

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429170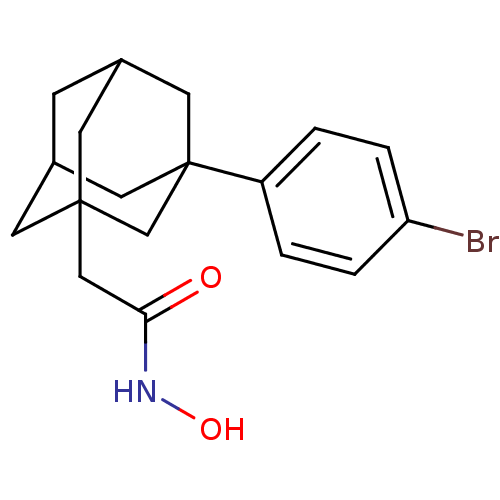 (CHEMBL2336715) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAP-66mer (141-206aa) as substrate by FRET ... | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429171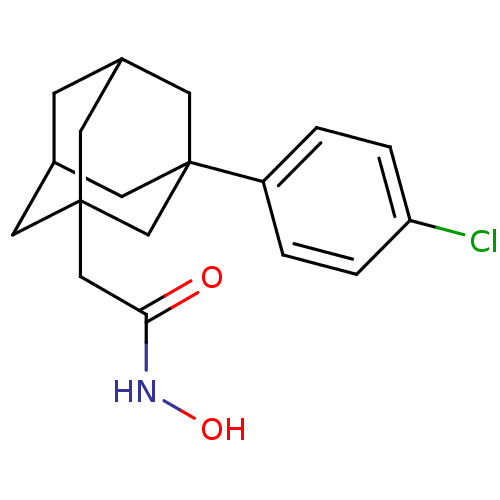 (CHEMBL2336714) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAP-66mer (141-206aa) as substrate by FRET ... | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429172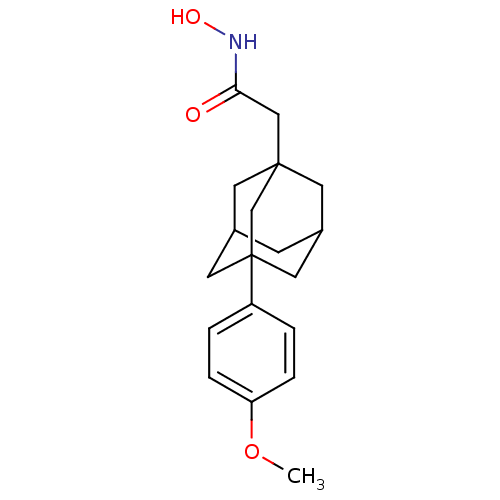 (CHEMBL2336713) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAP-66mer (141-206aa) as substrate by FRET ... | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429169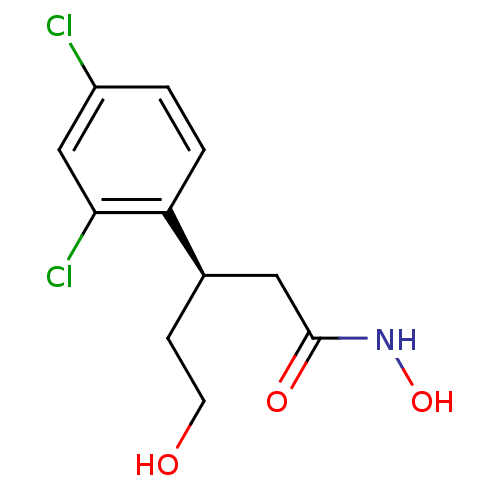 (CHEMBL2336719) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAP-66mer (141-206aa) as substrate by FRET ... | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM23274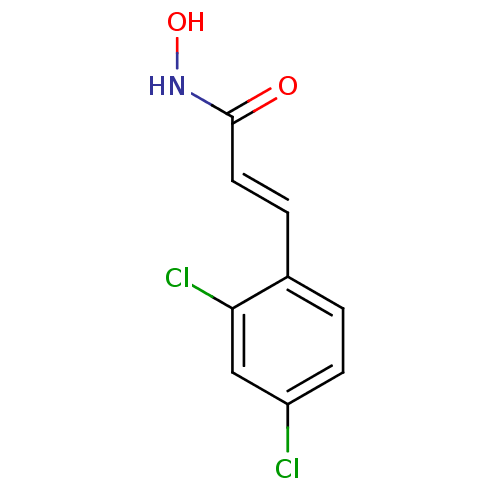 ((2E)-3-(2,4-dichlorophenyl)-N-hydroxyprop-2-enamid...) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | DrugBank MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAP-66mer (141-206aa) as substrate by FRET ... | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM23274 ((2E)-3-(2,4-dichlorophenyl)-N-hydroxyprop-2-enamid...) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | DrugBank MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of Clostridium botulinum BoNT/A using SNAP-25 (141-206) as substrate by HPLC analysis | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01006 BindingDB Entry DOI: 10.7270/Q2WD446P | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM23274 ((2E)-3-(2,4-dichlorophenyl)-N-hydroxyprop-2-enamid...) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | DrugBank MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of Clostridium botulinum BoNT/A light chain | Citation and Details Article DOI: 10.1039/d1md00089f BindingDB Entry DOI: 10.7270/Q24T6P2K | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429174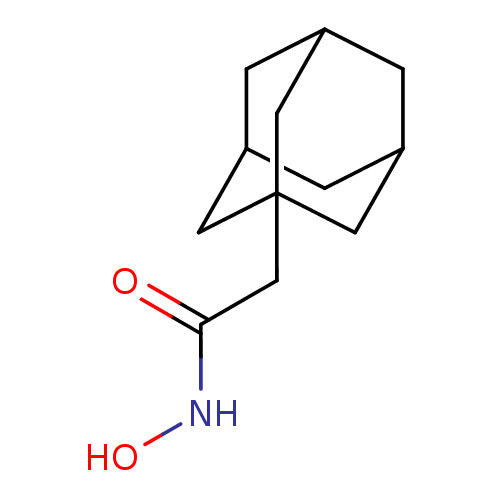 (1-Adamantyl N-Hydroxyacetamide | CHEMBL2336721) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem | PDB Article PubMed | 460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAP-66mer (141-206aa) as substrate by FRET ... | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429173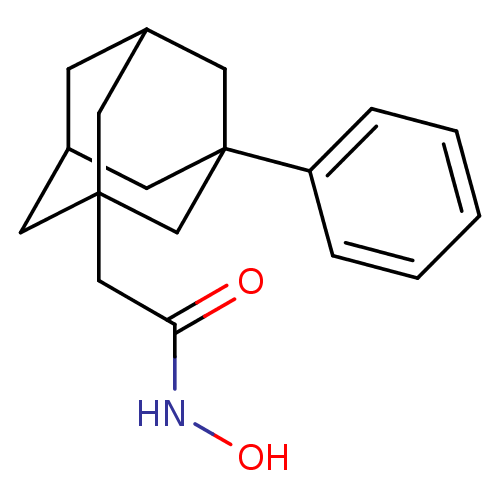 (CHEMBL2336709) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 750 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAP-66mer (141-206aa) as