

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394056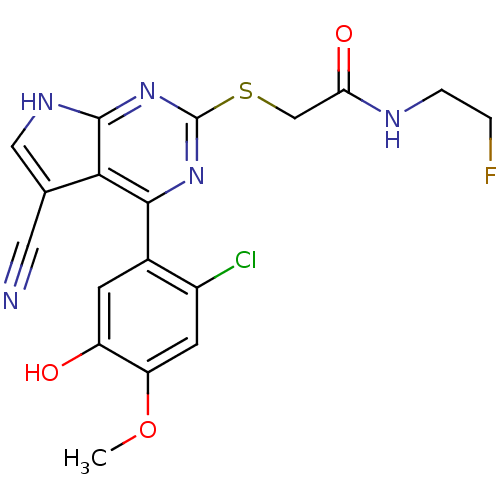 (CHEMBL2158577) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394058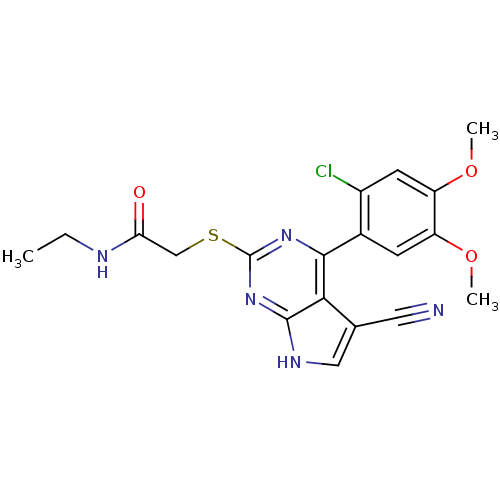 (CHEMBL2158570) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394065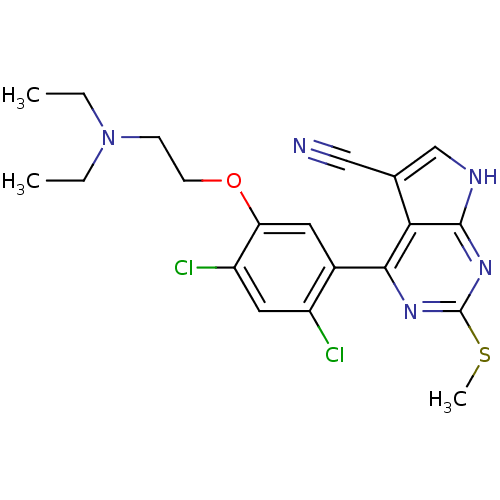 (CHEMBL2158626) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394064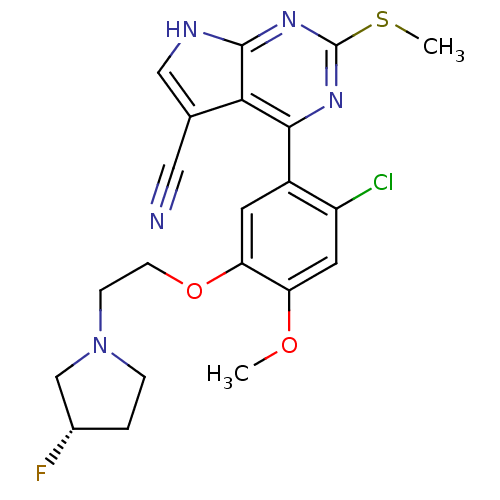 (CHEMBL2158627) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394059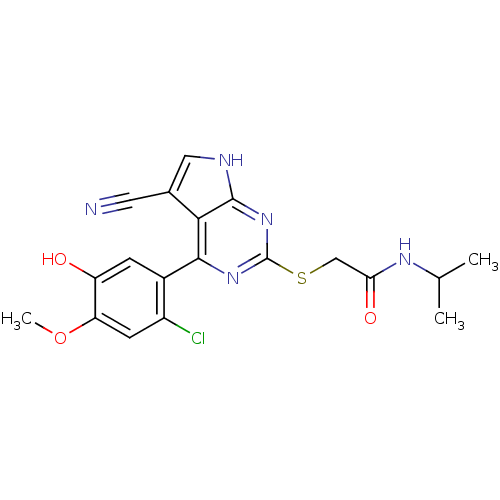 (CHEMBL2158569) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394066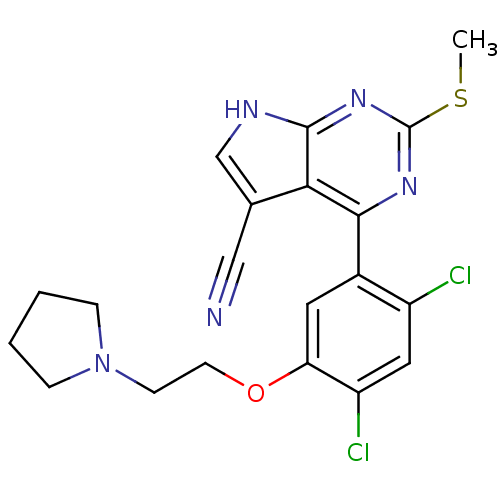 (CHEMBL2158625) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394057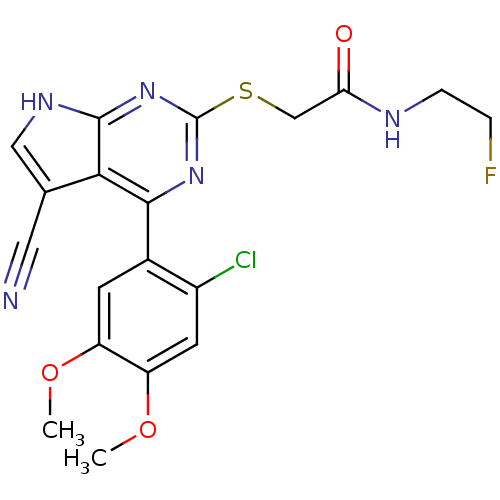 (CHEMBL2158576) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394062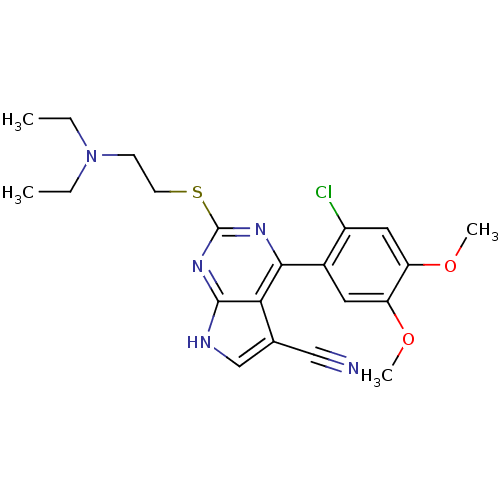 (CHEMBL2158630) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394055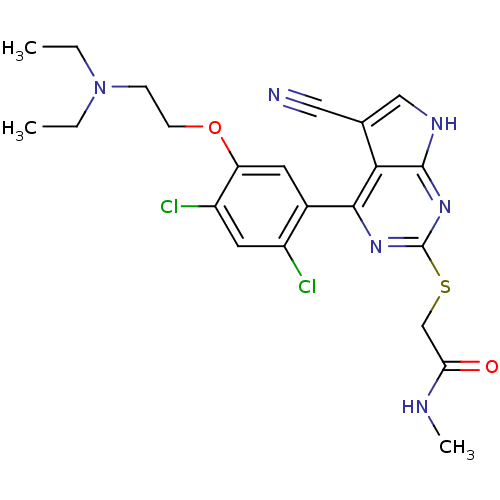 (CHEMBL2158563) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394063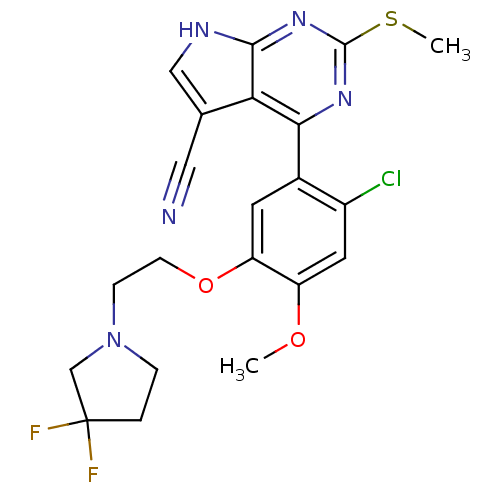 (CHEMBL2158628) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394061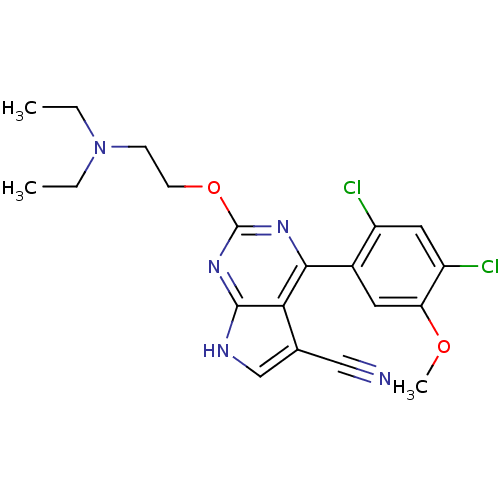 (CHEMBL2158008) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 62 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kynureninase (Homo sapiens (Human)) | BDBM50069730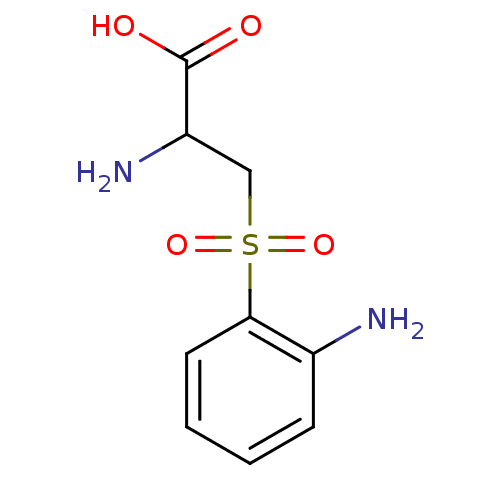 (2-Amino-3-(2-amino-benzenesulfonyl)-propionic acid...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Compound was tested for the binding affinity against bacterial kynureninase | Bioorg Med Chem Lett 8: 133-8 (1999) BindingDB Entry DOI: 10.7270/Q2639NXW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock 70 kDa protein 1A (Homo sapiens (Human)) | BDBM82124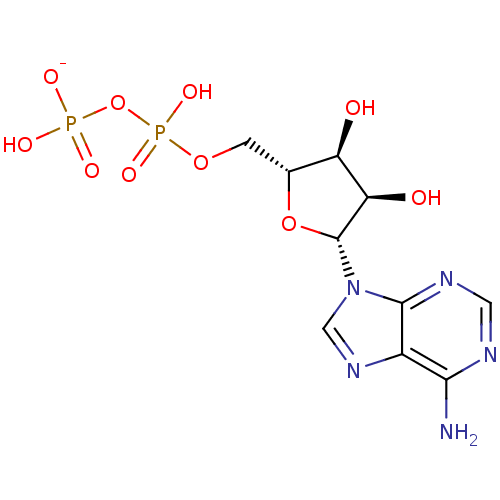 (adenosine-derived inhibitor (Grp78), 1) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Similars | Article PubMed | 110 | -39.