12.13,(.01,.36,;.81,1.67,;2.35,1.63,;3.09,.28,;3.16,2.94,;2.42,4.3,;.88,4.34,;.08,3.02,;-1.46,3.02,;-1.94,4.49,;-1.03,5.73,;-3.48,4.49,;-4.72,5.39,;-4.72,6.93,;-5.97,4.49,;-5.5,3.02,;-3.95,3.02,;-2.71,2.12,;-2.71,.58,;4.7,2.9,;6.24,2.9,)| Show InChI InChI=1S/C14H12ClN3O3/c1-7-9(3-2-8(6-16)11(7)15)18-13(20)12-10(19)4-5-17(12)14(18)21/h2-3,10,19-20H,4-5H2,1H3/t10-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Androgen receptor
(Homo sapiens (Human)) | BDBM29322
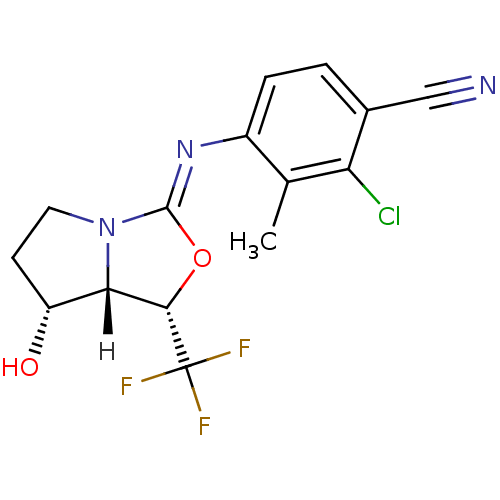
(oxazolidin-2-imine, 6e)Show SMILES [H][C@@]12[C@H](O)CCN1\C(O[C@@H]2C(F)(F)F)=N\c1ccc(C#N)c(Cl)c1C |r| Show InChI InChI=1S/C15H13ClF3N3O2/c1-7-9(3-2-8(6-20)11(7)16)21-14-22-5-4-10(23)12(22)13(24-14)15(17,18)19/h2-3,10,12-13,23H,4-5H2,1H3/b21-14-/t10-,12+,13+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.30 | -48.8 | n/a | n/a | 1.10 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... |
J Med Chem 52: 2794-8 (2009)
Article DOI: 10.1021/jm801583j
BindingDB Entry DOI: 10.7270/Q2W66J30 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205100
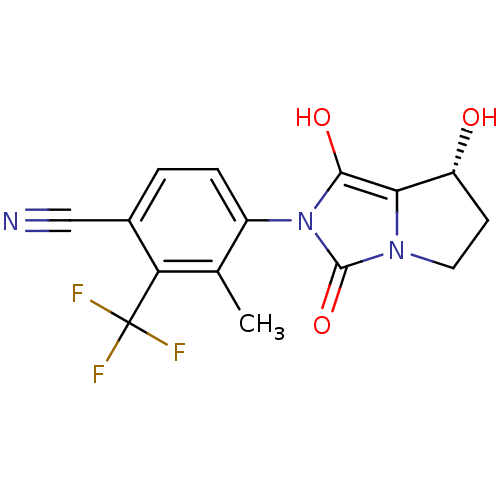
(4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydro-1H-pyrr...)Show SMILES Cc1c(ccc(C#N)c1C(F)(F)F)-n1c(O)c2[C@H](O)CCn2c1=O |wU:17.18,(-.05,-49.74,;.73,-48.41,;-.03,-47.07,;.75,-45.75,;2.28,-45.75,;3.05,-47.09,;4.59,-47.09,;6.13,-47.1,;2.27,-48.42,;3.03,-49.77,;3.79,-51.1,;1.69,-50.53,;4.38,-49.01,;-1.57,-47.07,;-2.46,-45.82,;-1.98,-44.35,;-3.93,-46.28,;-5.39,-45.8,;-5.86,-44.33,;-6.31,-47.04,;-5.4,-48.29,;-3.94,-47.82,;-2.48,-48.3,;-2.01,-49.77,)| Show InChI InChI=1S/C15H12F3N3O3/c1-7-9(3-2-8(6-19)11(7)15(16,17)18)21-13(23)12-10(22)4-5-20(12)14(21)24/h2-3,10,22-23H,4-5H2,1H3/t10-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205113
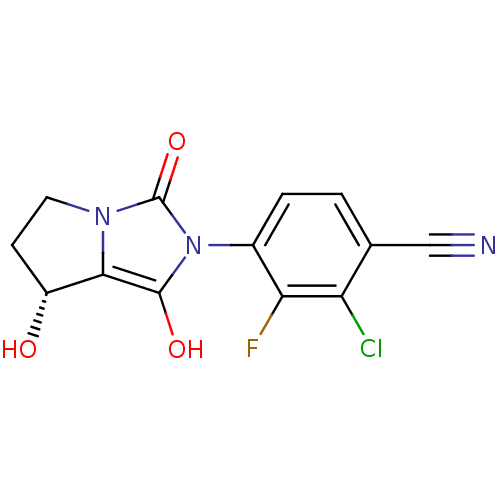
(2-chloro-3-fluoro-4-((7R,7aS)-7-hydroxy-1,3-dioxo-...)Show SMILES O[C@@H]1CCn2c1c(O)n(-c1ccc(C#N)c(Cl)c1F)c2=O |wU:1.0,(-7.12,-24.7,;-6.65,-26.17,;-7.57,-27.42,;-6.66,-28.66,;-5.2,-28.19,;-5.19,-26.66,;-3.72,-26.19,;-3.24,-24.73,;-2.83,-27.44,;-1.29,-27.45,;-.52,-26.12,;1.02,-26.12,;1.79,-27.47,;3.33,-27.47,;4.87,-27.48,;1.01,-28.8,;1.77,-30.14,;-.53,-28.78,;-1.31,-30.11,;-3.74,-28.68,;-3.27,-30.15,)| Show InChI InChI=1S/C13H9ClFN3O3/c14-9-6(5-16)1-2-7(10(9)15)18-12(20)11-8(19)3-4-17(11)13(18)21/h1-2,8,19-20H,3-4H2/t8-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18171

(4-[(7R,7aS)-7-hydroxy-1,3-dioxo-hexahydro-1H-pyrro...)Show SMILES O[C@@H]1CCn2c1c(O)n(-c1ccc(C#N)c3ccccc13)c2=O |r,wU:1.0,(-7.31,4.33,;-6.4,3.09,;-6.88,1.62,;-5.63,.72,;-4.39,1.62,;-4.86,3.09,;-3.62,3.99,;-3.62,5.53,;-2.37,3.09,;-.83,3.09,;.03,4.36,;1.57,4.25,;2.24,2.87,;3.78,2.76,;5.32,2.76,;1.38,1.59,;2.05,.21,;1.19,-1.07,;-.35,-.96,;-1.02,.43,;-.16,1.7,;-2.85,1.62,;-1.94,.38,)| Show InChI InChI=1S/C17H13N3O3/c18-9-10-5-6-13(12-4-2-1-3-11(10)12)20-16(22)15-14(21)7-8-19(15)17(20)23/h1-6,14,21-22H,7-8H2/t14-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 3.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Androgen receptor
