 Found 317 hits with Last Name = 'centrella' and Initial = 'pa'
Found 317 hits with Last Name = 'centrella' and Initial = 'pa' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM8610

(1-[4-(4-{[(2R,4S)-2-(2,4-dichlorophenyl)-2-(1H-imi...)Show SMILES [H][C@]1(COc2ccc(cc2)N2CCN(CC2)C(C)=O)CO[C@@](Cn2ccnc2)(O1)c1ccc(Cl)cc1Cl |r| Show InChI InChI=1S/C26H28Cl2N4O4/c1-19(33)31-10-12-32(13-11-31)21-3-5-22(6-4-21)34-15-23-16-35-26(36-23,17-30-9-8-29-18-30)24-7-2-20(27)14-25(24)28/h2-9,14,18,23H,10-13,15-17H2,1H3/t23-,26-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 activity in human liver microsomes using midazolam as a substrate by LC/MS analysis |
Drug Metab Dispos 41: 814-26 (2013)
Article DOI: 10.1124/dmd.112.048355
BindingDB Entry DOI: 10.7270/Q2NC62XH |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM8610

(1-[4-(4-{[(2R,4S)-2-(2,4-dichlorophenyl)-2-(1H-imi...)Show SMILES [H][C@]1(COc2ccc(cc2)N2CCN(CC2)C(C)=O)CO[C@@](Cn2ccnc2)(O1)c1ccc(Cl)cc1Cl |r| Show InChI InChI=1S/C26H28Cl2N4O4/c1-19(33)31-10-12-32(13-11-31)21-3-5-22(6-4-21)34-15-23-16-35-26(36-23,17-30-9-8-29-18-30)24-7-2-20(27)14-25(24)28/h2-9,14,18,23H,10-13,15-17H2,1H3/t23-,26-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 activity in human liver microsomes using nifedipine as a substrate by LC/MS analysis |
Drug Metab Dispos 41: 814-26 (2013)
Article DOI: 10.1124/dmd.112.048355
BindingDB Entry DOI: 10.7270/Q2NC62XH |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM8610

(1-[4-(4-{[(2R,4S)-2-(2,4-dichlorophenyl)-2-(1H-imi...)Show SMILES [H][C@]1(COc2ccc(cc2)N2CCN(CC2)C(C)=O)CO[C@@](Cn2ccnc2)(O1)c1ccc(Cl)cc1Cl |r| Show InChI InChI=1S/C26H28Cl2N4O4/c1-19(33)31-10-12-32(13-11-31)21-3-5-22(6-4-21)34-15-23-16-35-26(36-23,17-30-9-8-29-18-30)24-7-2-20(27)14-25(24)28/h2-9,14,18,23H,10-13,15-17H2,1H3/t23-,26-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 49 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 activity in human liver microsomes using testosterone as a substrate by LC/MS analysis |
Drug Metab Dispos 41: 814-26 (2013)
Article DOI: 10.1124/dmd.112.048355
BindingDB Entry DOI: 10.7270/Q2NC62XH |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50568221
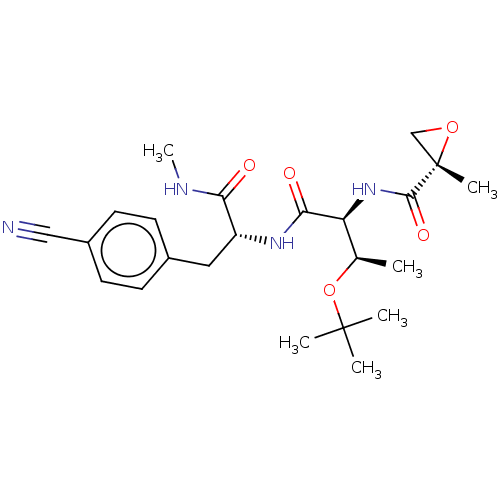
(CHEMBL4854947)Show SMILES CNC(=O)[C@@H](Cc1ccc(cc1)C#N)NC(=O)[C@@H](NC(=O)[C@]1(C)CO1)[C@@H](C)OC(C)(C)C |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 1.25E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His-tagged BTK expressed in baculovirus infected in Sf9 cells assessed as inhibitory constant incubated fo... |
Citation and Details
Article DOI: 10.1016/j.bmc.2021.116223
BindingDB Entry DOI: 10.7270/Q2M04964 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50568222
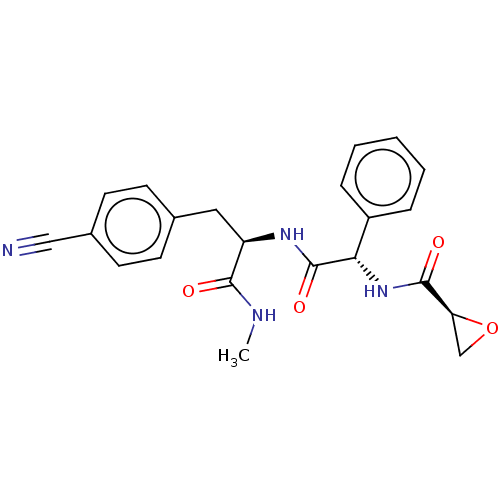
(CHEMBL4867490)Show SMILES CNC(=O)[C@@H](Cc1ccc(cc1)C#N)NC(=O)[C@@H](NC(=O)[C@@H]1CO1)c1ccccc1 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.93E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His-tagged BTK expressed in baculovirus infected in Sf9 cells assessed as inhibitory constant incubated fo... |
Citation and Details
Article DOI: 10.1016/j.bmc.2021.116223
BindingDB Entry DOI: 10.7270/Q2M04964 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50568220
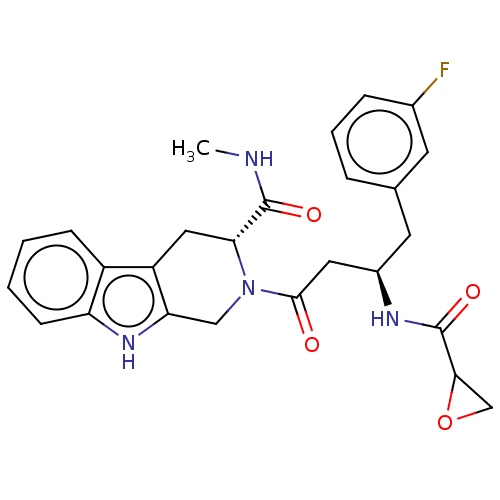
(CHEMBL4873655)Show SMILES CNC(=O)[C@H]1Cc2c(CN1C(=O)C[C@@H](Cc1cccc(F)c1)NC(=O)C1CO1)[nH]c1ccccc21 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 9.27E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His-tagged BTK expressed in baculovirus infected in Sf9 cells assessed as inhibitory constant incubated fo... |
Citation and Details
Article DOI: 10.1016/j.bmc.2021.116223
BindingDB Entry DOI: 10.7270/Q2M04964 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50088527
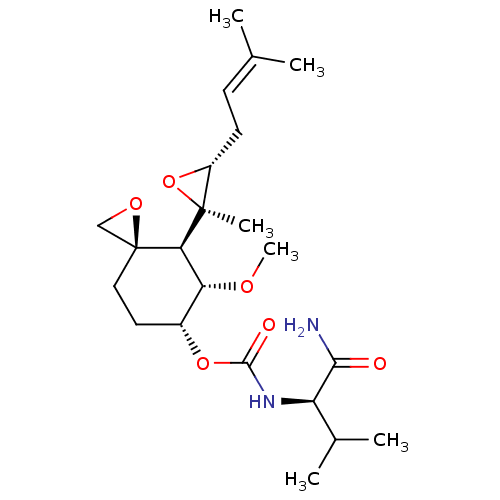
(CHEMBL3527358)Show SMILES [H][C@@]1([#6@H](-[#8]-[#6])-[#6@@H](-[#6]-[#6][C@]11[#6]-[#8]1)-[#8]-[#6](=O)-[#7]-[#6@H](-[#6](-[#6])-[#6])-[#6](-[#7])=O)[C@@]1([#6])[#8]-[#6@@H]1-[#6]\[#6]=[#6](\[#6])-[#6] |r| Show InChI InChI=1S/C22H36N2O6/c1-12(2)7-8-15-21(5,30-15)18-17(27-6)14(9-10-22(18)11-28-22)29-20(26)24-16(13(3)4)19(23)25/h7,13-18H,8-11H2,1-6H3,(H2,23,25)(H,24,26)/t14-,15-,16-,17-,18-,21+,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 activity in human liver microsomes using midazolam as a substrate by LC/MS analysis |
Drug Metab Dispos 41: 814-26 (2013)
Article DOI: 10.1124/dmd.112.048355
BindingDB Entry DOI: 10.7270/Q2NC62XH |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50088527

