 Found 239 hits with Last Name = 'conklyn' and Initial = 'm'
Found 239 hits with Last Name = 'conklyn' and Initial = 'm' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Leukotriene B4 receptor 1
(Homo sapiens (Human)) | BDBM50286748
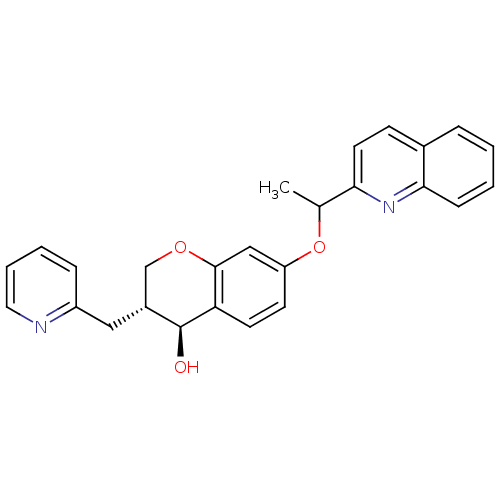
((3R,4S)-3-Pyridin-2-ylmethyl-7-(1-quinolin-2-yl-et...)Show SMILES CC(Oc1ccc2[C@@H](O)[C@H](Cc3ccccn3)COc2c1)c1ccc2ccccc2n1 Show InChI InChI=1S/C26H24N2O3/c1-17(23-12-9-18-6-2-3-8-24(18)28-23)31-21-10-11-22-25(15-21)30-16-19(26(22)29)14-20-7-4-5-13-27-20/h2-13,15,17,19,26,29H,14,16H2,1H3/t17?,19-,26+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against human neutrophil leukotriene B4 (LTB4) |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1
(Homo sapiens (Human)) | BDBM50001610