(Homo sapiens (Human)) | BDBM18171

(4-[(7R,7aS)-7-hydroxy-1,3-dioxo-hexahydro-1H-pyrro...)Show SMILES O[C@@H]1CCn2c1c(O)n(-c1ccc(C#N)c3ccccc13)c2=O |r,wU:1.0,(-7.31,4.33,;-6.4,3.09,;-6.88,1.62,;-5.63,.72,;-4.39,1.62,;-4.86,3.09,;-3.62,3.99,;-3.62,5.53,;-2.37,3.09,;-.83,3.09,;.03,4.36,;1.57,4.25,;2.24,2.87,;3.78,2.76,;5.32,2.76,;1.38,1.59,;2.05,.21,;1.19,-1.07,;-.35,-.96,;-1.02,.43,;-.16,1.7,;-2.85,1.62,;-1.94,.38,)| Show InChI InChI=1S/C17H13N3O3/c18-9-10-5-6-13(12-4-2-1-3-11(10)12)20-16(22)15-14(21)7-8-19(15)17(20)23/h1-6,14,21-22H,7-8H2/t14-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 3.20 | -48.0 | n/a | n/a | 2.30 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 49: 7596-9 (2006)
Article DOI: 10.1021/jm061101w
BindingDB Entry DOI: 10.7270/Q2862DQ9 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205102
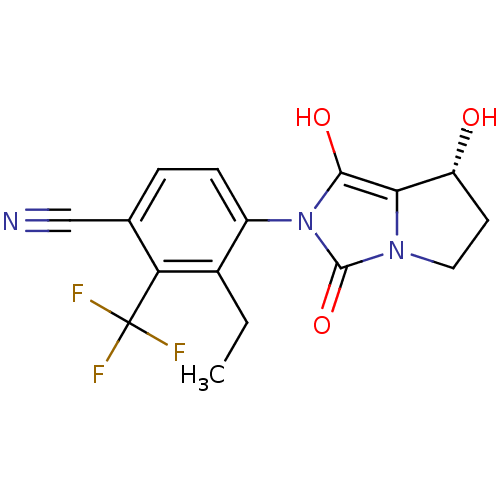
(3-ethyl-4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydro...)Show SMILES CCc1c(ccc(C#N)c1C(F)(F)F)-n1c(O)c2[C@H](O)CCn2c1=O |wU:18.19,(4.34,-4.72,;3.58,-3.37,;4.36,-2.05,;3.6,-.71,;4.37,.62,;5.91,.61,;6.68,-.73,;8.22,-.73,;9.76,-.74,;5.9,-2.06,;6.66,-3.4,;7.42,-4.73,;5.32,-4.17,;8,-2.65,;2.06,-.7,;1.16,.55,;1.65,2.01,;-.3,.08,;-1.76,.57,;-2.23,2.04,;-2.68,-.68,;-1.77,-1.93,;-.31,-1.46,;1.15,-1.94,;1.62,-3.41,)| Show InChI InChI=1S/C16H14F3N3O3/c1-2-9-10(4-3-8(7-20)12(9)16(17,18)19)22-14(24)13-11(23)5-6-21(13)15(22)25/h3-4,11,23-24H,2,5-6H2,1H3/t11-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18167
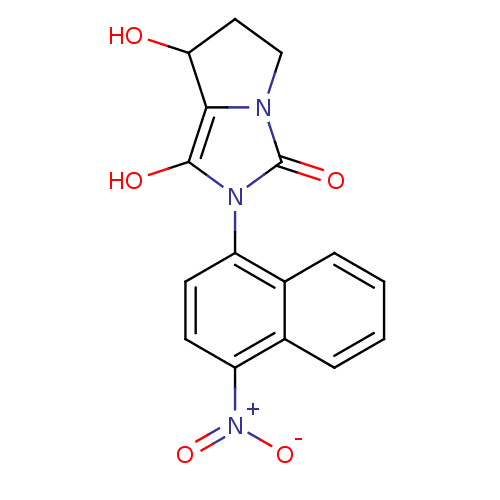
(4-(Tetrahydro-7-hydroxy-1,3-dioxo-1H-pyrrolo[1,2-c...)Show SMILES OC1CCn2c1c(O)n(-c1ccc([N+]([O-])=O)c3ccccc13)c2=O |(-7.31,4.33,;-6.4,3.09,;-6.88,1.62,;-5.63,.72,;-4.39,1.62,;-4.86,3.09,;-3.62,3.99,;-3.62,5.53,;-2.37,3.09,;-.83,3.09,;.03,4.36,;1.57,4.25,;2.24,2.87,;3.78,2.76,;4.45,1.37,;4.64,4.03,;1.38,1.59,;2.05,.21,;1.19,-1.07,;-.35,-.96,;-1.02,.43,;-.16,1.7,;-2.85,1.62,;-1.94,.38,)| Show InChI InChI=1S/C16H13N3O5/c20-13-7-8-17-14(13)15(21)18(16(17)22)11-5-6-12(19(23)24)10-4-2-1-3-9(10)11/h1-6,13,20-21H,7-8H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.5 | -47.8 | n/a | n/a | 4.40 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 49: 7596-9 (2006)
Article DOI: 10.1021/jm061101w
BindingDB Entry DOI: 10.7270/Q2862DQ9 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205103
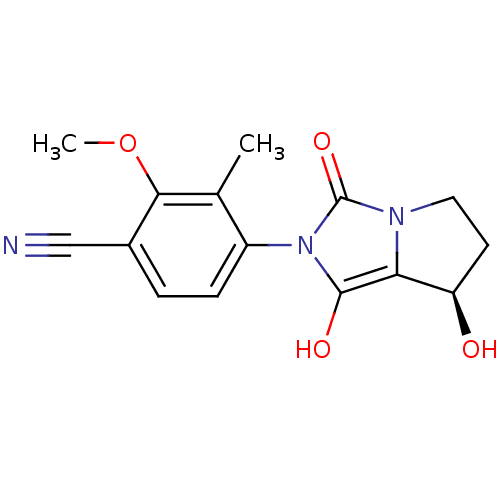
