 Found 237 hits with Last Name = 'devine' and Initial = 'a'
Found 237 hits with Last Name = 'devine' and Initial = 'a' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50127246

(8-[Amino-(4-chloro-phenyl)-(3-methyl-3H-imidazol-4...)Show SMILES Cn1cncc1C(N)(c1ccc(Cl)cc1)c1cc2CCn3c2c(c1)c(cc3=O)-c1cccc(Cl)c1 Show InChI InChI=1S/C28H22Cl2N4O/c1-33-16-32-15-25(33)28(31,19-5-7-21(29)8-6-19)20-11-18-9-10-34-26(35)14-23(24(13-20)27(18)34)17-3-2-4-22(30)12-17/h2-8,11-16H,9-10,31H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Inhibition of Geranylgeranylprotein transferase-I at 10 uM |
Bioorg Med Chem Lett 13: 1543-7 (2003)
BindingDB Entry DOI: 10.7270/Q2M61JNS |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50127246

(8-[Amino-(4-chloro-phenyl)-(3-methyl-3H-imidazol-4...)Show SMILES Cn1cncc1C(N)(c1ccc(Cl)cc1)c1cc2CCn3c2c(c1)c(cc3=O)-c1cccc(Cl)c1 Show InChI InChI=1S/C28H22Cl2N4O/c1-33-16-32-15-25(33)28(31,19-5-7-21(29)8-6-19)20-11-18-9-10-34-26(35)14-23(24(13-20)27(18)34)17-3-2-4-22(30)12-17/h2-8,11-16H,9-10,31H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-FPP incorporation into biotinylated laminB peptide by farnesyl transferase |
Bioorg Med Chem Lett 13: 1543-7 (2003)
BindingDB Entry DOI: 10.7270/Q2M61JNS |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50126335
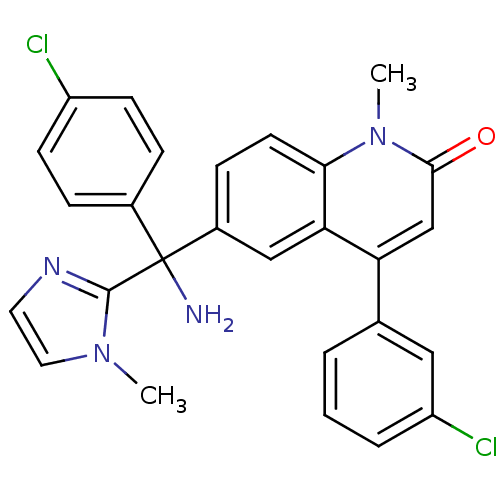
(6-[(R)-Amino-(4-chloro-phenyl)-(3-methyl-3H-imidaz...)Show SMILES Cn1ccnc1C(N)(c1ccc(Cl)cc1)c1ccc2n(C)c(=O)cc(-c3cccc(Cl)c3)c2c1 Show InChI InChI=1S/C27H22Cl2N4O/c1-32-13-12-31-26(32)27(30,18-6-9-20(28)10-7-18)19-8-11-24-23(15-19)22(16-25(34)33(24)2)17-4-3-5-21(29)14-17/h3-16H,30H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| MMDB
PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-FPP incorporation into biotinylated laminB peptide by farnesyl transferase |
Bioorg Med Chem Lett 13: 1543-7 (2003)
BindingDB Entry DOI: 10.7270/Q2M61JNS |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM14434
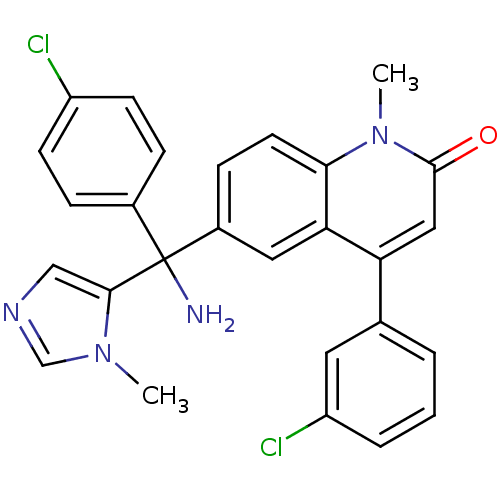
(6-[amino(4-chlorophenyl)(1-methyl-1H-imidazol-5-yl...)Show SMILES Cn1cncc1C(N)(c1ccc(Cl)cc1)c1ccc2n(C)c(=O)cc(-c3cccc(Cl)c3)c2c1 Show InChI InChI=1S/C27H22Cl2N4O/c1-32-16-31-15-25(32)27(30,18-6-9-20(28)10-7-18)19-8-11-24-23(13-19)22(14-26(34)33(24)2)17-4-3-5-21(29)12-17/h3-16H,30H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| n/a | n/a | 0.860 | n/a | n/a | n/a | n/a | 7.5 | 22 |
Janssen Research Foundation
| Assay Description
Compounds were tested as inhibitors of FTase in vitro using purified human enzyme to catalyze the reaction between [3H] FPP and a biotinylated peptid... |
Cancer Res 61: 131-7 (2001)
BindingDB Entry DOI: 10.7270/Q20C4T2R |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50127248
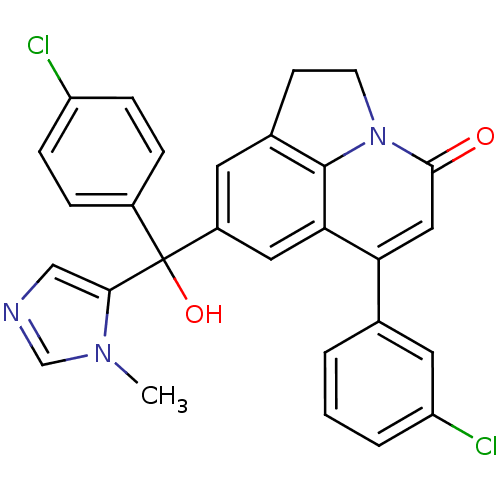
(6-(3-Chloro-phenyl)-8-[(4-chloro-phenyl)-hydroxy-(...)Show SMILES Cn1cncc1C(O)(c1ccc(Cl)cc1)c1cc2CCn3c2c(c1)c(cc3=O)-c1cccc(Cl)c1 Show InChI InChI=1S/C28H21Cl2N3O2/c1-32-16-31-15-25(32)28(35,19-5-7-21(29)8-6-19)20-11-18-9-10-33-26(34)14-23(24(13-20)27(18)33)17-3-2-4-22(30)12-17/h2-8,11-16,35H,9-10H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-FPP incorporation into biotinylated laminB peptide by farnesyl transferase |
Bioorg Med Chem Lett 13: 1543-7 (2003)
BindingDB Entry DOI: 10.7270/Q2M61JNS |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50179206
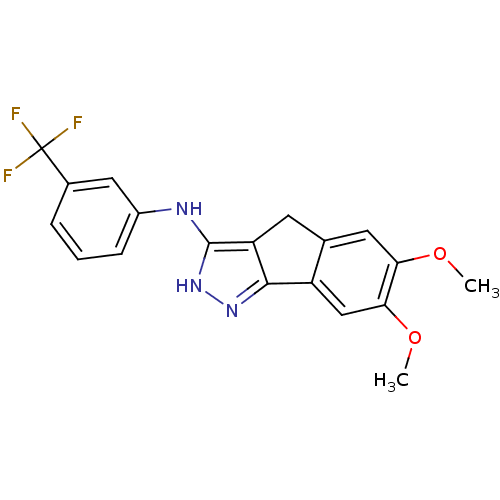
(3-trifluoromethyl-N-(6,7-dimethoxy-2,4-dihydroinde...)Show SMILES COc1cc2Cc3c(Nc4cccc(c4)C(F)(F)F)[nH]nc3-c2cc1OC Show InChI InChI=1S/C19H16F3N3O2/c1-26-15-7-10-6-14-17(13(10)9-16(15)27-2)24-25-18(14)23-12-5-3-4-11(8-12)19(20,21)22/h3-5,7-9H,6H2,1-2H3,(H2,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Antiproliferative activity against PDGF-BB stimulated HCASMC |
J Med Chem 48: 8163-73 (2005)
Article DOI: 10.1021/jm050680m
BindingDB Entry DOI: 10.7270/Q2W095HW |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM13534