(7-[3-(4-Acetyl-3-methoxy-2-propyl-phenoxy)-propoxy...)Show SMILES CCCc1c(OCCCOc2ccc3CCC(Oc3c2CCC)C(O)=O)ccc(C(C)=O)c1OC Show InChI InChI=1S/C28H36O7/c1-5-8-21-23(13-10-19-11-14-25(28(30)31)35-26(19)21)33-16-7-17-34-24-15-12-20(18(3)29)27(32-4)22(24)9-6-2/h10,12-13,15,25H,5-9,11,14,16-17H2,1-4H3,(H,30,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro antagonist activity against leukotriene B4 (LTB4) receptor in [3H]-LTB4 human neutrophil receptor binding assay |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1
(Homo sapiens (Human)) | BDBM50286747
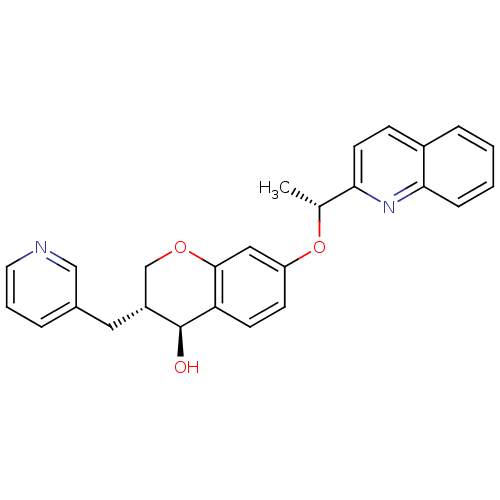
((3R,4S)-3-Pyridin-3-ylmethyl-7-((R)-1-quinolin-2-y...)Show SMILES C[C@@H](Oc1ccc2[C@@H](O)[C@H](Cc3cccnc3)COc2c1)c1ccc2ccccc2n1 Show InChI InChI=1S/C26H24N2O3/c1-17(23-11-8-19-6-2-3-7-24(19)28-23)31-21-9-10-22-25(14-21)30-16-20(26(22)29)13-18-5-4-12-27-15-18/h2-12,14-15,17,20,26,29H,13,16H2,1H3/t17-,20-,26+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against human neutrophil leukotriene B4 (LTB4) |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50286750
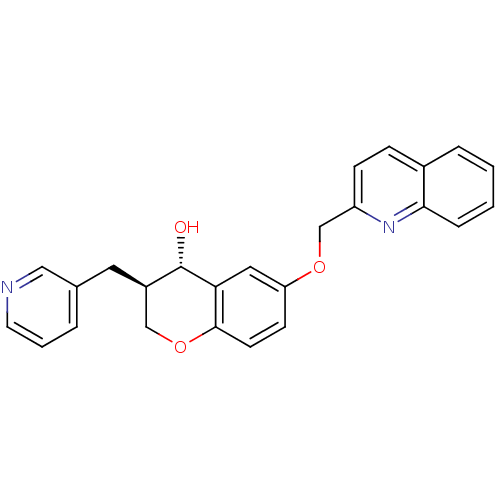
((3R,4S)-3-Pyridin-3-ylmethyl-6-(quinolin-2-ylmetho...)Show SMILES O[C@H]1[C@H](Cc2cccnc2)COc2ccc(OCc3ccc4ccccc4n3)cc12 Show InChI InChI=1S/C25H22N2O3/c28-25-19(12-17-4-3-11-26-14-17)15-30-24-10-9-21(13-22(24)25)29-16-20-8-7-18-5-1-2-6-23(18)27-20/h1-11,13-14,19,25,28H,12,15-16H2/t19-,25+/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against leukotriene D4 receptor |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50286746
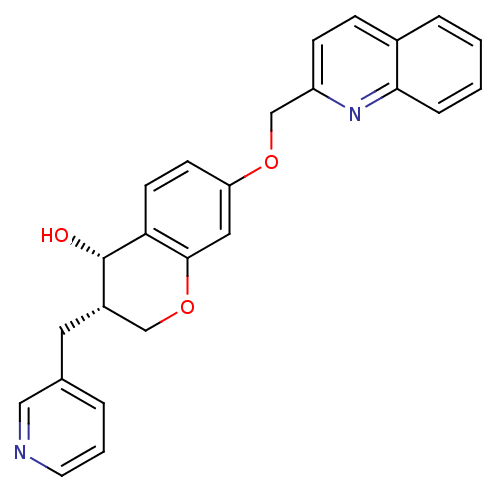
((3S,4S)-3-Pyridin-3-ylmethyl-7-(quinolin-2-ylmetho...)Show SMILES O[C@H]1[C@@H](Cc2cccnc2)COc2cc(OCc3ccc4ccccc4n3)ccc12 Show InChI InChI=1S/C25H22N2O3/c28-25-19(12-17-4-3-11-26-14-17)15-30-24-13-21(9-10-22(24)25)29-16-20-8-7-18-5-1-2-6-23(18)27-20/h1-11,13-14,19,25,28H,12,15-16H2/t19-,25-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against leukotriene D4 receptor |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50284680
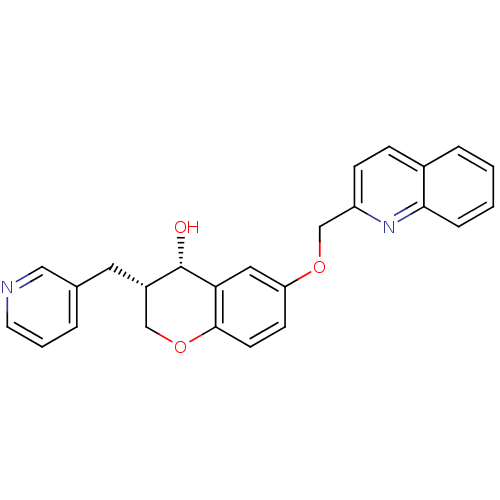
((3S,4S)-3-Pyridin-3-ylmethyl-6-(quinolin-2-ylmetho...)Show SMILES O[C@H]1[C@@H](Cc2cccnc2)COc2ccc(OCc3ccc4ccccc4n3)cc12 Show InChI InChI=1S/C25H22N2O3/c28-25-19(12-17-4-3-11-26-14-17)15-30-24-10-9-21(13-22(24)25)29-16-20-8-7-18-5-1-2-6-23(18)27-20/h1-11,13-14,19,25,28H,12,15-16H2/t19-,25-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against human leukotriene D4 (LTD4) |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1
(Homo sapiens (Human)) | BDBM50286743
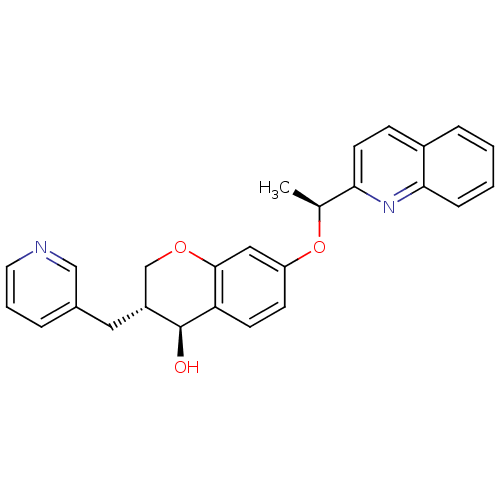
((3R,4S)-3-Pyridin-3-ylmethyl-7-((S)-1-quinolin-2-y...)Show SMILES C[C@H](Oc1ccc2[C@@H](O)[C@H](Cc3cccnc3)COc2c1)c1ccc2ccccc2n1 Show InChI InChI=1S/C26H24N2O3/c1-17(23-11-8-19-6-2-3-7-24(19)28-23)31-21-9-10-22-25(14-21)30-16-20(26(22)29)13-18-5-4-12-27-15-18/h2-12,14-15,17,20,26,29H,13,16H2,1H3/t17-,20+,26-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against human neutrophil leukotriene B4 (LTB4) |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1
(Homo sapiens (Human)) | BDBM50286745