(CHEMBL3527358)Show SMILES [H][C@@]1([#6@H](-[#8]-[#6])-[#6@@H](-[#6]-[#6][C@]11[#6]-[#8]1)-[#8]-[#6](=O)-[#7]-[#6@H](-[#6](-[#6])-[#6])-[#6](-[#7])=O)[C@@]1([#6])[#8]-[#6@@H]1-[#6]\[#6]=[#6](\[#6])-[#6] |r| Show InChI InChI=1S/C22H36N2O6/c1-12(2)7-8-15-21(5,30-15)18-17(27-6)14(9-10-22(18)11-28-22)29-20(26)24-16(13(3)4)19(23)25/h7,13-18H,8-11H2,1-6H3,(H2,23,25)(H,24,26)/t14-,15-,16-,17-,18-,21+,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.87E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 activity in human liver microsomes using testosterone as a substrate by LC/MS analysis |
Drug Metab Dispos 41: 814-26 (2013)
Article DOI: 10.1124/dmd.112.048355
BindingDB Entry DOI: 10.7270/Q2NC62XH |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50088527

(CHEMBL3527358)Show SMILES [H][C@@]1([#6@H](-[#8]-[#6])-[#6@@H](-[#6]-[#6][C@]11[#6]-[#8]1)-[#8]-[#6](=O)-[#7]-[#6@H](-[#6](-[#6])-[#6])-[#6](-[#7])=O)[C@@]1([#6])[#8]-[#6@@H]1-[#6]\[#6]=[#6](\[#6])-[#6] |r| Show InChI InChI=1S/C22H36N2O6/c1-12(2)7-8-15-21(5,30-15)18-17(27-6)14(9-10-22(18)11-28-22)29-20(26)24-16(13(3)4)19(23)25/h7,13-18H,8-11H2,1-6H3,(H2,23,25)(H,24,26)/t14-,15-,16-,17-,18-,21+,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.03E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 activity in human liver microsomes using nifedipine as a substrate by LC/MS analysis |
Drug Metab Dispos 41: 814-26 (2013)
Article DOI: 10.1124/dmd.112.048355
BindingDB Entry DOI: 10.7270/Q2NC62XH |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM225238
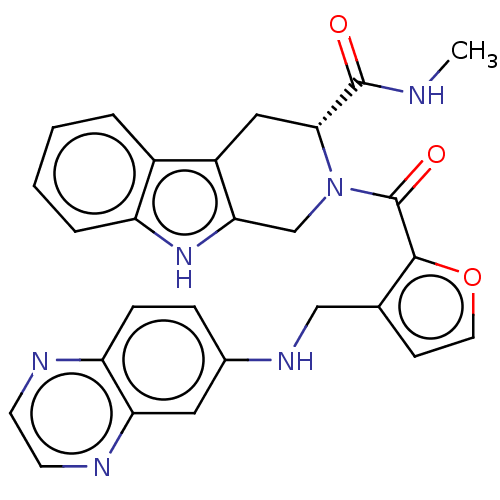
(BTK inhibitor, 3)Show SMILES CNC(=O)[C@H]1Cc2c(CN1C(=O)c1occc1CNc1ccc3nccnc3c1)[nH]c1ccccc21 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 0.550 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant wild type human N-terminal His6-tagged BTK expressed in baculovirus incubated for 20 mins by TR-FRET based competitive bind... |
Citation and Details
Article DOI: 10.1016/j.bmc.2021.116223
BindingDB Entry DOI: 10.7270/Q2M04964 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM225238

(BTK inhibitor, 3)Show SMILES CNC(=O)[C@H]1Cc2c(CN1C(=O)c1occc1CNc1ccc3nccnc3c1)[nH]c1ccccc21 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 0.550 | n/a | n/a | n/a | n/a | n/a | 30 |
X-Chem Pharmaceuticals
| Assay Description
The BTK time-resolved FRET-based competitive binding assay and cell-based BTK assays have been previously described.[Xu et al., J.Pharmacol. Exp. The... |
Chembiochem 18: 864-871 (2017)
Article DOI: 10.1002/cbic.201600573
BindingDB Entry DOI: 10.7270/Q22J69Q5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Bifunctional epoxide hydrolase 2
(Homo sapiens (Human)) | BDBM50566635
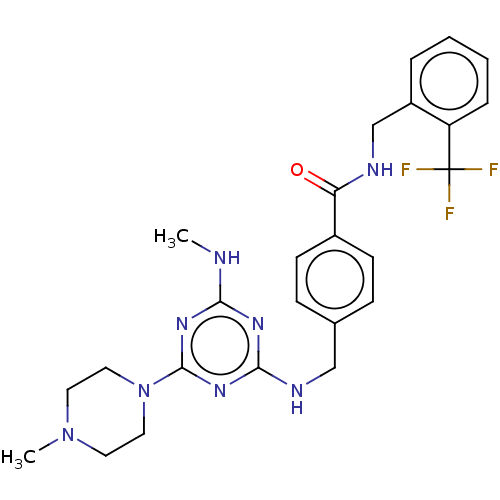
(CHEMBL4875337)Show SMILES CNc1nc(NCc2ccc(cc2)C(=O)NCc2ccccc2C(F)(F)F)nc(n1)N1CCN(C)CC1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human sEH using EnzChek as substrate preincubated with enzyme for 5 mins followed by substrate addition and measured every 30 secs for ... |
Citation and Details
Article DOI: 10.1016/j.bmc.2021.116216
BindingDB Entry DOI: 10.7270/Q22J6GNB |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50168737