(CHEMBL572878 | N-[4-({4-[(3-methyl-1H-pyrazol-5-yl...)Show SMILES CN1CCN(CC1)c1cc(Nc2cc(C)n[nH]2)nc(Sc2ccc(NC(=O)C3CC3)cc2)n1 Show InChI InChI=1S/C23H28N8OS/c1-15-13-20(29-28-15)25-19-14-21(31-11-9-30(2)10-12-31)27-23(26-19)33-18-7-5-17(6-8-18)24-22(32)16-3-4-16/h5-8,13-14,16H,3-4,9-12H2,1-2H3,(H,24,32)(H2,25,26,27,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | 7.0 | 22 |
Astex
| Assay Description
Assays for Aurora kinase was performed in a DELFIA format. Aurora enzyme was incubated with test compound and cross-tide substrate in the reaction bu... |
J Med Chem 52: 379-88 (2009)
Article DOI: 10.1021/jm800984v
BindingDB Entry DOI: 10.7270/Q2P55KT7 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aurora kinase A
(Homo sapiens (Human)) | BDBM27083

(1-(2,6-difluorophenyl)-3-{3-[5-(morpholin-4-ylmeth...)Show SMILES Fc1cccc(F)c1NC(=O)Nc1c[nH]nc1-c1nc2ccc(CN3CCOCC3)cc2[nH]1 Show InChI InChI=1S/C22H21F2N7O2/c23-14-2-1-3-15(24)19(14)29-22(32)28-18-11-25-30-20(18)21-26-16-5-4-13(10-17(16)27-21)12-31-6-8-33-9-7-31/h1-5,10-11H,6-9,12H2,(H,25,30)(H,26,27)(H2,28,29,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | 7.0 | 22 |
Astex
| Assay Description
Assays for Aurora kinase was performed in a DELFIA format. Aurora enzyme was incubated with test compound and cross-tide substrate in the reaction bu... |
J Med Chem 52: 379-88 (2009)
Article DOI: 10.1021/jm800984v
BindingDB Entry DOI: 10.7270/Q2P55KT7 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50127252
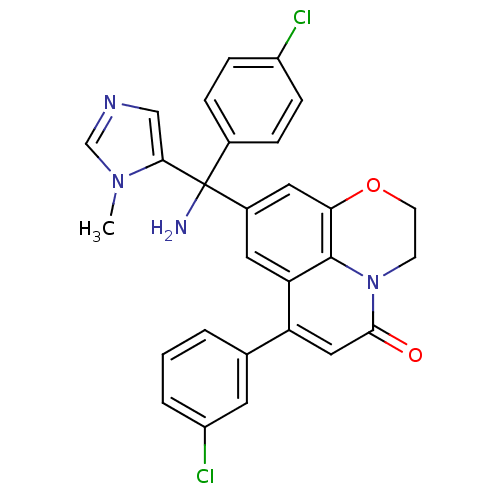
(8-[Amino-(4-chloro-phenyl)-(3-methyl-3H-imidazol-4...)Show SMILES Cn1cncc1C(N)(c1ccc(Cl)cc1)c1cc2OCCn3c2c(c1)c(cc3=O)-c1cccc(Cl)c1 Show InChI InChI=1S/C28H22Cl2N4O2/c1-33-16-32-15-25(33)28(31,18-5-7-20(29)8-6-18)19-12-23-22(17-3-2-4-21(30)11-17)14-26(35)34-9-10-36-24(13-19)27(23)34/h2-8,11-16H,9-10,31H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-FPP incorporation into biotinylated laminB peptide by farnesyl transferase |
Bioorg Med Chem Lett 13: 1543-7 (2003)
BindingDB Entry DOI: 10.7270/Q2M61JNS |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50127254
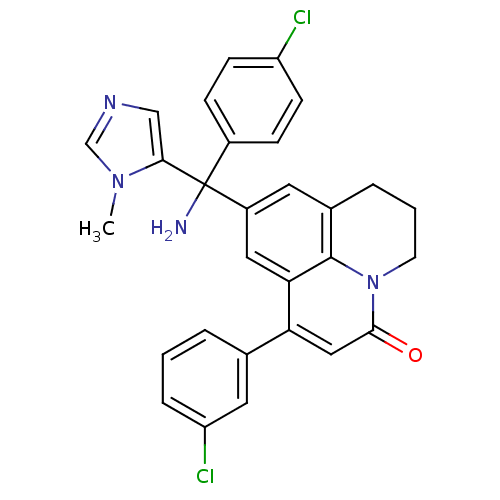
(9-[Amino-(4-chloro-phenyl)-(3-methyl-3H-imidazol-4...)Show SMILES Cn1cncc1C(N)(c1ccc(Cl)cc1)c1cc2CCCn3c2c(c1)c(cc3=O)-c1cccc(Cl)c1 Show InChI InChI=1S/C29H24Cl2N4O/c1-34-17-33-16-26(34)29(32,20-7-9-22(30)10-8-20)21-12-19-5-3-11-35-27(36)15-24(25(14-21)28(19)35)18-4-2-6-23(31)13-18/h2,4,6-10,12-17H,3,5,11,32H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-FPP incorporation into biotinylated laminB peptide by farnesyl transferase |
Bioorg Med Chem Lett 13: 1543-7 (2003)
BindingDB Entry DOI: 10.7270/Q2M61JNS |
More data for this
Ligand-Target Pair | |
Amine oxidase [flavin-containing] A
(Homo sapiens (Human)) | BDBM15581
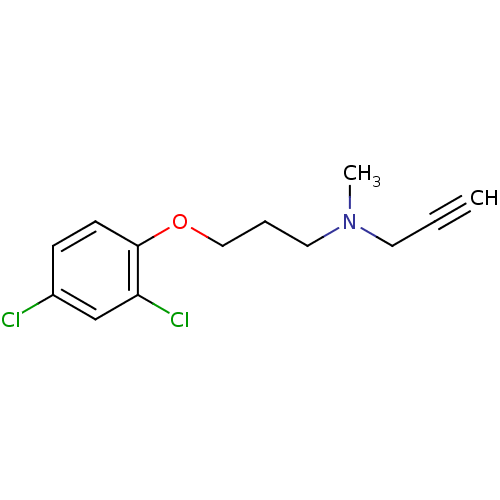
(CHEMBL8706 | CLG | CLORGILINE | Clorgyline | N-[3-...)Show InChI InChI=1S/C13H15Cl2NO/c1-3-7-16(2)8-4-9-17-13-6-5-11(14)10-12(13)15/h1,5-6,10H,4,7-9H2,2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of British Columbia
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant MAO-A expressed in baculovirus infected BTI-TN-5B1-4 insect cells using tyramine as substrate preincubated for 1 hr f... |
J Med Chem 61: 7043-7064 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01588
BindingDB Entry DOI: 10.7270/Q2VT1VSP |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aurora kinase B
(Homo sapiens (Human)) | BDBM27083