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd. | Assay Description The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... | J Med Chem 54: 4034-41 (2011) Article DOI: 10.1021/jm101625x BindingDB Entry DOI: 10.7270/Q2R49P83 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock 70 kDa protein 1A (Homo sapiens (Human)) | BDBM32378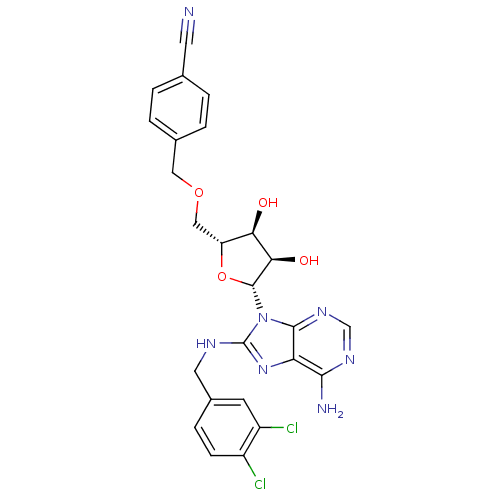 (adenosine-derived inhibitor (Grp78), 13 | adenosin...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase MCE MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | 120 | -39.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd. | Assay Description The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... | J Med Chem 54: 4034-41 (2011) Article DOI: 10.1021/jm101625x BindingDB Entry DOI: 10.7270/Q2R49P83 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394060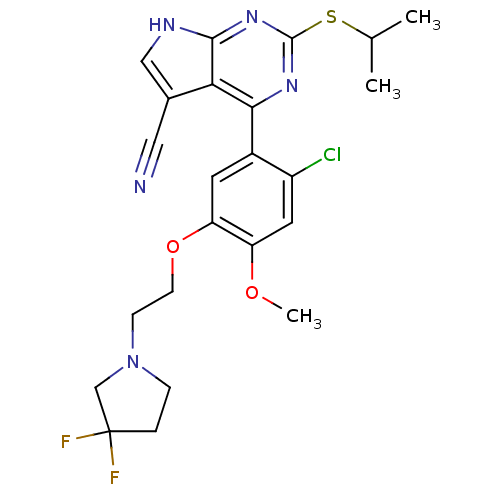 (CHEMBL2158565) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 172 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394079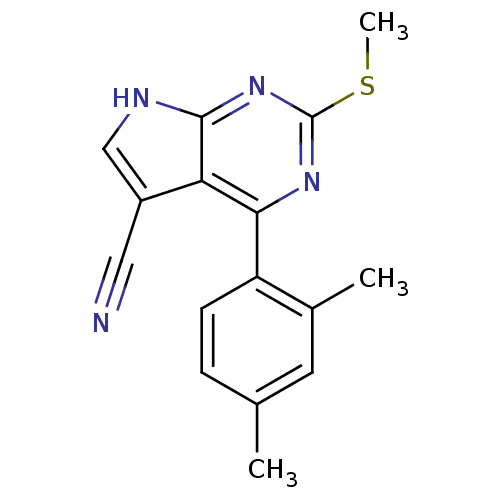 (CHEMBL2158581) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 195 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394077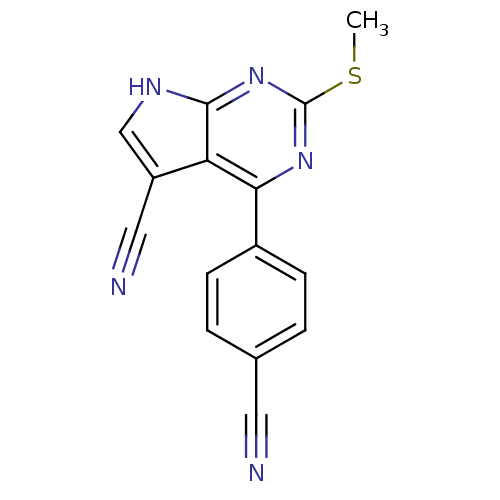 (CHEMBL2158583) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 204 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock 70 kDa protein 1A (Homo sapiens (Human)) | BDBM32381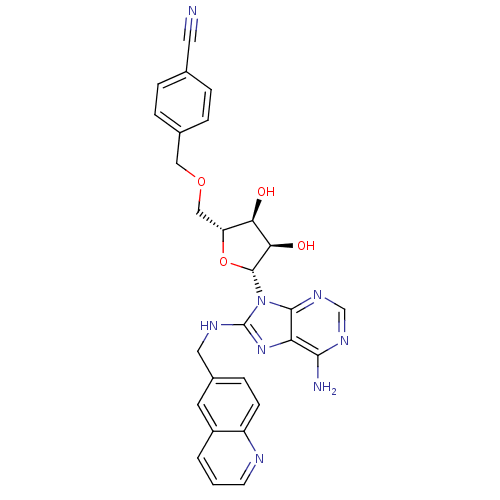 (adenosine-derived inhibitor (Grp78), 14 | adenosin...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | 310 | -37.1 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd. | Assay Description The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... | J Med Chem 54: 4034-41 (2011) Article DOI: 10.1021/jm101625x BindingDB Entry DOI: 10.7270/Q2R49P83 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394078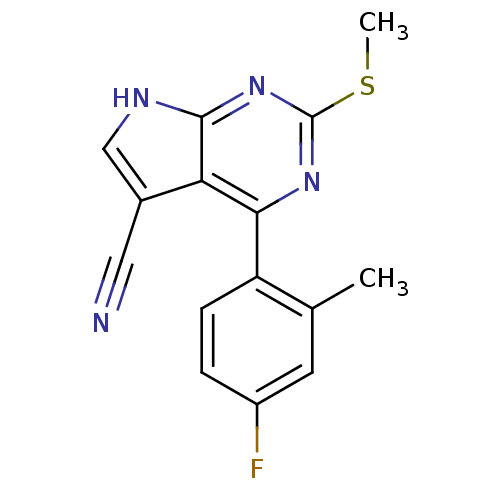 (CHEMBL2158582) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 358 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock 70 kDa protein 1A (Homo sapiens (Human)) | BDBM82127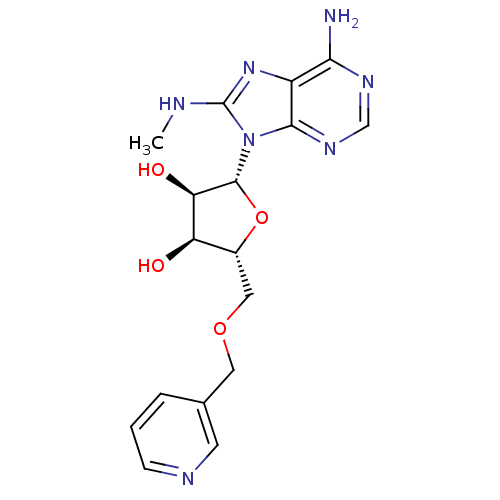 ((2R,3R,4S,5R)-2-(6-Amino-8-methylaminopurin-9-yl)-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 450 | -36.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd. | Assay Description The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... | J Med Chem 54: 4034-41 (2011) Article DOI: 10.1021/jm101625x BindingDB Entry DOI: 10.7270/Q2R49P83 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394069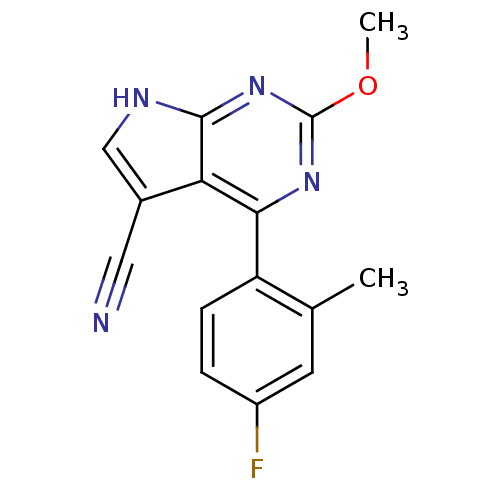 (CHEMBL2158622) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 628 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394085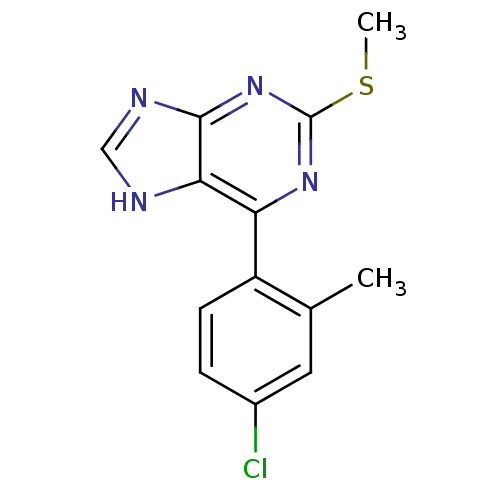 (CHEMBL2158635) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 661 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock 70 kDa protein 1A (Homo sapiens (Human)) | BDBM32377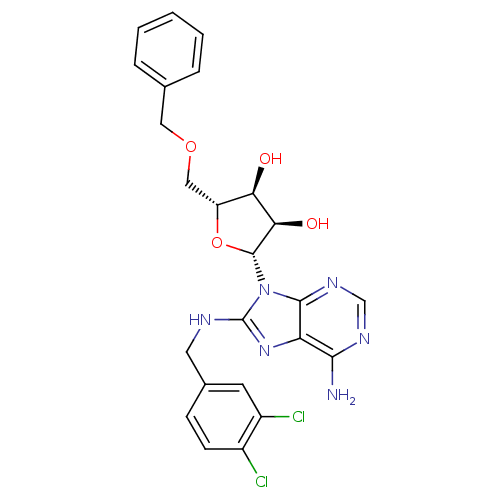 (adenosine-derived inhibitor (Grp78), 12 | adenosin...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 1.05E+3 | -34.1 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd. | Assay Description The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... | J Med Chem 54: 4034-41 (2011) Article DOI: 10.1021/jm101625x BindingDB Entry DOI: 10.7270/Q2R49P83 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock 70 kDa protein 1A (Homo sapiens (Human)) | BDBM82125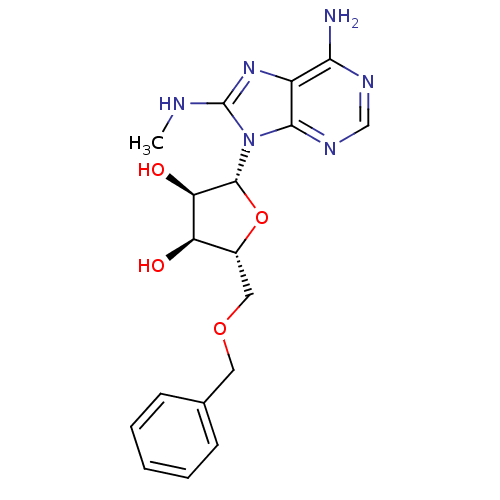 ((2R,3R,4S,5R)-2-(6-Amino-8-methylaminopurin-9-yl)-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 1.10E+3 | -34.0 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd. | Assay Description The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... | J Med Chem 54: 4034-41 (2011) Article DOI: 10.1021/jm101625x BindingDB Entry DOI: 10.7270/Q2R49P83 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock 70 kDa protein 1A (Homo sapiens (Human)) | BDBM32375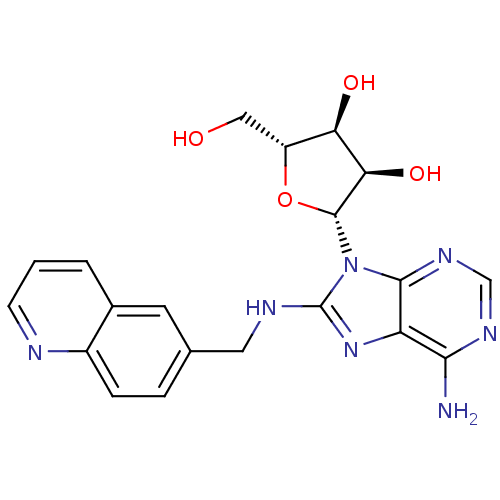 (adenosine-derived inhibitor (Grp78), 9 | adenosine...