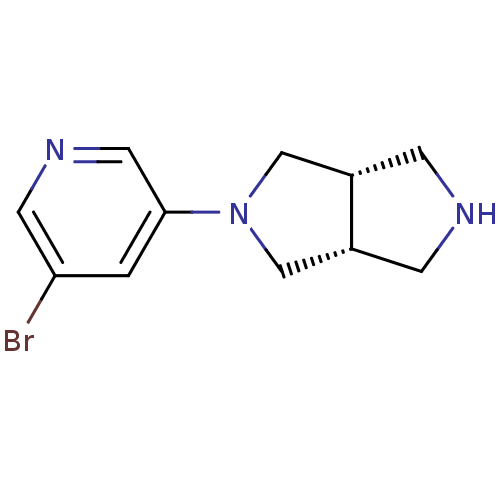Found 2115 hits with Last Name = 'li' and Initial = 'jh'
Found 2115 hits with Last Name = 'li' and Initial = 'jh' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
5-hydroxytryptamine receptor 2A
(Rattus norvegicus (rat)) | BDBM50451055
(CHEMBL4205290)Show InChI InChI=1S/C19H24N4O2/c1-25-18-8-3-2-7-17(18)23-14-12-22(13-15-23)11-10-21-19(24)16-6-4-5-9-20-16/h2-9H,10-15H2,1H3,(H,21,24) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0224 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dipartimento di Farmacia Universit£ di Napoli "Federico II" Via D. Montesano
Curated by ChEMBL
| Assay Description
Displacement of [3H]ketanserin from serotonin 5-HT2A receptor in Sprague-Dawley rat brain cortex homogenates incubated for 15 mins by liquid scintill... |
Bioorg Med Chem 25: 5820-5837 (2017)
Article DOI: 10.1016/j.bmc.2017.09.018
BindingDB Entry DOI: 10.7270/Q2VD721F |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM50279404
(CHEMBL447388 | cis-2-(5-Ethoxy-3-pyridinyl)octahyd...)Show InChI InChI=1S/C13H19N3O/c1-2-17-13-3-12(6-15-7-13)16-8-10-4-14-5-11(10)9-16/h3,6-7,10-11,14H,2,4-5,8-9H2,1H3/t10-,11+ | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from alpha4beta2 nicotinic acetylcholine receptor in rat brain minus cerebellum membrane |
J Med Chem 52: 4126-41 (2009)
Article DOI: 10.1021/jm900249k
BindingDB Entry DOI: 10.7270/Q2GT5P3R |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM50279405
(CHEMBL488772 | cis-3-(5-Propyloxy-3-pyridinyl)-3,7...)Show InChI InChI=1S/C14H21N3O/c1-2-3-18-14-4-13(7-16-8-14)17-9-11-5-15-6-12(11)10-17/h4,7-8,11-12,15H,2-3,5-6,9-10H2,1H3/t11-,12+ | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0410 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from alpha4beta2 nicotinic acetylcholine receptor in rat brain minus cerebellum membrane |
J Med Chem 52: 4126-41 (2009)
Article DOI: 10.1021/jm900249k
BindingDB Entry DOI: 10.7270/Q2GT5P3R |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Rattus norvegicus (rat)) | BDBM50451039
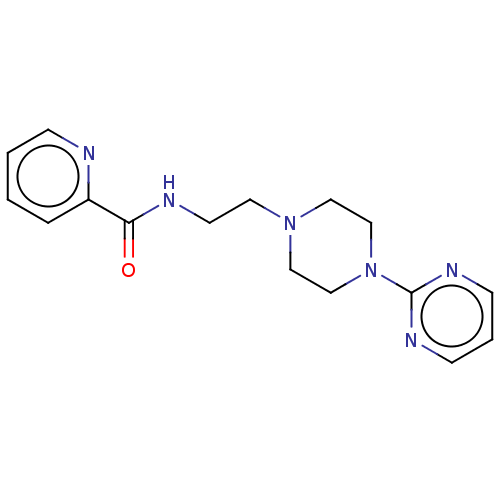
(CHEMBL4209954)Show InChI InChI=1S/C16H20N6O/c23-15(14-4-1-2-5-17-14)18-8-9-21-10-12-22(13-11-21)16-19-6-3-7-20-16/h1-7H,8-13H2,(H,18,23) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dipartimento di Farmacia Universit£ di Napoli "Federico II" Via D. Montesano
Curated by ChEMBL
| Assay Description
Displacement of [3H]-8-OH-DPAT from serotonin 5-HT1A receptor in Sprague-Dawley rat brain cortex homogenates incubated for 30 mins by liquid scintill... |
Bioorg Med Chem 25: 5820-5837 (2017)
Article DOI: 10.1016/j.bmc.2017.09.018
BindingDB Entry DOI: 10.7270/Q2VD721F |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM50049757

