

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Endothelin-converting enzyme 1 (Homo sapiens (Human)) | BDBM50035236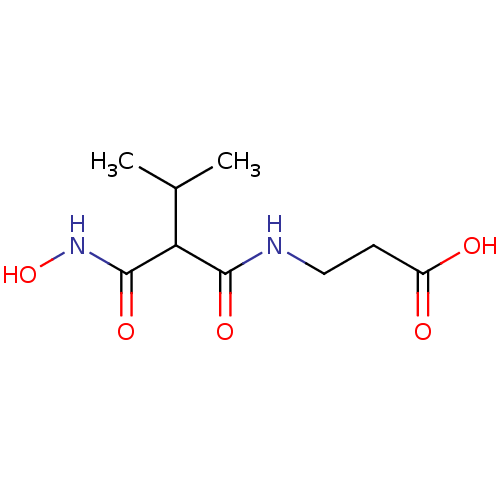 (3-(2-Hydroxycarbamoyl-3-methyl-butyrylamino)-propi...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Laboratories Curated by ChEMBL | Assay Description Inhibitory activity against human bronchiolar smooth muscle Endothelin-converting enzyme 1 | J Med Chem 38: 2119-29 (1995) BindingDB Entry DOI: 10.7270/Q2D79C2M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neprilysin (Oryctolagus cuniculus (rabbit)) | BDBM50035247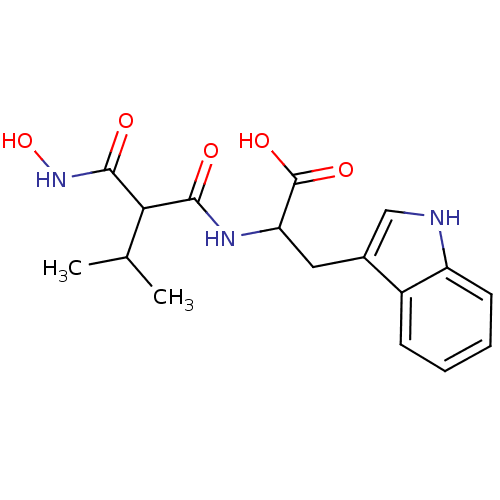 (2-(2-Hydroxycarbamoyl-3-methyl-butyrylamino)-3-(1H...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Laboratories Curated by ChEMBL | Assay Description Inhibitory activity against big ET-1 of Neutral endopeptidase | J Med Chem 38: 2119-29 (1995) BindingDB Entry DOI: 10.7270/Q2D79C2M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neprilysin (Oryctolagus cuniculus (rabbit)) | BDBM50035246 (2-(2-Hydroxycarbamoyl-4-methyl-pentanoylamino)-3-(...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Laboratories Curated by ChEMBL | Assay Description Inhibitory activity against Leu-enkeph of Neutral endopeptidase | J Med Chem 38: 2119-29 (1995) BindingDB Entry DOI: 10.7270/Q2D79C2M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-alpha (Homo sapiens (Human)) | BDBM81917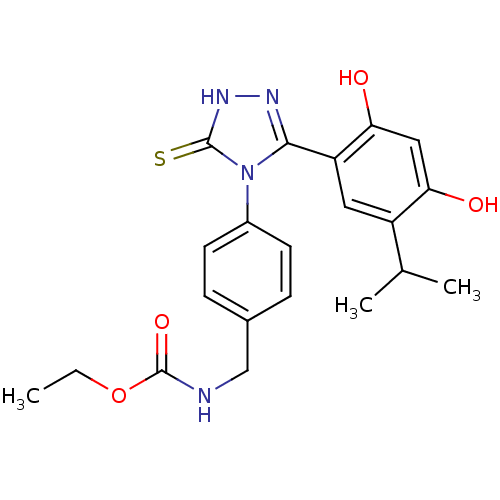 (BX-2819) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0400 | n/a | n/a | n/a | n/a | 7.4 | n/a |