(4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydro-1H-pyrr...)Show SMILES COc1c(C)c(ccc1C#N)-n1c(O)c2[C@H](O)CCn2c1=O |wU:15.16,(.83,-2.32,;1.61,-.99,;.85,.35,;-.69,.37,;-1.47,-.96,;-1.45,1.7,;-.68,3.03,;.86,3.03,;1.63,1.68,;3.17,1.68,;4.71,1.67,;-2.99,1.71,;-3.89,2.96,;-3.4,4.42,;-5.35,2.49,;-6.81,2.98,;-7.28,4.45,;-7.73,1.73,;-6.83,.49,;-5.36,.96,;-3.9,.47,;-3.44,-1,)| Show InChI InChI=1S/C15H15N3O4/c1-8-10(4-3-9(7-16)13(8)22-2)18-14(20)12-11(19)5-6-17(12)15(18)21/h3-4,11,19-20H,5-6H2,1-2H3/t11-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205104
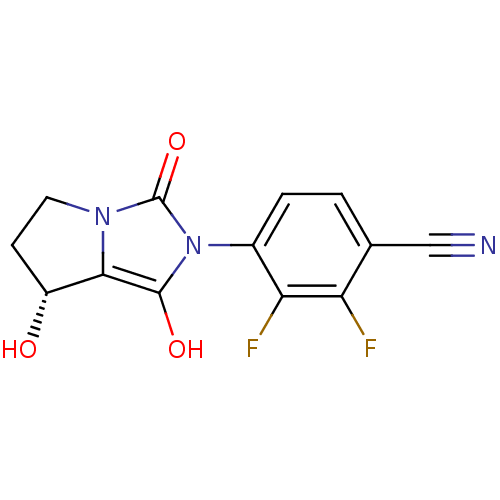
(2,3-difluoro-4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetra...)Show SMILES O[C@@H]1CCn2c1c(O)n(-c1ccc(C#N)c(F)c1F)c2=O |wU:1.0,(16.12,-15.47,;16.59,-16.94,;15.67,-18.19,;16.58,-19.44,;18.04,-18.97,;18.05,-17.43,;19.52,-16.97,;20,-15.5,;20.41,-18.21,;21.95,-18.22,;22.73,-16.89,;24.26,-16.9,;25.03,-18.24,;26.57,-18.24,;28.11,-18.25,;24.25,-19.57,;25.01,-20.91,;22.71,-19.56,;21.93,-20.89,;19.5,-19.45,;19.97,-20.92,)| Show InChI InChI=1S/C13H9F2N3O3/c14-9-6(5-16)1-2-7(10(9)15)18-12(20)11-8(19)3-4-17(11)13(18)21/h1-2,8,19-20H,3-4H2/t8-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18166
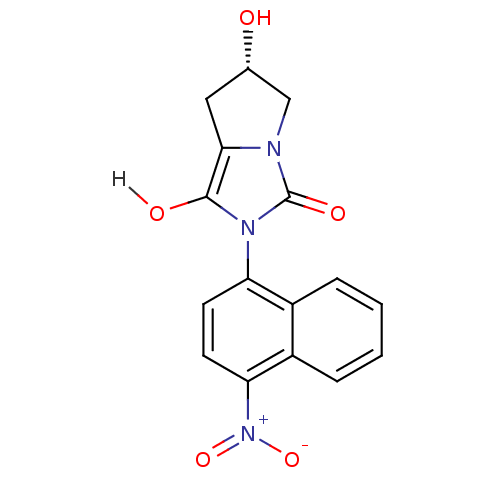
((6S)-6-hydroxy-2-(4-nitronaphthalen-1-yl)-hexahydr...)Show SMILES O[C@H]1Cc2c(O)n(-c3ccc([N+]([O-])=O)c4ccccc34)c(=O)n2C1 |r,wU:1.0,(-8.34,1.15,;-6.88,1.62,;-6.4,3.09,;-4.86,3.09,;-3.62,3.99,;-3.62,5.53,;-2.37,3.09,;-.83,3.09,;.03,4.36,;1.57,4.25,;2.24,2.87,;3.78,2.76,;4.45,1.37,;4.64,4.03,;1.38,1.59,;2.05,.21,;1.19,-1.07,;-.35,-.96,;-1.02,.43,;-.16,1.7,;-2.85,1.62,;-1.94,.38,;-4.39,1.62,;-5.63,.72,)| Show InChI InChI=1S/C16H13N3O5/c20-9-7-14-15(21)18(16(22)17(14)8-9)12-5-6-13(19(23)24)11-4-2-1-3-10(11)12/h1-6,9,20-21H,7-8H2/t9-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4.30 | -47.3 | n/a | n/a | 265 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 49: 7596-9 (2006)
Article DOI: 10.1021/jm061101w
BindingDB Entry DOI: 10.7270/Q2862DQ9 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205098

(4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydro-1H-pyrr...)Show SMILES Cc1c(C)c(ccc1C#N)-n1c(O)c2[C@H](O)CCn2c1=O |wU:14.15,(1.66,-40.91,;.9,-39.57,;-.64,-39.56,;-1.42,-40.88,;-1.4,-38.22,;-.62,-36.89,;.91,-36.9,;1.68,-38.24,;3.22,-38.24,;4.76,-38.25,;-2.94,-38.21,;-3.83,-36.96,;-3.35,-35.5,;-5.3,-37.43,;-6.76,-36.94,;-7.23,-35.47,;-7.68,-38.19,;-6.77,-39.44,;-5.31,-38.97,;-3.85,-39.45,;-3.38,-40.92,)| Show InChI InChI=1S/C15H15N3O3/c1-8-9(2)11(4-3-10(8)7-16)18-14(20)13-12(19)5-6-17(13)15(18)21/h3-4,12,19-20H,5-6H2,1-2H3/t12-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50204085