(()-2-(6-Chloro-pyridin-3-yl)-7-aza-bicyclo[2.2.1]h...)Show InChI InChI=1S/C11H13ClN2/c12-11-4-1-7(6-13-11)9-5-8-2-3-10(9)14-8/h1,4,6,8-10,14H,2-3,5H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.0470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from alpha4beta2 nicotinic acetylcholine receptor in rat brain minus cerebellum membrane |
J Med Chem 52: 4126-41 (2009)
Article DOI: 10.1021/jm900249k
BindingDB Entry DOI: 10.7270/Q2GT5P3R |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM50278878
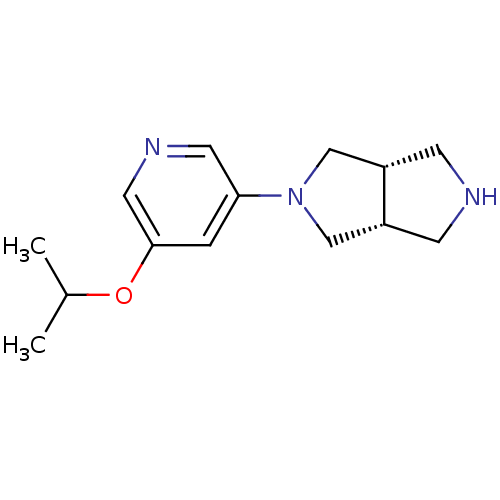
(CHEMBL502567 | cis-3-(5-Isopropyloxy-3-pyridinyl)-...)Show InChI InChI=1S/C14H21N3O/c1-10(2)18-14-3-13(6-16-7-14)17-8-11-4-15-5-12(11)9-17/h3,6-7,10-12,15H,4-5,8-9H2,1-2H3/t11-,12+ | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from alpha4beta2 nicotinic acetylcholine receptor in rat brain minus cerebellum membrane |
J Med Chem 52: 4126-41 (2009)
Article DOI: 10.1021/jm900249k
BindingDB Entry DOI: 10.7270/Q2GT5P3R |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM50278877
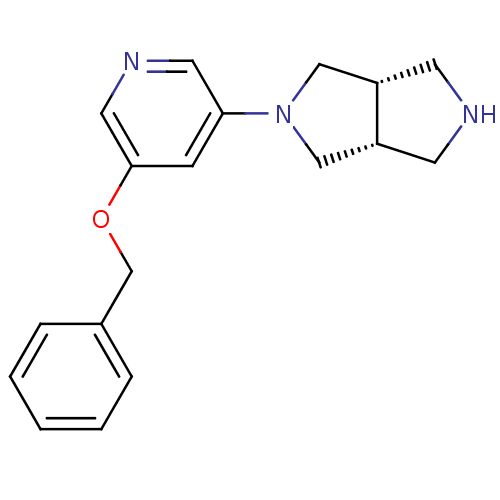
(CHEMBL524911 | cis-2-(5-(Benzyloxy)pyridin-3-yl)oc...)Show SMILES C(Oc1cncc(c1)N1C[C@@H]2CNC[C@@H]2C1)c1ccccc1 |r| Show InChI InChI=1S/C18H21N3O/c1-2-4-14(5-3-1)13-22-18-6-17(9-20-10-18)21-11-15-7-19-8-16(15)12-21/h1-6,9-10,15-16,19H,7-8,11-13H2/t15-,16+ | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from alpha4beta2 nicotinic acetylcholine receptor in rat brain minus cerebellum membrane |
J Med Chem 52: 4126-41 (2009)
Article DOI: 10.1021/jm900249k
BindingDB Entry DOI: 10.7270/Q2GT5P3R |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM50279402
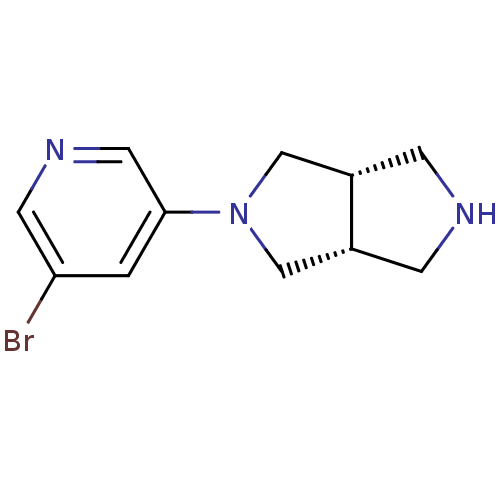
(CHEMBL486511 | cis-2-(5-Bromopyridin-3-yl)-octahyd...)Show InChI InChI=1S/C11H14BrN3/c12-10-1-11(5-14-4-10)15-6-8-2-13-3-9(8)7-15/h1,4-5,8-9,13H,2-3,6-7H2/t8-,9+ | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from alpha4beta2 nicotinic acetylcholine receptor in rat brain minus cerebellum membrane |
J Med Chem 52: 4126-41 (2009)
Article DOI: 10.1021/jm900249k
BindingDB Entry DOI: 10.7270/Q2GT5P3R |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM520
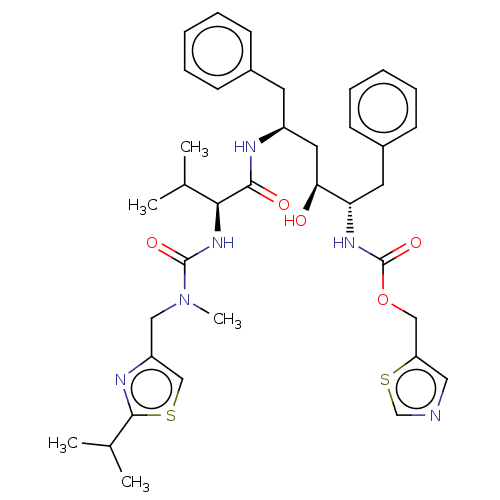
(1,3-thiazol-5-ylmethyl N-[(2S,3S,5S)-3-hydroxy-5-[...)Show SMILES CC(C)[C@H](NC(=O)N(C)Cc1csc(n1)C(C)C)C(=O)N[C@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)OCc1cncs1)Cc1ccccc1 |r| Show InChI InChI=1S/C37H48N6O5S2/c1-24(2)33(42-36(46)43(5)20-29-22-49-35(40-29)25(3)4)34(45)39-28(16-26-12-8-6-9-13-26)18-32(44)31(17-27-14-10-7-11-15-27)41-37(47)48-21-30-19-38-23-50-30/h6-15,19,22-25,28,31-33,44H,16-18,20-21H2,1-5H3,(H,39,45)(H,41,47)(H,42,46)/t28-,31-,32-,33-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
PubMed
| 0.0620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Dissociation constant obtained by inhibition of Wild-type protease |
J Med Chem 43: 3386-99 (2000)
BindingDB Entry DOI: 10.7270/Q23J3DP4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM519
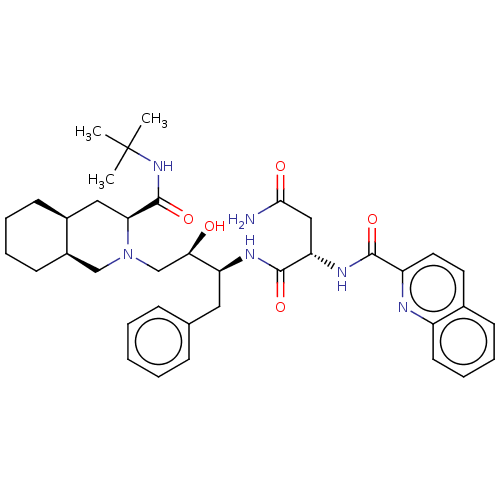
((2S)-N-[(2S,3R)-4-[(3S,4aS,8aS)-3-(tert-butylcarba...)Show SMILES [H][C@@]12CCCC[C@]1([H])CN(C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)c1ccc3ccccc3n1)[C@@H](C2)C(=O)NC(C)(C)C |r| Show InChI InChI=1S/C38H50N6O5/c1-38(2,3)43-37(49)32-20-26-14-7-8-15-27(26)22-44(32)23-33(45)30(19-24-11-5-4-6-12-24)41-36(48)31(21-34(39)46)42-35(47)29-18-17-25-13-9-10-16-28(25)40-29/h4-6,9-13,16-18,26-27,30-33,45H,7-8,14-15,19-23H2,1-3H3,(H2,39,46)(H,41,48)(H,42,47)(H,43,49)/t26-,27+,30-,31-,32-,33+/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 0.0620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Dissociation constant obtained by inhibition of Wild-type protease |
J Med Chem 43: 3386-99 (2000)
BindingDB Entry DOI: 10.7270/Q23J3DP4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Neuronal acetylcholine receptor subunit alpha-7
(Rattus norvegicus (Rat)) | BDBM50319933
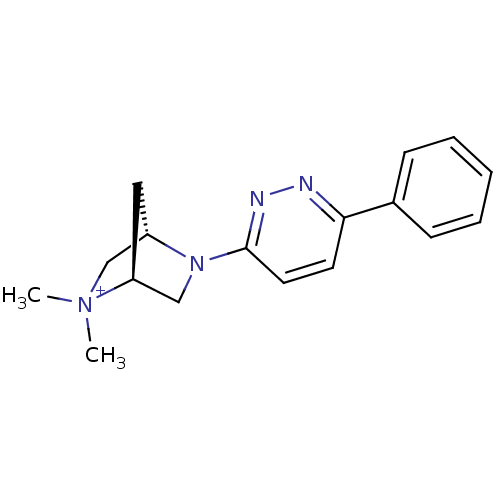
((1S,4S)-2,2-dimethyl-5-(6-phenylpyridazin-3-yl)-5-...)Show SMILES C[N+]1(C)C[C@@H]2C[C@H]1CN2c1ccc(nn1)-c1ccccc1 |r| Show InChI InChI=1S/C17H21N4/c1-21(2)12-14-10-15(21)11-20(14)17-9-8-16(18-19-17)13-6-4-3-5-7-13/h3-9,14-15H,10-12H2,1-2H3/q+1/t14-,15-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0660 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Park
Curated by ChEMBL
| Assay Description
Displacement of [3H]A585539 from alpha7 nACHR in rat brain homogenate |
Bioorg Med Chem Lett 20: 3636-9 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.105
BindingDB Entry DOI: 10.7270/Q2MC905N |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM50279403
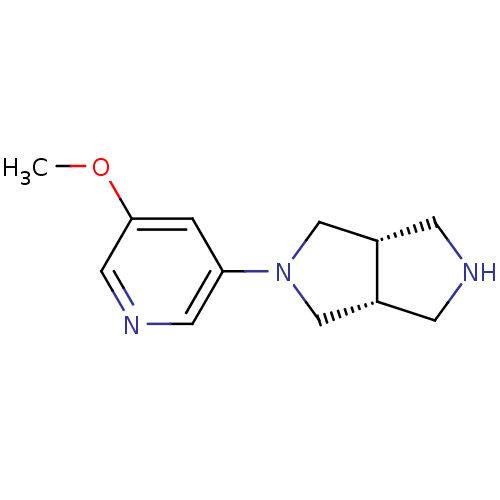
(CHEMBL486512 | cis-2-(5-Methoxy-3-pyridinyl)octahy...)Show InChI InChI=1S/C12H17N3O/c1-16-12-2-11(5-14-6-12)15-7-9-3-13-4-10(9)8-15/h2,5-6,9-10,13H,3-4,7-8H2,1H3/t9-,10+ | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0810 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from alpha4beta2 nicotinic acetylcholine receptor in rat brain minus cerebellum membrane |
J Med Chem 52: 4126-41 (2009)
Article DOI: 10.1021/jm900249k
BindingDB Entry DOI: 10.7270/Q2GT5P3R |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50454822
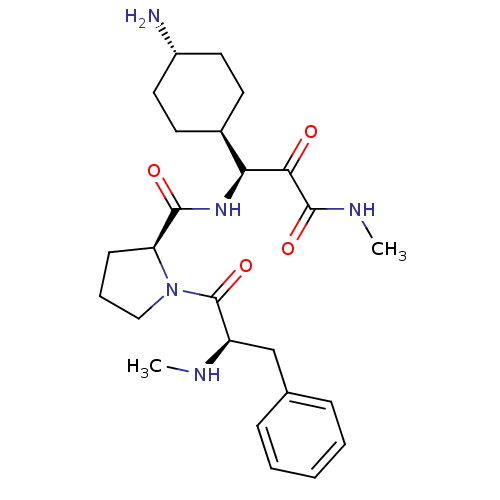
(CHEMBL2062141 | L-370518)Show SMILES [H][C@@](NC(=O)[C@@H]1CCCN1C(=O)[C@@H](Cc1ccccc1)NC)(C(=O)C(=O)NC)[C@@]1([H])CC[C@H](N)CC1 |wU:1.0,wD:12.13,5.4,32.34,28.30,(9.54,-15.25,;8.45,-16.34,;7.42,-17.49,;5.92,-17.17,;5.44,-15.7,;4.89,-18.31,;5.21,-19.82,;3.87,-20.59,;2.73,-19.56,;3.35,-18.15,;2.58,-16.82,;3.35,-15.48,;1.04,-16.82,;.27,-15.48,;1.04,-14.15,;2.58,-14.15,;3.35,-12.82,;2.58,-11.48,;1.04,-11.48,;.27,-12.82,;.27,-18.15,;-1.27,-18.15,;7.98,-14.88,;6.47,-14.56,;9.01,-13.73,;10.51,-14.05,;8.53,-12.27,;9.56,-11.12,;9.96,-16.66,;8.87,-17.75,;10.99,-15.52,;12.5,-15.84,;12.97,-17.3,;14.48,-17.62,;11.94,-18.45,;10.44,-18.13,)| Show InChI InChI=1S/C25H37N5O4/c1-27-19(15-16-7-4-3-5-8-16)25(34)30-14-6-9-20(30)23(32)29-21(22(31)24(33)28-2)17-10-12-18(26)13-11-17/h3-5,7-8,17-21,27H,6,9-15,26H2,1-2H3,(H,28,33)(H,29,32)/t17-,18-,19-,20+,21+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards thrombin was determined |
J Med Chem 41: 401-6 (1998)
Article DOI: 10.1021/jm9705014
BindingDB Entry DOI: 10.7270/Q2H995V2 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50057828