Bayer Healthcare, | Assay Description To assess the affinity of compounds binding to Hsp90, we measured their ability to compete with the binding of a fluorescent analog of GA (GM-Bodipy)... | Chem Biol Drug Des 74: 43-50 (2009) Article DOI: 10.1111/j.1747-0285.2009.00833.x BindingDB Entry DOI: 10.7270/Q2KW5DJW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neprilysin (Oryctolagus cuniculus (rabbit)) | BDBM50035246 (2-(2-Hydroxycarbamoyl-4-methyl-pentanoylamino)-3-(...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Laboratories Curated by ChEMBL | Assay Description Inhibitory activity against big ET-1 of Neutral endopeptidase | J Med Chem 38: 2119-29 (1995) BindingDB Entry DOI: 10.7270/Q2D79C2M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-alpha (Homo sapiens (Human)) | BDBM81914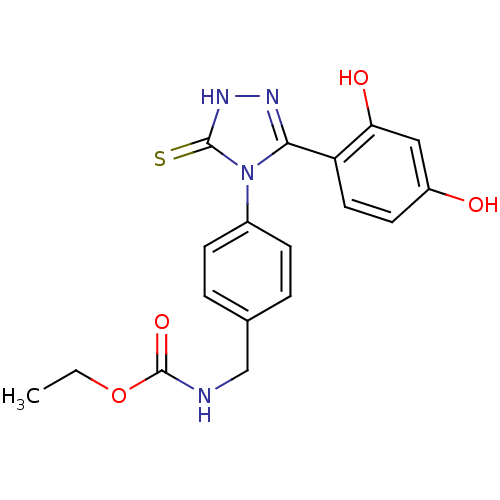 (Ethyl carbamate analog, 3) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | 7.4 | n/a |
Bayer Healthcare, | Assay Description To assess the affinity of compounds binding to Hsp90, we measured their ability to compete with the binding of a fluorescent analog of GA (GM-Bodipy)... | Chem Biol Drug Des 74: 43-50 (2009) Article DOI: 10.1111/j.1747-0285.2009.00833.x BindingDB Entry DOI: 10.7270/Q2KW5DJW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-alpha (Homo sapiens (Human)) | BDBM81916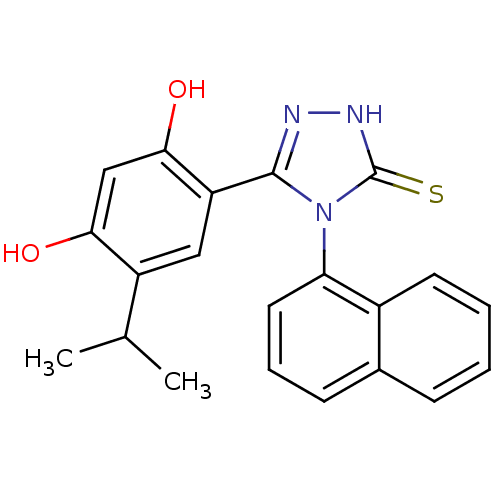 (lspropyl analog, 5) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | 7.4 | n/a |
Bayer Healthcare, | Assay Description To assess the affinity of compounds binding to Hsp90, we measured their ability to compete with the binding of a fluorescent analog of GA (GM-Bodipy)... | Chem Biol Drug Des 74: 43-50 (2009) Article DOI: 10.1111/j.1747-0285.2009.00833.x BindingDB Entry DOI: 10.7270/Q2KW5DJW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205961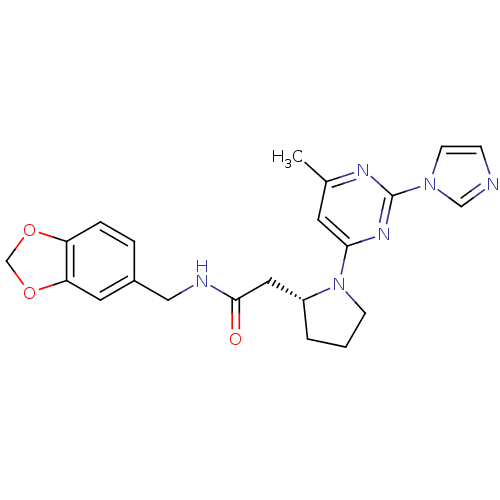 (2-((R)-1-(2-(1H-imidazol-1-yl)-6-methylpyrimidin-4...