((2R,3R)-1-[4-(2-Chloro-4-fluoro-benzyloxy)-benzene...)Show SMILES C[C@@]1(O)CCCN([C@H]1C(=O)NO)S(=O)(=O)c1ccc(OCc2ccc(F)cc2Cl)cc1 Show InChI InChI=1S/C20H22ClFN2O6S/c1-20(26)9-2-10-24(18(20)19(25)23-27)31(28,29)16-7-5-15(6-8-16)30-12-13-3-4-14(22)11-17(13)21/h3-8,11,18,26-27H,2,9-10,12H2,1H3,(H,23,25)/t18-,20+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of MMP-13 using 5-FAM-TPGPLGL[Dap- (DNP)]ARRK(5-TAMRA)-amide as substrate after 45 mins |
J Med Chem 55: 7061-79 (2012)
Article DOI: 10.1021/jm300449x
BindingDB Entry DOI: 10.7270/Q2RX9D6T |
More data for this
Ligand-Target Pair | |
Bifunctional epoxide hydrolase 2
(Homo sapiens (Human)) | BDBM50566638
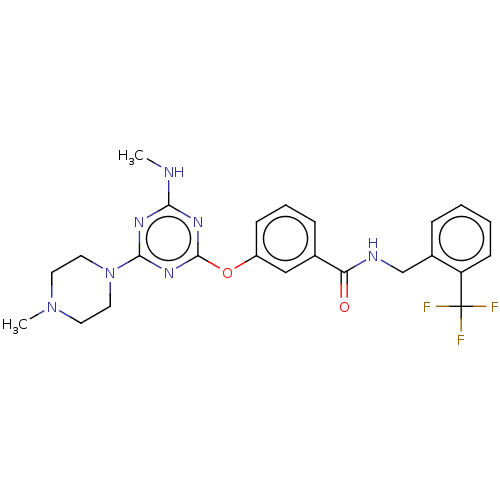
(CHEMBL4862566)Show SMILES CNc1nc(Oc2cccc(c2)C(=O)NCc2ccccc2C(F)(F)F)nc(n1)N1CCN(C)CC1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human sEH using EnzChek as substrate preincubated with enzyme for 5 mins followed by substrate addition and measured every 30 secs for ... |
Citation and Details
Article DOI: 10.1016/j.bmc.2021.116216
BindingDB Entry DOI: 10.7270/Q22J6GNB |
More data for this
Ligand-Target Pair | |
Disintegrin and metalloproteinase domain-containing protein 17
(Homo sapiens (Human)) | BDBM50168737

((2R,3R)-1-[4-(2-Chloro-4-fluoro-benzyloxy)-benzene...)Show SMILES C[C@@]1(O)CCCN([C@H]1C(=O)NO)S(=O)(=O)c1ccc(OCc2ccc(F)cc2Cl)cc1 Show InChI InChI=1S/C20H22ClFN2O6S/c1-20(26)9-2-10-24(18(20)19(25)23-27)31(28,29)16-7-5-15(6-8-16)30-12-13-3-4-14(22)11-17(13)21/h3-8,11,18,26-27H,2,9-10,12H2,1H3,(H,23,25)/t18-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of TACE using Mca-PLAQAV-Dpa-RSSSR-NH2 as substrate preincubated 15 mins measured every 30 sec for 30 mins |
J Med Chem 55: 7061-79 (2012)
Article DOI: 10.1021/jm300449x
BindingDB Entry DOI: 10.7270/Q2RX9D6T |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM36463
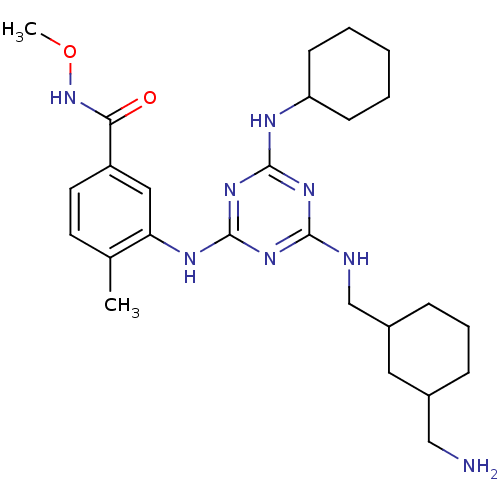
(3-(4-((3-(Aminomethyl)cyclohexyl)methylamino)-6-(3...)Show SMILES CONC(=O)c1ccc(C)c(Nc2nc(NCC3CCCC(CN)C3)nc(NC3CCCCC3)n2)c1 Show InChI InChI=1S/C26H40N8O2/c1-17-11-12-20(23(35)34-36-2)14-22(17)30-26-32-24(28-16-19-8-6-7-18(13-19)15-27)31-25(33-26)29-21-9-4-3-5-10-21/h11-12,14,18-19,21H,3-10,13,15-16,27H2,1-2H3,(H,34,35)(H3,28,29,30,31,32,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | <2 | n/a | n/a | n/a | n/a | 9.5 | 16 |
Praecis Pharmaceuticals
| Assay Description
Selection of DNA-encoded libraries (DELs), which are covalent attachment of encoding double stranded DNA to small-molecule created using a combinatio... |
Nat Chem Biol 5: 647-54 (2009)
Article DOI: 10.1038/nchembio.211
BindingDB Entry DOI: 10.7270/Q2MP51NX |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM36462
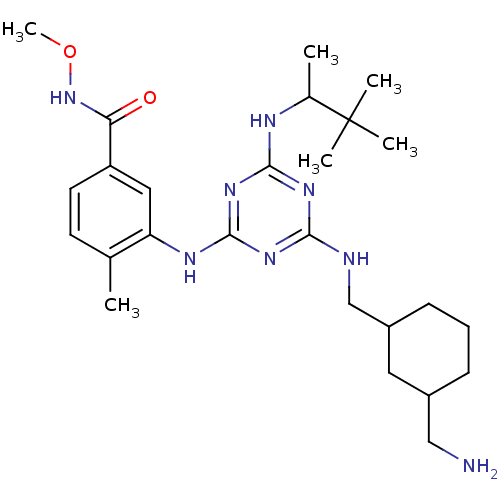
(3-(4-((3-(Aminomethyl)cyclohexyl)methylamino)-6-(3...)Show SMILES CONC(=O)c1ccc(C)c(Nc2nc(NCC3CCCC(CN)C3)nc(NC(C)C(C)(C)C)n2)c1 Show InChI InChI=1S/C26H42N8O2/c1-16-10-11-20(22(35)34-36-6)13-21(16)30-25-32-23(28-15-19-9-7-8-18(12-19)14-27)31-24(33-25)29-17(2)26(3,4)5/h10-11,13,17-19H,7-9,12,14-15,27H2,1-6H3,(H,34,35)(H3,28,29,30,31,32,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <2 | n/a | n/a | n/a | n/a | 9.5 | 16 |
Praecis Pharmaceuticals
| Assay Description
Selection of DNA-encoded libraries (DELs), which are covalent attachment of encoding double stranded DNA to small-molecule created using a combinatio... |
Nat Chem Biol 5: 647-54 (2009)
Article DOI: 10.1038/nchembio.211
BindingDB Entry DOI: 10.7270/Q2MP51NX |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448867