((3S,4S)-3-Pyridin-2-ylmethyl-7-(1-quinolin-2-yl-et...)Show SMILES CC(Oc1ccc2[C@@H](O)[C@@H](Cc3ccccn3)COc2c1)c1ccc2ccccc2n1 Show InChI InChI=1S/C26H24N2O3/c1-17(23-12-9-18-6-2-3-8-24(18)28-23)31-21-10-11-22-25(15-21)30-16-19(26(22)29)14-20-7-4-5-13-27-20/h2-13,15,17,19,26,29H,14,16H2,1H3/t17?,19-,26-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 3.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against human neutrophil leukotriene B4 (LTB4) |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50286744
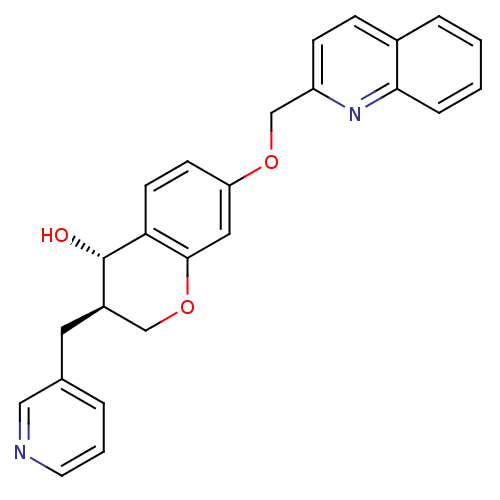
((3R,4S)-3-Pyridin-3-ylmethyl-7-(quinolin-2-ylmetho...)Show SMILES O[C@H]1[C@H](Cc2cccnc2)COc2cc(OCc3ccc4ccccc4n3)ccc12 Show InChI InChI=1S/C25H22N2O3/c28-25-19(12-17-4-3-11-26-14-17)15-30-24-13-21(9-10-22(24)25)29-16-20-8-7-18-5-1-2-6-23(18)27-20/h1-11,13-14,19,25,28H,12,15-16H2/t19-,25+/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 4.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against leukotriene D4 receptor |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1
(Homo sapiens (Human)) | BDBM50286749
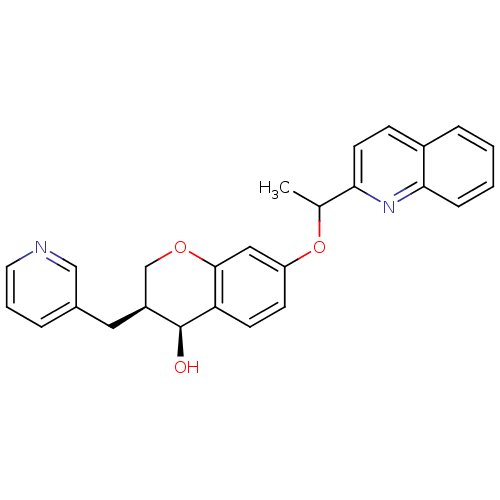
((3S,4S)-3-Pyridin-3-ylmethyl-7-(1-quinolin-2-yl-et...)Show SMILES CC(Oc1ccc2[C@@H](O)[C@@H](Cc3cccnc3)COc2c1)c1ccc2ccccc2n1 Show InChI InChI=1S/C26H24N2O3/c1-17(23-11-8-19-6-2-3-7-24(19)28-23)31-21-9-10-22-25(14-21)30-16-20(26(22)29)13-18-5-4-12-27-15-18/h2-12,14-15,17,20,26,29H,13,16H2,1H3/t17?,20-,26-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 5.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against human neutrophil leukotriene B4 (LTB4) |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1
(Homo sapiens (Human)) | BDBM50286750

((3R,4S)-3-Pyridin-3-ylmethyl-6-(quinolin-2-ylmetho...)Show SMILES O[C@H]1[C@H](Cc2cccnc2)COc2ccc(OCc3ccc4ccccc4n3)cc12 Show InChI InChI=1S/C25H22N2O3/c28-25-19(12-17-4-3-11-26-14-17)15-30-24-10-9-21(13-22(24)25)29-16-20-8-7-18-5-1-2-6-23(18)27-20/h1-11,13-14,19,25,28H,12,15-16H2/t19-,25+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 1.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against human neutrophil leukotriene B4 (LTB4) |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1
(Homo sapiens (Human)) | BDBM50286746

((3S,4S)-3-Pyridin-3-ylmethyl-7-(quinolin-2-ylmetho...)Show SMILES O[C@H]1[C@@H](Cc2cccnc2)COc2cc(OCc3ccc4ccccc4n3)ccc12 Show InChI InChI=1S/C25H22N2O3/c28-25-19(12-17-4-3-11-26-14-17)15-30-24-13-21(9-10-22(24)25)29-16-20-8-7-18-5-1-2-6-23(18)27-20/h1-11,13-14,19,25,28H,12,15-16H2/t19-,25-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 3.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against human neutrophil leukotriene B4 (LTB4) |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1
(Homo sapiens (Human)) | BDBM50284680

((3S,4S)-3-Pyridin-3-ylmethyl-6-(quinolin-2-ylmetho...)Show SMILES O[C@H]1[C@@H](Cc2cccnc2)COc2ccc(OCc3ccc4ccccc4n3)cc12 Show InChI InChI=1S/C25H22N2O3/c28-25-19(12-17-4-3-11-26-14-17)15-30-24-10-9-21(13-22(24)25)29-16-20-8-7-18-5-1-2-6-23(18)27-20/h1-11,13-14,19,25,28H,12,15-16H2/t19-,25-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 3.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against human neutrophil leukotriene B4 (LTB4) |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1
(Homo sapiens (Human)) | BDBM50286744

((3R,4S)-3-Pyridin-3-ylmethyl-7-(quinolin-2-ylmetho...)Show SMILES O[C@H]1[C@H](Cc2cccnc2)COc2cc(OCc3ccc4ccccc4n3)ccc12 Show InChI InChI=1S/C25H22N2O3/c28-25-19(12-17-4-3-11-26-14-17)15-30-24-13-21(9-10-22(24)25)29-16-20-8-7-18-5-1-2-6-23(18)27-20/h1-11,13-14,19,25,28H,12,15-16H2/t19-,25+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 3.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity was evaluated against leukotriene B4 (LTB4) in guinea pig binding assay |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50286745

((3S,4S)-3-Pyridin-2-ylmethyl-7-(1-quinolin-2-yl-et...)Show SMILES CC(Oc1ccc2[C@@H](O)[C@@H](Cc3ccccn3)COc2c1)c1ccc2ccccc2n1 Show InChI InChI=1S/C26H24N2O3/c1-17(23-12-9-18-6-2-3-8-24(18)28-23)31-21-10-11-22-25(15-21)30-16-19(26(22)29)14-20-7-4-5-13-27-20/h2-13,15,17,19,26,29H,14,16H2,1H3/t17?,19-,26-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against leukotriene D4 receptor |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50286743