(CHEMBL3960606)Show SMILES CC(C)(O)c1cn(c(Cc2ccccc2Cl)n1)-c1ccc(cc1)-c1ccc(CO)c(c1)S(C)(=O)=O Show InChI InChI=1S/C27H27ClN2O4S/c1-27(2,32)25-16-30(26(29-25)15-20-6-4-5-7-23(20)28)22-12-10-18(11-13-22)19-8-9-21(17-31)24(14-19)35(3,33)34/h4-14,16,31-32H,15,17H2,1-3H3 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Displacement of [3H]-24,25-epoxycholesterol from human LXRbeta/RXRalpha expressed in baculovirus infected Sf9 cells by scintillation proximity analys... |
ACS Med Chem Lett 7: 1207-1212 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00234
BindingDB Entry DOI: 10.7270/Q2V98B13 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18172
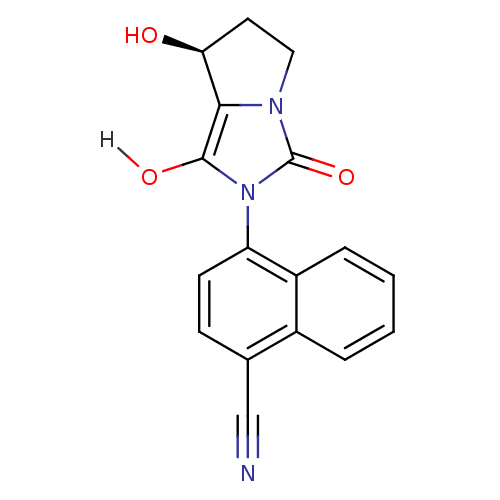
(4-[(7S,7aR)-7-hydroxy-1,3-dioxo-hexahydro-1H-pyrro...)Show SMILES O[C@H]1CCn2c1c(O)n(-c1ccc(C#N)c3ccccc13)c2=O |r,wD:1.0,(-7.31,4.33,;-6.4,3.09,;-6.88,1.62,;-5.63,.72,;-4.39,1.62,;-4.86,3.09,;-3.62,3.99,;-3.62,5.53,;-2.37,3.09,;-.83,3.09,;.03,4.36,;1.57,4.25,;2.24,2.87,;3.78,2.76,;5.32,2.76,;1.38,1.59,;2.05,.21,;1.19,-1.07,;-.35,-.96,;-1.02,.43,;-.16,1.7,;-2.85,1.62,;-1.94,.38,)| Show InChI InChI=1S/C17H13N3O3/c18-9-10-5-6-13(12-4-2-1-3-11(10)12)20-16(22)15-14(21)7-8-19(15)17(20)23/h1-6,14,21-22H,7-8H2/t14-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 5.30 | -46.8 | 60 | n/a | 1.30E+3 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 49: 7596-9 (2006)
Article DOI: 10.1021/jm061101w
BindingDB Entry DOI: 10.7270/Q2862DQ9 |
More data for this
Ligand-Target Pair | |
Androgen receptor
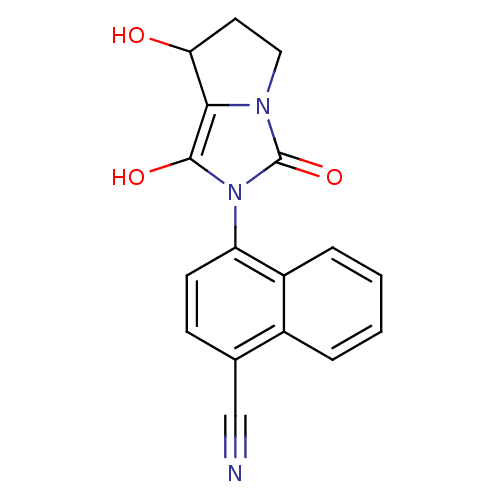
(Homo sapiens (Human)) | BDBM18170
(4-{7-hydroxy-1,3-dioxo-hexahydro-1H-pyrrolo[1,2-a]...)Show SMILES OC1CCn2c1c(O)n(-c1ccc(C#N)c3ccccc13)c2=O |(-7.31,4.33,;-6.4,3.09,;-6.88,1.62,;-5.63,.72,;-4.39,1.62,;-4.86,3.09,;-3.62,3.99,;-3.62,5.53,;-2.37,3.09,;-.83,3.09,;.03,4.36,;1.57,4.25,;2.24,2.87,;3.78,2.76,;5.32,2.76,;1.38,1.59,;2.05,.21,;1.19,-1.07,;-.35,-.96,;-1.02,.43,;-.16,1.7,;-2.85,1.62,;-1.94,.38,)| Show InChI InChI=1S/C17H13N3O3/c18-9-10-5-6-13(12-4-2-1-3-11(10)12)20-16(22)15-14(21)7-8-19(15)17(20)23/h1-6,14,21-22H,7-8H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.5 | -46.7 | n/a | n/a | 6.30 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 49: 7596-9 (2006)
Article DOI: 10.1021/jm061101w
BindingDB Entry DOI: 10.7270/Q2862DQ9 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18174
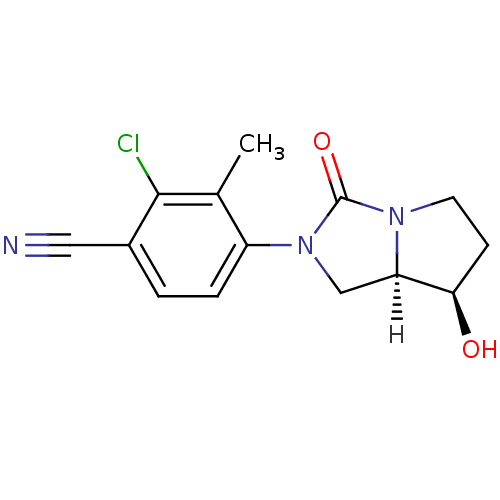
(4-[(7R,7aR)-7-hydroxy-3-oxo-hexahydro-1H-pyrrolo[1...)Show SMILES [H][C@]12CN(C(=O)N1CC[C@H]2O)c1ccc(C#N)c(Cl)c1C |r| Show InChI InChI=1S/C14H14ClN3O2/c1-8-10(3-2-9(6-16)13(8)15)18-7-11-12(19)4-5-17(11)14(18)20/h2-3,11-12,19H,4-5,7H2,1H3/t11-,12-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 6 | -46.5 | n/a | n/a | 6.40 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... |
J Med Chem 52: 2794-8 (2009)
Article DOI: 10.1021/jm801583j
BindingDB Entry DOI: 10.7270/Q2W66J30 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50204079