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 1.29E+3 | -33.6 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd. | Assay Description The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... | J Med Chem 54: 4034-41 (2011) Article DOI: 10.1021/jm101625x BindingDB Entry DOI: 10.7270/Q2R49P83 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Heat shock 70 kDa protein 1A (Homo sapiens (Human)) | BDBM32376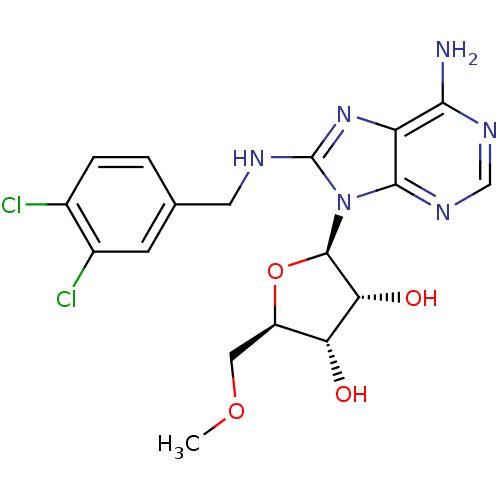 (adenosine-derived inhibitor (Grp78), 11 | adenosin...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 1.44E+3 | -33.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd. | Assay Description The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... | J Med Chem 54: 4034-41 (2011) Article DOI: 10.1021/jm101625x BindingDB Entry DOI: 10.7270/Q2R49P83 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394067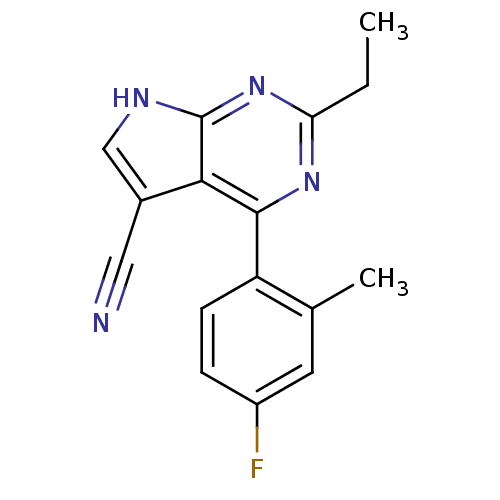 (CHEMBL2158624) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 1.72E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock 70 kDa protein 1A (Homo sapiens (Human)) | BDBM82128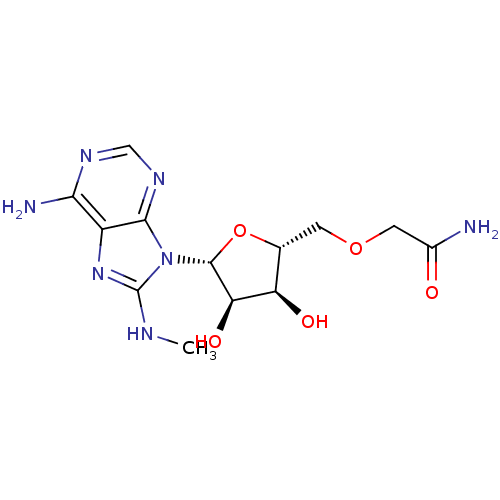 (2-[(2R,3S,4R,5R)-5-(6-Amino-8-methylaminopurin-9-y...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | 1.85E+3 | -32.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd. | Assay Description The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... | J Med Chem 54: 4034-41 (2011) Article DOI: 10.1021/jm101625x BindingDB Entry DOI: 10.7270/Q2R49P83 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Protein arginine N-methyltransferase 5 (Homo sapiens (Human)) | BDBM50572983 (CHEMBL4848846) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Covalent