(CHEMBL3125235)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(=O)NCc2ccccn2)n(CC)n1 |r| Show InChI InChI=1S/C28H30N6O4/c1-3-18-13-24(34(4-2)32-18)27(36)31-20-14-23(26(35)30-15-19-9-7-8-12-29-19)33(16-20)28(37)22-17-38-25-11-6-5-10-21(22)25/h5-13,17,20,23H,3-4,14-16H2,1-2H3,(H,30,35)(H,31,36)/t20-,23+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50091691
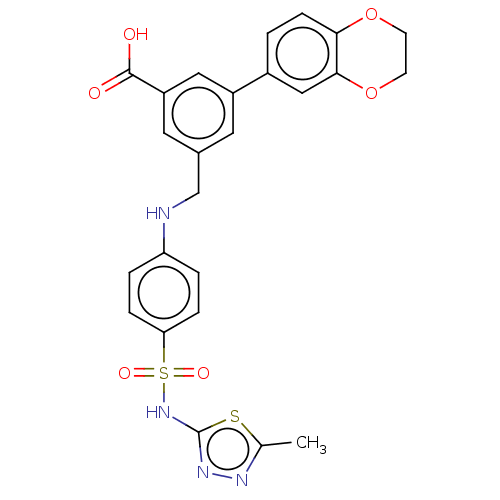
(CHEMBL3582356)Show SMILES Cc1nnc(NS(=O)(=O)c2ccc(NCc3cc(cc(c3)-c3ccc4OCCOc4c3)C(O)=O)cc2)s1 Show InChI InChI=1S/C25H22N4O6S2/c1-15-27-28-25(36-15)29-37(32,33)21-5-3-20(4-6-21)26-14-16-10-18(12-19(11-16)24(30)31)17-2-7-22-23(13-17)35-9-8-34-22/h2-7,10-13,26H,8-9,14H2,1H3,(H,28,29)(H,30,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) |
ACS Med Chem Lett 6: 531-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00025
BindingDB Entry DOI: 10.7270/Q2BP04JF |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448871

(CHEMBL3125101)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(=O)NCc2ccc3COB(O)c3c2)n(CC)n1 |r| Show InChI InChI=1S/C30H32BN5O6/c1-3-20-12-26(36(4-2)34-20)29(38)33-21-13-25(28(37)32-14-18-9-10-19-16-42-31(40)24(19)11-18)35(15-21)30(39)23-17-41-27-8-6-5-7-22(23)27/h5-12,17,21,25,40H,3-4,13-16H2,1-2H3,(H,32,37)(H,33,38)/t21-,25+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50091689
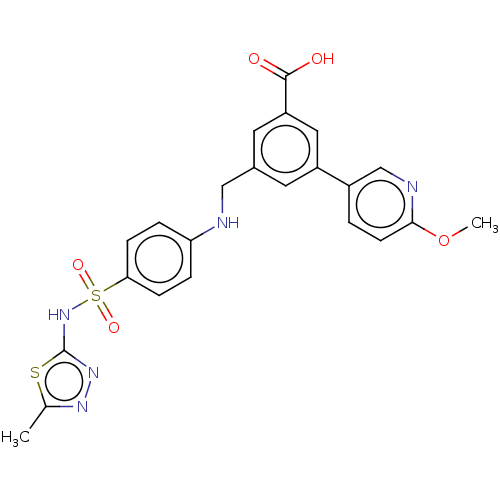
(CHEMBL3582354)Show SMILES COc1ccc(cn1)-c1cc(CNc2ccc(cc2)S(=O)(=O)Nc2nnc(C)s2)cc(c1)C(O)=O Show InChI InChI=1S/C23H21N5O5S2/c1-14-26-27-23(34-14)28-35(31,32)20-6-4-19(5-7-20)24-12-15-9-17(11-18(10-15)22(29)30)16-3-8-21(33-2)25-13-16/h3-11,13,24H,12H2,1-2H3,(H,27,28)(H,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) |
ACS Med Chem Lett 6: 531-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00025
BindingDB Entry DOI: 10.7270/Q2BP04JF |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448888

(CHEMBL3125253)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(=O)Nc2cnn(CC)n2)n(C)n1 |r| Show InChI InChI=1S/C25H28N8O4/c1-4-15-10-19(31(3)29-15)23(34)27-16-11-20(24(35)28-22-12-26-33(5-2)30-22)32(13-16)25(36)18-14-37-21-9-7-6-8-17(18)21/h6-10,12,14,16,20H,4-5,11,13H2,1-3H3,(H,27,34)(H,28,30,35)/t16-,20+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM225238