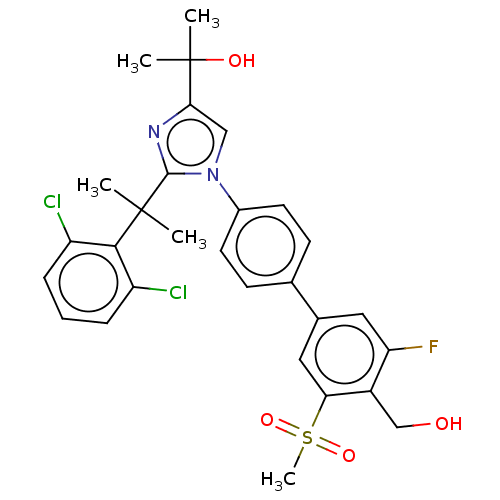
(CHEMBL3935187)Show SMILES CC(C)(O)c1cn(c(n1)C(C)(C)c1c(Cl)cccc1Cl)-c1ccc(cc1)-c1cc(F)c(CO)c(c1)S(C)(=O)=O Show InChI InChI=1S/C29H29Cl2FN2O4S/c1-28(2,26-21(30)7-6-8-22(26)31)27-33-25(29(3,4)36)15-34(27)19-11-9-17(10-12-19)18-13-23(32)20(16-35)24(14-18)39(5,37)38/h6-15,35-36H,16H2,1-5H3 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Displacement of [3H]-24,25-epoxycholesterol from human LXRbeta/RXRalpha expressed in baculovirus infected Sf9 cells by scintillation proximity analys... |
ACS Med Chem Lett 7: 1207-1212 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00234
BindingDB Entry DOI: 10.7270/Q2V98B13 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205107
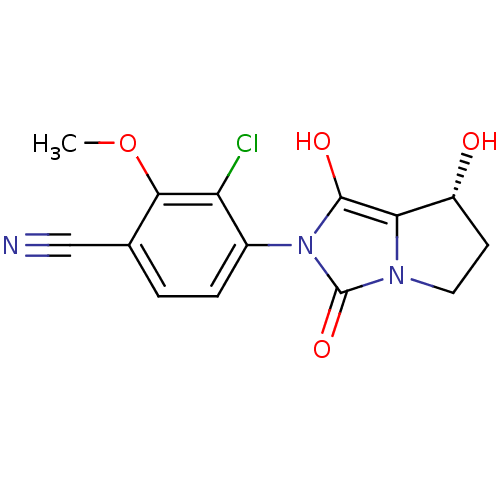
(3-chloro-4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydr...)Show SMILES COc1c(Cl)c(ccc1C#N)-n1c(O)c2[C@H](O)CCn2c1=O |wU:15.16,(1.24,-38.75,;2.02,-37.42,;1.26,-36.08,;-.28,-36.06,;-1.06,-37.39,;-1.04,-34.73,;-.27,-33.4,;1.27,-33.4,;2.04,-34.75,;3.58,-34.75,;5.12,-34.76,;-2.58,-34.72,;-3.48,-33.47,;-2.99,-32.01,;-4.94,-33.94,;-6.4,-33.45,;-6.87,-31.98,;-7.32,-34.7,;-6.42,-35.94,;-4.95,-35.47,;-3.49,-35.96,;-3.02,-37.43,)| Show InChI InChI=1S/C14H12ClN3O4/c1-22-12-7(6-16)2-3-8(10(12)15)18-13(20)11-9(19)4-5-17(11)14(18)21/h2-3,9,19-20H,4-5H2,1H3/t9-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 6.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205101
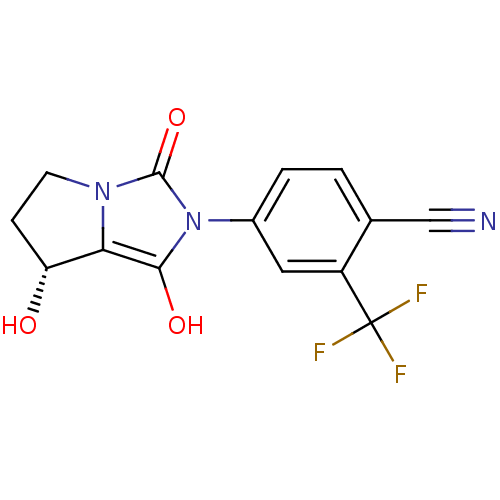
(4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydro-1H-pyrr...)Show SMILES O[C@@H]1CCn2c1c(O)n(-c1ccc(C#N)c(c1)C(F)(F)F)c2=O Show InChI InChI=1S/C14H10F3N3O3/c15-14(16,17)9-5-8(2-1-7(9)6-18)20-12(22)11-10(21)3-4-19(11)13(20)23/h1-2,5,10,21-22H,3-4H2/t10-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205110
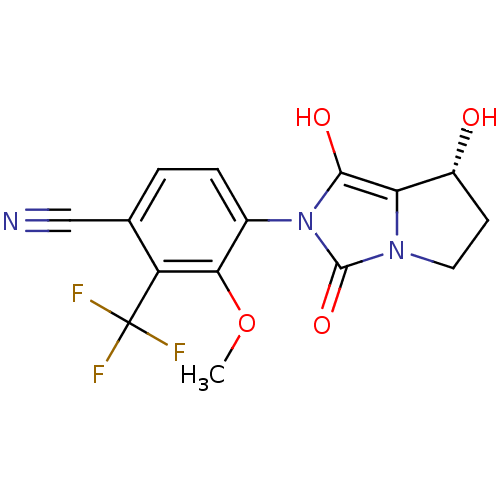
(4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydro-1H-pyrr...)Show SMILES COc1c(ccc(C#N)c1C(F)(F)F)-n1c(O)c2[C@H](O)CCn2c1=O |wU:18.19,(13.93,-47.01,;13.17,-45.67,;13.95,-44.34,;13.19,-43,;13.96,-41.68,;15.5,-41.68,;16.27,-43.03,;17.81,-43.03,;19.35,-43.03,;15.49,-44.36,;16.25,-45.7,;17.01,-47.03,;14.91,-46.46,;17.6,-44.95,;11.65,-43,;10.76,-41.75,;11.24,-40.29,;9.29,-42.22,;7.83,-41.73,;7.36,-40.26,;6.91,-42.97,;7.82,-44.22,;9.28,-43.75,;10.74,-44.24,;11.21,-45.71,)| Show InChI InChI=1S/C15H12F3N3O4/c1-25-12-8(3-2-7(6-19)10(12)15(16,17)18)21-13(23)11-9(22)4-5-20(11)14(21)24/h2-3,9,22-23H,4-5H2,1H3/t9-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 7.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205112
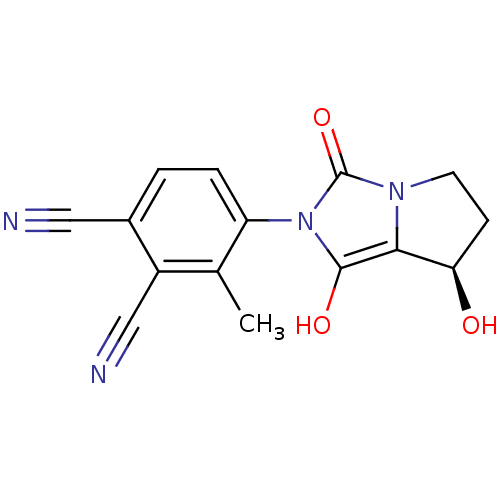