inhibition of human PRMT5 assessed as initial binding constant by LC-MS analysis | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00507 BindingDB Entry DOI: 10.7270/Q26977DH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394073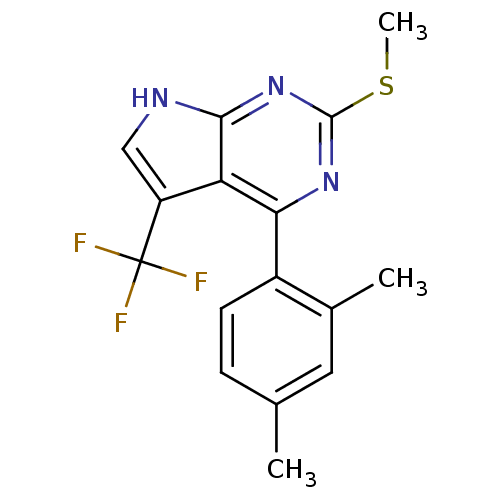 (CHEMBL2158618) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.64E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394070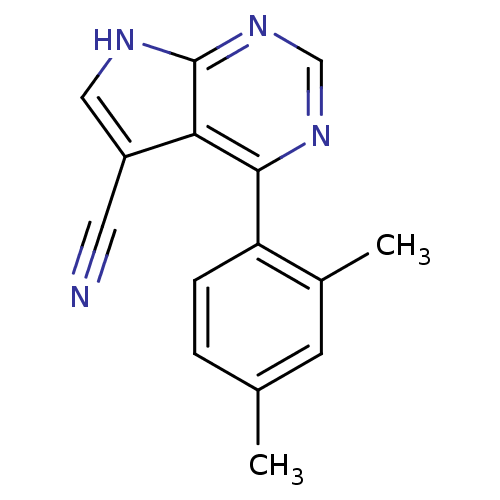 (CHEMBL2158621) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 2.83E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock 70 kDa protein 1A (Homo sapiens (Human)) | BDBM82126 ((2R,3R,4S,5R)-2-(6-Amino-8-methylaminopurin-9-yl)-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 4.07E+3 | -30.8 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd. | Assay Description The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... | J Med Chem 54: 4034-41 (2011) Article DOI: 10.1021/jm101625x BindingDB Entry DOI: 10.7270/Q2R49P83 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394068 (CHEMBL2158623) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 4.07E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock 70 kDa protein 1A (Homo sapiens (Human)) | BDBM32373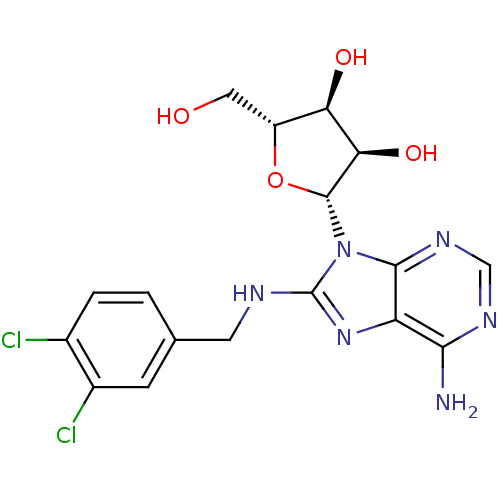 (adenosine-derived inhibitor (Grp78), 8 | adenosine...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | DrugBank MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | 4.31E+3 | -30.6 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd. | Assay Description The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... | J Med Chem 54: 4034-41 (2011) Article DOI: 10.1021/jm101625x BindingDB Entry DOI: 10.7270/Q2R49P83 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Heat shock 70 kDa protein 1A (Homo sapiens (Human)) | BDBM82129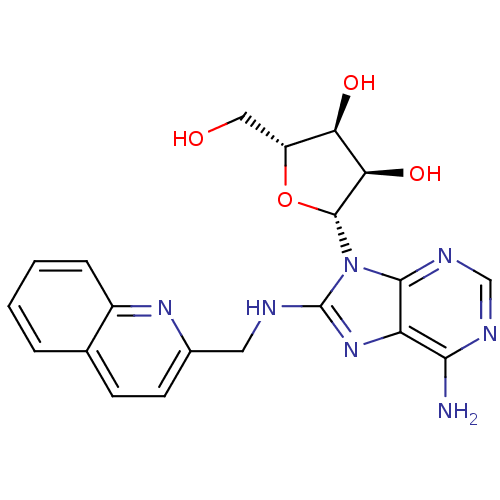 ((2R,3R,4S,5R)-2-{6-Amino-8-[(quinolin-2-ylmethyl)a...