(BTK inhibitor, 3)Show SMILES CNC(=O)[C@H]1Cc2c(CN1C(=O)c1occc1CNc1ccc3nccnc3c1)[nH]c1ccccc21 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His-tagged BTK C481S mutant expressed in baculovirus infected in Sf9 cells incubated for 1 hr by TR-FRET a... |
Citation and Details
Article DOI: 10.1016/j.bmc.2021.116223
BindingDB Entry DOI: 10.7270/Q2M04964 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Bifunctional epoxide hydrolase 2
(Homo sapiens (Human)) | BDBM50539700
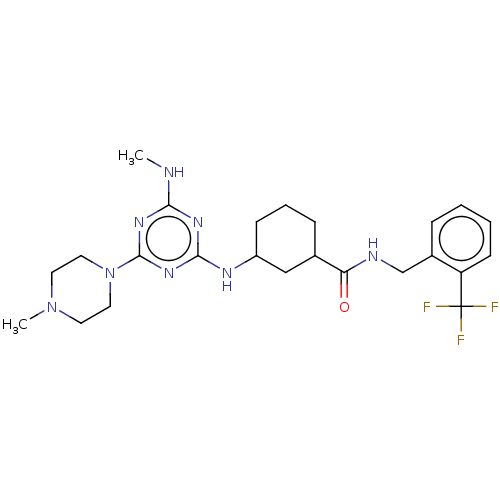
(CHEMBL4637413)Show SMILES CNc1nc(NC2CCCC(C2)C(=O)NCc2ccccc2C(F)(F)F)nc(n1)N1CCN(C)CC1 Show InChI InChI=1S/C24H33F3N8O/c1-28-21-31-22(33-23(32-21)35-12-10-34(2)11-13-35)30-18-8-5-7-16(14-18)20(36)29-15-17-6-3-4-9-19(17)24(25,26)27/h3-4,6,9,16,18H,5,7-8,10-15H2,1-2H3,(H,29,36)(H2,28,30,31,32,33) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human sEH using EnzChek as substrate preincubated with enzyme for 5 mins followed by substrate addition and measured every 30 secs for ... |
Citation and Details
Article DOI: 10.1016/j.bmc.2021.116216
BindingDB Entry DOI: 10.7270/Q22J6GNB |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448880
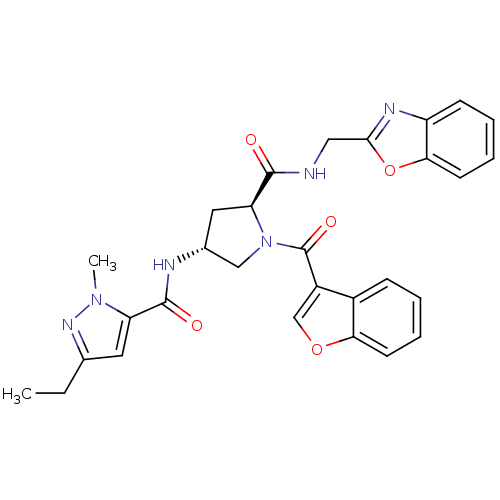
(CHEMBL3125261)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(=O)NCc2nc3ccccc3o2)n(C)n1 |r| Show InChI InChI=1S/C29H28N6O5/c1-3-17-12-22(34(2)33-17)28(37)31-18-13-23(27(36)30-14-26-32-21-9-5-7-11-25(21)40-26)35(15-18)29(38)20-16-39-24-10-6-4-8-19(20)24/h4-12,16,18,23H,3,13-15H2,1-2H3,(H,30,36)(H,31,37)/t18-,23+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448875

(CHEMBL3125266)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(=O)NCC(=O)OC)n(CC)n1 |r| Show InChI InChI=1S/C25H29N5O6/c1-4-15-10-20(30(5-2)28-15)24(33)27-16-11-19(23(32)26-12-22(31)35-3)29(13-16)25(34)18-14-36-21-9-7-6-8-17(18)21/h6-10,14,16,19H,4-5,11-13H2,1-3H3,(H,26,32)(H,27,33)/t16-,19+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |
A disintegrin and metalloproteinase with thrombospondin motifs 4
(Homo sapiens (Human)) | BDBM50168737

((2R,3R)-1-[4-(2-Chloro-4-fluoro-benzyloxy)-benzene...)Show SMILES C[C@@]1(O)CCCN([C@H]1C(=O)NO)S(=O)(=O)c1ccc(OCc2ccc(F)cc2Cl)cc1 Show InChI InChI=1S/C20H22ClFN2O6S/c1-20(26)9-2-10-24(18(20)19(25)23-27)31(28,29)16-7-5-15(6-8-16)30-12-13-3-4-14(22)11-17(13)21/h3-8,11,18,26-27H,2,9-10,12H2,1H3,(H,23,25)/t18-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of ADAMTS-4 using WAAG-3R as substrate preincubated for 15 mins measured after 1 hr |
J Med Chem 55: 7061-79 (2012)
Article DOI: 10.1021/jm300449x
BindingDB Entry DOI: 10.7270/Q2RX9D6T |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448868

(CHEMBL3125104)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(=O)NCCO)n(CC)n1 |r| Show InChI InChI=1S/C24H29N5O5/c1-3-15-11-20(29(4-2)27-15)23(32)26-16-12-19(22(31)25-9-10-30)28(13-16)24(33)18-14-34-21-8-6-5-7-17(18)21/h5-8,11,14,16,19,30H,3-4,9-10,12-13H2,1-2H3,(H,25,31)(H,26,32)/t16-,19+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |
Bifunctional epoxide hydrolase 2
(Homo sapiens (Human)) | BDBM50566636

(CHEMBL4870025)Show SMILES CNc1nc(NCc2cc(oc2C)C(=O)NCc2ccccc2C(F)(F)F)nc(n1)N1CCN(C)CC1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human sEH using EnzChek as substrate preincubated with enzyme for 5 mins followed by substrate addition and measured every 30 secs for ... |
Citation and Details
Article DOI: 10.1016/j.bmc.2021.116216
BindingDB Entry DOI: 10.7270/Q22J6GNB |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM225238

(BTK inhibitor, 3)Show SMILES CNC(=O)[C@H]1Cc2c(CN1C(=O)c1occc1CNc1ccc3nccnc3c1)[nH]c1ccccc21 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 3.80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human N-terminal His-tagged BTK expressed in baculovirus infected in Sf9 cells incubated for 1 hr by TR-FRET assay |
Citation and Details
Article DOI: 10.1016/j.bmc.2021.116223
BindingDB Entry DOI: 10.7270/Q2M04964 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448870
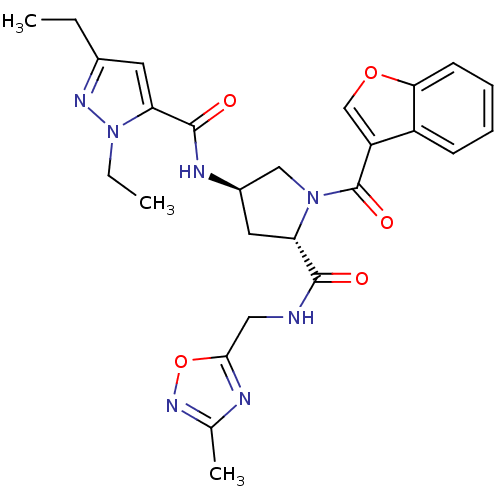
(CHEMBL3125102)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(=O)NCc2nc(C)no2)n(CC)n1 |r| Show InChI InChI=1S/C26H29N7O5/c1-4-16-10-21(33(5-2)30-16)25(35)29-17-11-20(24(34)27-12-23-28-15(3)31-38-23)32(13-17)26(36)19-14-37-22-9-7-6-8-18(19)22/h6-10,14,17,20H,4-5,11-13H2,1-3H3,(H,27,34)(H,29,35)/t17-,20+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448885