(4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydro-1H-pyrr...)Show SMILES Cc1c(ccc(C#N)c1C#N)-n1c(O)c2[C@H](O)CCn2c1=O |wU:15.16,(-.55,-20.76,;.23,-19.43,;-.53,-18.1,;.25,-16.77,;1.78,-16.77,;2.56,-18.12,;4.1,-18.12,;5.64,-18.12,;1.77,-19.45,;2.53,-20.79,;3.29,-22.13,;-2.07,-18.09,;-2.96,-16.84,;-2.48,-15.38,;-4.43,-17.31,;-5.89,-16.82,;-6.36,-15.35,;-6.8,-18.07,;-5.9,-19.31,;-4.44,-18.84,;-2.98,-19.33,;-2.51,-20.8,)| Show InChI InChI=1S/C15H12N4O3/c1-8-10(7-17)9(6-16)2-3-11(8)19-14(21)13-12(20)4-5-18(13)15(19)22/h2-3,12,20-21H,4-5H2,1H3/t12-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205099

(4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydro-1H-pyrr...)Show SMILES COc1c(C)c(ccc1-n1c(O)c2[C@H](O)CCn2c1=O)C#N |wU:13.14,(-2.46,-47.7,;-3.22,-46.35,;-2.44,-45.03,;-.9,-45.04,;-.14,-46.38,;-.12,-43.71,;-.89,-42.37,;-2.42,-42.36,;-3.2,-43.69,;-4.74,-43.68,;-5.63,-42.44,;-5.15,-40.97,;-7.1,-42.9,;-8.56,-42.41,;-9.03,-40.94,;-9.47,-43.66,;-8.57,-44.91,;-7.11,-44.44,;-5.65,-44.92,;-5.18,-46.39,;1.42,-43.71,;2.97,-43.72,)| Show InChI InChI=1S/C15H15N3O4/c1-8-9(7-16)3-4-10(13(8)22-2)18-14(20)12-11(19)5-6-17(12)15(18)21/h3-4,11,19-20H,5-6H2,1-2H3/t11-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205115
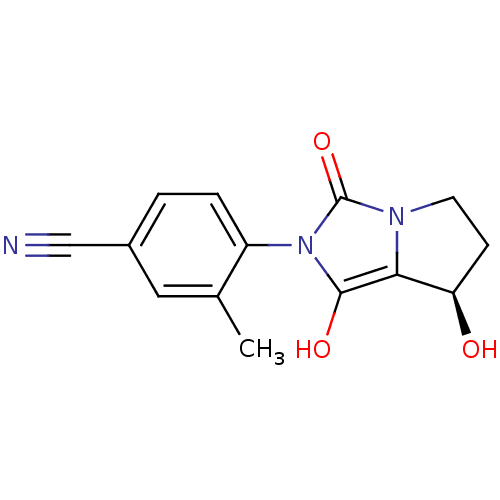
(4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydro-1H-pyrr...)Show SMILES Cc1cc(ccc1-n1c(O)c2[C@H](O)CCn2c1=O)C#N |wU:11.12,(19.99,-30.33,;20.77,-29,;22.31,-29.02,;23.09,-27.69,;22.32,-26.34,;20.79,-26.34,;20.01,-27.66,;18.47,-27.66,;17.58,-26.41,;18.06,-24.95,;16.11,-26.88,;14.65,-26.39,;14.18,-24.92,;13.73,-27.63,;14.64,-28.88,;16.1,-28.41,;17.56,-28.9,;18.03,-30.37,;24.63,-27.69,;26.18,-27.69,)| Show InChI InChI=1S/C14H13N3O3/c1-8-6-9(7-15)2-3-10(8)17-13(19)12-11(18)4-5-16(12)14(17)20/h2-3,6,11,18-19H,4-5H2,1H3/t11-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-alpha/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50204078

(CHEMBL3926292)Show SMILES CC(C)(O)c1cn(c(n1)C(C)(C)c1ccccc1Cl)-c1ccc(cc1F)-c1cc(F)c(CO)c(c1)S(C)(=O)=O Show InChI InChI=1S/C29H29ClF2N2O4S/c1-28(2,20-8-6-7-9-21(20)30)27-33-26(29(3,4)36)15-34(27)24-11-10-17(12-23(24)32)18-13-22(31)19(16-35)25(14-18)39(5,37)38/h6-15,35-36H,16H2,1-5H3 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Displacement of [3H]-24,25-epoxycholesterol from human LXRalpha/RXRalpha expressed in baculovirus infected Sf9 cells by scintillation proximity analy... |
ACS Med Chem Lett 7: 1207-1212 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00234
BindingDB Entry DOI: 10.7270/Q2V98B13 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM29325
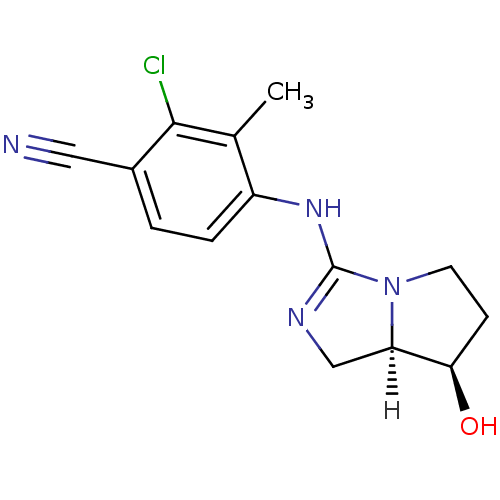