((S)-1-((R)-2-Amino-3,3-diphenyl-propionyl)-pyrroli...)Show SMILES N[C@H](C(c1ccccc1)c1ccccc1)C(=O)N1CCC[C@H]1C(=O)NCC1CCC(N)CC1 |wU:21.24,wD:1.0,(8.37,-6.36,;9.7,-5.59,;9.7,-4.05,;11.04,-3.28,;12.58,-3.28,;13.35,-1.94,;12.56,-.61,;11.02,-.62,;10.27,-1.96,;8.35,-3.28,;8.35,-1.74,;7.03,-.97,;5.69,-1.74,;5.69,-3.28,;7.03,-4.05,;11.05,-6.36,;11.05,-7.9,;12.36,-5.59,;12.52,-4.05,;14.03,-3.73,;14.8,-5.05,;13.77,-6.2,;14.1,-7.71,;12.96,-8.74,;15.57,-8.18,;16.72,-7.15,;18.17,-7.61,;18.09,-8.95,;19.1,-10.37,;17.98,-11.46,;19.06,-12.54,;18.09,-10.21,;17.07,-8.71,)| Show InChI InChI=1S/C27H36N4O2/c28-22-15-13-19(14-16-22)18-30-26(32)23-12-7-17-31(23)27(33)25(29)24(20-8-3-1-4-9-20)21-10-5-2-6-11-21/h1-6,8-11,19,22-25H,7,12-18,28-29H2,(H,30,32)/t19?,22?,23-,25+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards thrombin was determined |
J Med Chem 41: 401-6 (1998)
Article DOI: 10.1021/jm9705014
BindingDB Entry DOI: 10.7270/Q2H995V2 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM50279369
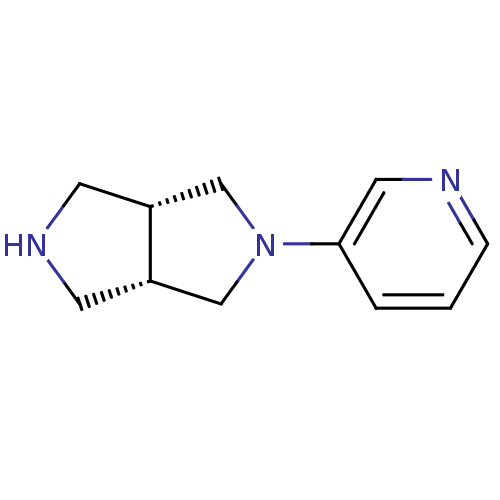
(CHEMBL460562 | cis-2-(3-Pyridinyl)octahydropyrrolo...)Show InChI InChI=1S/C11H15N3/c1-2-11(6-12-3-1)14-7-9-4-13-5-10(9)8-14/h1-3,6,9-10,13H,4-5,7-8H2/t9-,10+ | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from alpha4beta2 nicotinic acetylcholine receptor in rat brain minus cerebellum membrane |
J Med Chem 52: 4126-41 (2009)
Article DOI: 10.1021/jm900249k
BindingDB Entry DOI: 10.7270/Q2GT5P3R |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50366785
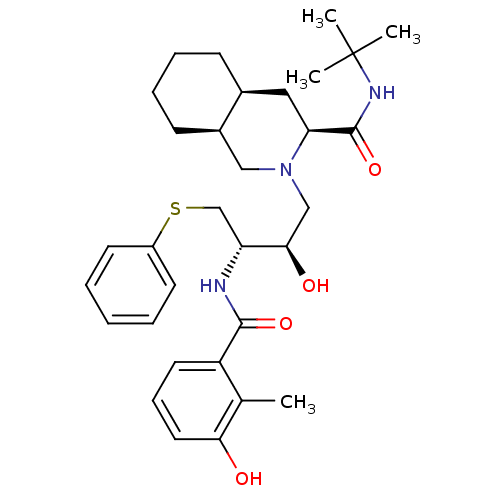
(NELFINAVIR)Show SMILES Cc1c(O)cccc1C(=O)N[C@H](CSc1ccccc1)[C@H](O)CN1C[C@H]2CCCC[C@H]2C[C@H]1C(=O)NC(C)(C)C |r| Show InChI InChI=1S/C32H45N3O4S/c1-21-25(15-10-16-28(21)36)30(38)33-26(20-40-24-13-6-5-7-14-24)29(37)19-35-18-23-12-9-8-11-22(23)17-27(35)31(39)34-32(2,3)4/h5-7,10,13-16,22-23,26-27,29,36-37H,8-9,11-12,17-20H2,1-4H3,(H,33,38)(H,34,39)/t22-,23+,26+,27-,29+/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Dissociation constant obtained by inhibition of Wild-type protease |
J Med Chem 43: 3386-99 (2000)
BindingDB Entry DOI: 10.7270/Q23J3DP4 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM50278881
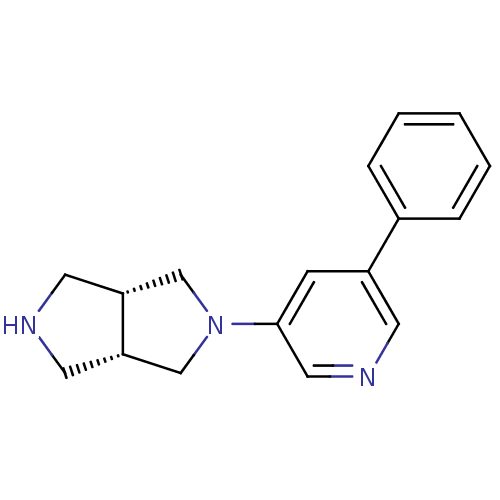
(CHEMBL496667 | cis-2-(5-Phenylpyridin-3-yl)-octahy...)Show SMILES C1NC[C@@H]2CN(C[C@H]12)c1cncc(c1)-c1ccccc1 |r| Show InChI InChI=1S/C17H19N3/c1-2-4-13(5-3-1)14-6-17(10-19-7-14)20-11-15-8-18-9-16(15)12-20/h1-7,10,15-16,18H,8-9,11-12H2/t15-,16+ | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from alpha4beta2 nicotinic acetylcholine receptor in rat brain minus cerebellum membrane |
J Med Chem 52: 4126-41 (2009)
Article DOI: 10.1021/jm900249k
BindingDB Entry DOI: 10.7270/Q2GT5P3R |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Rattus norvegicus (rat)) | BDBM50451054
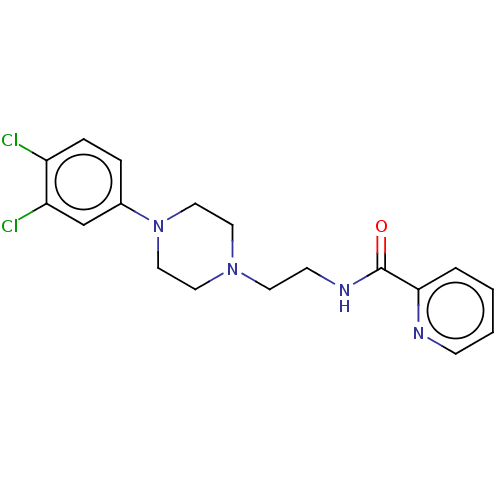
(CHEMBL4208744)Show InChI InChI=1S/C18H20Cl2N4O/c19-15-5-4-14(13-16(15)20)24-11-9-23(10-12-24)8-7-22-18(25)17-3-1-2-6-21-17/h1-6,13H,7-12H2,(H,22,25) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.183 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dipartimento di Farmacia Universit£ di Napoli "Federico II" Via D. Montesano
Curated by ChEMBL
| Assay Description
Displacement of [3H]-8-OH-DPAT from serotonin 5-HT1A receptor in Sprague-Dawley rat brain cortex homogenates incubated for 30 mins by liquid scintill... |
Bioorg Med Chem 25: 5820-5837 (2017)
Article DOI: 10.1016/j.bmc.2017.09.018
BindingDB Entry DOI: 10.7270/Q2VD721F |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 2
(Homo sapiens (Human)) | BDBM50231578

(CHEMBL3349022)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O |r| Show InChI InChI=1S/C190H287N55O57/c1-16-94(9)149(180(296)235-129(81-141(194)255)169(285)227-124(74-93(7)8)172(288)240-150(95(10)17-2)181(297)241-151(100(15)248)182(298)222-116(32-23-67-209-190(203)204)156(272)221-118(57-60-140(193)254)161(277)219-114(30-21-65-207-188(199)200)157(273)224-121(152(196)268)76-102-39-49-108(250)50-40-102)239-173(289)127(79-105-45-55-111(253)56-46-105)230-168(284)128(80-106-86-205-90-211-106)231-159(275)115(31-22-66-208-189(201)202)220-165(281)123(73-92(5)6)225-155(271)97(12)213-174(290)134(88-246)237-167(283)126(78-104-43-53-110(252)54-44-104)229-166(282)125(77-103-41-51-109(251)52-42-103)228-158(274)113(29-20-64-206-187(197)198)217-153(269)96(11)212-163(279)122(72-91(3)4)226-170(286)131(84-147(264)265)233-162(278)119(59-62-145(260)261)218-154(270)98(13)214-177(293)137-34-25-68-242(137)183(299)99(14)215-164(280)130(83-146(262)263)232-160(276)117(58-61-144(258)259)216-143(257)87-210-176(292)136-33-24-70-244(136)186(302)133(82-142(195)256)236-171(287)132(85-148(266)267)234-178(294)139-36-27-71-245(139)185(301)120(28-18-19-63-191)223-175(291)135(89-247)238-179(295)138-35-26-69-243(138)184(300)112(192)75-101-37-47-107(249)48-38-101/h37-56,86,90-100,112-139,149-151,246-253H,16-36,57-85,87-89,191-192H2,1-15H3,(H2,193,254)(H2,194,255)(H2,195,256)(H2,196,268)(H,205,211)(H,210,292)(H,212,279)(H,213,290)(H,214,293)(H,215,280)(H,216,257)(H,217,269)(H,218,270)(H,219,277)(H,220,281)(H,221,272)(H,222,298)(H,223,291)(H,224,273)(H,225,271)(H,226,286)(H,227,285)(H,228,274)(H,229,282)(H,230,284)(H,231,275)(H,232,276)(H,233,278)(H,234,294)(H,235,296)(H,236,287)(H,237,283)(H,238,295)(H,239,289)(H,240,288)(H,241,297)(H,258,259)(H,260,261)(H,262,263)(H,264,265)(H,266,267)(H4,197,198,206)(H4,199,200,207)(H4,201,202,208)(H4,203,204,209)/t94-,95-,96-,97-,98-,99-,100+,112-,113-,114-,115-,116-,117-,118-,119-,120+,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,139-,149-,150-,151-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Salk Institute
Curated by ChEMBL
| Assay Description
Affinity against Neuropeptide Y2 receptor on SK-N-BE2 human neuroblastoma cells |
J Med Chem 36: 3802-8 (1994)
BindingDB Entry DOI: 10.7270/Q28K784Q |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM50279371
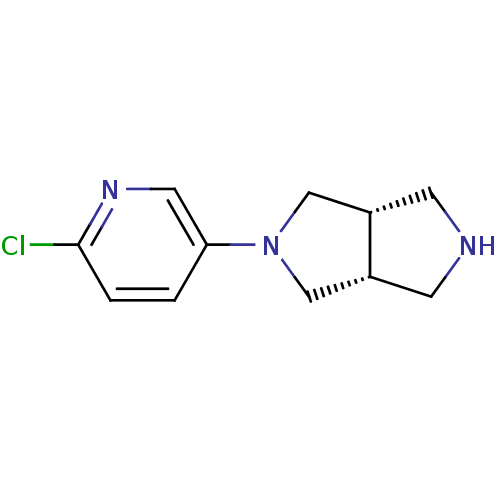
(CHEMBL484831 | cis-2-(6-Chloro-3-pyridinyl)octahyd...)Show InChI InChI=1S/C11H14ClN3/c12-11-2-1-10(5-14-11)15-6-8-3-13-4-9(8)7-15/h1-2,5,8-9,13H,3-4,6-7H2/t8-,9+ | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from alpha4beta2 nicotinic acetylcholine receptor in rat brain minus cerebellum membrane |
J Med Chem 52: 4126-41 (2009)
Article DOI: 10.1021/jm900249k
BindingDB Entry DOI: 10.7270/Q2GT5P3R |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 2
(Homo sapiens (Human)) | BDBM50231578

(CHEMBL3349022)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O |r| Show InChI InChI=1S/C190H287N55O57/c1-16-94(9)149(180(296)235-129(81-141(194)255)169(285)227-124(74-93(7)8)172(288)240-150(95(10)17-2)181(297)241-151(100(15)248)182(298)222-116(32-23-67-209-190(203)204)156(272)221-118(57-60-140(193)254)161(277)219-114(30-21-65-207-188(199)200)157(273)224-121(152(196)268)76-102-39-49-108(250)50-40-102)239-173(289)127(79-105-45-55-111(253)56-46-105)230-168(284)128(80-106-86-205-90-211-106)231-159(275)115(31-22-66-208-189(201)202)220-165(281)123(73-92(5)6)225-155(271)97(12)213-174(290)134(88-246)237-167(283)126(78-104-43-53-110(252)54-44-104)229-166(282)125(77-103-41-51-109(251)52-42-103)228-158(274)113(29-20-64-206-187(197)198)217-153(269)96(11)212-163(279)122(72-91(3)4)226-170(286)131(84-147(264)265)233-162(278)119(59-62-145(260)261)218-154(270)98(13)214-177(293)137-34-25-68-242(137)183(299)99(14)215-164(280)130(83-146(262)263)232-160(276)117(58-61-144(258)259)216-143(257)87-210-176(292)136-33-24-70-244(136)186(302)133(82-142(195)256)236-171(287)132(85-148(266)267)234-178(294)139-36-27-71-245(139)185(301)120(28-18-19-63-191)223-175(291)135(89-247)238-179(295)138-35-26-69-243(138)184(300)112(192)75-101-37-47-107(249)48-38-101/h37-56,86,90-100,112-139,149-151,246-253H,16-36,57-85,87-89,191-192H2,1-15H3,(H2,193,254)(H2,194,255)(H2,195,256)(H2,196,268)(H,205,211)(H,210,292)(H,212,279)(H,213,290)(H,214,293)(H,215,280)(H,216,257)(H,217,269)(H,218,270)(H,219,277)(H,220,281)(H,221,272)(H,222,298)(H,223,291)(H,224,273)(H,225,271)(H,226,286)(H,227,285)(H,228,274)(H,229,282)(H,230,284)(H,231,275)(H,232,276)(H,233,278)(H,234,294)(H,235,296)(H,236,287)(H,237,283)(H,238,295)(H,239,289)(H,240,288)(H,241,297)(H,258,259)(H,260,261)(H,262,263)(H,264,265)(H,266,267)(H4,197,198,206)(H4,199,200,207)(H4,201,202,208)(H4,203,204,209)/t94-,95-,96-,97-,98-,99-,100+,112-,113-,114-,115-,116-,117-,118-,119-,120+,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,139-,149-,150-,151-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Salk Institute
Curated by ChEMBL
| Assay Description
Affinity against Neuropeptide Y2 receptor on SK-N-BE2 human neuroblastoma cells |
J Med Chem 36: 3802-8 (1994)
BindingDB Entry DOI: 10.7270/Q28K784Q |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 2
(Homo sapiens (Human)) | BDBM50231578