(CHEMBL3125256)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(=O)Nc2ccn(Cc3c(F)cccc3F)n2)n(C)n1 |r| Show InChI InChI=1S/C31H29F2N7O4/c1-3-18-13-25(38(2)36-18)29(41)34-19-14-26(40(15-19)31(43)22-17-44-27-10-5-4-7-20(22)27)30(42)35-28-11-12-39(37-28)16-21-23(32)8-6-9-24(21)33/h4-13,17,19,26H,3,14-16H2,1-2H3,(H,34,41)(H,35,37,42)/t19-,26+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448866
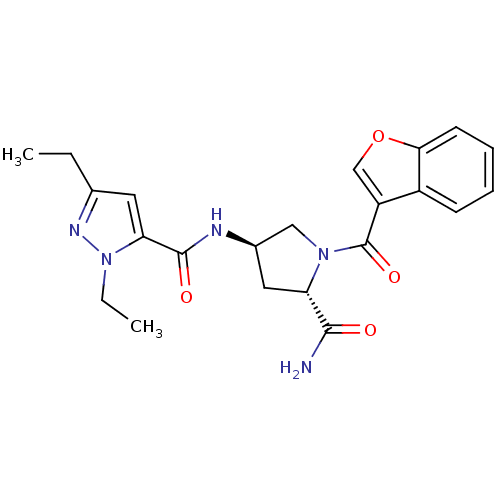
(CHEMBL3125270)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(N)=O)n(CC)n1 |r| Show InChI InChI=1S/C22H25N5O4/c1-3-13-9-18(27(4-2)25-13)21(29)24-14-10-17(20(23)28)26(11-14)22(30)16-12-31-19-8-6-5-7-15(16)19/h5-9,12,14,17H,3-4,10-11H2,1-2H3,(H2,23,28)(H,24,29)/t14-,17+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448869
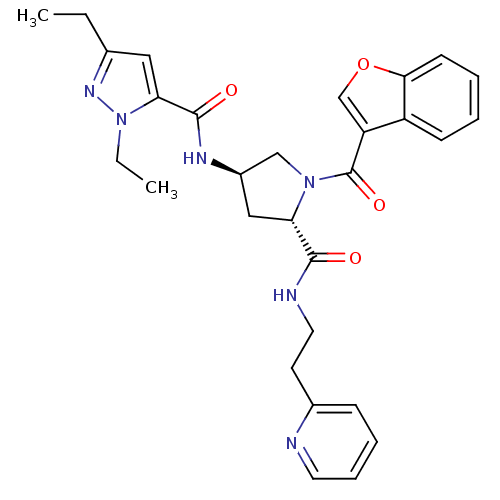
(CHEMBL3125103)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(=O)NCCc2ccccn2)n(CC)n1 |r| Show InChI InChI=1S/C29H32N6O4/c1-3-19-15-25(35(4-2)33-19)28(37)32-21-16-24(27(36)31-14-12-20-9-7-8-13-30-20)34(17-21)29(38)23-18-39-26-11-6-5-10-22(23)26/h5-11,13,15,18,21,24H,3-4,12,14,16-17H2,1-2H3,(H,31,36)(H,32,37)/t21-,24+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50091692

(CHEMBL3582357)Show SMILES Cc1nnc(NS(=O)(=O)c2ccc(NCc3cc(cc(c3)-c3ccc4OCOc4c3)C(O)=O)cc2)s1 Show InChI InChI=1S/C24H20N4O6S2/c1-14-26-27-24(35-14)28-36(31,32)20-5-3-19(4-6-20)25-12-15-8-17(10-18(9-15)23(29)30)16-2-7-21-22(11-16)34-13-33-21/h2-11,25H,12-13H2,1H3,(H,27,28)(H,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) |
ACS Med Chem Lett 6: 531-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00025
BindingDB Entry DOI: 10.7270/Q2BP04JF |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50091696
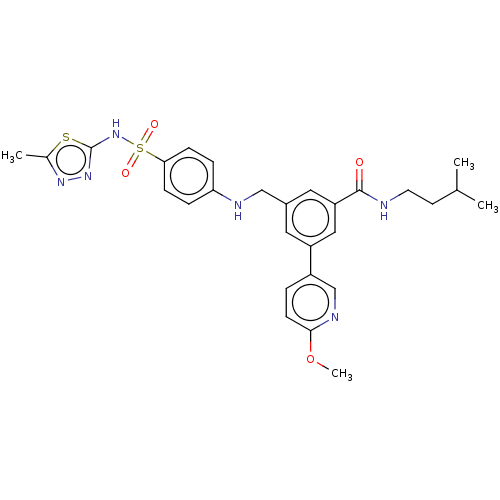
(CHEMBL3582351)Show SMILES COc1ccc(cn1)-c1cc(CNc2ccc(cc2)S(=O)(=O)Nc2nnc(C)s2)cc(c1)C(=O)NCCC(C)C Show InChI InChI=1S/C28H32N6O4S2/c1-18(2)11-12-29-27(35)23-14-20(13-22(15-23)21-5-10-26(38-4)31-17-21)16-30-24-6-8-25(9-7-24)40(36,37)34-28-33-32-19(3)39-28/h5-10,13-15,17-18,30H,11-12,16H2,1-4H3,(H,29,35)(H,33,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) |
ACS Med Chem Lett 6: 531-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00025
BindingDB Entry DOI: 10.7270/Q2BP04JF |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50091695
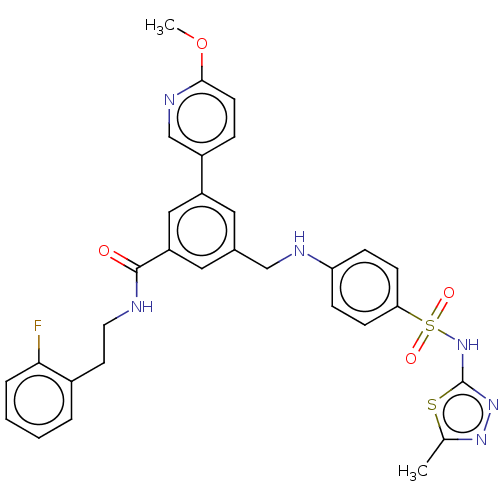
(CHEMBL3582350)Show SMILES COc1ccc(cn1)-c1cc(CNc2ccc(cc2)S(=O)(=O)Nc2nnc(C)s2)cc(c1)C(=O)NCCc1ccccc1F Show InChI InChI=1S/C31H29FN6O4S2/c1-20-36-37-31(43-20)38-44(40,41)27-10-8-26(9-11-27)34-18-21-15-24(23-7-12-29(42-2)35-19-23)17-25(16-21)30(39)33-14-13-22-5-3-4-6-28(22)32/h3-12,15-17,19,34H,13-14,18H2,1-2H3,(H,33,39)(H,37,38) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.40 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) |
ACS Med Chem Lett 6: 531-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00025
BindingDB Entry DOI: 10.7270/Q2BP04JF |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448864