(1-(2,6-difluorophenyl)-3-{3-[5-(morpholin-4-ylmeth...)Show SMILES Fc1cccc(F)c1NC(=O)Nc1c[nH]nc1-c1nc2ccc(CN3CCOCC3)cc2[nH]1 Show InChI InChI=1S/C22H21F2N7O2/c23-14-2-1-3-15(24)19(14)29-22(32)28-18-11-25-30-20(18)21-26-16-5-4-13(10-17(16)27-21)12-31-6-8-33-9-7-31/h1-5,10-11H,6-9,12H2,(H,25,30)(H,26,27)(H2,28,29,32) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Astex
| Assay Description
Assays for Aurora kinase was performed in a DELFIA format. Aurora enzyme was incubated with test compound and cross-tide substrate in the reaction bu... |
J Med Chem 52: 379-88 (2009)
Article DOI: 10.1021/jm800984v
BindingDB Entry DOI: 10.7270/Q2P55KT7 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM27082
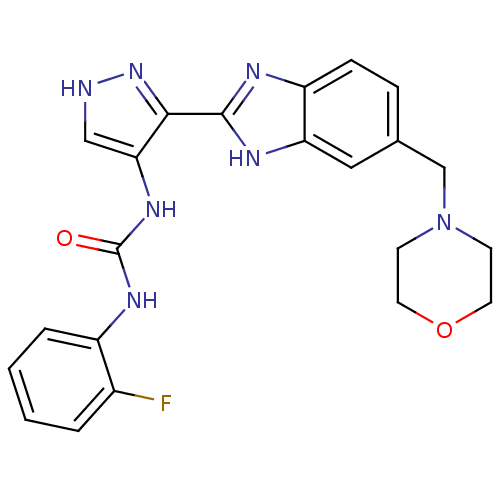
(1-(2-fluorophenyl)-3-{3-[5-(morpholin-4-ylmethyl)-...)Show SMILES Fc1ccccc1NC(=O)Nc1c[nH]nc1-c1nc2ccc(CN3CCOCC3)cc2[nH]1 Show InChI InChI=1S/C22H22FN7O2/c23-15-3-1-2-4-16(15)27-22(31)28-19-12-24-29-20(19)21-25-17-6-5-14(11-18(17)26-21)13-30-7-9-32-10-8-30/h1-6,11-12H,7-10,13H2,(H,24,29)(H,25,26)(H2,27,28,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.80 | n/a | n/a | n/a | n/a | 7.0 | 22 |
Astex
| Assay Description
Assays for Aurora kinase was performed in a DELFIA format. Aurora enzyme was incubated with test compound and cross-tide substrate in the reaction bu... |
J Med Chem 52: 379-88 (2009)
Article DOI: 10.1021/jm800984v
BindingDB Entry DOI: 10.7270/Q2P55KT7 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50179207

(3-fluoro-N-(6,7-dimethoxy-2,4-dihydroindeno[1,2-c]...)Show InChI InChI=1S/C18H16FN3O2/c1-23-15-7-10-6-14-17(13(10)9-16(15)24-2)21-22-18(14)20-12-5-3-4-11(19)8-12/h3-5,7-9H,6H2,1-2H3,(H2,20,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Antiproliferative activity against PDGF-BB stimulated HCASMC |
J Med Chem 48: 8163-73 (2005)
Article DOI: 10.1021/jm050680m
BindingDB Entry DOI: 10.7270/Q2W095HW |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50179212
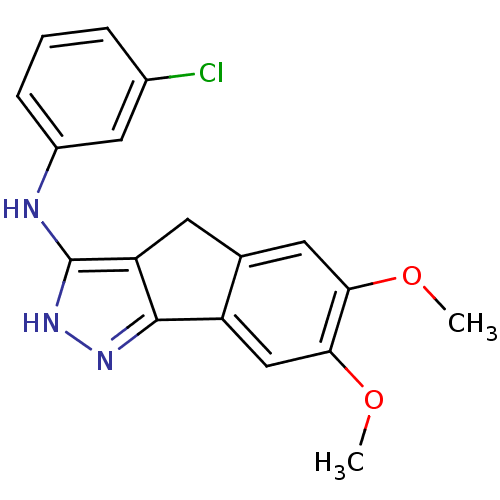
(3-chloro-N-(6,7-dimethoxy-2,4-dihydroindeno[1,2-c]...)Show InChI InChI=1S/C18H16ClN3O2/c1-23-15-7-10-6-14-17(13(10)9-16(15)24-2)21-22-18(14)20-12-5-3-4-11(19)8-12/h3-5,7-9H,6H2,1-2H3,(H2,20,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDGFRbeta kinase |
J Med Chem 48: 8163-73 (2005)
Article DOI: 10.1021/jm050680m
BindingDB Entry DOI: 10.7270/Q2W095HW |
More data for this
Ligand-Target Pair | |
Cyclin-A2/Cyclin-dependent kinase 2
(Homo sapiens (Human)) | BDBM24644
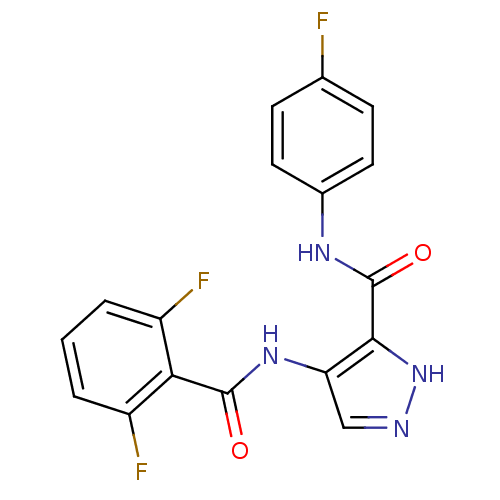
(4-N-(2,6-difluorobenzene)-3-N-(4-fluorophenyl)-1H-...)Show SMILES Fc1ccc(NC(=O)c2[nH]ncc2NC(=O)c2c(F)cccc2F)cc1 Show InChI InChI=1S/C17H11F3N4O2/c18-9-4-6-10(7-5-9)22-17(26)15-13(8-21-24-15)23-16(25)14-11(19)2-1-3-12(14)20/h1-8H,(H,21,24)(H,22,26)(H,23,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | 7.2 | 22 |
Astex
| Assay Description
CDK2/cyclin A activity was determined using a radiometric assay to measure the incorporation of gamma-phosphate from [gamma-33P]-ATP into histone H1.... |
J Med Chem 51: 4986-99 (2008)
Article DOI: 10.1021/jm800382h
BindingDB Entry DOI: 10.7270/Q24X563W |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aurora kinase A
(Homo sapiens (Human)) | BDBM27079