(CHEMBL3349022)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O |r| Show InChI InChI=1S/C190H287N55O57/c1-16-94(9)149(180(296)235-129(81-141(194)255)169(285)227-124(74-93(7)8)172(288)240-150(95(10)17-2)181(297)241-151(100(15)248)182(298)222-116(32-23-67-209-190(203)204)156(272)221-118(57-60-140(193)254)161(277)219-114(30-21-65-207-188(199)200)157(273)224-121(152(196)268)76-102-39-49-108(250)50-40-102)239-173(289)127(79-105-45-55-111(253)56-46-105)230-168(284)128(80-106-86-205-90-211-106)231-159(275)115(31-22-66-208-189(201)202)220-165(281)123(73-92(5)6)225-155(271)97(12)213-174(290)134(88-246)237-167(283)126(78-104-43-53-110(252)54-44-104)229-166(282)125(77-103-41-51-109(251)52-42-103)228-158(274)113(29-20-64-206-187(197)198)217-153(269)96(11)212-163(279)122(72-91(3)4)226-170(286)131(84-147(264)265)233-162(278)119(59-62-145(260)261)218-154(270)98(13)214-177(293)137-34-25-68-242(137)183(299)99(14)215-164(280)130(83-146(262)263)232-160(276)117(58-61-144(258)259)216-143(257)87-210-176(292)136-33-24-70-244(136)186(302)133(82-142(195)256)236-171(287)132(85-148(266)267)234-178(294)139-36-27-71-245(139)185(301)120(28-18-19-63-191)223-175(291)135(89-247)238-179(295)138-35-26-69-243(138)184(300)112(192)75-101-37-47-107(249)48-38-101/h37-56,86,90-100,112-139,149-151,246-253H,16-36,57-85,87-89,191-192H2,1-15H3,(H2,193,254)(H2,194,255)(H2,195,256)(H2,196,268)(H,205,211)(H,210,292)(H,212,279)(H,213,290)(H,214,293)(H,215,280)(H,216,257)(H,217,269)(H,218,270)(H,219,277)(H,220,281)(H,221,272)(H,222,298)(H,223,291)(H,224,273)(H,225,271)(H,226,286)(H,227,285)(H,228,274)(H,229,282)(H,230,284)(H,231,275)(H,232,276)(H,233,278)(H,234,294)(H,235,296)(H,236,287)(H,237,283)(H,238,295)(H,239,289)(H,240,288)(H,241,297)(H,258,259)(H,260,261)(H,262,263)(H,264,265)(H,266,267)(H4,197,198,206)(H4,199,200,207)(H4,201,202,208)(H4,203,204,209)/t94-,95-,96-,97-,98-,99-,100+,112-,113-,114-,115-,116-,117-,118-,119-,120+,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,139-,149-,150-,151-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Salk Institute
Curated by ChEMBL
| Assay Description
Affinity against Neuropeptide Y2 receptor on SK-N-BE2 human neuroblastoma cells |
J Med Chem 36: 3802-8 (1994)
BindingDB Entry DOI: 10.7270/Q28K784Q |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 2
(Homo sapiens (Human)) | BDBM50231578

(CHEMBL3349022)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O |r| Show InChI InChI=1S/C190H287N55O57/c1-16-94(9)149(180(296)235-129(81-141(194)255)169(285)227-124(74-93(7)8)172(288)240-150(95(10)17-2)181(297)241-151(100(15)248)182(298)222-116(32-23-67-209-190(203)204)156(272)221-118(57-60-140(193)254)161(277)219-114(30-21-65-207-188(199)200)157(273)224-121(152(196)268)76-102-39-49-108(250)50-40-102)239-173(289)127(79-105-45-55-111(253)56-46-105)230-168(284)128(80-106-86-205-90-211-106)231-159(275)115(31-22-66-208-189(201)202)220-165(281)123(73-92(5)6)225-155(271)97(12)213-174(290)134(88-246)237-167(283)126(78-104-43-53-110(252)54-44-104)229-166(282)125(77-103-41-51-109(251)52-42-103)228-158(274)113(29-20-64-206-187(197)198)217-153(269)96(11)212-163(279)122(72-91(3)4)226-170(286)131(84-147(264)265)233-162(278)119(59-62-145(260)261)218-154(270)98(13)214-177(293)137-34-25-68-242(137)183(299)99(14)215-164(280)130(83-146(262)263)232-160(276)117(58-61-144(258)259)216-143(257)87-210-176(292)136-33-24-70-244(136)186(302)133(82-142(195)256)236-171(287)132(85-148(266)267)234-178(294)139-36-27-71-245(139)185(301)120(28-18-19-63-191)223-175(291)135(89-247)238-179(295)138-35-26-69-243(138)184(300)112(192)75-101-37-47-107(249)48-38-101/h37-56,86,90-100,112-139,149-151,246-253H,16-36,57-85,87-89,191-192H2,1-15H3,(H2,193,254)(H2,194,255)(H2,195,256)(H2,196,268)(H,205,211)(H,210,292)(H,212,279)(H,213,290)(H,214,293)(H,215,280)(H,216,257)(H,217,269)(H,218,270)(H,219,277)(H,220,281)(H,221,272)(H,222,298)(H,223,291)(H,224,273)(H,225,271)(H,226,286)(H,227,285)(H,228,274)(H,229,282)(H,230,284)(H,231,275)(H,232,276)(H,233,278)(H,234,294)(H,235,296)(H,236,287)(H,237,283)(H,238,295)(H,239,289)(H,240,288)(H,241,297)(H,258,259)(H,260,261)(H,262,263)(H,264,265)(H,266,267)(H4,197,198,206)(H4,199,200,207)(H4,201,202,208)(H4,203,204,209)/t94-,95-,96-,97-,98-,99-,100+,112-,113-,114-,115-,116-,117-,118-,119-,120+,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,139-,149-,150-,151-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Salk Institute
Curated by ChEMBL
| Assay Description
Affinity against Neuropeptide Y2 receptor on SK-N-BE2 human neuroblastoma cells |
J Med Chem 36: 3802-8 (1994)
BindingDB Entry DOI: 10.7270/Q28K784Q |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50319928
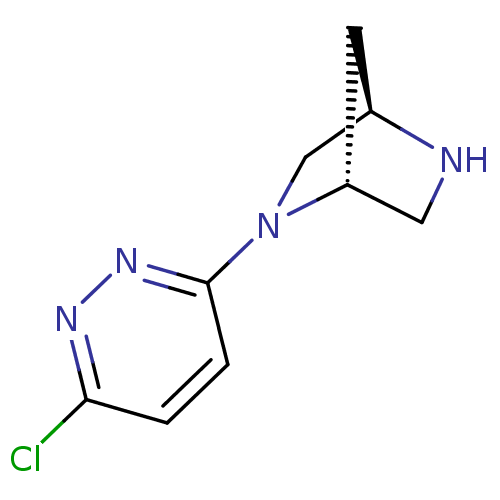
((1R,4R)-2-(6-chloropyridazin-3-yl)-2,5-diazabicycl...)Show InChI InChI=1S/C9H11ClN4/c10-8-1-2-9(13-12-8)14-5-6-3-7(14)4-11-6/h1-2,6-7,11H,3-5H2/t6-,7-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Park
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from alpha4beta2 nACHR in rat brain homogenate |
Bioorg Med Chem Lett 20: 3636-9 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.105
BindingDB Entry DOI: 10.7270/Q2MC905N |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-7
(Rattus norvegicus (Rat)) | BDBM50319964
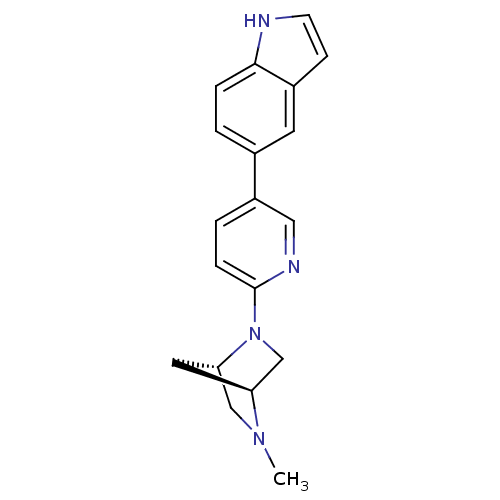
(5-(6-((1S,4S)-5-methyl-2,5-diazabicyclo[2.2.1]hept...)Show SMILES CN1C[C@@H]2C[C@H]1CN2c1ccc(cn1)-c1ccc2[nH]ccc2c1 |r| Show InChI InChI=1S/C19H20N4/c1-22-11-17-9-16(22)12-23(17)19-5-3-15(10-21-19)13-2-4-18-14(8-13)6-7-20-18/h2-8,10,16-17,20H,9,11-12H2,1H3/t16-,17-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Park
Curated by ChEMBL
| Assay Description
Displacement of [3H]A585539 from alpha7 nACHR in rat brain homogenate |
Bioorg Med Chem Lett 20: 3636-9 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.105
BindingDB Entry DOI: 10.7270/Q2MC905N |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM517