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205952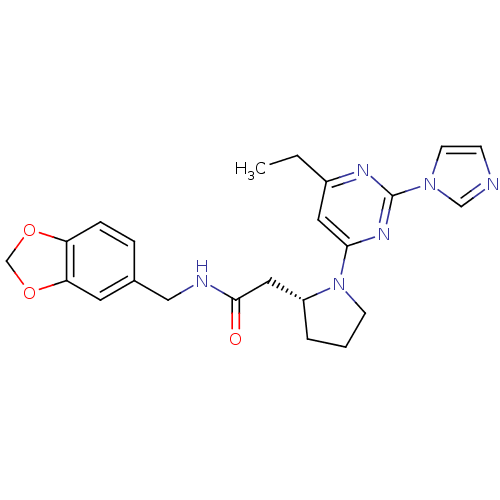 (2-((R)-1-(2-(1H-imidazol-1-yl)-6-methylpyrimidin-4...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-alpha (Homo sapiens (Human)) | BDBM81915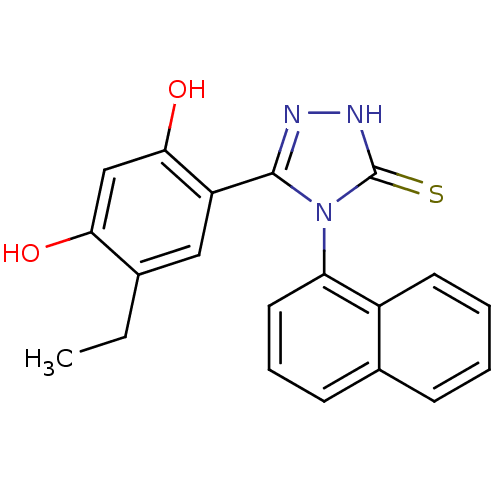 (Ethyl analog, 4) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | 7.4 | n/a |
Bayer Healthcare, | Assay Description To assess the affinity of compounds binding to Hsp90, we measured their ability to compete with the binding of a fluorescent analog of GA (GM-Bodipy)... | Chem Biol Drug Des 74: 43-50 (2009) Article DOI: 10.1111/j.1747-0285.2009.00833.x BindingDB Entry DOI: 10.7270/Q2KW5DJW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin-converting enzyme 1 (Homo sapiens (Human)) | BDBM50035247 (2-(2-Hydroxycarbamoyl-3-methyl-butyrylamino)-3-(1H...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Laboratories Curated by ChEMBL | Assay Description Inhibitory activity against human bronchiolar smooth muscle Endothelin-converting enzyme 1 | J Med Chem 38: 2119-29 (1995) BindingDB Entry DOI: 10.7270/Q2D79C2M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205910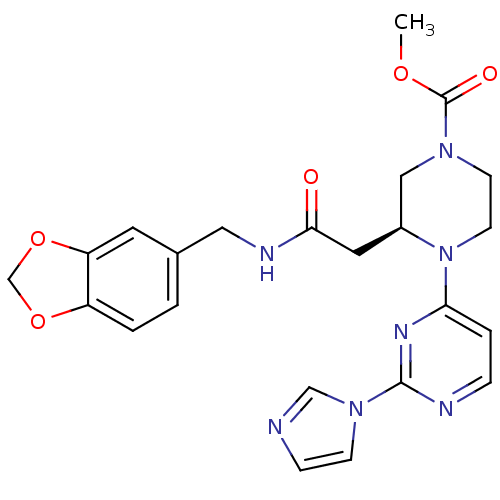 ((3S)-methyl 4-(2-(1H-imidazol-1-yl)pyrimidin-4-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 0.240 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Endothelin-converting enzyme 1 (Homo sapiens (Human)) | BDBM50035246 (2-(2-Hydroxycarbamoyl-4-methyl-pentanoylamino)-3-(...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.25 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Laboratories Curated by ChEMBL | Assay Description Inhibitory activity against human bronchiolar smooth muscle Endothelin-converting enzyme 1 | J Med Chem 38: 2119-29 (1995) BindingDB Entry DOI: 10.7270/Q2D79C2M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205950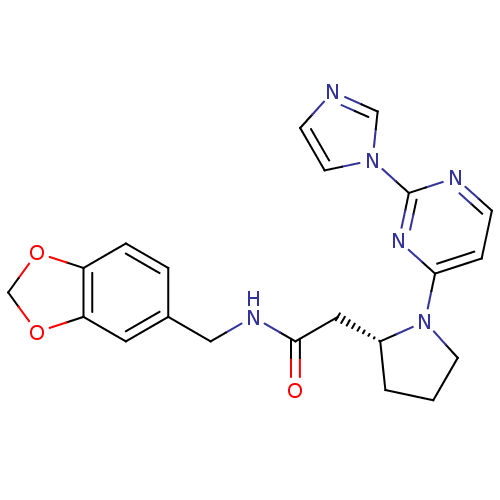 (2-((R)-1-(2-(1H-imidazol-1-yl)pyrimidin-4-yl)pyrro...