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | 4.32E+3 | -30.6 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd. | Assay Description The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... | J Med Chem 54: 4034-41 (2011) Article DOI: 10.1021/jm101625x BindingDB Entry DOI: 10.7270/Q2R49P83 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Heat shock 70 kDa protein 1A (Homo sapiens (Human)) | BDBM32370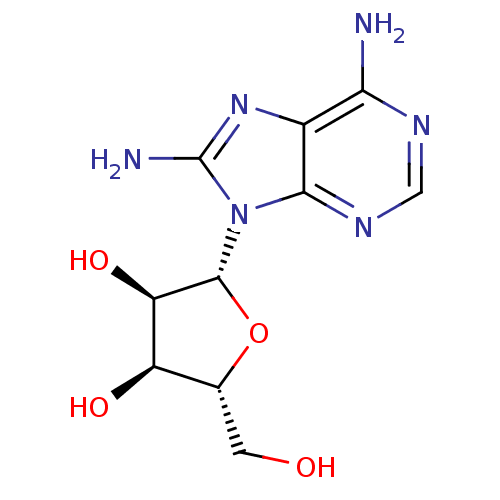 (adenosine-derived inhibitor (Grp78), 2 | adenosine...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase MCE MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 4.46E+3 | -30.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd. | Assay Description The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... | J Med Chem 54: 4034-41 (2011) Article DOI: 10.1021/jm101625x BindingDB Entry DOI: 10.7270/Q2R49P83 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394076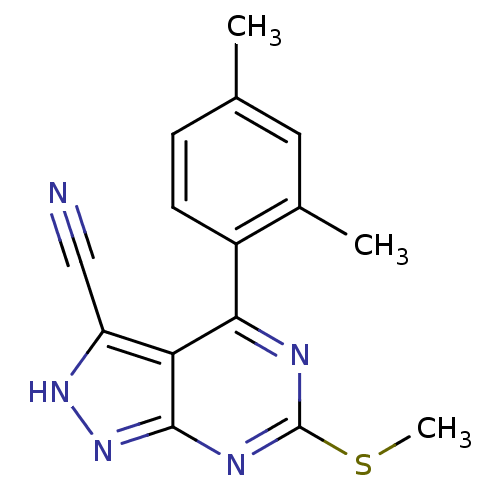 (CHEMBL2158584) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 5.23E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein arginine N-methyltransferase 5 (Homo sapiens (Human)) | BDBM50572968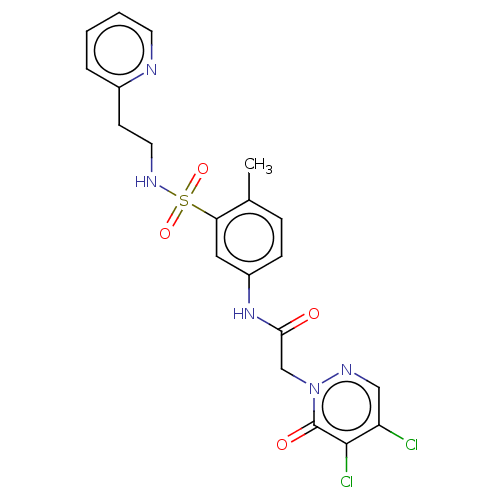 (CHEMBL4862851) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 8.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Covalent inhibition of human PRMT5 assessed as initial binding constant by LC-MS analysis | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00507 BindingDB Entry DOI: 10.7270/Q26977DH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock 70 kDa protein 1A (Homo sapiens (Human)) | BDBM32371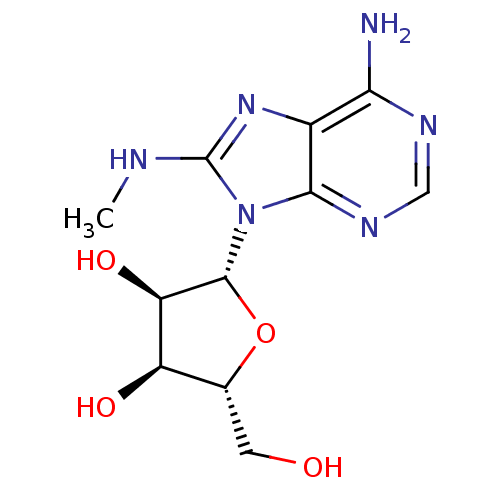 (adenosine-derived inhibitor (Grp78), 3 | adenosine...