((2S)-1-[(2S,4R)-4-benzyl-2-hydroxy-4-{[(1S,2R)-2-h...)Show SMILES CC(C)(C)NC(=O)[C@@H]1CN(Cc2cccnc2)CCN1C[C@@H](O)C[C@@H](Cc1ccccc1)C(=O)N[C@@H]1[C@H](O)Cc2ccccc12 |r| Show InChI InChI=1S/C36H47N5O4/c1-36(2,3)39-35(45)31-24-40(22-26-12-9-15-37-21-26)16-17-41(31)23-29(42)19-28(18-25-10-5-4-6-11-25)34(44)38-33-30-14-8-7-13-27(30)20-32(33)43/h4-15,21,28-29,31-33,42-43H,16-20,22-24H2,1-3H3,(H,38,44)(H,39,45)/t28-,29+,31+,32-,33+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Dissociation constant obtained by inhibition of Wild-type protease |
J Med Chem 43: 3386-99 (2000)
BindingDB Entry DOI: 10.7270/Q23J3DP4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Neuronal acetylcholine receptor subunit alpha-7
(Rattus norvegicus (Rat)) | BDBM50279059
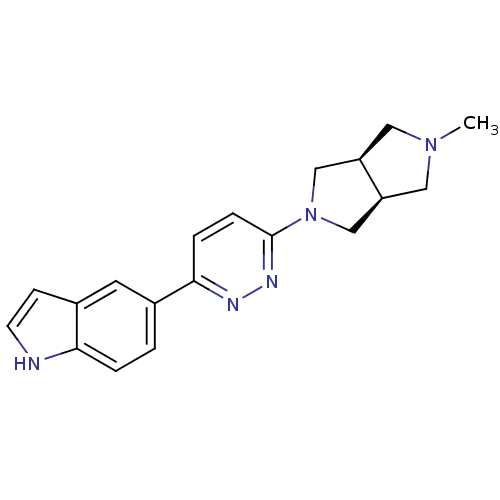
(CHEMBL496851 | cis-5-[6-(5-Methyl-hexahydro-pyrrol...)Show SMILES CN1C[C@H]2CN(C[C@H]2C1)c1ccc(nn1)-c1ccc2[nH]ccc2c1 |r| Show InChI InChI=1S/C19H21N5/c1-23-9-15-11-24(12-16(15)10-23)19-5-4-18(21-22-19)13-2-3-17-14(8-13)6-7-20-17/h2-8,15-16,20H,9-12H2,1H3/t15-,16+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]A585539 from alpha7 nicotinic acetylcholine receptor in rat brain minus cerebellum membrane |
J Med Chem 52: 4126-41 (2009)
Article DOI: 10.1021/jm900249k
BindingDB Entry DOI: 10.7270/Q2GT5P3R |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM50279401
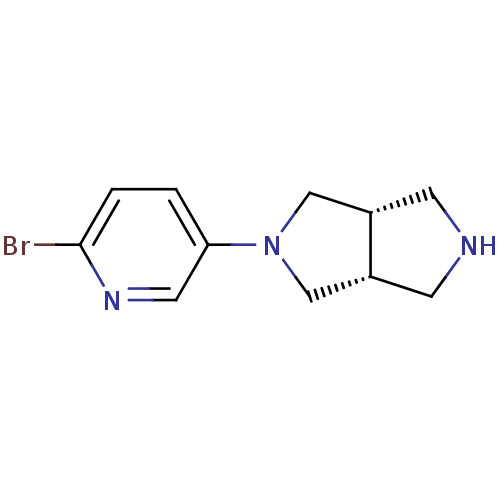
(CHEMBL498943 | cis-5-(6-Bromo-pyridin-3-yl)-hexahy...)Show InChI InChI=1S/C11H14BrN3/c12-11-2-1-10(5-14-11)15-6-8-3-13-4-9(8)7-15/h1-2,5,8-9,13H,3-4,6-7H2/t8-,9+ | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from alpha4beta2 nicotinic acetylcholine receptor in rat brain minus cerebellum membrane |
J Med Chem 52: 4126-41 (2009)
Article DOI: 10.1021/jm900249k
BindingDB Entry DOI: 10.7270/Q2GT5P3R |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-7
(Rattus norvegicus (Rat)) | BDBM50319944
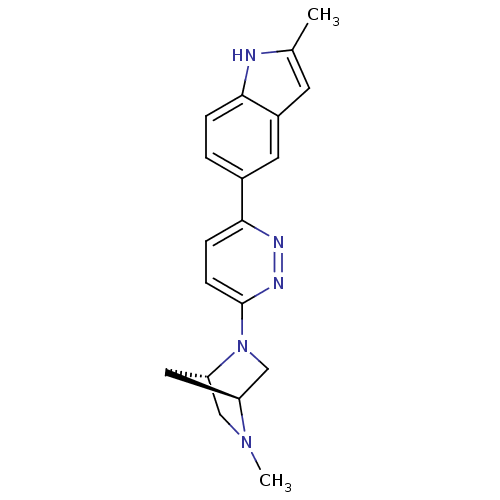
(2-methyl-5-(6-((1S,4S)-5-methyl-2,5-diazabicyclo[2...)Show SMILES CN1C[C@@H]2C[C@H]1CN2c1ccc(nn1)-c1ccc2[nH]c(C)cc2c1 |r| Show InChI InChI=1S/C19H21N5/c1-12-7-14-8-13(3-4-17(14)20-12)18-5-6-19(22-21-18)24-11-15-9-16(24)10-23(15)2/h3-8,15-16,20H,9-11H2,1-2H3/t15-,16-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Park
Curated by ChEMBL
| Assay Description
Displacement of [3H]A585539 from alpha7 nACHR in rat brain homogenate |
Bioorg Med Chem Lett 20: 3636-9 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.105
BindingDB Entry DOI: 10.7270/Q2MC905N |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-7
(Rattus norvegicus (Rat)) | BDBM50319963
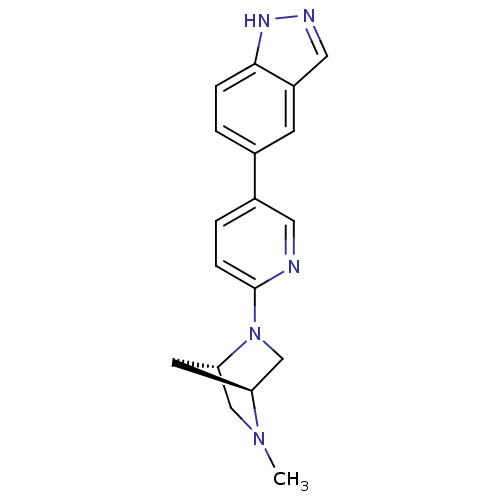
(5-(6-((1S,4S)-5-methyl-2,5-diazabicyclo[2.2.1]hept...)Show SMILES CN1C[C@@H]2C[C@H]1CN2c1ccc(cn1)-c1ccc2[nH]ncc2c1 |r| Show InChI InChI=1S/C18H19N5/c1-22-10-16-7-15(22)11-23(16)18-5-3-13(8-19-18)12-2-4-17-14(6-12)9-20-21-17/h2-6,8-9,15-16H,7,10-11H2,1H3,(H,20,21)/t15-,16-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Park
Curated by ChEMBL
| Assay Description
Displacement of [3H]A585539 from alpha7 nACHR in rat brain homogenate |
Bioorg Med Chem Lett 20: 3636-9 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.105
BindingDB Entry DOI: 10.7270/Q2MC905N |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 2
(Homo sapiens (Human)) | BDBM50005530
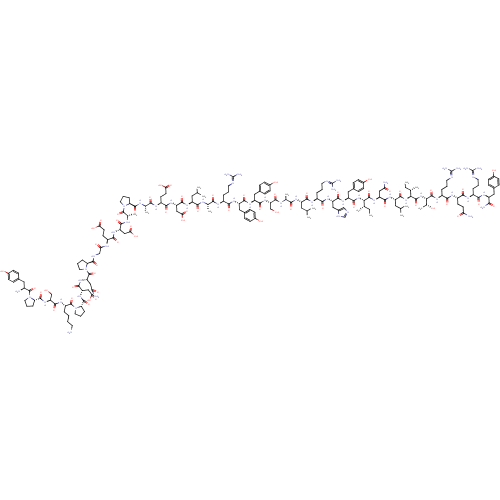
(CHEMBL267633 | D-Tyr-Pro-Ser-Lys-Pro-Asp-Asn-Pro-G...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O |r| Show InChI InChI=1S/C190H287N55O57/c1-16-94(9)149(180(296)235-129(81-141(194)255)169(285)227-124(74-93(7)8)172(288)240-150(95(10)17-2)181(297)241-151(100(15)248)182(298)222-116(32-23-67-209-190(203)204)156(272)221-118(57-60-140(193)254)161(277)219-114(30-21-65-207-188(199)200)157(273)224-121(152(196)268)76-102-39-49-108(250)50-40-102)239-173(289)127(79-105-45-55-111(253)56-46-105)230-168(284)128(80-106-86-205-90-211-106)231-159(275)115(31-22-66-208-189(201)202)220-165(281)123(73-92(5)6)225-155(271)97(12)213-174(290)134(88-246)237-167(283)126(78-104-43-53-110(252)54-44-104)229-166(282)125(77-103-41-51-109(251)52-42-103)228-158(274)113(29-20-64-206-187(197)198)217-153(269)96(11)212-163(279)122(72-91(3)4)226-170(286)131(84-147(264)265)233-162(278)119(59-62-145(260)261)218-154(270)98(13)214-177(293)137-34-25-68-242(137)183(299)99(14)215-164(280)130(83-146(262)263)232-160(276)117(58-61-144(258)259)216-143(257)87-210-176(292)136-33-24-70-244(136)186(302)133(82-142(195)256)236-171(287)132(85-148(266)267)234-178(294)139-36-27-71-245(139)185(301)120(28-18-19-63-191)223-175(291)135(89-247)238-179(295)138-35-26-69-243(138)184(300)112(192)75-101-37-47-107(249)48-38-101/h37-56,86,90-100,112-139,149-151,246-253H,16-36,57-85,87-89,191-192H2,1-15H3,(H2,193,254)(H2,194,255)(H2,195,256)(H2,196,268)(H,205,211)(H,210,292)(H,212,279)(H,213,290)(H,214,293)(H,215,280)(H,216,257)(H,217,269)(H,218,270)(H,219,277)(H,220,281)(H,221,272)(H,222,298)(H,223,291)(H,224,273)(H,225,271)(H,226,286)(H,227,285)(H,228,274)(H,229,282)(H,230,284)(H,231,275)(H,232,276)(H,233,278)(H,234,294)(H,235,296)(H,236,287)(H,237,283)(H,238,295)(H,239,289)(H,240,288)(H,241,297)(H,258,259)(H,260,261)(H,262,263)(H,264,265)(H,266,267)(H4,197,198,206)(H4,199,200,207)(H4,201,202,208)(H4,203,204,209)/t94-,95-,96-,97-,98-,99-,100+,112-,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,139-,149-,150-,151-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Salk Institute
Curated by ChEMBL
| Assay Description
Affinity against Neuropeptide Y2 receptor on SK-N-BE2 human neuroblastoma cells |
J Med Chem 36: 3802-8 (1994)
BindingDB Entry DOI: 10.7270/Q28K784Q |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50069189
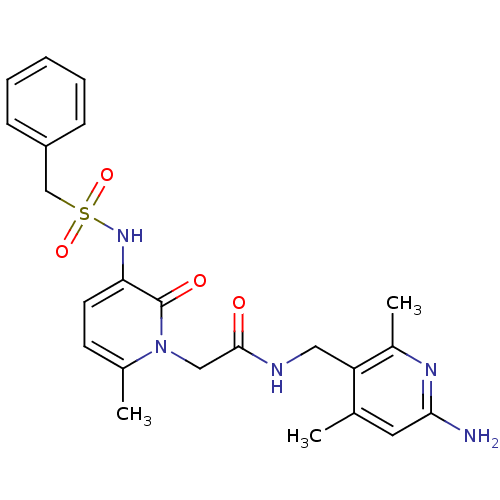
(CHEMBL353431 | N-(6-Amino-2,4-dimethyl-pyridin-3-y...)Show SMILES Cc1cc(N)nc(C)c1CNC(=O)Cn1c(C)ccc(NS(=O)(=O)Cc2ccccc2)c1=O Show InChI InChI=1S/C23H27N5O4S/c1-15-11-21(24)26-17(3)19(15)12-25-22(29)13-28-16(2)9-10-20(23(28)30)27-33(31,32)14-18-7-5-4-6-8-18/h4-11,27H,12-14H2,1-3H3,(H2,24,26)(H,25,29) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards thrombin was evaluated |
Bioorg Med Chem Lett 8: 817-22 (1999)
BindingDB Entry DOI: 10.7270/Q2JH3K9Z |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Rattus norvegicus (rat)) | BDBM50145625

(CHEMBL3763633)Show InChI InChI=1S/C18H21ClN4O/c19-16-1-3-17(4-2-16)23-13-11-22(12-14-23)10-9-21-18(24)15-5-7-20-8-6-15/h1-8H,9-14H2,(H,21,24) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.344 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dipartimento di Farmacia Universit£ di Napoli "Federico II" Via D. Montesano
Curated by ChEMBL
| Assay Description
Displacement of [3H]-8-OH-DPAT from serotonin 5-HT1A receptor in Sprague-Dawley rat brain cortex homogenates incubated for 30 mins by liquid scintill... |
Bioorg Med Chem 25: 5820-5837 (2017)
Article DOI: 10.1016/j.bmc.2017.09.018
BindingDB Entry DOI: 10.7270/Q2VD721F |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50067795