(CHEMBL3125276)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(=O)NCC(=O)OC)n(C)n1 |r| Show InChI InChI=1S/C24H27N5O6/c1-4-14-9-18(28(2)27-14)23(32)26-15-10-19(22(31)25-11-21(30)34-3)29(12-15)24(33)17-13-35-20-8-6-5-7-16(17)20/h5-9,13,15,19H,4,10-12H2,1-3H3,(H,25,31)(H,26,32)/t15-,19+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448845

(CHEMBL3125240)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(=O)NCc2ccc(OC(F)(F)F)cc2)n(C)n1 |r| Show InChI InChI=1S/C29H28F3N5O5/c1-3-18-12-23(36(2)35-18)27(39)34-19-13-24(26(38)33-14-17-8-10-20(11-9-17)42-29(30,31)32)37(15-19)28(40)22-16-41-25-7-5-4-6-21(22)25/h4-12,16,19,24H,3,13-15H2,1-2H3,(H,33,38)(H,34,39)/t19-,24+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448886
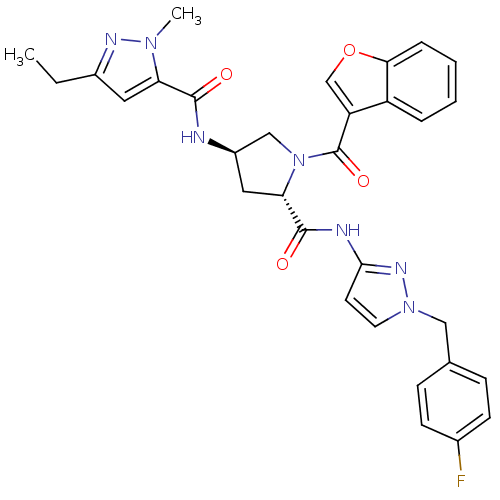
(CHEMBL3125255)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(=O)Nc2ccn(Cc3ccc(F)cc3)n2)n(C)n1 |r| Show InChI InChI=1S/C31H30FN7O4/c1-3-21-14-25(37(2)35-21)29(40)33-22-15-26(39(17-22)31(42)24-18-43-27-7-5-4-6-23(24)27)30(41)34-28-12-13-38(36-28)16-19-8-10-20(32)11-9-19/h4-14,18,22,26H,3,15-17H2,1-2H3,(H,33,40)(H,34,36,41)/t22-,26+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448882

(CHEMBL3125259)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(=O)NCc2ncco2)n(C)n1 |r| Show InChI InChI=1S/C25H26N6O5/c1-3-15-10-19(30(2)29-15)24(33)28-16-11-20(23(32)27-12-22-26-8-9-35-22)31(13-16)25(34)18-14-36-21-7-5-4-6-17(18)21/h4-10,14,16,20H,3,11-13H2,1-2H3,(H,27,32)(H,28,33)/t16-,20+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |
Bifunctional epoxide hydrolase 2
(Homo sapiens (Human)) | BDBM50566637
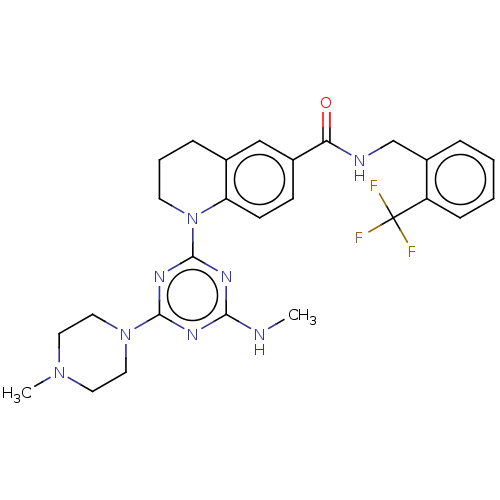
(CHEMBL4871885)Show SMILES CNc1nc(nc(n1)N1CCCc2cc(ccc12)C(=O)NCc1ccccc1C(F)(F)F)N1CCN(C)CC1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human sEH using EnzChek as substrate preincubated with enzyme for 5 mins followed by substrate addition and measured every 30 secs for ... |
Citation and Details
Article DOI: 10.1016/j.bmc.2021.116216
BindingDB Entry DOI: 10.7270/Q22J6GNB |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448838
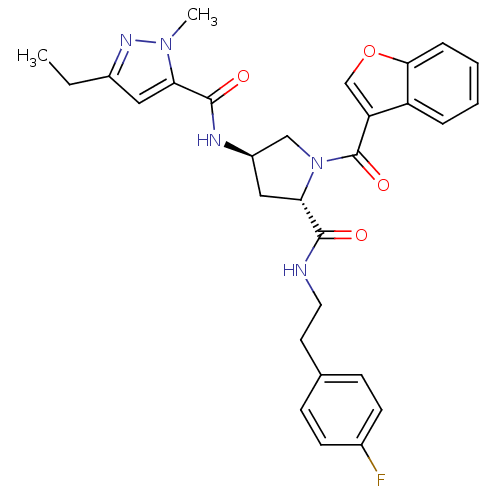
(CHEMBL3125247)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(=O)NCCc2ccc(F)cc2)n(C)n1 |r| Show InChI InChI=1S/C29H30FN5O4/c1-3-20-14-24(34(2)33-20)28(37)32-21-15-25(27(36)31-13-12-18-8-10-19(30)11-9-18)35(16-21)29(38)23-17-39-26-7-5-4-6-22(23)26/h4-11,14,17,21,25H,3,12-13,15-16H2,1-2H3,(H,31,36)(H,32,37)/t21-,25+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |
A disintegrin and metalloproteinase with thrombospondin motifs 5
(Homo sapiens (Human)) | BDBM50168737

((2R,3R)-1-[4-(2-Chloro-4-fluoro-benzyloxy)-benzene...)Show SMILES C[C@@]1(O)CCCN([C@H]1C(=O)NO)S(=O)(=O)c1ccc(OCc2ccc(F)cc2Cl)cc1 Show InChI InChI=1S/C20H22ClFN2O6S/c1-20(26)9-2-10-24(18(20)19(25)23-27)31(28,29)16-7-5-15(6-8-16)30-12-13-3-4-14(22)11-17(13)21/h3-8,11,18,26-27H,2,9-10,12H2,1H3,(H,23,25)/t18-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human ADAMTS-5 expressed in CHO cells using WAAG-3R as substrate preincubated for 15 mins measured after 1 hr by FRET assay |
J Med Chem 55: 7061-79 (2012)
Article DOI: 10.1021/jm300449x
BindingDB Entry DOI: 10.7270/Q2RX9D6T |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50091695