((3R,4S)-3-Pyridin-3-ylmethyl-7-((S)-1-quinolin-2-y...)Show SMILES C[C@H](Oc1ccc2[C@@H](O)[C@H](Cc3cccnc3)COc2c1)c1ccc2ccccc2n1 Show InChI InChI=1S/C26H24N2O3/c1-17(23-11-8-19-6-2-3-7-24(19)28-23)31-21-9-10-22-25(14-21)30-16-20(26(22)29)13-18-5-4-12-27-15-18/h2-12,14-15,17,20,26,29H,13,16H2,1H3/t17-,20+,26-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against leukotriene D4 receptor |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50286748

((3R,4S)-3-Pyridin-2-ylmethyl-7-(1-quinolin-2-yl-et...)Show SMILES CC(Oc1ccc2[C@@H](O)[C@H](Cc3ccccn3)COc2c1)c1ccc2ccccc2n1 Show InChI InChI=1S/C26H24N2O3/c1-17(23-12-9-18-6-2-3-8-24(18)28-23)31-21-10-11-22-25(15-21)30-16-19(26(22)29)14-20-7-4-5-13-27-20/h2-13,15,17,19,26,29H,14,16H2,1H3/t17?,19-,26+/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against leukotriene D4 receptor |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50286747

((3R,4S)-3-Pyridin-3-ylmethyl-7-((R)-1-quinolin-2-y...)Show SMILES C[C@@H](Oc1ccc2[C@@H](O)[C@H](Cc3cccnc3)COc2c1)c1ccc2ccccc2n1 Show InChI InChI=1S/C26H24N2O3/c1-17(23-11-8-19-6-2-3-7-24(19)28-23)31-21-9-10-22-25(14-21)30-16-20(26(22)29)13-18-5-4-12-27-15-18/h2-12,14-15,17,20,26,29H,13,16H2,1H3/t17-,20-,26+/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against leukotriene D4 receptor |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50286749

((3S,4S)-3-Pyridin-3-ylmethyl-7-(1-quinolin-2-yl-et...)Show SMILES CC(Oc1ccc2[C@@H](O)[C@@H](Cc3cccnc3)COc2c1)c1ccc2ccccc2n1 Show InChI InChI=1S/C26H24N2O3/c1-17(23-11-8-19-6-2-3-7-24(19)28-23)31-21-9-10-22-25(14-21)30-16-20(26(22)29)13-18-5-4-12-27-15-18/h2-12,14-15,17,20,26,29H,13,16H2,1H3/t17?,20-,26-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against leukotriene D4 receptor |
Bioorg Med Chem Lett 5: 887-892 (1995)
Article DOI: 10.1016/0960-894X(95)00135-G
BindingDB Entry DOI: 10.7270/Q2J966BX |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 1
(Homo sapiens (Human)) | BDBM50144428

(CHEMBL66159 | Quinoxaline-2-carboxylic acid ((1S,2...)Show SMILES CC(C)(F)CCC(C[C@H](O)[C@H](Cc1ccccc1)NC(=O)c1cnc2ccccc2n1)C(=O)NN Show InChI InChI=1S/C26H32FN5O3/c1-26(2,27)13-12-18(24(34)32-28)15-23(33)21(14-17-8-4-3-5-9-17)31-25(35)22-16-29-19-10-6-7-11-20(19)30-22/h3-11,16,18,21,23,33H,12-15,28H2,1-2H3,(H,31,35)(H,32,34)/t18?,21-,23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CCL3 induced chemotaxis in human T lymphocytes |
Bioorg Med Chem Lett 14: 2175-9 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.022
BindingDB Entry DOI: 10.7270/Q24B30SR |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 1
(Homo sapiens (Human)) | BDBM50144400

(CHEMBL304358 | Quinoxaline-2-carboxylic acid ((1S,...)Show SMILES CC(C)CC[C@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)c1cnc2ccccc2n1)C(N)=O Show InChI InChI=1S/C26H32N4O3/c1-17(2)12-13-19(25(27)32)15-24(31)22(14-18-8-4-3-5-9-18)30-26(33)23-16-28-20-10-6-7-11-21(20)29-23/h3-11,16-17,19,22,24,31H,12-15H2,1-2H3,(H2,27,32)(H,30,33)/t19-,22+,24+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CCL3 induced chemotaxis in human T lymphocytes |
Bioorg Med Chem Lett 14: 2175-9 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.022
BindingDB Entry DOI: 10.7270/Q24B30SR |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215854

(CHEMBL301829)Show SMILES O[C@@H]1[C@@H](Cc2ccccc2)COc2cc(ccc12)-c1cc(ccc1C(O)=O)C(F)(F)F Show InChI InChI=1S/C24H19F3O4/c25-24(26,27)17-7-9-18(23(29)30)20(12-17)15-6-8-19-21(11-15)31-13-16(22(19)28)10-14-4-2-1-3-5-14/h1-9,11-12,16,22,28H,10,13H2,(H,29,30)/t16-,22+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4-induced chemotaxis of isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215854

(CHEMBL301829)Show SMILES O[C@@H]1[C@@H](Cc2ccccc2)COc2cc(ccc12)-c1cc(ccc1C(O)=O)C(F)(F)F Show InChI InChI=1S/C24H19F3O4/c25-24(26,27)17-7-9-18(23(29)30)20(12-17)15-6-8-19-21(11-15)31-13-16(22(19)28)10-14-4-2-1-3-5-14/h1-9,11-12,16,22,28H,10,13H2,(H,29,30)/t16-,22+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4-induced chemotaxis of isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215854