(CHEMBL138855 | N-(6-Amino-2,4-dimethyl-pyridin-3-y...)Show SMILES Cc1cc(N)nc(C)c1CNC(=O)Cn1c(C)cnc(NCCc2ccccc2)c1=O Show InChI InChI=1S/C23H28N6O2/c1-15-11-20(24)28-17(3)19(15)13-26-21(30)14-29-16(2)12-27-22(23(29)31)25-10-9-18-7-5-4-6-8-18/h4-8,11-12H,9-10,13-14H2,1-3H3,(H2,24,28)(H,25,27)(H,26,30) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against thrombin (IIa) was determined |
J Med Chem 41: 4466-74 (1998)
Article DOI: 10.1021/jm980368v
BindingDB Entry DOI: 10.7270/Q2KK99X0 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-7
(Rattus norvegicus (Rat)) | BDBM50319940
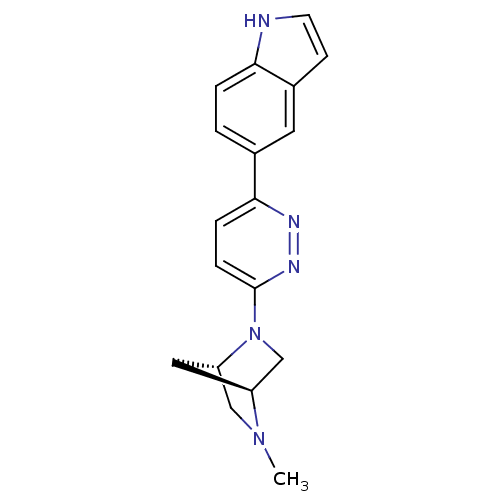
(5-(6-((1S,4S)-5-methyl-2,5-diazabicyclo[2.2.1]hept...)Show SMILES CN1C[C@@H]2C[C@H]1CN2c1ccc(nn1)-c1ccc2[nH]ccc2c1 |r| Show InChI InChI=1S/C18H19N5/c1-22-10-15-9-14(22)11-23(15)18-5-4-17(20-21-18)12-2-3-16-13(8-12)6-7-19-16/h2-8,14-15,19H,9-11H2,1H3/t14-,15-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Park
Curated by ChEMBL
| Assay Description
Displacement of [3H]A585539 from alpha7 nACHR in rat brain homogenate |
Bioorg Med Chem Lett 20: 3636-9 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.105
BindingDB Entry DOI: 10.7270/Q2MC905N |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 2
(Homo sapiens (Human)) | BDBM50231578

(CHEMBL3349022)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O |r| Show InChI InChI=1S/C190H287N55O57/c1-16-94(9)149(180(296)235-129(81-141(194)255)169(285)227-124(74-93(7)8)172(288)240-150(95(10)17-2)181(297)241-151(100(15)248)182(298)222-116(32-23-67-209-190(203)204)156(272)221-118(57-60-140(193)254)161(277)219-114(30-21-65-207-188(199)200)157(273)224-121(152(196)268)76-102-39-49-108(250)50-40-102)239-173(289)127(79-105-45-55-111(253)56-46-105)230-168(284)128(80-106-86-205-90-211-106)231-159(275)115(31-22-66-208-189(201)202)220-165(281)123(73-92(5)6)225-155(271)97(12)213-174(290)134(88-246)237-167(283)126(78-104-43-53-110(252)54-44-104)229-166(282)125(77-103-41-51-109(251)52-42-103)228-158(274)113(29-20-64-206-187(197)198)217-153(269)96(11)212-163(279)122(72-91(3)4)226-170(286)131(84-147(264)265)233-162(278)119(59-62-145(260)261)218-154(270)98(13)214-177(293)137-34-25-68-242(137)183(299)99(14)215-164(280)130(83-146(262)263)232-160(276)117(58-61-144(258)259)216-143(257)87-210-176(292)136-33-24-70-244(136)186(302)133(82-142(195)256)236-171(287)132(85-148(266)267)234-178(294)139-36-27-71-245(139)185(301)120(28-18-19-63-191)223-175(291)135(89-247)238-179(295)138-35-26-69-243(138)184(300)112(192)75-101-37-47-107(249)48-38-101/h37-56,86,90-100,112-139,149-151,246-253H,16-36,57-85,87-89,191-192H2,1-15H3,(H2,193,254)(H2,194,255)(H2,195,256)(H2,196,268)(H,205,211)(H,210,292)(H,212,279)(H,213,290)(H,214,293)(H,215,280)(H,216,257)(H,217,269)(H,218,270)(H,219,277)(H,220,281)(H,221,272)(H,222,298)(H,223,291)(H,224,273)(H,225,271)(H,226,286)(H,227,285)(H,228,274)(H,229,282)(H,230,284)(H,231,275)(H,232,276)(H,233,278)(H,234,294)(H,235,296)(H,236,287)(H,237,283)(H,238,295)(H,239,289)(H,240,288)(H,241,297)(H,258,259)(H,260,261)(H,262,263)(H,264,265)(H,266,267)(H4,197,198,206)(H4,199,200,207)(H4,201,202,208)(H4,203,204,209)/t94-,95-,96-,97-,98-,99-,100+,112-,113-,114-,115-,116-,117-,118-,119-,120+,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,139-,149-,150-,151-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Salk Institute
Curated by ChEMBL
| Assay Description
Affinity against Neuropeptide Y2 receptor on SK-N-BE2 human neuroblastoma cells |
J Med Chem 36: 3802-8 (1994)
BindingDB Entry DOI: 10.7270/Q28K784Q |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50067796
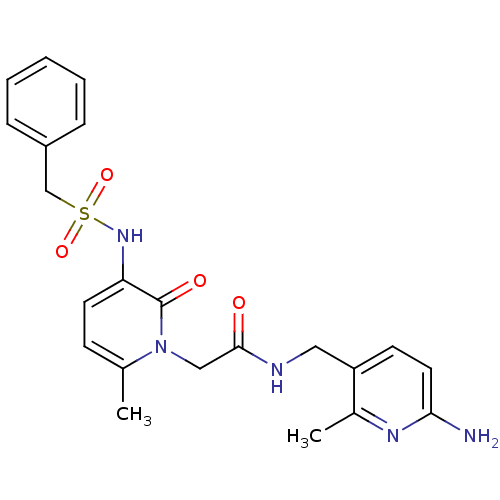
(CHEMBL11157 | L-374087 | N-((6-amino-2-methylpyrid...)Show SMILES Cc1nc(N)ccc1CNC(=O)Cn1c(C)ccc(NS(=O)(=O)Cc2ccccc2)c1=O Show InChI InChI=1S/C22H25N5O4S/c1-15-8-10-19(26-32(30,31)14-17-6-4-3-5-7-17)22(29)27(15)13-21(28)24-12-18-9-11-20(23)25-16(18)2/h3-11,26H,12-14H2,1-2H3,(H2,23,25)(H,24,28) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards thrombin was evaluated |
Bioorg Med Chem Lett 8: 817-22 (1999)
BindingDB Entry DOI: 10.7270/Q2JH3K9Z |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50069190

(CHEMBL287614 | N-(1-Carbamimidoyl-piperidin-4-ylme...)Show SMILES Cc1ccc(NS(=O)(=O)Cc2ccccc2)c(=O)n1CC(=O)NCC1CCN(CC1)C(N)=N Show InChI InChI=1S/C22H30N6O4S/c1-16-7-8-19(26-33(31,32)15-18-5-3-2-4-6-18)21(30)28(16)14-20(29)25-13-17-9-11-27(12-10-17)22(23)24/h2-8,17,26H,9-15H2,1H3,(H3,23,24)(H,25,29) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards thrombin was evaluated |
Bioorg Med Chem Lett 8: 817-22 (1999)
BindingDB Entry DOI: 10.7270/Q2JH3K9Z |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50067796

(CHEMBL11157 | L-374087 | N-((6-amino-2-methylpyrid...)Show SMILES Cc1nc(N)ccc1CNC(=O)Cn1c(C)ccc(NS(=O)(=O)Cc2ccccc2)c1=O Show InChI InChI=1S/C22H25N5O4S/c1-15-8-10-19(26-32(30,31)14-17-6-4-3-5-7-17)22(29)27(15)13-21(28)24-12-18-9-11-20(23)25-16(18)2/h3-11,26H,12-14H2,1-2H3,(H2,23,25)(H,24,28) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against thrombin (IIa) was determined |
J Med Chem 41: 4466-74 (1998)
Article DOI: 10.1021/jm980368v
BindingDB Entry DOI: 10.7270/Q2KK99X0 |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 2
(Homo sapiens (Human)) | BDBM50231578

(CHEMBL3349022)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O |r| Show InChI InChI=1S/C190H287N55O57/c1-16-94(9)149(180(296)235-129(81-141(194)255)169(285)227-124(74-93(7)8)172(288)240-150(95(10)17-2)181(297)241-151(100(15)248)182(298)222-116(32-23-67-209-190(203)204)156(272)221-118(57-60-140(193)254)161(277)219-114(30-21-65-207-188(199)200)157(273)224-121(152(196)268)76-102-39-49-108(250)50-40-102)239-173(289)127(79-105-45-55-111(253)56-46-105)230-168(284)128(80-106-86-205-90-211-106)231-159(275)115(31-22-66-208-189(201)202)220-165(281)123(73-92(5)6)225-155(271)97(12)213-174(290)134(88-246)237-167(283)126(78-104-43-53-110(252)54-44-104)229-166(282)125(77-103-41-51-109(251)52-42-103)228-158(274)113(29-20-64-206-187(197)198)217-153(269)96(11)212-163(279)122(72-91(3)4)226-170(286)131(84-147(264)265)233-162(278)119(59-62-145(260)261)218-154(270)98(13)214-177(293)137-34-25-68-242(137)183(299)99(14)215-164(280)130(83-146(262)263)232-160(276)117(58-61-144(258)259)216-143(257)87-210-176(292)136-33-24-70-244(136)186(302)133(82-142(195)256)236-171(287)132(85-148(266)267)234-178(294)139-36-27-71-245(139)185(301)120(28-18-19-63-191)223-175(291)135(89-247)238-179(295)138-35-26-69-243(138)184(300)112(192)75-101-37-47-107(249)48-38-101/h37-56,86,90-100,112-139,149-151,246-253H,16-36,57-85,87-89,191-192H2,1-15H3,(H2,193,254)(H2,194,255)(H2,195,256)(H2,196,268)(H,205,211)(H,210,292)(H,212,279)(H,213,290)(H,214,293)(H,215,280)(H,216,257)(H,217,269)(H,218,270)(H,219,277)(H,220,281)(H,221,272)(H,222,298)(H,223,291)(H,224,273)(H,225,271)(H,226,286)(H,227,285)(H,228,274)(H,229,282)(H,230,284)(H,231,275)(H,232,276)(H,233,278)(H,234,294)(H,235,296)(H,236,287)(H,237,283)(H,238,295)(H,239,289)(H,240,288)(H,241,297)(H,258,259)(H,260,261)(H,262,263)(H,264,265)(H,266,267)(H4,197,198,206)(H4,199,200,207)(H4,201,202,208)(H4,203,204,209)/t94-,95-,96-,97-,98-,99-,100+,112-,113-,114-,115-,116-,117-,118-,119-,120+,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,139-,149-,150-,151-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Salk Institute
Curated by ChEMBL
| Assay Description
Affinity against Neuropeptide Y2 receptor on SK-N-BE2 human neuroblastoma cells |
J Med Chem 36: 3802-8 (1994)
BindingDB Entry DOI: 10.7270/Q28K784Q |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 2
(Homo sapiens (Human)) | BDBM50231578