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205937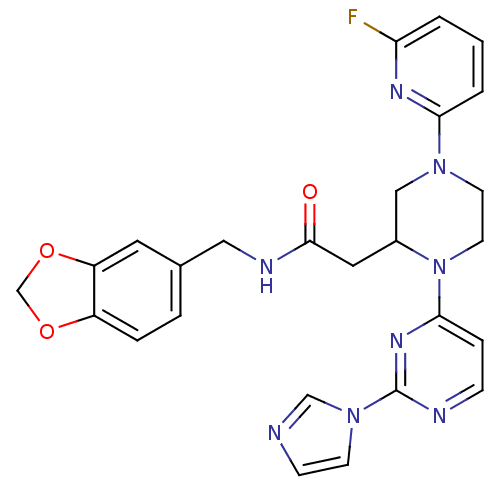 (CHEMBL385325 | N-[(1,3-benzodioxol-5-yl)methyl]-1-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205947 ((2R)-1-(2-(1H-imidazol-1-yl)-6-methylpyrimidin-4-y...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Heat shock protein HSP 90-alpha (Homo sapiens (Human)) | BDBM81912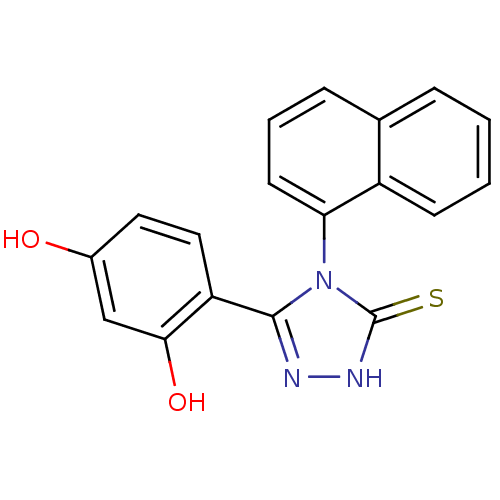 (DC23 | Resorcinol analog, 1) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | 7.4 | n/a |
Bayer Healthcare, | Assay Description To assess the affinity of compounds binding to Hsp90, we measured their ability to compete with the binding of a fluorescent analog of GA (GM-Bodipy)... | Chem Biol Drug Des 74: 43-50 (2009) Article DOI: 10.1111/j.1747-0285.2009.00833.x BindingDB Entry DOI: 10.7270/Q2KW5DJW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50111438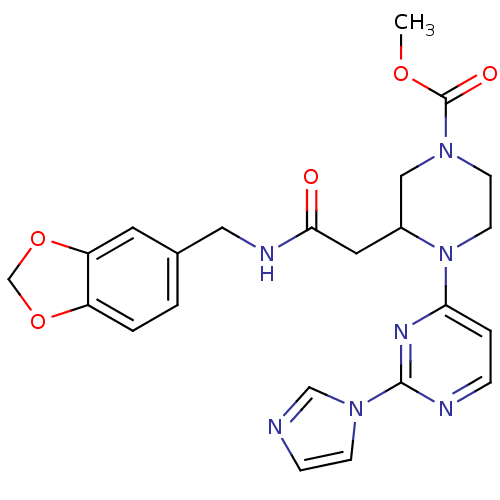 (3-{[(Benzo[1,3]dioxol-5-ylmethyl)-carbamoyl]-methy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 0.380 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Neprilysin (Oryctolagus cuniculus (rabbit)) | BDBM50035247 (2-(2-Hydroxycarbamoyl-3-methyl-butyrylamino)-3-(1H...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Laboratories Curated by ChEMBL | Assay Description Inhibitory activity against Leu-enkeph of Neutral endopeptidase | J Med Chem 38: 2119-29 (1995) BindingDB Entry DOI: 10.7270/Q2D79C2M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205911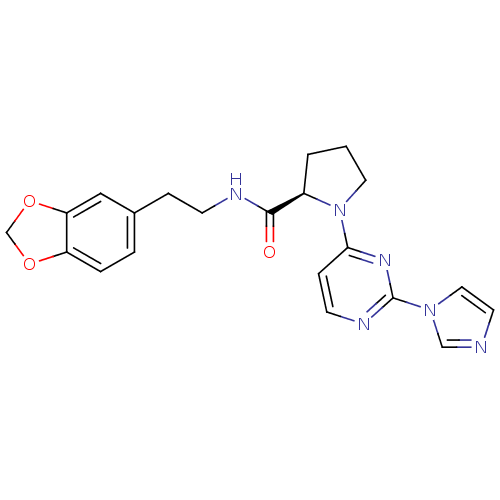 (2-((R)-1-(2-(1H-imidazol-1-yl)-6-methylpyrimidin-4...