(N-{3-[5-(morpholin-4-ylmethyl)-1H-1,3-benzodiazol-...)Show SMILES O=C(Nc1c[nH]nc1-c1nc2ccc(CN3CCOCC3)cc2[nH]1)c1ccccc1 Show InChI InChI=1S/C22H22N6O2/c29-22(16-4-2-1-3-5-16)26-19-13-23-27-20(19)21-24-17-7-6-15(12-18(17)25-21)14-28-8-10-30-11-9-28/h1-7,12-13H,8-11,14H2,(H,23,27)(H,24,25)(H,26,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | 7.0 | 22 |
Astex
| Assay Description
Assays for Aurora kinase was performed in a DELFIA format. Aurora enzyme was incubated with test compound and cross-tide substrate in the reaction bu... |
J Med Chem 52: 379-88 (2009)
Article DOI: 10.1021/jm800984v
BindingDB Entry DOI: 10.7270/Q2P55KT7 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50127255
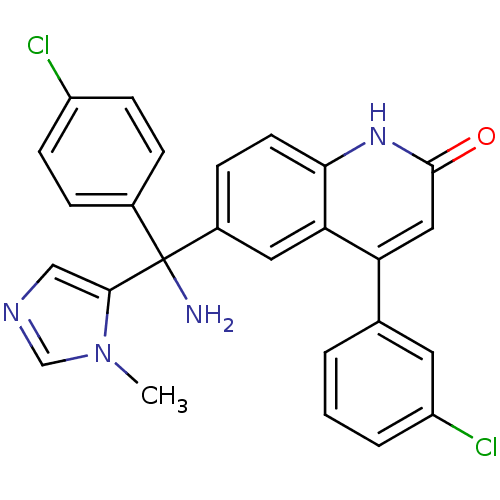
(6-[Amino-(4-chloro-phenyl)-(3-methyl-3H-imidazol-4...)Show SMILES Cn1cncc1C(N)(c1ccc(Cl)cc1)c1ccc2[nH]c(=O)cc(-c3cccc(Cl)c3)c2c1 Show InChI InChI=1S/C26H20Cl2N4O/c1-32-15-30-14-24(32)26(29,17-5-8-19(27)9-6-17)18-7-10-23-22(12-18)21(13-25(33)31-23)16-3-2-4-20(28)11-16/h2-15H,29H2,1H3,(H,31,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-FPP incorporation into biotinylated laminB peptide by farnesyl transferase |
Bioorg Med Chem Lett 13: 1543-7 (2003)
BindingDB Entry DOI: 10.7270/Q2M61JNS |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50127247
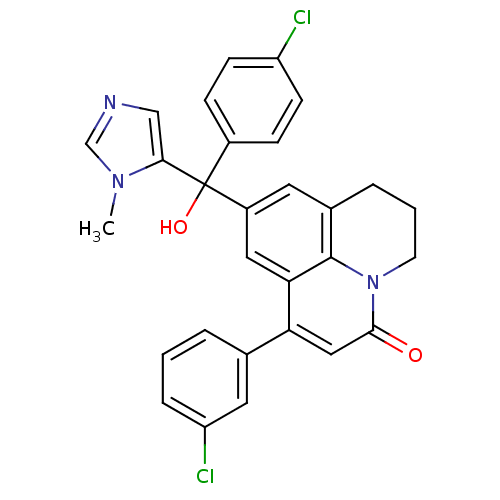
(1-(3-Chloro-phenyl)-9-[(4-chloro-phenyl)-hydroxy-(...)Show SMILES Cn1cncc1C(O)(c1ccc(Cl)cc1)c1cc2CCCn3c2c(c1)c(cc3=O)-c1cccc(Cl)c1 Show InChI InChI=1S/C29H23Cl2N3O2/c1-33-17-32-16-26(33)29(36,20-7-9-22(30)10-8-20)21-12-19-5-3-11-34-27(35)15-24(25(14-21)28(19)34)18-4-2-6-23(31)13-18/h2,4,6-10,12-17,36H,3,5,11H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-FPP incorporation into biotinylated laminB peptide by farnesyl transferase |
Bioorg Med Chem Lett 13: 1543-7 (2003)
BindingDB Entry DOI: 10.7270/Q2M61JNS |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50179207

(3-fluoro-N-(6,7-dimethoxy-2,4-dihydroindeno[1,2-c]...)Show InChI InChI=1S/C18H16FN3O2/c1-23-15-7-10-6-14-17(13(10)9-16(15)24-2)21-22-18(14)20-12-5-3-4-11(19)8-12/h3-5,7-9H,6H2,1-2H3,(H2,20,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDGFRbeta kinase |
J Med Chem 48: 8163-73 (2005)
Article DOI: 10.1021/jm050680m
BindingDB Entry DOI: 10.7270/Q2W095HW |
More data for this
Ligand-Target Pair | |
Amine oxidase [flavin-containing] B
(Homo sapiens (Human)) | BDBM50462966

(CHEMBL4239843)Show InChI InChI=1S/C20H23NO/c1-4-14-21(3)17(2)15-18-10-12-20(13-11-18)22-16-19-8-6-5-7-9-19/h1,5-13,17H,14-16H2,2-3H3/t17-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of British Columbia
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant MAO-B expressed in baculovirus infected BTI-TN-5B1-4 insect cells using benzylamine as substrate preincubated for 1 h... |
J Med Chem 61: 7043-7064 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01588
BindingDB Entry DOI: 10.7270/Q2VT1VSP |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50179211
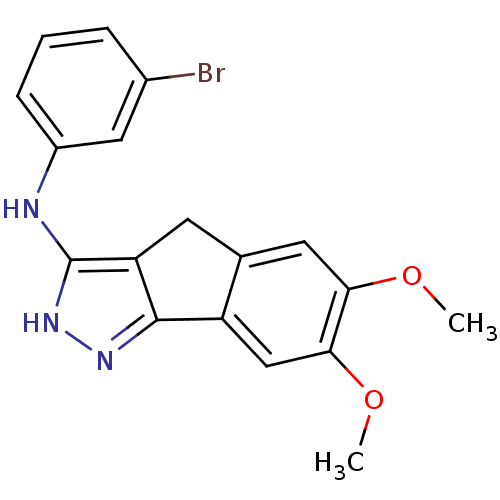
(3-bromo-N-(6,7-dimethoxy-2,4-dihydroindeno[1,2-c]p...)Show InChI InChI=1S/C18H16BrN3O2/c1-23-15-7-10-6-14-17(13(10)9-16(15)24-2)21-22-18(14)20-12-5-3-4-11(19)8-12/h3-5,7-9H,6H2,1-2H3,(H2,20,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDGFRbeta kinase |
J Med Chem 48: 8163-73 (2005)
Article DOI: 10.1021/jm050680m
BindingDB Entry DOI: 10.7270/Q2W095HW |
More data for this
Ligand-Target Pair | |
Aurora kinase B
(Homo sapiens (Human)) | BDBM27084

(1-cyclohexyl-3-{3-[5-(morpholin-4-ylmethyl)-1H-1,3...)Show SMILES O=C(NC1CCCCC1)Nc1c[nH]nc1-c1nc2ccc(CN3CCOCC3)cc2[nH]1 Show InChI InChI=1S/C22H29N7O2/c30-22(24-16-4-2-1-3-5-16)27-19-13-23-28-20(19)21-25-17-7-6-15(12-18(17)26-21)14-29-8-10-31-11-9-29/h6-7,12-13,16H,1-5,8-11,14H2,(H,23,28)(H,25,26)(H2,24,27,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.20 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Astex
| Assay Description
Assays for Aurora kinase was performed in a DELFIA format. Aurora enzyme was incubated with test compound and cross-tide substrate in the reaction bu... |
J Med Chem 52: 379-88 (2009)
Article DOI: 10.1021/jm800984v
BindingDB Entry DOI: 10.7270/Q2P55KT7 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM27084