(CHEMBL3349022)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O |r| Show InChI InChI=1S/C190H287N55O57/c1-16-94(9)149(180(296)235-129(81-141(194)255)169(285)227-124(74-93(7)8)172(288)240-150(95(10)17-2)181(297)241-151(100(15)248)182(298)222-116(32-23-67-209-190(203)204)156(272)221-118(57-60-140(193)254)161(277)219-114(30-21-65-207-188(199)200)157(273)224-121(152(196)268)76-102-39-49-108(250)50-40-102)239-173(289)127(79-105-45-55-111(253)56-46-105)230-168(284)128(80-106-86-205-90-211-106)231-159(275)115(31-22-66-208-189(201)202)220-165(281)123(73-92(5)6)225-155(271)97(12)213-174(290)134(88-246)237-167(283)126(78-104-43-53-110(252)54-44-104)229-166(282)125(77-103-41-51-109(251)52-42-103)228-158(274)113(29-20-64-206-187(197)198)217-153(269)96(11)212-163(279)122(72-91(3)4)226-170(286)131(84-147(264)265)233-162(278)119(59-62-145(260)261)218-154(270)98(13)214-177(293)137-34-25-68-242(137)183(299)99(14)215-164(280)130(83-146(262)263)232-160(276)117(58-61-144(258)259)216-143(257)87-210-176(292)136-33-24-70-244(136)186(302)133(82-142(195)256)236-171(287)132(85-148(266)267)234-178(294)139-36-27-71-245(139)185(301)120(28-18-19-63-191)223-175(291)135(89-247)238-179(295)138-35-26-69-243(138)184(300)112(192)75-101-37-47-107(249)48-38-101/h37-56,86,90-100,112-139,149-151,246-253H,16-36,57-85,87-89,191-192H2,1-15H3,(H2,193,254)(H2,194,255)(H2,195,256)(H2,196,268)(H,205,211)(H,210,292)(H,212,279)(H,213,290)(H,214,293)(H,215,280)(H,216,257)(H,217,269)(H,218,270)(H,219,277)(H,220,281)(H,221,272)(H,222,298)(H,223,291)(H,224,273)(H,225,271)(H,226,286)(H,227,285)(H,228,274)(H,229,282)(H,230,284)(H,231,275)(H,232,276)(H,233,278)(H,234,294)(H,235,296)(H,236,287)(H,237,283)(H,238,295)(H,239,289)(H,240,288)(H,241,297)(H,258,259)(H,260,261)(H,262,263)(H,264,265)(H,266,267)(H4,197,198,206)(H4,199,200,207)(H4,201,202,208)(H4,203,204,209)/t94-,95-,96-,97-,98-,99-,100+,112-,113-,114-,115-,116-,117-,118-,119-,120+,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,139-,149-,150-,151-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Salk Institute
Curated by ChEMBL
| Assay Description
Affinity against Neuropeptide Y2 receptor on SK-N-BE2 human neuroblastoma cells |
J Med Chem 36: 3802-8 (1994)
BindingDB Entry DOI: 10.7270/Q28K784Q |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50067798

(CHEMBL336438 | N-(6-Amino-2-methyl-pyridin-3-ylmet...)Show SMILES C[C@H](Cc1ccccc1)Nc1ncc(C)n(CC(=O)NCc2ccc(N)nc2C)c1=O Show InChI InChI=1S/C23H28N6O2/c1-15(11-18-7-5-4-6-8-18)27-22-23(31)29(16(2)12-26-22)14-21(30)25-13-19-9-10-20(24)28-17(19)3/h4-10,12,15H,11,13-14H2,1-3H3,(H2,24,28)(H,25,30)(H,26,27)/t15-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.520 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against thrombin (IIa) was determined |
J Med Chem 41: 4466-74 (1998)
Article DOI: 10.1021/jm980368v
BindingDB Entry DOI: 10.7270/Q2KK99X0 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50319962
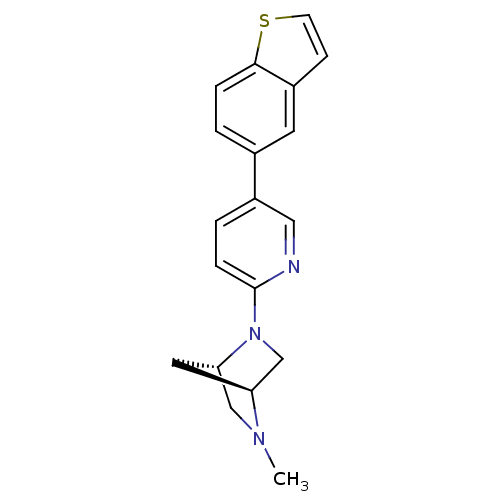
((1S,4S)-2-(5-(benzo[b]thiophen-5-yl)pyridin-2-yl)-...)Show SMILES CN1C[C@@H]2C[C@H]1CN2c1ccc(cn1)-c1ccc2sccc2c1 |r| Show InChI InChI=1S/C19H19N3S/c1-21-11-17-9-16(21)12-22(17)19-5-3-15(10-20-19)13-2-4-18-14(8-13)6-7-23-18/h2-8,10,16-17H,9,11-12H2,1H3/t16-,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Park
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetidile from human ERG by whole-cell patch clamp |
Bioorg Med Chem Lett 20: 3636-9 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.105
BindingDB Entry DOI: 10.7270/Q2MC905N |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50319955
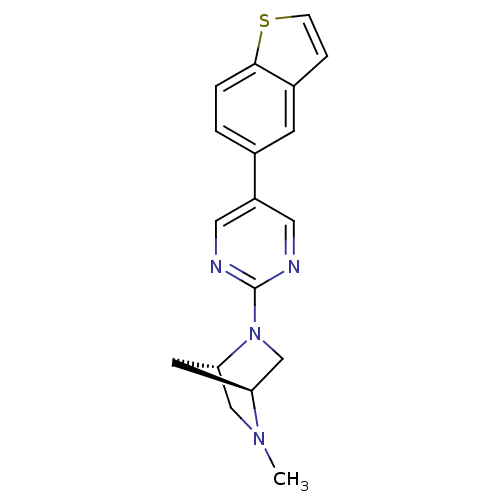
((1S,4S)-2-(5-(benzo[b]thiophen-5-yl)pyrimidin-2-yl...)Show SMILES CN1C[C@@H]2C[C@H]1CN2c1ncc(cn1)-c1ccc2sccc2c1 |r| Show InChI InChI=1S/C18H18N4S/c1-21-10-16-7-15(21)11-22(16)18-19-8-14(9-20-18)12-2-3-17-13(6-12)4-5-23-17/h2-6,8-9,15-16H,7,10-11H2,1H3/t15-,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.560 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Park
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetidile from human ERG by whole-cell patch clamp |
Bioorg Med Chem Lett 20: 3636-9 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.105
BindingDB Entry DOI: 10.7270/Q2MC905N |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 2
(Homo sapiens (Human)) | BDBM50231578

(CHEMBL3349022)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O |r| Show InChI InChI=1S/C190H287N55O57/c1-16-94(9)149(180(296)235-129(81-141(194)255)169(285)227-124(74-93(7)8)172(288)240-150(95(10)17-2)181(297)241-151(100(15)248)182(298)222-116(32-23-67-209-190(203)204)156(272)221-118(57-60-140(193)254)161(277)219-114(30-21-65-207-188(199)200)157(273)224-121(152(196)268)76-102-39-49-108(250)50-40-102)239-173(289)127(79-105-45-55-111(253)56-46-105)230-168(284)128(80-106-86-205-90-211-106)231-159(275)115(31-22-66-208-189(201)202)220-165(281)123(73-92(5)6)225-155(271)97(12)213-174(290)134(88-246)237-167(283)126(78-104-43-53-110(252)54-44-104)229-166(282)125(77-103-41-51-109(251)52-42-103)228-158(274)113(29-20-64-206-187(197)198)217-153(269)96(11)212-163(279)122(72-91(3)4)226-170(286)131(84-147(264)265)233-162(278)119(59-62-145(260)261)218-154(270)98(13)214-177(293)137-34-25-68-242(137)183(299)99(14)215-164(280)130(83-146(262)263)232-160(276)117(58-61-144(258)259)216-143(257)87-210-176(292)136-33-24-70-244(136)186(302)133(82-142(195)256)236-171(287)132(85-148(266)267)234-178(294)139-36-27-71-245(139)185(301)120(28-18-19-63-191)223-175(291)135(89-247)238-179(295)138-35-26-69-243(138)184(300)112(192)75-101-37-47-107(249)48-38-101/h37-56,86,90-100,112-139,149-151,246-253H,16-36,57-85,87-89,191-192H2,1-15H3,(H2,193,254)(H2,194,255)(H2,195,256)(H2,196,268)(H,205,211)(H,210,292)(H,212,279)(H,213,290)(H,214,293)(H,215,280)(H,216,257)(H,217,269)(H,218,270)(H,219,277)(H,220,281)(H,221,272)(H,222,298)(H,223,291)(H,224,273)(H,225,271)(H,226,286)(H,227,285)(H,228,274)(H,229,282)(H,230,284)(H,231,275)(H,232,276)(H,233,278)(H,234,294)(H,235,296)(H,236,287)(H,237,283)(H,238,295)(H,239,289)(H,240,288)(H,241,297)(H,258,259)(H,260,261)(H,262,263)(H,264,265)(H,266,267)(H4,197,198,206)(H4,199,200,207)(H4,201,202,208)(H4,203,204,209)/t94-,95-,96-,97-,98-,99-,100+,112-,113-,114-,115-,116-,117-,118-,119-,120+,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,139-,149-,150-,151-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Salk Institute
Curated by ChEMBL
| Assay Description
Affinity against Neuropeptide Y2 receptor on SK-N-BE2 human neuroblastoma cells |
J Med Chem 36: 3802-8 (1994)
BindingDB Entry DOI: 10.7270/Q28K784Q |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 2
(Homo sapiens (Human)) | BDBM50231578