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.480 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205932 (CHEMBL373623 | N-[(1,3-benzodioxol-5-yl)methyl]-1-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.480 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205956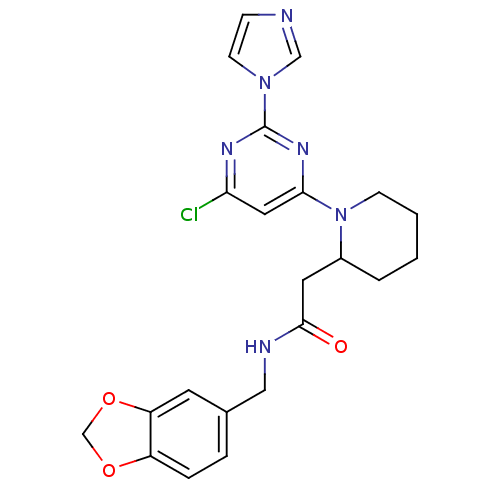 (CHEMBL223788 | N-(1,3-benzodioxol-5-ylmethyl)-1-[6...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205926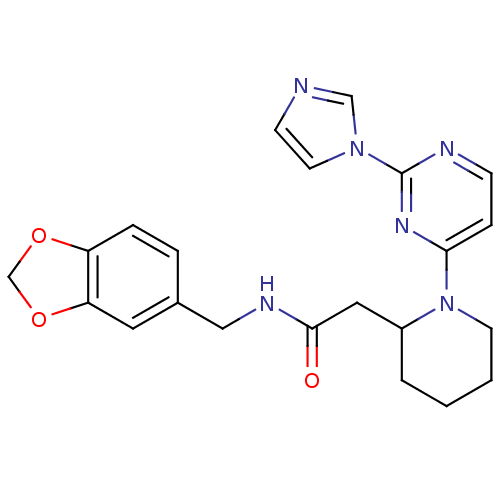 (CHEMBL442041 | N-[(1,3-benzodioxol-5-yl)methyl]-1-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205905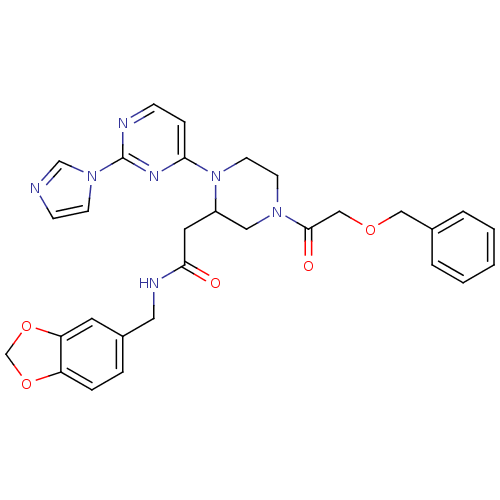 (CHEMBL376713 | N-[(1,3-benzodioxol-5-yl)methyl]-1-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Heat shock protein HSP 90-alpha (Homo sapiens (Human)) | BDBM81913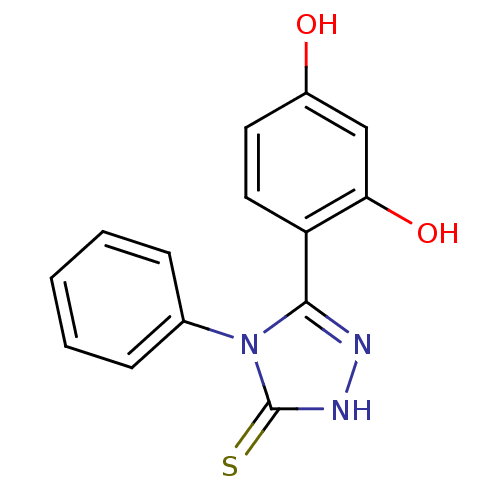 (Unsubstituted phenyl ring analog, 2) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | 7.4 | n/a |
Bayer Healthcare, | Assay Description To assess the affinity of compounds binding to Hsp90, we measured their ability to compete with the binding of a fluorescent analog of GA (GM-Bodipy)... | Chem Biol Drug Des 74: 43-50 (2009) Article DOI: 10.1111/j.1747-0285.2009.00833.x BindingDB Entry DOI: 10.7270/Q2KW5DJW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205957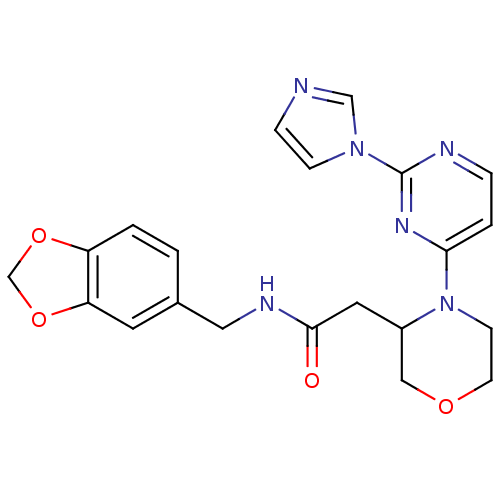 (CHEMBL220932 | N-[(1,3-benzodioxol-5-yl)methyl]-4-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205954 (CHEMBL385334 | N-[(1,3-benzodioxol-5-yl)methyl]-1-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205907 ((2R)-N-(2-(benzo[d][1,3]dioxol-5-yl)ethyl)-1-(6-et...