(1-cyclohexyl-3-{3-[5-(morpholin-4-ylmethyl)-1H-1,3...)Show SMILES O=C(NC1CCCCC1)Nc1c[nH]nc1-c1nc2ccc(CN3CCOCC3)cc2[nH]1 Show InChI InChI=1S/C22H29N7O2/c30-22(24-16-4-2-1-3-5-16)27-19-13-23-28-20(19)21-25-17-7-6-15(12-18(17)26-21)14-29-8-10-31-11-9-29/h6-7,12-13,16H,1-5,8-11,14H2,(H,23,28)(H,25,26)(H2,24,27,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.70 | n/a | n/a | n/a | n/a | 7.0 | 22 |
Astex
| Assay Description
Assays for Aurora kinase was performed in a DELFIA format. Aurora enzyme was incubated with test compound and cross-tide substrate in the reaction bu... |
J Med Chem 52: 379-88 (2009)
Article DOI: 10.1021/jm800984v
BindingDB Entry DOI: 10.7270/Q2P55KT7 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Aurora kinase A
(Homo sapiens (Human)) | BDBM27078
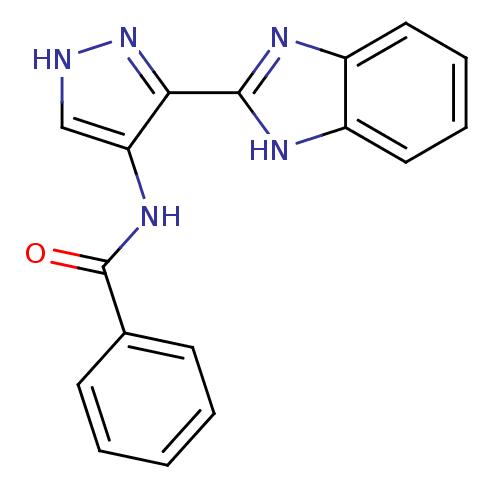
(N-[3-(1H-1,3-benzodiazol-2-yl)-1H-pyrazol-4-yl]ben...)Show InChI InChI=1S/C17H13N5O/c23-17(11-6-2-1-3-7-11)21-14-10-18-22-15(14)16-19-12-8-4-5-9-13(12)20-16/h1-10H,(H,18,22)(H,19,20)(H,21,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 5.90 | n/a | n/a | n/a | n/a | 7.0 | 22 |
Astex
| Assay Description
Assays for Aurora kinase was performed in a DELFIA format. Aurora enzyme was incubated with test compound and cross-tide substrate in the reaction bu... |
J Med Chem 52: 379-88 (2009)
Article DOI: 10.1021/jm800984v
BindingDB Entry DOI: 10.7270/Q2P55KT7 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
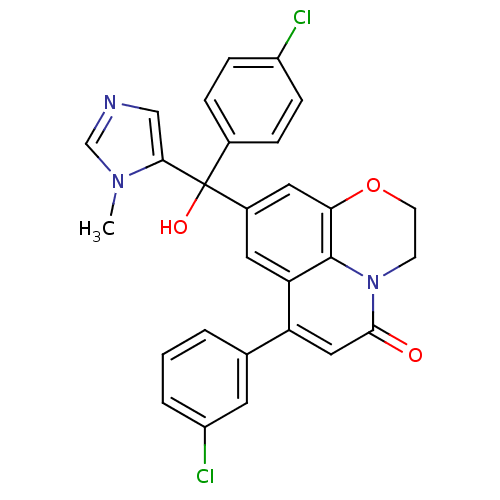
(Homo sapiens (Human)) | BDBM50127249
(6-(3-Chloro-phenyl)-8-[(4-chloro-phenyl)-hydroxy-(...)Show SMILES Cn1cncc1C(O)(c1ccc(Cl)cc1)c1cc2OCCn3c2c(c1)c(cc3=O)-c1cccc(Cl)c1 Show InChI InChI=1S/C28H21Cl2N3O3/c1-32-16-31-15-25(32)28(35,18-5-7-20(29)8-6-18)19-12-23-22(17-3-2-4-21(30)11-17)14-26(34)33-9-10-36-24(13-19)27(23)33/h2-8,11-16,35H,9-10H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-FPP incorporation into biotinylated laminB peptide by farnesyl transferase |
Bioorg Med Chem Lett 13: 1543-7 (2003)
BindingDB Entry DOI: 10.7270/Q2M61JNS |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
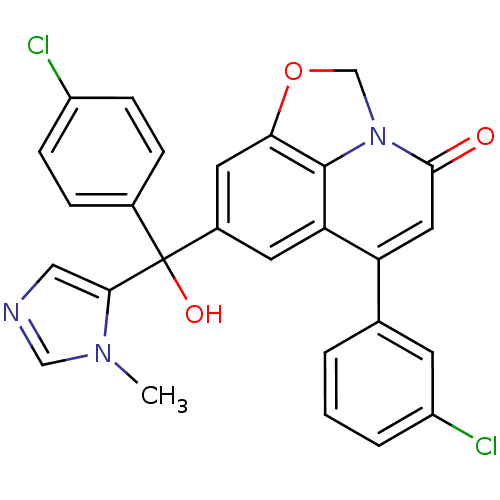
(Homo sapiens (Human)) | BDBM50127257
(6-(3-Chloro-phenyl)-8-[(4-chloro-phenyl)-hydroxy-(...)Show SMILES Cn1cncc1C(O)(c1ccc(Cl)cc1)c1cc2OCn3c2c(c1)c(cc3=O)-c1cccc(Cl)c1 Show InChI InChI=1S/C27H19Cl2N3O3/c1-31-14-30-13-24(31)27(34,17-5-7-19(28)8-6-17)18-10-22-21(16-3-2-4-20(29)9-16)12-25(33)32-15-35-23(11-18)26(22)32/h2-14,34H,15H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-FPP incorporation into biotinylated laminB peptide by farnesyl transferase |
Bioorg Med Chem Lett 13: 1543-7 (2003)
BindingDB Entry DOI: 10.7270/Q2M61JNS |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50127250
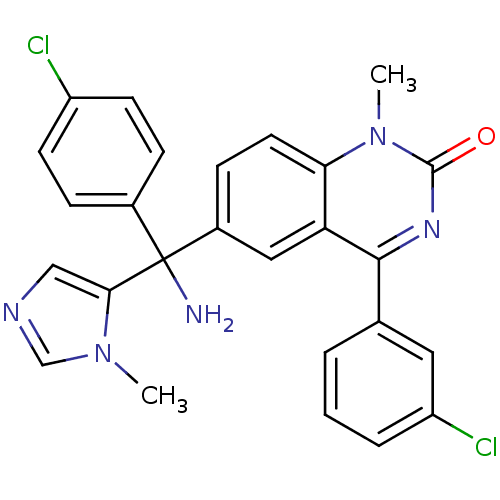
(6-[Amino-(4-chloro-phenyl)-(3-methyl-3H-imidazol-4...)Show SMILES Cn1cncc1C(N)(c1ccc(Cl)cc1)c1ccc2n(C)c(=O)nc(-c3cccc(Cl)c3)c2c1 Show InChI InChI=1S/C26H21Cl2N5O/c1-32-15-30-14-23(32)26(29,17-6-9-19(27)10-7-17)18-8-11-22-21(13-18)24(31-25(34)33(22)2)16-4-3-5-20(28)12-16/h3-15H,29H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| n/a | n/a | 6.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-FPP incorporation into biotinylated laminB peptide by farnesyl transferase |
Bioorg Med Chem Lett 13: 1543-7 (2003)
BindingDB Entry DOI: 10.7270/Q2M61JNS |
More data for this
Ligand-Target Pair | |
Amine oxidase [flavin-containing] B
(Homo sapiens (Human)) | BDBM15579
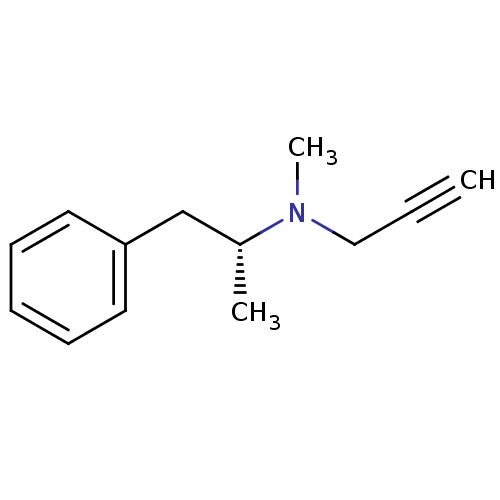
(CHEMBL972 | DEPRENYL | L-Deprenyl | N-methyl-N-[(2...)Show InChI InChI=1S/C13H17N/c1-4-10-14(3)12(2)11-13-8-6-5-7-9-13/h1,5-9,12H,10-11H2,2-3H3/t12-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of British Columbia
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant MAO-B expressed in baculovirus infected BTI-TN-5B1-4 insect cells using benzylamine as substrate preincubated for 1 h... |
J Med Chem 61: 7043-7064 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01588
BindingDB Entry DOI: 10.7270/Q2VT1VSP |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM14434