(CHEMBL301829)Show SMILES O[C@@H]1[C@@H](Cc2ccccc2)COc2cc(ccc12)-c1cc(ccc1C(O)=O)C(F)(F)F Show InChI InChI=1S/C24H19F3O4/c25-24(26,27)17-7-9-18(23(29)30)20(12-17)15-6-8-19-21(11-15)31-13-16(22(19)28)10-14-4-2-1-3-5-14/h1-9,11-12,16,22,28H,10,13H2,(H,29,30)/t16-,22+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4-induced chemotaxis of isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215738
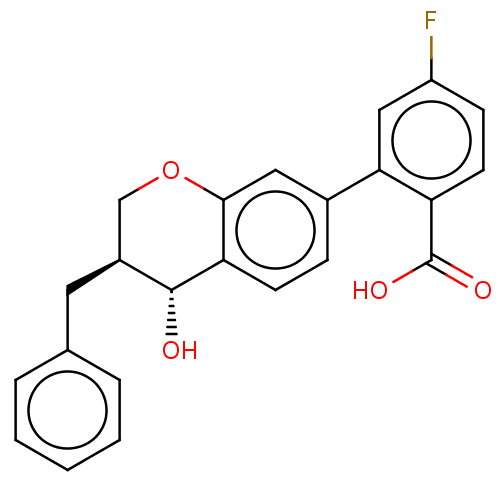
(CHEMBL52675)Show SMILES O[C@@H]1[C@@H](Cc2ccccc2)COc2cc(ccc12)-c1cc(F)ccc1C(O)=O Show InChI InChI=1S/C23H19FO4/c24-17-7-9-18(23(26)27)20(12-17)15-6-8-19-21(11-15)28-13-16(22(19)25)10-14-4-2-1-3-5-14/h1-9,11-12,16,22,25H,10,13H2,(H,26,27)/t16-,22+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4 binding to LTB receptors on isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215740

(CHEMBL48906)Show SMILES O[C@@H]1[C@@H](Cc2ccc(cc2)-c2ccccc2)COc2cc(ccc12)-c1cc(F)ccc1C(O)=O Show InChI InChI=1S/C29H23FO4/c30-23-11-13-24(29(32)33)26(16-23)21-10-12-25-27(15-21)34-17-22(28(25)31)14-18-6-8-20(9-7-18)19-4-2-1-3-5-19/h1-13,15-16,22,28,31H,14,17H2,(H,32,33)/t22-,28+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4 binding to LTB receptors on isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215740

(CHEMBL48906)Show SMILES O[C@@H]1[C@@H](Cc2ccc(cc2)-c2ccccc2)COc2cc(ccc12)-c1cc(F)ccc1C(O)=O Show InChI InChI=1S/C29H23FO4/c30-23-11-13-24(29(32)33)26(16-23)21-10-12-25-27(15-21)34-17-22(28(25)31)14-18-6-8-20(9-7-18)19-4-2-1-3-5-19/h1-13,15-16,22,28,31H,14,17H2,(H,32,33)/t22-,28+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4 binding to LTB receptors on isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215738

(CHEMBL52675)Show SMILES O[C@@H]1[C@@H](Cc2ccccc2)COc2cc(ccc12)-c1cc(F)ccc1C(O)=O Show InChI InChI=1S/C23H19FO4/c24-17-7-9-18(23(26)27)20(12-17)15-6-8-19-21(11-15)28-13-16(22(19)25)10-14-4-2-1-3-5-14/h1-9,11-12,16,22,25H,10,13H2,(H,26,27)/t16-,22+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4-induced chemotaxis of isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215855
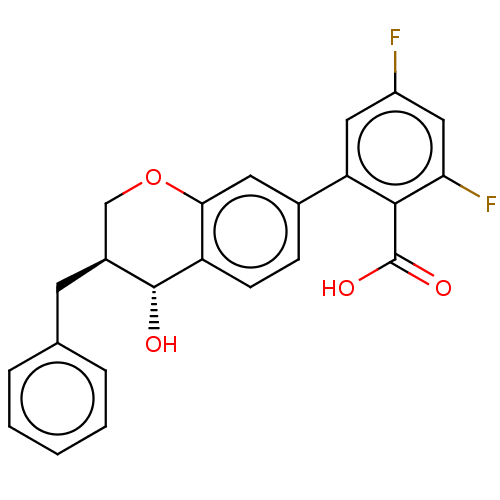
(CHEMBL51467)Show SMILES O[C@@H]1[C@@H](Cc2ccccc2)COc2cc(ccc12)-c1cc(F)cc(F)c1C(O)=O Show InChI InChI=1S/C23H18F2O4/c24-16-10-18(21(23(27)28)19(25)11-16)14-6-7-17-20(9-14)29-12-15(22(17)26)8-13-4-2-1-3-5-13/h1-7,9-11,15,22,26H,8,12H2,(H,27,28)/t15-,22+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4 binding to LTB receptors on isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215738