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.520 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205973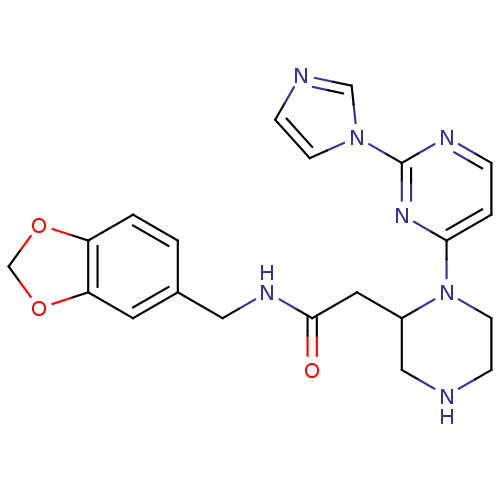 (CHEMBL411343 | N-[(1,3-benzodioxol-5-yl)methyl]-1-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.550 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205938 (CHEMBL424928 | N-[(1,3-benzodioxol-5-yl)methyl]-1-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.580 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205909 (CHEMBL223782 | N-(1,3-benzodioxol-5-ylmethyl)-4-(2...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.670 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205968 (CHEMBL373881 | N-[(1,3-benzodioxol-5-yl)methyl]-1-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205930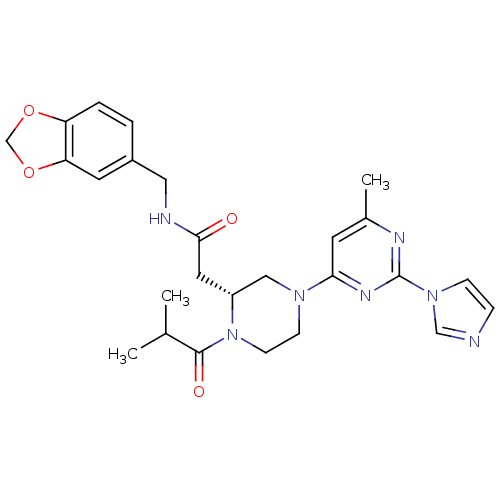 (2-((R)-4-(2-(1H-imidazol-1-yl)-6-methylpyrimidin-4...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.75 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205904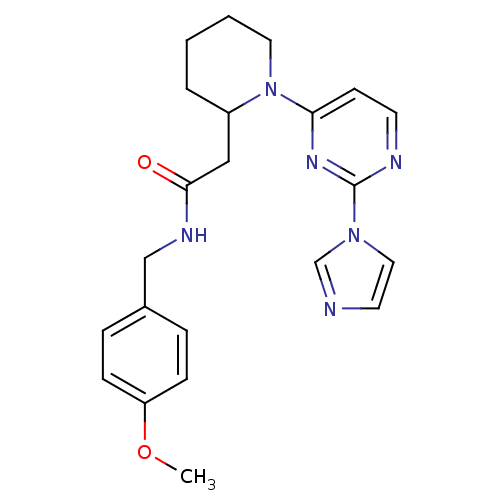 (1-[2-(1H-imidazol-1-yl)-4-pyrimidinyl]-N-[(4-metho...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205971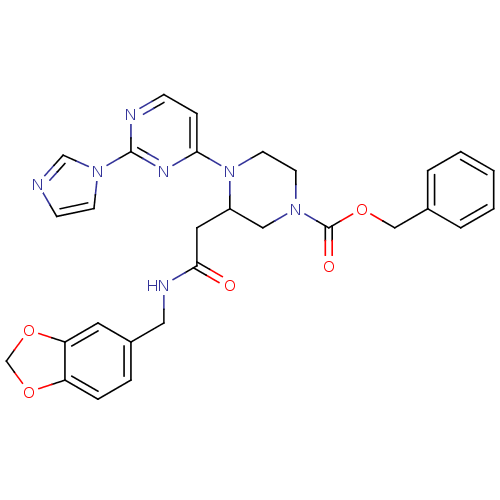 (CHEMBL223792 | N-[(1,3-benzodioxol-5-yl)methyl]-1-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin-converting enzyme 1 (Homo sapiens (Human)) | BDBM50251742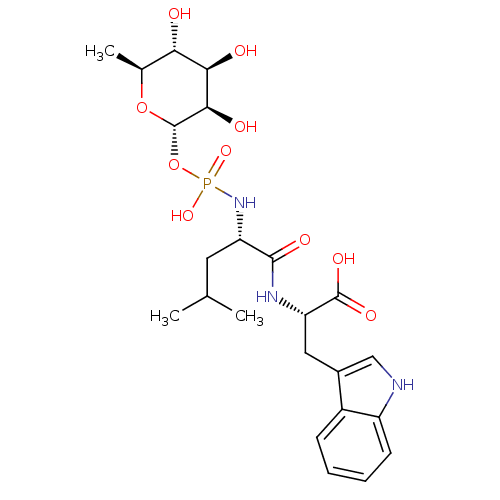 ((3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyl-tetrahydro...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB PubMed | n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Laboratories Curated by ChEMBL | Assay Description Inhibitory activity against human bronchiolar smooth muscle Endothelin-converting enzyme 1 | J Med Chem 38: 2119-29 (1995) BindingDB Entry DOI: 10.7270/Q2D79C2M | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205964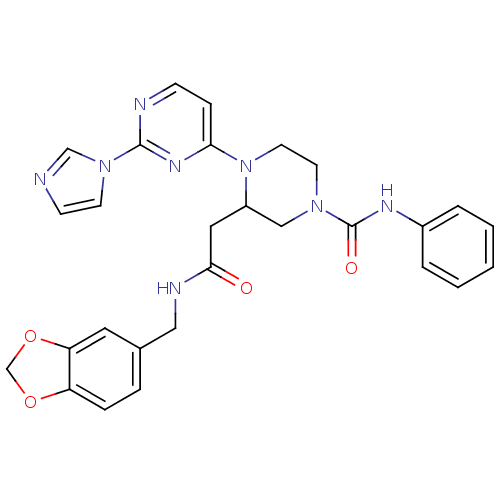 (CHEMBL374319 | N-(1,3-benzodioxol-5-ylmethyl)-1-[2...