(CHEMBL3349022)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O |r| Show InChI InChI=1S/C190H287N55O57/c1-16-94(9)149(180(296)235-129(81-141(194)255)169(285)227-124(74-93(7)8)172(288)240-150(95(10)17-2)181(297)241-151(100(15)248)182(298)222-116(32-23-67-209-190(203)204)156(272)221-118(57-60-140(193)254)161(277)219-114(30-21-65-207-188(199)200)157(273)224-121(152(196)268)76-102-39-49-108(250)50-40-102)239-173(289)127(79-105-45-55-111(253)56-46-105)230-168(284)128(80-106-86-205-90-211-106)231-159(275)115(31-22-66-208-189(201)202)220-165(281)123(73-92(5)6)225-155(271)97(12)213-174(290)134(88-246)237-167(283)126(78-104-43-53-110(252)54-44-104)229-166(282)125(77-103-41-51-109(251)52-42-103)228-158(274)113(29-20-64-206-187(197)198)217-153(269)96(11)212-163(279)122(72-91(3)4)226-170(286)131(84-147(264)265)233-162(278)119(59-62-145(260)261)218-154(270)98(13)214-177(293)137-34-25-68-242(137)183(299)99(14)215-164(280)130(83-146(262)263)232-160(276)117(58-61-144(258)259)216-143(257)87-210-176(292)136-33-24-70-244(136)186(302)133(82-142(195)256)236-171(287)132(85-148(266)267)234-178(294)139-36-27-71-245(139)185(301)120(28-18-19-63-191)223-175(291)135(89-247)238-179(295)138-35-26-69-243(138)184(300)112(192)75-101-37-47-107(249)48-38-101/h37-56,86,90-100,112-139,149-151,246-253H,16-36,57-85,87-89,191-192H2,1-15H3,(H2,193,254)(H2,194,255)(H2,195,256)(H2,196,268)(H,205,211)(H,210,292)(H,212,279)(H,213,290)(H,214,293)(H,215,280)(H,216,257)(H,217,269)(H,218,270)(H,219,277)(H,220,281)(H,221,272)(H,222,298)(H,223,291)(H,224,273)(H,225,271)(H,226,286)(H,227,285)(H,228,274)(H,229,282)(H,230,284)(H,231,275)(H,232,276)(H,233,278)(H,234,294)(H,235,296)(H,236,287)(H,237,283)(H,238,295)(H,239,289)(H,240,288)(H,241,297)(H,258,259)(H,260,261)(H,262,263)(H,264,265)(H,266,267)(H4,197,198,206)(H4,199,200,207)(H4,201,202,208)(H4,203,204,209)/t94-,95-,96-,97-,98-,99-,100+,112-,113-,114-,115-,116-,117-,118-,119-,120+,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,139-,149-,150-,151-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Salk Institute
Curated by ChEMBL
| Assay Description
Affinity against Neuropeptide Y2 receptor on SK-N-BE2 human neuroblastoma cells |
J Med Chem 36: 3802-8 (1994)
BindingDB Entry DOI: 10.7270/Q28K784Q |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 2
(Homo sapiens (Human)) | BDBM50231576
(CHEMBL2311081)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O |r| Show InChI InChI=1S/C190H287N55O57/c1-16-94(9)149(180(296)235-129(81-141(194)255)169(285)227-124(74-93(7)8)172(288)240-150(95(10)17-2)181(297)241-151(100(15)248)182(298)222-116(32-23-67-209-190(203)204)156(272)221-118(57-60-140(193)254)161(277)219-114(30-21-65-207-188(199)200)157(273)224-121(152(196)268)76-102-39-49-108(250)50-40-102)239-173(289)127(79-105-45-55-111(253)56-46-105)230-168(284)128(80-106-86-205-90-211-106)231-159(275)115(31-22-66-208-189(201)202)220-165(281)123(73-92(5)6)225-155(271)97(12)213-174(290)134(88-246)237-167(283)126(78-104-43-53-110(252)54-44-104)229-166(282)125(77-103-41-51-109(251)52-42-103)228-158(274)113(29-20-64-206-187(197)198)217-153(269)96(11)212-163(279)122(72-91(3)4)226-170(286)131(84-147(264)265)233-162(278)119(59-62-145(260)261)218-154(270)98(13)214-177(293)137-34-25-68-242(137)183(299)99(14)215-164(280)130(83-146(262)263)232-160(276)117(58-61-144(258)259)216-143(257)87-210-176(292)136-33-24-70-244(136)186(302)133(82-142(195)256)236-171(287)132(85-148(266)267)234-178(294)139-36-27-71-245(139)185(301)120(28-18-19-63-191)223-175(291)135(89-247)238-179(295)138-35-26-69-243(138)184(300)112(192)75-101-37-47-107(249)48-38-101/h37-56,86,90-100,112-139,149-151,246-253H,16-36,57-85,87-89,191-192H2,1-15H3,(H2,193,254)(H2,194,255)(H2,195,256)(H2,196,268)(H,205,211)(H,210,292)(H,212,279)(H,213,290)(H,214,293)(H,215,280)(H,216,257)(H,217,269)(H,218,270)(H,219,277)(H,220,281)(H,221,272)(H,222,298)(H,223,291)(H,224,273)(H,225,271)(H,226,286)(H,227,285)(H,228,274)(H,229,282)(H,230,284)(H,231,275)(H,232,276)(H,233,278)(H,234,294)(H,235,296)(H,236,287)(H,237,283)(H,238,295)(H,239,289)(H,240,288)(H,241,297)(H,258,259)(H,260,261)(H,262,263)(H,264,265)(H,266,267)(H4,197,198,206)(H4,199,200,207)(H4,201,202,208)(H4,203,204,209)/t94-,95-,96-,97-,98-,99-,100+,112-,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138+,139-,149-,150-,151-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Salk Institute
Curated by ChEMBL
| Assay Description
Affinity against Neuropeptide Y2 receptor on SK-N-BE2 human neuroblastoma cells |
J Med Chem 36: 3802-8 (1994)
BindingDB Entry DOI: 10.7270/Q28K784Q |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM50278880
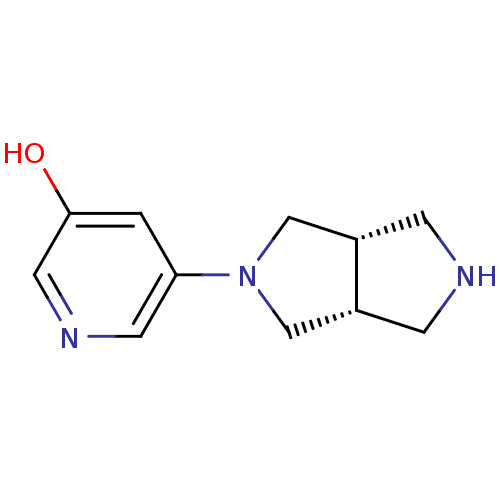
(CHEMBL502841 | cis-2-(5-Hydroxy-3-pyridinyl)octahy...)Show InChI InChI=1S/C11H15N3O/c15-11-1-10(4-13-5-11)14-6-8-2-12-3-9(8)7-14/h1,4-5,8-9,12,15H,2-3,6-7H2/t8-,9+ | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from alpha4beta2 nicotinic acetylcholine receptor in rat brain minus cerebellum membrane |
J Med Chem 52: 4126-41 (2009)
Article DOI: 10.1021/jm900249k
BindingDB Entry DOI: 10.7270/Q2GT5P3R |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2C
(Rattus norvegicus (Rat)) | BDBM50451040
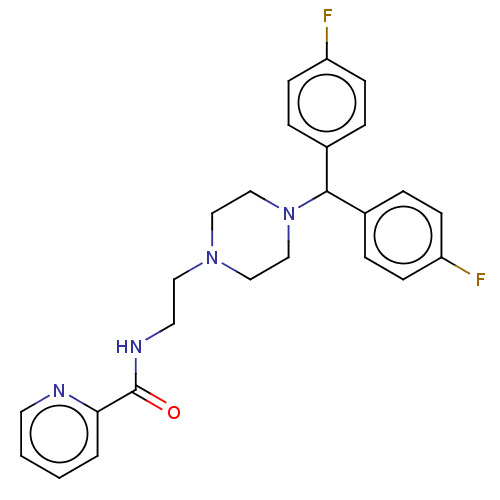
(CHEMBL4218771)Show SMILES Cl.Fc1ccc(cc1)C(N1CCN(CCNC(=O)c2ccccn2)CC1)c1ccc(F)cc1 Show InChI InChI=1S/C25H26F2N4O/c26-21-8-4-19(5-9-21)24(20-6-10-22(27)11-7-20)31-17-15-30(16-18-31)14-13-29-25(32)23-3-1-2-12-28-23/h1-12,24H,13-18H2,(H,29,32) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dipartimento di Farmacia Universit£ di Napoli "Federico II" Via D. Montesano
Curated by ChEMBL
| Assay Description
Displacement of [3H]mesulergine from serotonin 5-HT2C receptor in Sprague-Dawley rat brain cortex homogenates incubated for 15 mins by liquid scintil... |
Bioorg Med Chem 25: 5820-5837 (2017)
Article DOI: 10.1016/j.bmc.2017.09.018
BindingDB Entry DOI: 10.7270/Q2VD721F |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 2
(Homo sapiens (Human)) | BDBM50042810
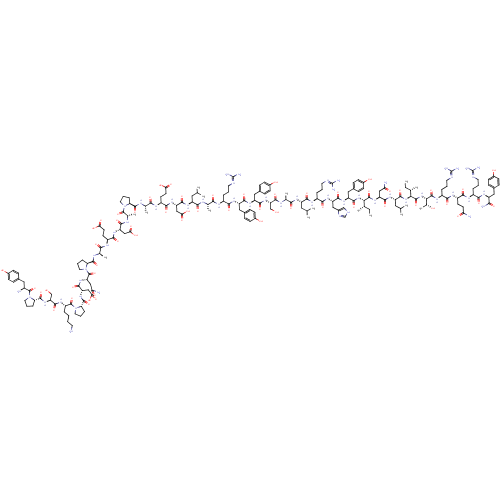
(CHEMBL437863 | Tyr-Pro-Ser-Lys-Pro-Asp-Asn-Pro-D-A...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1c[nH]cn1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](C)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O Show InChI InChI=1S/C191H289N55O57/c1-17-94(9)149(181(297)236-130(82-142(195)256)170(286)228-125(75-93(7)8)173(289)241-150(95(10)18-2)182(298)242-151(101(16)249)183(299)223-117(33-24-68-210-191(204)205)157(273)222-118(58-61-141(194)255)161(277)220-115(31-22-66-208-189(200)201)158(274)225-122(152(197)268)77-103-40-50-109(251)51-41-103)240-174(290)128(80-106-46-56-112(254)57-47-106)231-169(285)129(81-107-87-206-90-211-107)232-160(276)116(32-23-67-209-190(202)203)221-166(282)124(74-92(5)6)226-156(272)97(12)213-175(291)135(88-247)238-168(284)127(79-105-44-54-111(253)55-45-105)230-167(283)126(78-104-42-52-110(252)53-43-104)229-159(275)114(30-21-65-207-188(198)199)217-153(269)96(11)212-164(280)123(73-91(3)4)227-171(287)132(85-147(264)265)234-163(279)120(60-63-145(260)261)219-154(270)98(13)214-177(293)137-34-25-69-243(137)184(300)100(15)216-165(281)131(84-146(262)263)233-162(278)119(59-62-144(258)259)218-155(271)99(14)215-178(294)138-35-27-72-246(138)187(303)134(83-143(196)257)237-172(288)133(86-148(266)267)235-179(295)140-37-28-71-245(140)186(302)121(29-19-20-64-192)224-176(292)136(89-248)239-180(296)139-36-26-70-244(139)185(301)113(193)76-102-38-48-108(250)49-39-102/h38-57,87,90-101,113-140,149-151,247-254H,17-37,58-86,88-89,192-193H2,1-16H3,(H2,194,255)(H2,195,256)(H2,196,257)(H2,197,268)(H,206,211)(H,212,280)(H,213,291)(H,214,293)(H,215,294)(H,216,281)(H,217,269)(H,218,271)(H,219,270)(H,220,277)(H,221,282)(H,222,273)(H,223,299)(H,224,292)(H,225,274)(H,226,272)(H,227,287)(H,228,286)(H,229,275)(H,230,283)(H,231,285)(H,232,276)(H,233,278)(H,234,279)(H,235,295)(H,236,297)(H,237,288)(H,238,284)(H,239,296)(H,240,290)(H,241,289)(H,242,298)(H,258,259)(H,260,261)(H,262,263)(H,264,265)(H,266,267)(H4,198,199,207)(H4,200,201,208)(H4,202,203,209)(H4,204,205,210)/t94-,95-,96-,97-,98-,99+,100-,101+,113-,114-,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,139-,140-,149-,150-,151-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Salk Institute
Curated by ChEMBL
| Assay Description
Affinity against Neuropeptide Y2 receptor on SK-N-BE2 human neuroblastoma cells |
J Med Chem 36: 3802-8 (1994)
BindingDB Entry DOI: 10.7270/Q28K784Q |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data