(guanidine derivative, 11)Show SMILES [H][C@]12CN=C(Nc3ccc(C#N)c(Cl)c3C)N1CC[C@H]2O |r,t:3| Show InChI InChI=1S/C14H15ClN4O/c1-8-10(3-2-9(6-16)13(8)15)18-14-17-7-11-12(20)4-5-19(11)14/h2-3,11-12,20H,4-5,7H2,1H3,(H,17,18)/t11-,12-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 10 | -45.2 | n/a | n/a | 1.80E+3 | n/a | n/a | 7.6 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Binding assays were conducted by incubating test compound at various concentrations with [3H]DHT with human cancer epithelial breast cell lines MDAMB... |
J Med Chem 52: 2794-8 (2009)
Article DOI: 10.1021/jm801583j
BindingDB Entry DOI: 10.7270/Q2W66J30 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50204077
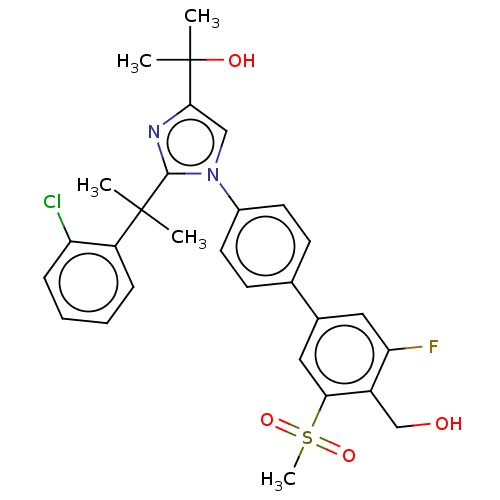
(CHEMBL3944154)Show SMILES CC(C)(O)c1cn(c(n1)C(C)(C)c1ccccc1Cl)-c1ccc(cc1)-c1cc(F)c(CO)c(c1)S(C)(=O)=O Show InChI InChI=1S/C29H30ClFN2O4S/c1-28(2,22-8-6-7-9-23(22)30)27-32-26(29(3,4)35)16-33(27)20-12-10-18(11-13-20)19-14-24(31)21(17-34)25(15-19)38(5,36)37/h6-16,34-35H,17H2,1-5H3 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Displacement of [3H]-24,25-epoxycholesterol from human LXRbeta/RXRalpha expressed in baculovirus infected Sf9 cells by scintillation proximity analys... |
ACS Med Chem Lett 7: 1207-1212 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00234
BindingDB Entry DOI: 10.7270/Q2V98B13 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50204075
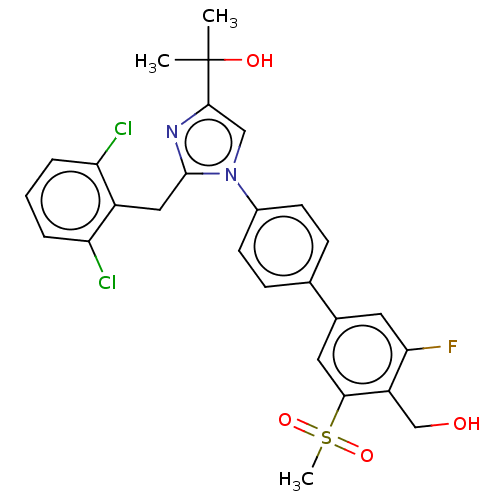
(CHEMBL3980683)Show SMILES CC(C)(O)c1cn(c(Cc2c(Cl)cccc2Cl)n1)-c1ccc(cc1)-c1cc(F)c(CO)c(c1)S(C)(=O)=O Show InChI InChI=1S/C27H25Cl2FN2O4S/c1-27(2,34)25-14-32(26(31-25)13-19-21(28)5-4-6-22(19)29)18-9-7-16(8-10-18)17-11-23(30)20(15-33)24(12-17)37(3,35)36/h4-12,14,33-34H,13,15H2,1-3H3 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Displacement of [3H]-24,25-epoxycholesterol from human LXRbeta/RXRalpha expressed in baculovirus infected Sf9 cells by scintillation proximity analys... |
ACS Med Chem Lett 7: 1207-1212 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00234
BindingDB Entry DOI: 10.7270/Q2V98B13 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50204076

(CHEMBL3890276)Show SMILES CC(C)(O)c1cn(c(n1)C(C)(C)c1c(Cl)cccc1Cl)-c1ccc(cc1)-c1cccc(c1)S(C)(=O)=O Show InChI InChI=1S/C28H28Cl2N2O3S/c1-27(2,25-22(29)10-7-11-23(25)30)26-31-24(28(3,4)33)17-32(26)20-14-12-18(13-15-20)19-8-6-9-21(16-19)36(5,34)35/h6-17,33H,1-5H3 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Displacement of [3H]-24,25-epoxycholesterol from human LXRbeta/RXRalpha expressed in baculovirus infected Sf9 cells by scintillation proximity analys... |
ACS Med Chem Lett 7: 1207-1212 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00234
BindingDB Entry DOI: 10.7270/Q2V98B13 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50204072
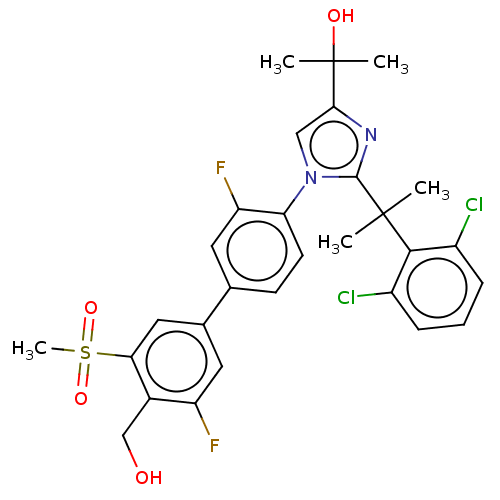