(CHEMBL52675)Show SMILES O[C@@H]1[C@@H](Cc2ccccc2)COc2cc(ccc12)-c1cc(F)ccc1C(O)=O Show InChI InChI=1S/C23H19FO4/c24-17-7-9-18(23(26)27)20(12-17)15-6-8-19-21(11-15)28-13-16(22(19)25)10-14-4-2-1-3-5-14/h1-9,11-12,16,22,25H,10,13H2,(H,26,27)/t16-,22+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4-induced chemotaxis of isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 1
(Homo sapiens (Human)) | BDBM50144420
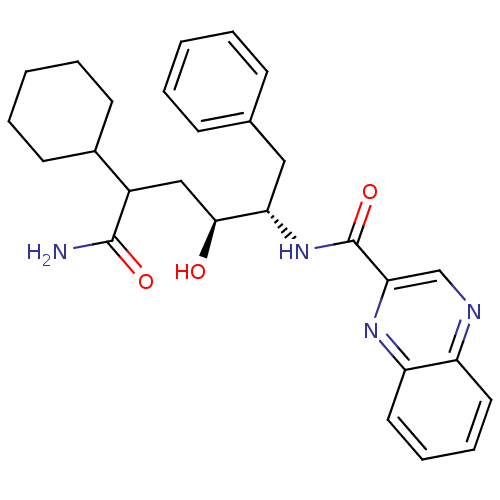
(CHEMBL68145 | Quinoxaline-2-carboxylic acid ((1S,2...)Show SMILES NC(=O)C(C[C@H](O)[C@H](Cc1ccccc1)NC(=O)c1cnc2ccccc2n1)C1CCCCC1 Show InChI InChI=1S/C27H32N4O3/c28-26(33)20(19-11-5-2-6-12-19)16-25(32)23(15-18-9-3-1-4-10-18)31-27(34)24-17-29-21-13-7-8-14-22(21)30-24/h1,3-4,7-10,13-14,17,19-20,23,25,32H,2,5-6,11-12,15-16H2,(H2,28,33)(H,31,34)/t20?,23-,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CCL3 induced chemotaxis in human T lymphocytes |
Bioorg Med Chem Lett 14: 2175-9 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.022
BindingDB Entry DOI: 10.7270/Q24B30SR |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50037218
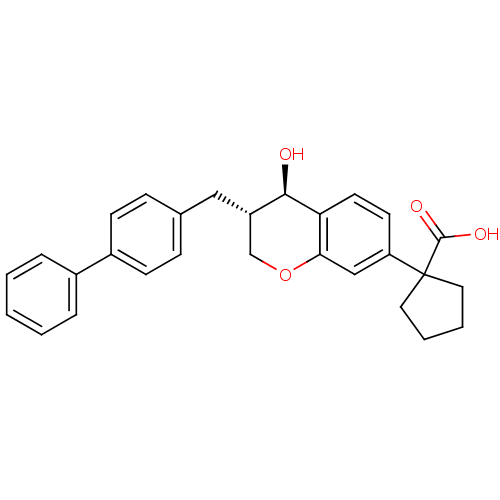
(1-((3S,4R)-3-Biphenyl-4-ylmethyl-4-hydroxy-chroman...)Show SMILES O[C@@H]1[C@@H](Cc2ccc(cc2)-c2ccccc2)COc2cc(ccc12)C1(CCCC1)C(O)=O Show InChI InChI=1S/C28H28O4/c29-26-22(16-19-8-10-21(11-9-19)20-6-2-1-3-7-20)18-32-25-17-23(12-13-24(25)26)28(27(30)31)14-4-5-15-28/h1-3,6-13,17,22,26,29H,4-5,14-16,18H2,(H,30,31)/t22-,26+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 5.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4-induced CD11b up-regulation on isolated human neutrophils in whole blood |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215853

(CHEMBL417364)Show SMILES O[C@@H]1[C@@H](Cc2ccc(cc2)-c2ccccc2)COc2cc(ccc12)-c1cc(ccc1C(O)=O)C(F)(F)F Show InChI InChI=1S/C30H23F3O4/c31-30(32,33)23-11-13-24(29(35)36)26(16-23)21-10-12-25-27(15-21)37-17-22(28(25)34)14-18-6-8-20(9-7-18)19-4-2-1-3-5-19/h1-13,15-16,22,28,34H,14,17H2,(H,35,36)/t22-,28+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4-induced chemotaxis of isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215853

(CHEMBL417364)Show SMILES O[C@@H]1[C@@H](Cc2ccc(cc2)-c2ccccc2)COc2cc(ccc12)-c1cc(ccc1C(O)=O)C(F)(F)F Show InChI InChI=1S/C30H23F3O4/c31-30(32,33)23-11-13-24(29(35)36)26(16-23)21-10-12-25-27(15-21)37-17-22(28(25)34)14-18-6-8-20(9-7-18)19-4-2-1-3-5-19/h1-13,15-16,22,28,34H,14,17H2,(H,35,36)/t22-,28+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4-induced chemotaxis of isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50037218

(1-((3S,4R)-3-Biphenyl-4-ylmethyl-4-hydroxy-chroman...)Show SMILES O[C@@H]1[C@@H](Cc2ccc(cc2)-c2ccccc2)COc2cc(ccc12)C1(CCCC1)C(O)=O Show InChI InChI=1S/C28H28O4/c29-26-22(16-19-8-10-21(11-9-19)20-6-2-1-3-7-20)18-32-25-17-23(12-13-24(25)26)28(27(30)31)14-4-5-15-28/h1-3,6-13,17,22,26,29H,4-5,14-16,18H2,(H,30,31)/t22-,26+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 6.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Compound was evaluated for its ability to inhibit [3H]LTB4 binding to LTB4 receptors on guinea pig spleen membranes |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215854

(CHEMBL301829)Show SMILES O[C@@H]1[C@@H](Cc2ccccc2)COc2cc(ccc12)-c1cc(ccc1C(O)=O)C(F)(F)F Show InChI InChI=1S/C24H19F3O4/c25-24(26,27)17-7-9-18(23(29)30)20(12-17)15-6-8-19-21(11-15)31-13-16(22(19)28)10-14-4-2-1-3-5-14/h1-9,11-12,16,22,28H,10,13H2,(H,29,30)/t16-,22+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 6.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4 binding to LTB receptors on isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 1
(Homo sapiens (Human)) | BDBM50144431