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.870 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205965 (CHEMBL222364 | N-[(1,3-benzodioxol-5-yl)methyl]-1-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.930 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205931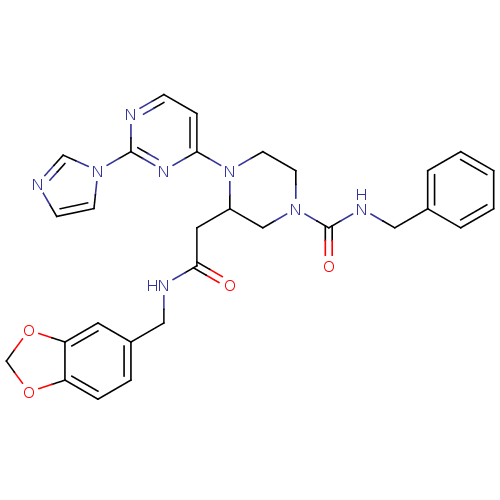 (CHEMBL223791 | N-(1,3-benzodioxol-5-ylmethyl)-1-[2...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.960 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205929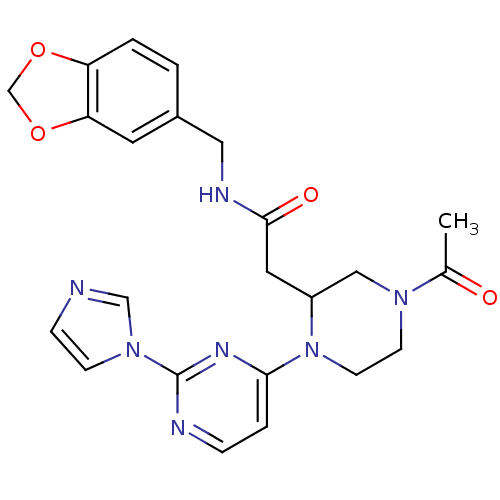 (4-(acetyl)-N-[(1,3-benzodioxol-5-yl)methyl]-1-[2-(...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205921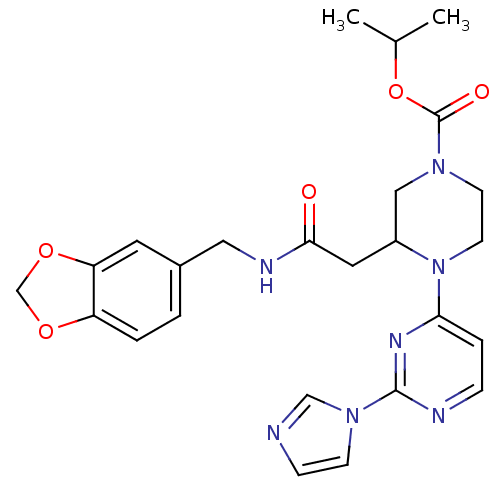 (3-[2-[(1,3-benzodioxol-5-ylmethyl)amino]-2-oxoethy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205920 (CHEMBL223634 | N-[(1,3-benzodioxol-5-yl)methyl]-1-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neprilysin (Oryctolagus cuniculus (rabbit)) | BDBM50035251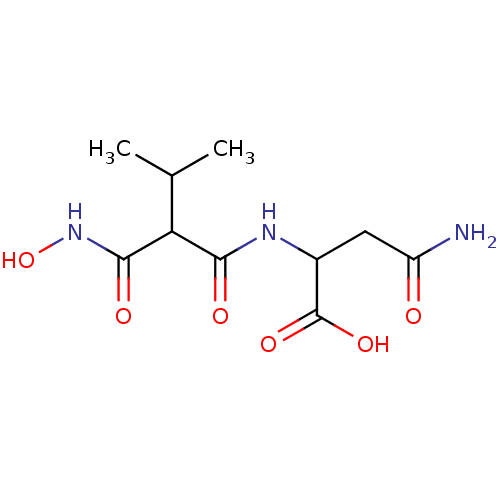 (2-(2-Hydroxycarbamoyl-3-methyl-butyrylamino)-succi...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Laboratories Curated by ChEMBL | Assay Description Inhibitory activity against Leu-enkeph of Neutral endopeptidase | J Med Chem 38: 2119-29 (1995) BindingDB Entry DOI: 10.7270/Q2D79C2M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205974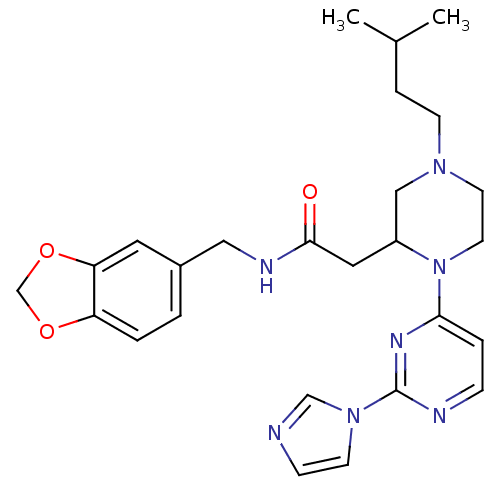 (CHEMBL375404 | N-[(1,3-benzodioxol-5-yl)methyl]-1-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neprilysin (Oryctolagus cuniculus (rabbit)) | BDBM50035236 (3-(2-Hydroxycarbamoyl-3-methyl-butyrylamino)-propi...