(CHEMBL3945199 | US10543183, Compound 12 | US109459...)Show SMILES CC(C)(O)c1cn(c(n1)C(C)(C)c1c(Cl)cccc1Cl)-c1ccc(cc1F)-c1cc(F)c(CO)c(c1)S(C)(=O)=O Show InChI InChI=1S/C29H28Cl2F2N2O4S/c1-28(2,26-19(30)7-6-8-20(26)31)27-34-25(29(3,4)37)14-35(27)23-10-9-16(11-22(23)33)17-12-21(32)18(15-36)24(13-17)40(5,38)39/h6-14,36-37H,15H2,1-5H3 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Displacement of [3H]-24,25-epoxycholesterol from human LXRbeta/RXRalpha expressed in baculovirus infected Sf9 cells by scintillation proximity analys... |
ACS Med Chem Lett 7: 1207-1212 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00234
BindingDB Entry DOI: 10.7270/Q2V98B13 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50204081

(CHEMBL3913162)Show SMILES CC(C)(O)c1cn(c(n1)C(C)(C)c1ccccc1Cl)-c1ccc(cc1)-c1ccc(CO)c(c1)S(C)(=O)=O Show InChI InChI=1S/C29H31ClN2O4S/c1-28(2,23-8-6-7-9-24(23)30)27-31-26(29(3,4)34)17-32(27)22-14-12-19(13-15-22)20-10-11-21(18-33)25(16-20)37(5,35)36/h6-17,33-34H,18H2,1-5H3 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Displacement of [3H]-24,25-epoxycholesterol from human LXRbeta/RXRalpha expressed in baculovirus infected Sf9 cells by scintillation proximity analys... |
ACS Med Chem Lett 7: 1207-1212 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00234
BindingDB Entry DOI: 10.7270/Q2V98B13 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50204082

(CHEMBL3945820)Show SMILES CC(c1nc(cn1-c1ccc(cc1F)-c1cc(F)c(CO)c(c1)S(C)(=O)=O)C(C)(C)O)c1c(Cl)cccc1Cl Show InChI InChI=1S/C28H26Cl2F2N2O4S/c1-15(26-19(29)6-5-7-20(26)30)27-33-25(28(2,3)36)13-34(27)23-9-8-16(10-22(23)32)17-11-21(31)18(14-35)24(12-17)39(4,37)38/h5-13,15,35-36H,14H2,1-4H3 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Displacement of [3H]-24,25-epoxycholesterol from human LXRbeta/RXRalpha expressed in baculovirus infected Sf9 cells by scintillation proximity analys... |
ACS Med Chem Lett 7: 1207-1212 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00234
BindingDB Entry DOI: 10.7270/Q2V98B13 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta/Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50204071
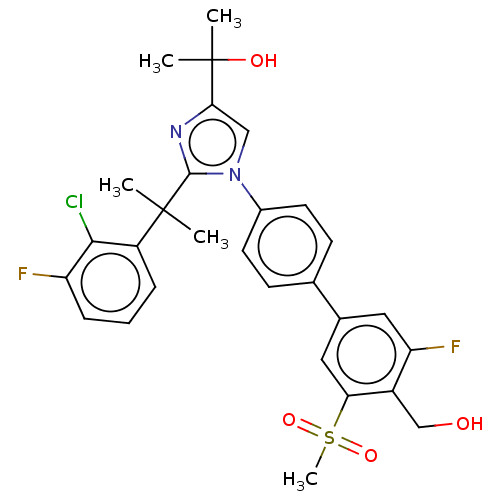
(CHEMBL3953927)Show SMILES CC(C)(O)c1cn(c(n1)C(C)(C)c1cccc(F)c1Cl)-c1ccc(cc1)-c1cc(F)c(CO)c(c1)S(C)(=O)=O Show InChI InChI=1S/C29H29ClF2N2O4S/c1-28(2,21-7-6-8-22(31)26(21)30)27-33-25(29(3,4)36)15-34(27)19-11-9-17(10-12-19)18-13-23(32)20(16-35)24(14-18)39(5,37)38/h6-15,35-36H,16H2,1-5H3 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Displacement of [3H]-24,25-epoxycholesterol from human LXRbeta/RXRalpha expressed in baculovirus infected Sf9 cells by scintillation proximity analys... |
ACS Med Chem Lett 7: 1207-1212 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00234
BindingDB Entry DOI: 10.7270/Q2V98B13 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data