(CHEMBL303673 | Quinoxaline-2-carboxylic acid [(1S,...)Show SMILES ONC(=O)C(C[C@H](O)[C@H](Cc1ccccc1)NC(=O)c1cnc2ccccc2n1)C1CCC(F)(F)CC1 Show InChI InChI=1S/C27H30F2N4O4/c28-27(29)12-10-18(11-13-27)19(25(35)33-37)15-24(34)22(14-17-6-2-1-3-7-17)32-26(36)23-16-30-20-8-4-5-9-21(20)31-23/h1-9,16,18-19,22,24,34,37H,10-15H2,(H,32,36)(H,33,35)/t19?,22-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CCL3 binding to C-C chemokine receptor type 1 |
Bioorg Med Chem Lett 14: 2175-9 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.022
BindingDB Entry DOI: 10.7270/Q24B30SR |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 1
(Homo sapiens (Human)) | BDBM50144428

(CHEMBL66159 | Quinoxaline-2-carboxylic acid ((1S,2...)Show SMILES CC(C)(F)CCC(C[C@H](O)[C@H](Cc1ccccc1)NC(=O)c1cnc2ccccc2n1)C(=O)NN Show InChI InChI=1S/C26H32FN5O3/c1-26(2,27)13-12-18(24(34)32-28)15-23(33)21(14-17-8-4-3-5-9-17)31-25(35)22-16-29-19-10-6-7-11-20(19)30-22/h3-11,16,18,21,23,33H,12-15,28H2,1-2H3,(H,31,35)(H,32,34)/t18?,21-,23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CCL3 induced chemotaxis in human T lymphocytes |
Bioorg Med Chem Lett 14: 2175-9 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.022
BindingDB Entry DOI: 10.7270/Q24B30SR |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215737

(CHEMBL50425)Show SMILES O[C@@H]1[C@@H](Cc2ccc(cc2)-c2ccccc2)COc2cc(ccc12)-c1cc(Cl)ccc1C(O)=O Show InChI InChI=1S/C29H23ClO4/c30-23-11-13-24(29(32)33)26(16-23)21-10-12-25-27(15-21)34-17-22(28(25)31)14-18-6-8-20(9-7-18)19-4-2-1-3-5-19/h1-13,15-16,22,28,31H,14,17H2,(H,32,33)/t22-,28+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4 binding to LTB receptors on isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215737

(CHEMBL50425)Show SMILES O[C@@H]1[C@@H](Cc2ccc(cc2)-c2ccccc2)COc2cc(ccc12)-c1cc(Cl)ccc1C(O)=O Show InChI InChI=1S/C29H23ClO4/c30-23-11-13-24(29(32)33)26(16-23)21-10-12-25-27(15-21)34-17-22(28(25)31)14-18-6-8-20(9-7-18)19-4-2-1-3-5-19/h1-13,15-16,22,28,31H,14,17H2,(H,32,33)/t22-,28+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4 binding to LTB receptors on isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 1
(Homo sapiens (Human)) | BDBM50144431

(CHEMBL303673 | Quinoxaline-2-carboxylic acid [(1S,...)Show SMILES ONC(=O)C(C[C@H](O)[C@H](Cc1ccccc1)NC(=O)c1cnc2ccccc2n1)C1CCC(F)(F)CC1 Show InChI InChI=1S/C27H30F2N4O4/c28-27(29)12-10-18(11-13-27)19(25(35)33-37)15-24(34)22(14-17-6-2-1-3-7-17)32-26(36)23-16-30-20-8-4-5-9-21(20)31-23/h1-9,16,18-19,22,24,34,37H,10-15H2,(H,32,36)(H,33,35)/t19?,22-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CCL3 induced chemotaxis in human T lymphocytes |
Bioorg Med Chem Lett 14: 2175-9 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.022
BindingDB Entry DOI: 10.7270/Q24B30SR |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 1
(Homo sapiens (Human)) | BDBM50144420

(CHEMBL68145 | Quinoxaline-2-carboxylic acid ((1S,2...)Show SMILES NC(=O)C(C[C@H](O)[C@H](Cc1ccccc1)NC(=O)c1cnc2ccccc2n1)C1CCCCC1 Show InChI InChI=1S/C27H32N4O3/c28-26(33)20(19-11-5-2-6-12-19)16-25(32)23(15-18-9-3-1-4-10-18)31-27(34)24-17-29-21-13-7-8-14-22(21)30-24/h1,3-4,7-10,13-14,17,19-20,23,25,32H,2,5-6,11-12,15-16H2,(H2,28,33)(H,31,34)/t20?,23-,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CCL3 binding to C-C chemokine receptor type 1 |
Bioorg Med Chem Lett 14: 2175-9 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.022
BindingDB Entry DOI: 10.7270/Q24B30SR |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 1
(Homo sapiens (Human)) | BDBM50144396

(CHEMBL69375 | Quinoxaline-2-carboxylic acid ((1S,2...)Show SMILES NC(=O)[C@@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)c1cnc2ccccc2n1)C1CCCCC1 Show InChI InChI=1S/C27H32N4O3/c28-26(33)20(19-11-5-2-6-12-19)16-25(32)23(15-18-9-3-1-4-10-18)31-27(34)24-17-29-21-13-7-8-14-22(21)30-24/h1,3-4,7-10,13-14,17,19-20,23,25,32H,2,5-6,11-12,15-16H2,(H2,28,33)(H,31,34)/t20-,23-,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CCL3 binding to C-C chemokine receptor type 1 |
Bioorg Med Chem Lett 14: 2169-73 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.021
BindingDB Entry DOI: 10.7270/Q2833RGN |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50037218