(6-[amino(4-chlorophenyl)(1-methyl-1H-imidazol-5-yl...)Show SMILES Cn1cncc1C(N)(c1ccc(Cl)cc1)c1ccc2n(C)c(=O)cc(-c3cccc(Cl)c3)c2c1 Show InChI InChI=1S/C27H22Cl2N4O/c1-32-16-31-15-25(32)27(30,18-6-9-20(28)10-7-18)19-8-11-24-23(13-19)22(14-26(34)33(24)2)17-4-3-5-21(29)12-17/h3-16H,30H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| n/a | n/a | 7.90 | n/a | n/a | n/a | n/a | 7.5 | 22 |
Janssen Research Foundation
| Assay Description
Compounds were tested as inhibitors of FTase in vitro using purified human enzyme to catalyze the reaction between [3H] FPP and a biotinylated peptid... |
Cancer Res 61: 131-7 (2001)
BindingDB Entry DOI: 10.7270/Q20C4T2R |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50179209
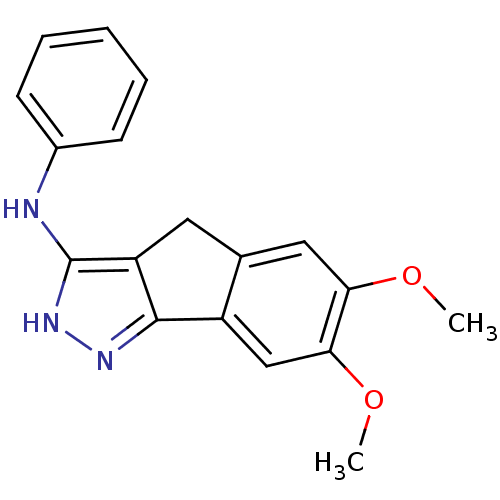
((6,7-dimethoxy-2,4-dihydroindeno[1,2-c]pyrazol-3-y...)Show InChI InChI=1S/C18H17N3O2/c1-22-15-9-11-8-14-17(13(11)10-16(15)23-2)20-21-18(14)19-12-6-4-3-5-7-12/h3-7,9-10H,8H2,1-2H3,(H2,19,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDGFRbeta kinase |
J Med Chem 48: 8163-73 (2005)
Article DOI: 10.1021/jm050680m
BindingDB Entry DOI: 10.7270/Q2W095HW |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50179209

((6,7-dimethoxy-2,4-dihydroindeno[1,2-c]pyrazol-3-y...)Show InChI InChI=1S/C18H17N3O2/c1-22-15-9-11-8-14-17(13(11)10-16(15)23-2)20-21-18(14)19-12-6-4-3-5-7-12/h3-7,9-10H,8H2,1-2H3,(H2,19,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDGFRbeta kinase |
J Med Chem 48: 8163-73 (2005)
Article DOI: 10.1021/jm050680m
BindingDB Entry DOI: 10.7270/Q2W095HW |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50179210
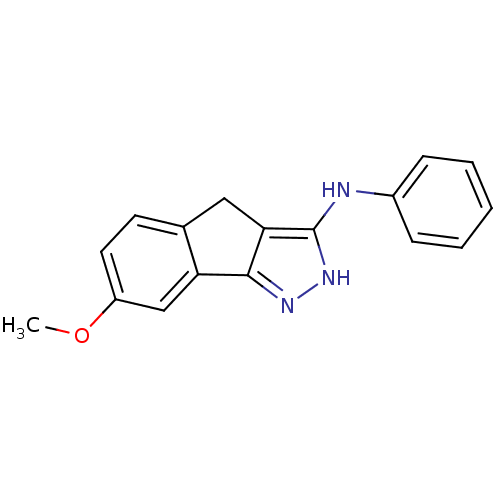
((7-methoxy-2,4-dihydroindeno[1,2-c]pyrazol-3-yl)ph...)Show InChI InChI=1S/C17H15N3O/c1-21-13-8-7-11-9-15-16(14(11)10-13)19-20-17(15)18-12-5-3-2-4-6-12/h2-8,10H,9H2,1H3,(H2,18,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Antiproliferative activity against PDGF-BB stimulated HCASMC |
J Med Chem 48: 8163-73 (2005)
Article DOI: 10.1021/jm050680m
BindingDB Entry DOI: 10.7270/Q2W095HW |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM27080

(4-fluoro-N-{3-[5-(morpholin-4-ylmethyl)-1H-1,3-ben...)Show SMILES Fc1ccc(cc1)C(=O)Nc1c[nH]nc1-c1nc2ccc(CN3CCOCC3)cc2[nH]1 Show InChI InChI=1S/C22H21FN6O2/c23-16-4-2-15(3-5-16)22(30)27-19-12-24-28-20(19)21-25-17-6-1-14(11-18(17)26-21)13-29-7-9-31-10-8-29/h1-6,11-12H,7-10,13H2,(H,24,28)(H,25,26)(H,27,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | 7.0 | 22 |
Astex
| Assay Description
Assays for Aurora kinase was performed in a DELFIA format. Aurora enzyme was incubated with test compound and cross-tide substrate in the reaction bu... |
J Med Chem 52: 379-88 (2009)
Article DOI: 10.1021/jm800984v
BindingDB Entry DOI: 10.7270/Q2P55KT7 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50179204

(3-methoxy-N-(6,7-dimethoxy-2,4-dihydroindeno[1,2-c...)Show SMILES COc1cccc(Nc2[nH]nc-3c2Cc2cc(OC)c(OC)cc-32)c1 Show InChI InChI=1S/C19H19N3O3/c1-23-13-6-4-5-12(9-13)20-19-15-7-11-8-16(24-2)17(25-3)10-14(11)18(15)21-22-19/h4-6,8-10H,7H2,1-3H3,(H2,20,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDGFRbeta kinase |
J Med Chem 48: 8163-73 (2005)
Article DOI: 10.1021/jm050680m
BindingDB Entry DOI: 10.7270/Q2W095HW |
More data for this
Ligand-Target Pair | |
Aurora kinase B
(Homo sapiens (Human)) | BDBM27082

(1-(2-fluorophenyl)-3-{3-[5-(morpholin-4-ylmethyl)-...)Show SMILES Fc1ccccc1NC(=O)Nc1c[nH]nc1-c1nc2ccc(CN3CCOCC3)cc2[nH]1 Show InChI InChI=1S/C22H22FN7O2/c23-15-3-1-2-4-16(15)27-22(31)28-19-12-24-29-20(19)21-25-17-6-5-14(11-18(17)26-21)13-30-7-9-32-10-8-30/h1-6,11-12H,7-10,13H2,(H,24,29)(H,25,26)(H2,27,28,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Astex
| Assay Description
Assays for Aurora kinase was performed in a DELFIA format. Aurora enzyme was incubated with test compound and cross-tide substrate in the reaction bu... |
J Med Chem 52: 379-88 (2009)
Article DOI: 10.1021/jm800984v
BindingDB Entry DOI: 10.7270/Q2P55KT7 |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM24644

(4-N-(2,6-difluorobenzene)-3-N-(4-fluorophenyl)-1H-...)Show SMILES Fc1ccc(NC(=O)c2[nH]ncc2NC(=O)c2c(F)cccc2F)cc1 Show InChI InChI=1S/C17H11F3N4O2/c18-9-4-6-10(7-5-9)22-17(26)15-13(8-21-24-15)23-16(25)14-11(19)2-1-3-12(14)20/h1-8H,(H,21,24)(H,22,26)(H,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Astex
| Assay Description
CDK1/cyclin B activity was determined using a radiometric assay to measure the incorporation of gamma-phosphate from [gamma-33P]-ATP into histone H1.... |
J Med Chem 51: 4986-99 (2008)
Article DOI: 10.1021/jm800382h
BindingDB Entry DOI: 10.7270/Q24X563W |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aurora kinase B
(Homo sapiens (Human)) | BDBM27086

(3-{3-[5-(morpholin-4-ylmethyl)-1H-1,3-benzodiazol-...)Show SMILES O=C(NC1CCOCC1)Nc1c[nH]nc1-c1nc2ccc(CN3CCOCC3)cc2[nH]1 Show InChI InChI=1S/C21H27N7O3/c29-21(23-15-3-7-30-8-4-15)26-18-12-22-27-19(18)20-24-16-2-1-14(11-17(16)25-20)13-28-5-9-31-10-6-28/h1-2,11-12,15H,3-10,13H2,(H,22,27)(H,24,25)(H2,23,26,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Astex
| Assay Description
Assays for Aurora kinase was performed in a DELFIA format. Aurora enzyme was incubated with test compound and cross-tide substrate in the reaction bu... |
J Med Chem 52: 379-88 (2009)
Article DOI: 10.1021/jm800984v
BindingDB Entry DOI: 10.7270/Q2P55KT7 |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50179204