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Laboratories Curated by ChEMBL | Assay Description Inhibitory activity against big ET-1 of Neutral endopeptidase | J Med Chem 38: 2119-29 (1995) BindingDB Entry DOI: 10.7270/Q2D79C2M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205924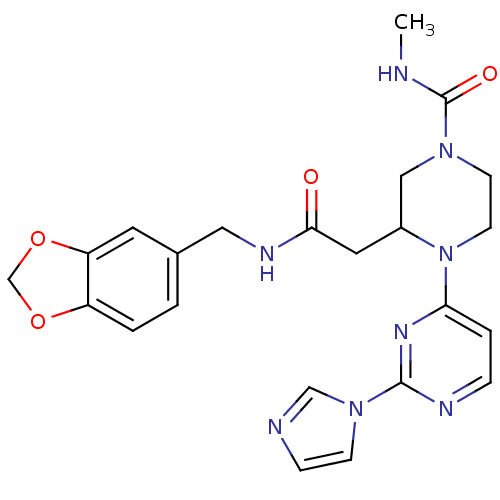 (CHEMBL223384 | N-[(1,3-benzodioxol-5-yl)methyl]-1-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neprilysin (Oryctolagus cuniculus (rabbit)) | BDBM50105264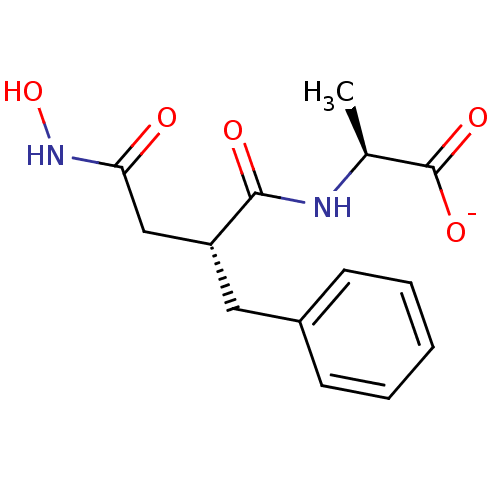 (2-(2-Benzyl-3-hydroxycarbamoyl-propionylamino)-pro...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase MCE PC cid PC sid PDB UniChem Similars | PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Laboratories Curated by ChEMBL | Assay Description Inhibitory activity against Leu-enkeph of Neutral endopeptidase | J Med Chem 38: 2119-29 (1995) BindingDB Entry DOI: 10.7270/Q2D79C2M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205976 (CHEMBL223838 | N-[(1,3-benzodioxol-5-yl)methyl]-4-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin-converting enzyme 1 (Homo sapiens (Human)) | BDBM50035239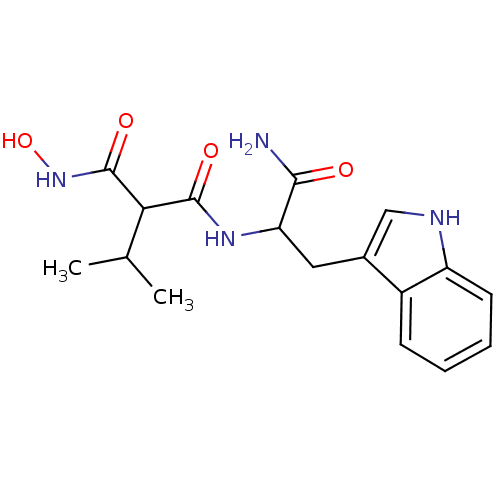 (CHEMBL63317 | N-[1-Carbamoyl-2-(1H-indol-3-yl)-eth...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Laboratories Curated by ChEMBL | Assay Description Inhibitory activity against human bronchiolar smooth muscle Endothelin-converting enzyme 1 | J Med Chem 38: 2119-29 (1995) BindingDB Entry DOI: 10.7270/Q2D79C2M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50205953 (3-[2-[(1,3-benzodioxol-5-ylmethyl)amino]-2-oxoethy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences Curated by ChEMBL | Assay Description Inhibition of cytokine-induced iNOS expressed in human A172 cells assessed as inhibition of NO formation | J Med Chem 50: 1146-57 (2007) Article DOI: 10.1021/jm061319i BindingDB Entry DOI: 10.7270/Q2SX6CWJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 247 total ) | Next | Last >> |