(CHEMBL3582350)Show SMILES COc1ccc(cn1)-c1cc(CNc2ccc(cc2)S(=O)(=O)Nc2nnc(C)s2)cc(c1)C(=O)NCCc1ccccc1F Show InChI InChI=1S/C31H29FN6O4S2/c1-20-36-37-31(43-20)38-44(40,41)27-10-8-26(9-11-27)34-18-21-15-24(23-7-12-29(42-2)35-19-23)17-25(16-21)30(39)33-14-13-22-5-3-4-6-28(22)32/h3-12,15-17,19,34H,13-14,18H2,1-2H3,(H,33,39)(H,37,38) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of wild type his-tagged PI3Kalpha (unknown origin) |
ACS Med Chem Lett 6: 531-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00025
BindingDB Entry DOI: 10.7270/Q2BP04JF |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448889

(CHEMBL3125252)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(=O)Nc2ccn(C)n2)n(C)n1 |r| Show InChI InChI=1S/C25H27N7O4/c1-4-15-11-19(31(3)28-15)23(33)26-16-12-20(24(34)27-22-9-10-30(2)29-22)32(13-16)25(35)18-14-36-21-8-6-5-7-17(18)21/h5-11,14,16,20H,4,12-13H2,1-3H3,(H,26,33)(H,27,29,34)/t16-,20+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448881
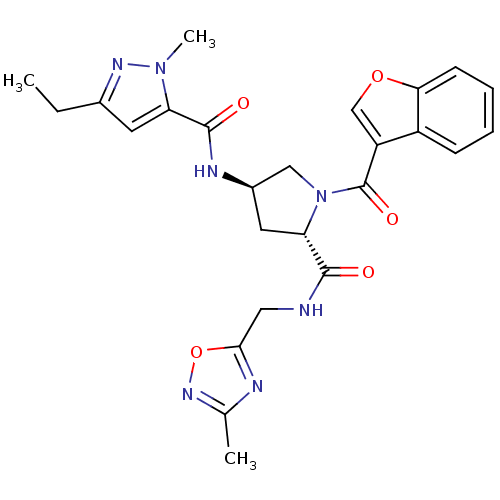
(CHEMBL3125260)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(=O)NCc2nc(C)no2)n(C)n1 |r| Show InChI InChI=1S/C25H27N7O5/c1-4-15-9-19(31(3)29-15)24(34)28-16-10-20(23(33)26-11-22-27-14(2)30-37-22)32(12-16)25(35)18-13-36-21-8-6-5-7-17(18)21/h5-9,13,16,20H,4,10-12H2,1-3H3,(H,26,33)(H,28,34)/t16-,20+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50091689

(CHEMBL3582354)Show SMILES COc1ccc(cn1)-c1cc(CNc2ccc(cc2)S(=O)(=O)Nc2nnc(C)s2)cc(c1)C(O)=O Show InChI InChI=1S/C23H21N5O5S2/c1-14-26-27-23(34-14)28-35(31,32)20-6-4-19(5-7-20)24-12-15-9-17(11-18(10-15)22(29)30)16-3-8-21(33-2)25-13-16/h3-11,13,24H,12H2,1-2H3,(H,27,28)(H,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 8.5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha H1047R mutant (unknown origin) |
ACS Med Chem Lett 6: 531-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00025
BindingDB Entry DOI: 10.7270/Q2BP04JF |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Bifunctional epoxide hydrolase 2
(Homo sapiens (Human)) | BDBM50566632
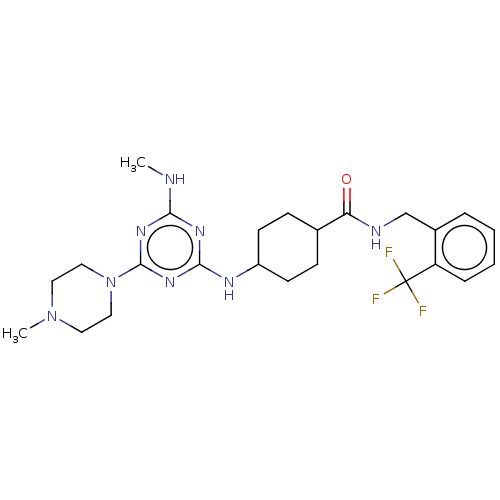
(CHEMBL4850549)Show SMILES CNc1nc(NC2CCC(CC2)C(=O)NCc2ccccc2C(F)(F)F)nc(n1)N1CCN(C)CC1 |(45.76,-44.82,;44.43,-45.6,;44.44,-47.14,;43.11,-47.91,;43.12,-49.45,;41.78,-50.22,;40.45,-49.45,;39.11,-50.22,;37.79,-49.45,;37.78,-47.91,;39.11,-47.13,;40.45,-47.91,;36.45,-47.15,;36.44,-45.61,;35.12,-47.92,;33.78,-47.16,;32.45,-47.93,;31.11,-47.17,;29.79,-47.94,;29.78,-49.49,;31.12,-50.26,;32.46,-49.48,;33.79,-50.25,;34.55,-48.91,;35.33,-50.24,;33.79,-51.79,;44.45,-50.22,;45.78,-49.45,;45.78,-47.91,;47.11,-50.22,;47.1,-51.76,;48.43,-52.53,;49.77,-51.77,;51.1,-52.55,;49.77,-50.23,;48.44,-49.45,)| | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human sEH using EnzChek as substrate preincubated with enzyme for 5 mins followed by substrate addition and measured every 30 secs for ... |
Citation and Details
Article DOI: 10.1016/j.bmc.2021.116216
BindingDB Entry DOI: 10.7270/Q22J6GNB |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH]
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50448890

(CHEMBL3125251)Show SMILES CCc1cc(C(=O)N[C@@H]2C[C@H](N(C2)C(=O)c2coc3ccccc23)C(=O)Nc2nc(C)co2)n(C)n1 |r| Show InChI InChI=1S/C25H26N6O5/c1-4-15-9-19(30(3)29-15)22(32)27-16-10-20(23(33)28-25-26-14(2)12-36-25)31(11-16)24(34)18-13-35-21-8-6-5-7-17(18)21/h5-9,12-13,16,20H,4,10-11H2,1-3H3,(H,27,32)(H,26,28,33)/t16-,20+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis full length biotinylated InhA (270 amino acids) using DDCoA as substrate assessed as NADH oxidation by fluor... |
J Med Chem 57: 1276-88 (2014)
Article DOI: 10.1021/jm401326j
BindingDB Entry DOI: 10.7270/Q2NP25XB |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data