(1-((3S,4R)-3-Biphenyl-4-ylmethyl-4-hydroxy-chroman...)Show SMILES O[C@@H]1[C@@H](Cc2ccc(cc2)-c2ccccc2)COc2cc(ccc12)C1(CCCC1)C(O)=O Show InChI InChI=1S/C28H28O4/c29-26-22(16-19-8-10-21(11-9-19)20-6-2-1-3-7-20)18-32-25-17-23(12-13-24(25)26)28(27(30)31)14-4-5-15-28/h1-3,6-13,17,22,26,29H,4-5,14-16,18H2,(H,30,31)/t22-,26+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 8.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4 binding to LTB receptors on isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 1
(Homo sapiens (Human)) | BDBM50144417
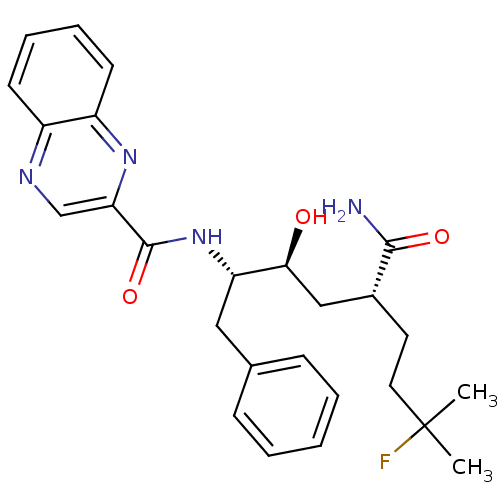
(CHEMBL68366 | Quinoxaline-2-carboxylic acid ((1S,2...)Show SMILES CC(C)(F)CC[C@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)c1cnc2ccccc2n1)C(N)=O Show InChI InChI=1S/C26H31FN4O3/c1-26(2,27)13-12-18(24(28)33)15-23(32)21(14-17-8-4-3-5-9-17)31-25(34)22-16-29-19-10-6-7-11-20(19)30-22/h3-11,16,18,21,23,32H,12-15H2,1-2H3,(H2,28,33)(H,31,34)/t18-,21+,23+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of CCL3 binding to C-C chemokine receptor type 1 |
Bioorg Med Chem Lett 14: 2175-9 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.022
BindingDB Entry DOI: 10.7270/Q24B30SR |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215739

(CHEMBL52374)Show SMILES OCc1ccc(cc1-c1ccc2[C@H](O)[C@@H](Cc3ccccc3)COc2c1)C(F)(F)F Show InChI InChI=1S/C24H21F3O3/c25-24(26,27)19-8-6-17(13-28)21(12-19)16-7-9-20-22(11-16)30-14-18(23(20)29)10-15-4-2-1-3-5-15/h1-9,11-12,18,23,28-29H,10,13-14H2/t18-,23+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4-induced chemotaxis of isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215861

(CHEMBL298724)Show SMILES O[C@@H]1[C@@H](Cc2ccccc2)COc2cc(ccc12)-c1cccc(F)c1C(O)=O Show InChI InChI=1S/C23H19FO4/c24-19-8-4-7-17(21(19)23(26)27)15-9-10-18-20(12-15)28-13-16(22(18)25)11-14-5-2-1-3-6-14/h1-10,12,16,22,25H,11,13H2,(H,26,27)/t16-,22+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4-induced chemotaxis of isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215861

(CHEMBL298724)Show SMILES O[C@@H]1[C@@H](Cc2ccccc2)COc2cc(ccc12)-c1cccc(F)c1C(O)=O Show InChI InChI=1S/C23H19FO4/c24-19-8-4-7-17(21(19)23(26)27)15-9-10-18-20(12-15)28-13-16(22(18)25)11-14-5-2-1-3-6-14/h1-10,12,16,22,25H,11,13H2,(H,26,27)/t16-,22+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4-induced chemotaxis of isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215861

(CHEMBL298724)Show SMILES O[C@@H]1[C@@H](Cc2ccccc2)COc2cc(ccc12)-c1cccc(F)c1C(O)=O Show InChI InChI=1S/C23H19FO4/c24-19-8-4-7-17(21(19)23(26)27)15-9-10-18-20(12-15)28-13-16(22(18)25)11-14-5-2-1-3-6-14/h1-10,12,16,22,25H,11,13H2,(H,26,27)/t16-,22+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4 binding to LTB receptors on isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |
Leukotriene B4 receptor 1/2
(Homo sapiens (Human)) | BDBM50215738

(CHEMBL52675)Show SMILES O[C@@H]1[C@@H](Cc2ccccc2)COc2cc(ccc12)-c1cc(F)ccc1C(O)=O Show InChI InChI=1S/C23H19FO4/c24-17-7-9-18(23(26)27)20(12-17)15-6-8-19-21(11-15)28-13-16(22(19)25)10-14-4-2-1-3-5-14/h1-9,11-12,16,22,25H,10,13H2,(H,26,27)/t16-,22+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc
Curated by ChEMBL
| Assay Description
Ability to inhibit LTB4 binding to LTB receptors on isolated human neutrophils. |
Bioorg Med Chem Lett 8: 1781-6 (1998)
BindingDB Entry DOI: 10.7270/Q2RR21F1 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data