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | 8.30E+3 | -29.0 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd. | Assay Description The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... | J Med Chem 54: 4034-41 (2011) Article DOI: 10.1021/jm101625x BindingDB Entry DOI: 10.7270/Q2R49P83 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394084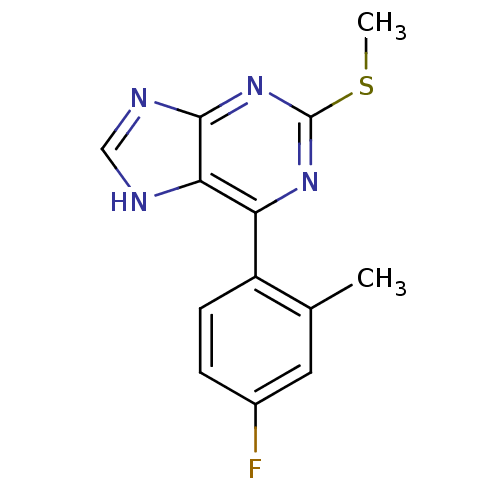 (CHEMBL2158631) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.28E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kynureninase (Rattus norvegicus) | BDBM50069730 (2-Amino-3-(2-amino-benzenesulfonyl)-propionic acid...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 1.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development Curated by ChEMBL | Assay Description Compound was tested for the binding affinity against kynureninase in rat liver | Bioorg Med Chem Lett 8: 133-8 (1999) BindingDB Entry DOI: 10.7270/Q2639NXW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein arginine N-methyltransferase 5 (Homo sapiens (Human)) | BDBM50572964 (CHEMBL4867592) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Covalent inhibition of human PRMT5 assessed as initial binding constant by LC-MS analysis | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00507 BindingDB Entry DOI: 10.7270/Q26977DH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394083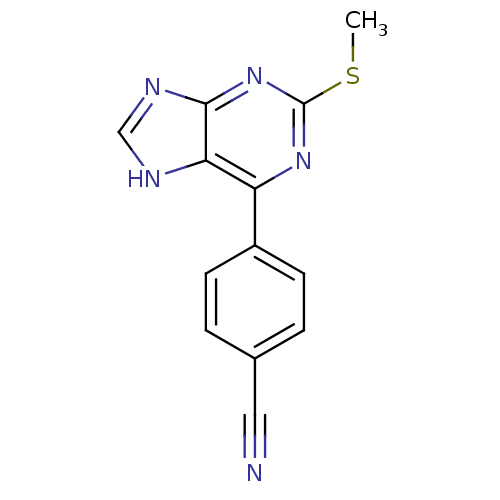 (CHEMBL2158632) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.45E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein arginine N-methyltransferase 5 (Homo sapiens (Human)) | BDBM50572989 (CHEMBL4846332) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 3.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Covalent inhibition of human PRMT5 assessed as initial binding constant by LC-MS analysis | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00507 BindingDB Entry DOI: 10.7270/Q26977DH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein arginine N-methyltransferase 5 (Homo sapiens (Human)) | BDBM50572991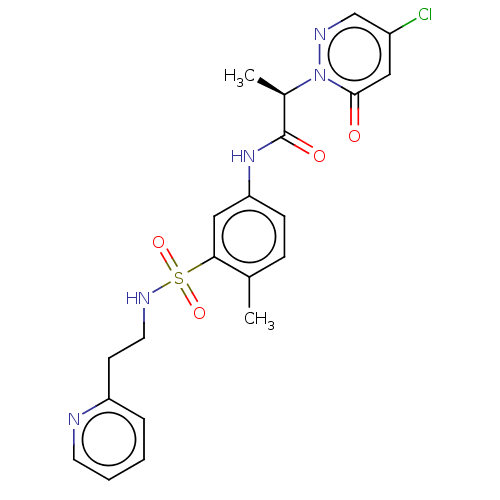 (CHEMBL4858967) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 4.