(3-methoxy-N-(6,7-dimethoxy-2,4-dihydroindeno[1,2-c...)Show SMILES COc1cccc(Nc2[nH]nc-3c2Cc2cc(OC)c(OC)cc-32)c1 Show InChI InChI=1S/C19H19N3O3/c1-23-13-6-4-5-12(9-13)20-19-15-7-11-8-16(24-2)17(25-3)10-14(11)18(15)21-22-19/h4-6,8-10H,7H2,1-3H3,(H2,20,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDGFRbeta kinase |
J Med Chem 48: 8163-73 (2005)
Article DOI: 10.1021/jm050680m
BindingDB Entry DOI: 10.7270/Q2W095HW |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50179216
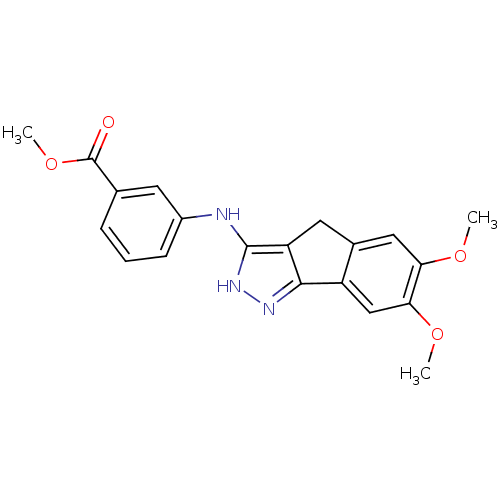
(3-carbomethoxy-N-(6,7-dimethoxy-2,4-dihydroindeno[...)Show SMILES COC(=O)c1cccc(Nc2[nH]nc-3c2Cc2cc(OC)c(OC)cc-32)c1 Show InChI InChI=1S/C20H19N3O4/c1-25-16-9-12-8-15-18(14(12)10-17(16)26-2)22-23-19(15)21-13-6-4-5-11(7-13)20(24)27-3/h4-7,9-10H,8H2,1-3H3,(H2,21,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Antiproliferative activity against PDGF-BB stimulated HCASMC |
J Med Chem 48: 8163-73 (2005)
Article DOI: 10.1021/jm050680m
BindingDB Entry DOI: 10.7270/Q2W095HW |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50179216

(3-carbomethoxy-N-(6,7-dimethoxy-2,4-dihydroindeno[...)Show SMILES COC(=O)c1cccc(Nc2[nH]nc-3c2Cc2cc(OC)c(OC)cc-32)c1 Show InChI InChI=1S/C20H19N3O4/c1-25-16-9-12-8-15-18(14(12)10-17(16)26-2)22-23-19(15)21-13-6-4-5-11(7-13)20(24)27-3/h4-7,9-10H,8H2,1-3H3,(H2,21,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Antiproliferative activity against PDGF-BB stimulated HCASMC |
J Med Chem 48: 8163-73 (2005)
Article DOI: 10.1021/jm050680m
BindingDB Entry DOI: 10.7270/Q2W095HW |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50179217
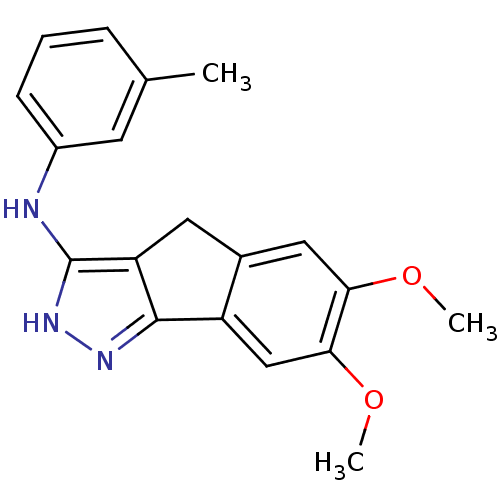
(3-methyl-N-(6,7-dimethoxy-2,4-dihydroindeno[1,2-c]...)Show InChI InChI=1S/C19H19N3O2/c1-11-5-4-6-13(7-11)20-19-15-8-12-9-16(23-2)17(24-3)10-14(12)18(15)21-22-19/h4-7,9-10H,8H2,1-3H3,(H2,20,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDGFRbeta kinase |
J Med Chem 48: 8163-73 (2005)
Article DOI: 10.1021/jm050680m
BindingDB Entry DOI: 10.7270/Q2W095HW |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM27081
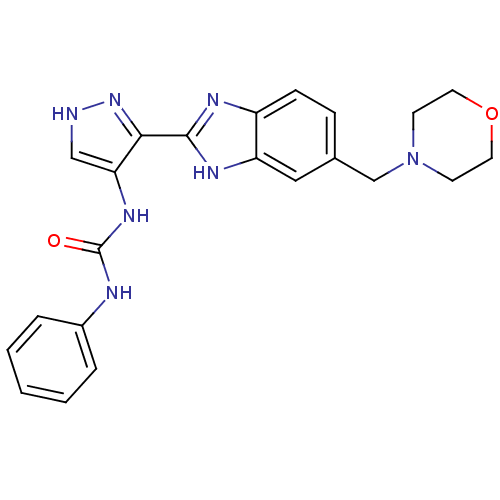
(3-{3-[5-(morpholin-4-ylmethyl)-1H-1,3-benzodiazol-...)Show SMILES O=C(Nc1c[nH]nc1-c1nc2ccc(CN3CCOCC3)cc2[nH]1)Nc1ccccc1 Show InChI InChI=1S/C22H23N7O2/c30-22(24-16-4-2-1-3-5-16)27-19-13-23-28-20(19)21-25-17-7-6-15(12-18(17)26-21)14-29-8-10-31-11-9-29/h1-7,12-13H,8-11,14H2,(H,23,28)(H,25,26)(H2,24,27,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | 7.0 | 22 |
Astex
| Assay Description
Assays for Aurora kinase was performed in a DELFIA format. Aurora enzyme was incubated with test compound and cross-tide substrate in the reaction bu... |
J Med Chem 52: 379-88 (2009)
Article DOI: 10.1021/jm800984v
BindingDB Entry DOI: 10.7270/Q2P55KT7 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-A2/Cyclin-dependent kinase 2
(Homo sapiens (Human)) | BDBM24646
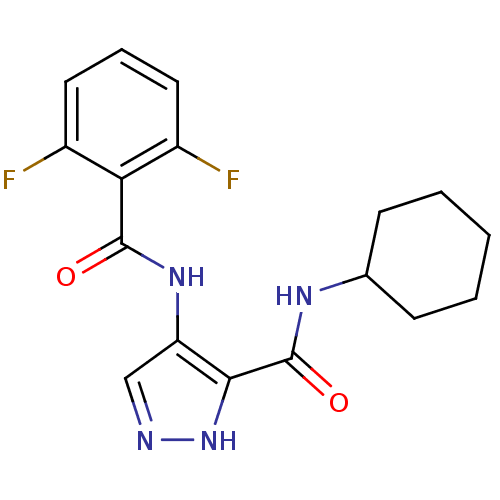
(3-N-cyclohexyl-4-N-(2,6-difluorobenzene)-1H-pyrazo...)Show InChI InChI=1S/C17H18F2N4O2/c18-11-7-4-8-12(19)14(11)16(24)22-13-9-20-23-15(13)17(25)21-10-5-2-1-3-6-10/h4,7-10H,1-3,5-6H2,(H,20,23)(H,21,25)(H,22,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | 7.2 | 22 |
Astex
| Assay Description
CDK2/cyclin A activity was determined using a radiometric assay to measure the incorporation of gamma-phosphate from [gamma-33P]-ATP into histone H1.... |
J Med Chem 51: 4986-99 (2008)
Article DOI: 10.1021/jm800382h
BindingDB Entry DOI: 10.7270/Q24X563W |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Aurora kinase A
(Homo sapiens (Human)) | BDBM27085
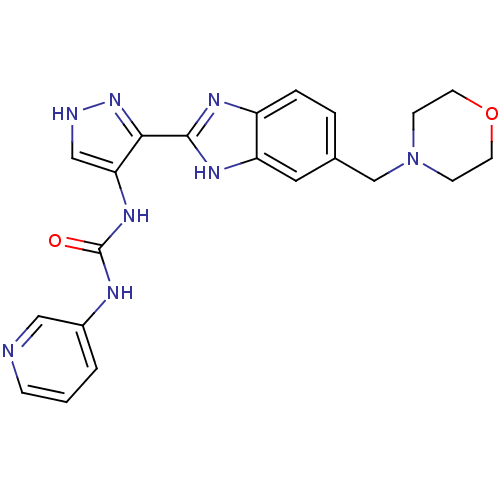
(3-{3-[5-(morpholin-4-ylmethyl)-1H-1,3-benzodiazol-...)Show SMILES O=C(Nc1c[nH]nc1-c1nc2ccc(CN3CCOCC3)cc2[nH]1)Nc1cccnc1 Show InChI InChI=1S/C21H22N8O2/c30-21(24-15-2-1-5-22-11-15)27-18-12-23-28-19(18)20-25-16-4-3-14(10-17(16)26-20)13-29-6-8-31-9-7-29/h1-5,10-12H,6-9,13H2,(H,23,28)(H,25,26)(H2,24,27,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | 7.0 | 22 |
Astex
| Assay Description
Assays for Aurora kinase was performed in a DELFIA format. Aurora enzyme was incubated with test compound and cross-tide substrate in the reaction bu... |
J Med Chem 52: 379-88 (2009)
Article DOI: 10.1021/jm800984v
BindingDB Entry DOI: 10.7270/Q2P55KT7 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Aurora kinase B
(Homo sapiens (Human)) | BDBM27079