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Covalent inhibition of human PRMT5 assessed as initial binding constant by LC-MS analysis | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00507 BindingDB Entry DOI: 10.7270/Q26977DH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein arginine N-methyltransferase 5 (Homo sapiens (Human)) | BDBM50572990 (CHEMBL4859105) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | 4.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Covalent inhibition of human PRMT5 assessed as initial binding constant by LC-MS analysis | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00507 BindingDB Entry DOI: 10.7270/Q26977DH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394072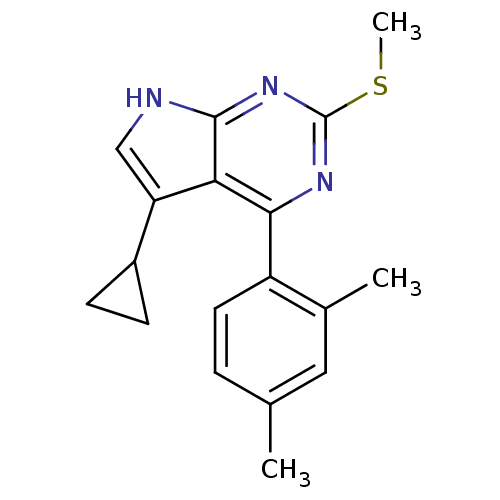 (CHEMBL2158619) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein arginine N-methyltransferase 5 (Homo sapiens (Human)) | BDBM50572988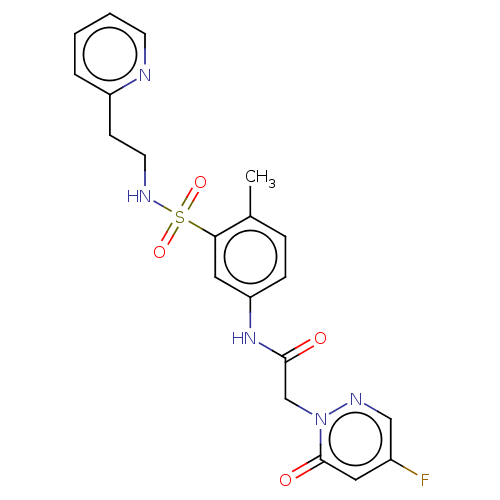 (CHEMBL4855695) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 5.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Covalent inhibition of human PRMT5 assessed as initial binding constant by LC-MS analysis | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00507 BindingDB Entry DOI: 10.7270/Q26977DH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein arginine N-methyltransferase 5 (Homo sapiens (Human)) | BDBM50572984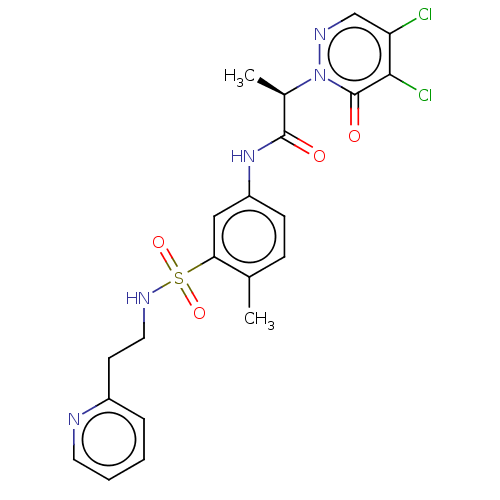 (CHEMBL4874198) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 7.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Covalent inhibition of human PRMT5 assessed as initial binding constant by LC-MS analysis | Citation and Details Article DOI: 10.1021/acs.jmedchem.1c00507 BindingDB Entry DOI: 10.7270/Q26977DH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394080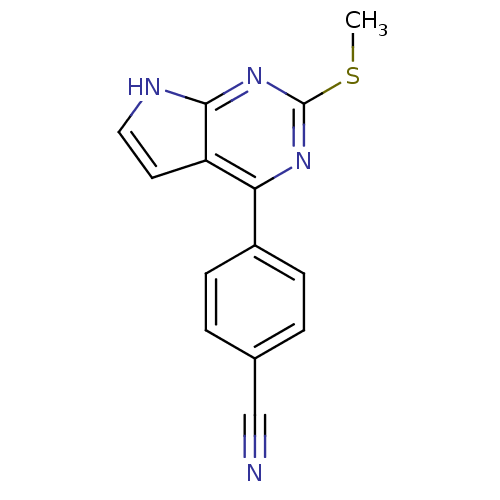 (CHEMBL2158580) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 943 total ) | Next | Last >> |