(N-{3-[5-(morpholin-4-ylmethyl)-1H-1,3-benzodiazol-...)Show SMILES O=C(Nc1c[nH]nc1-c1nc2ccc(CN3CCOCC3)cc2[nH]1)c1ccccc1 Show InChI InChI=1S/C22H22N6O2/c29-22(16-4-2-1-3-5-16)26-19-13-23-27-20(19)21-24-17-7-6-15(12-18(17)25-21)14-28-8-10-30-11-9-28/h1-7,12-13H,8-11,14H2,(H,23,27)(H,24,25)(H,26,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Astex
| Assay Description
Assays for Aurora kinase was performed in a DELFIA format. Aurora enzyme was incubated with test compound and cross-tide substrate in the reaction bu... |
J Med Chem 52: 379-88 (2009)
Article DOI: 10.1021/jm800984v
BindingDB Entry DOI: 10.7270/Q2P55KT7 |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50179208
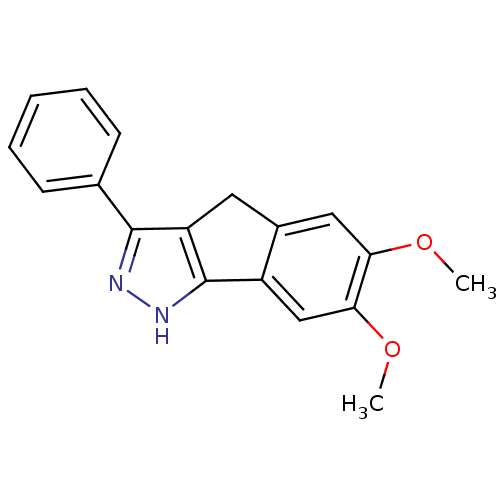
(6,7-dimethoxy-3-phenyl-1,4-dihydroindeno[1,2-c]pyr...)Show InChI InChI=1S/C18H16N2O2/c1-21-15-9-12-8-14-17(11-6-4-3-5-7-11)19-20-18(14)13(12)10-16(15)22-2/h3-7,9-10H,8H2,1-2H3,(H,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDGFRbeta kinase |
J Med Chem 48: 8163-73 (2005)
Article DOI: 10.1021/jm050680m
BindingDB Entry DOI: 10.7270/Q2W095HW |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50179208

(6,7-dimethoxy-3-phenyl-1,4-dihydroindeno[1,2-c]pyr...)Show InChI InChI=1S/C18H16N2O2/c1-21-15-9-12-8-14-17(11-6-4-3-5-7-11)19-20-18(14)13(12)10-16(15)22-2/h3-7,9-10H,8H2,1-2H3,(H,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Johnson & Johnson Pharmaceutical Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against PDGFRbeta kinase |
J Med Chem 48: 8163-73 (2005)
Article DOI: 10.1021/jm050680m
BindingDB Entry DOI: 10.7270/Q2W095HW |
More data for this
Ligand-Target Pair | |
Aurora kinase B
(Homo sapiens (Human)) | BDBM13534

(CHEMBL572878 | N-[4-({4-[(3-methyl-1H-pyrazol-5-yl...)Show SMILES CN1CCN(CC1)c1cc(Nc2cc(C)n[nH]2)nc(Sc2ccc(NC(=O)C3CC3)cc2)n1 Show InChI InChI=1S/C23H28N8OS/c1-15-13-20(29-28-15)25-19-14-21(31-11-9-30(2)10-12-31)27-23(26-19)33-18-7-5-17(6-8-18)24-22(32)16-3-4-16/h5-8,13-14,16H,3-4,9-12H2,1-2H3,(H,24,32)(H2,25,26,27,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Astex
| Assay Description
Assays for Aurora kinase was performed in a DELFIA format. Aurora enzyme was incubated with test compound and cross-tide substrate in the reaction bu... |
J Med Chem 52: 379-88 (2009)
Article DOI: 10.1021/jm800984v
BindingDB Entry DOI: 10.7270/Q2P55KT7 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-dependent kinase 5 activator 1
(Homo sapiens (Human)) | BDBM24654
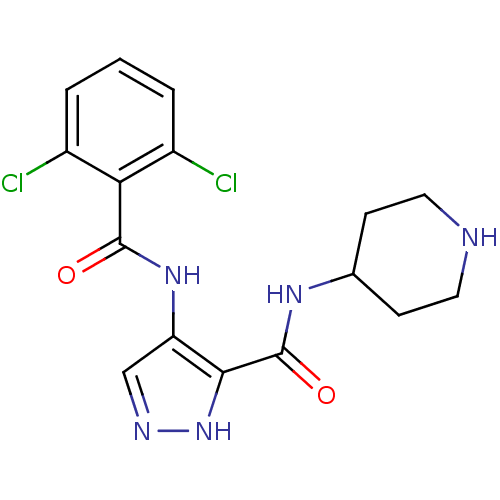
(4-N-(2,6-dichlorobenzene)-3-N-(piperidin-4-yl)-1H-...)Show SMILES Clc1cccc(Cl)c1C(=O)Nc1cn[nH]c1C(=O)NC1CCNCC1 Show InChI InChI=1S/C16H17Cl2N5O2/c17-10-2-1-3-11(18)13(10)15(24)22-12-8-20-23-14(12)16(25)21-9-4-6-19-7-5-9/h1-3,8-9,19H,4-7H2,(H,20,23)(H,21,25)(H,22,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Astex
| Assay Description
CDK5/p35 activity was determined in DELFIA format using a biotinylated Histone H1 peptide. The phosphorylated peptide was quantified using rabbit pho... |
J Med Chem 51: 4986-99 (2008)
Article DOI: 10.1021/jm800382h
BindingDB Entry DOI: 10.7270/Q24X563W |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data