substrate by FRET ... | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429168 (CHEMBL2336720) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 760 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAP-66mer (141-206aa) as substrate by FRET ... | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50445596 (CHEMBL3103447) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A light chain (1 to 425 amino acids) by LC-MS analysis | Bioorg Med Chem 22: 1208-17 (2014) Article DOI: 10.1016/j.bmc.2013.11.053 BindingDB Entry DOI: 10.7270/Q2W66N71 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50546871 (CHEMBL4745069) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Irreversible inhibition of Clostridium botulinum BoNT/A light chain expressed in Escherichia coli BL21 (DE3) using SNAPtide flp6 as substrate preincu... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01006 BindingDB Entry DOI: 10.7270/Q2WD446P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50445595 (CHEMBL3103453) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A light chain (1 to 425 amino acids) by LC-MS analysis | Bioorg Med Chem 22: 1208-17 (2014) Article DOI: 10.1016/j.bmc.2013.11.053 BindingDB Entry DOI: 10.7270/Q2W66N71 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50048539 (CHEMBL3309328) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem | Article PubMed | 7.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Irreversible inhibition of recombinant Clostridium botulinum N-terminal 6His-tagged BoNT/A (Met1 to Phe425 residues) catalytic domain expressed in Es... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01006 BindingDB Entry DOI: 10.7270/Q2WD446P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50546870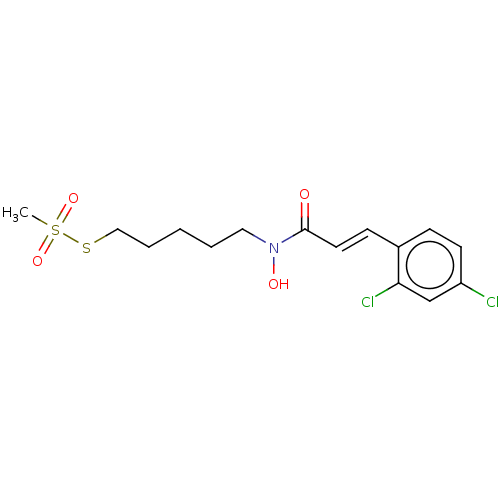 (CHEMBL4790141) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 8.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Irreversible inhibition of Clostridium botulinum BoNT/A light chain expressed in Escherichia coli BL21 (DE3) using SNAPtide flp6 as substrate preincu... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01006 BindingDB Entry DOI: 10.7270/Q2WD446P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50546872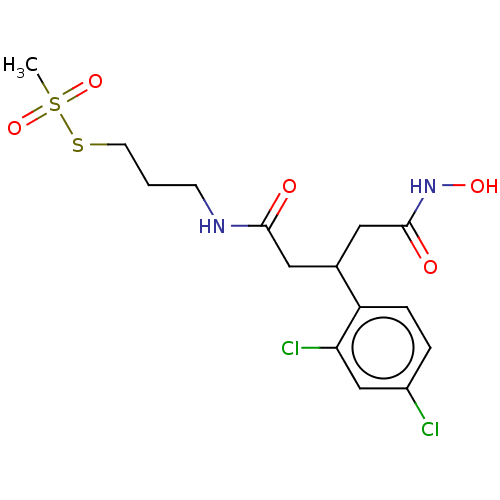 (CHEMBL4761825) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Time dependent inhibition of Clostridium botulinum BoNT/A light chain expressed in Escherichia coli BL21 (DE3) using SNAPtide flp6 as substrate by me... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01006 BindingDB Entry DOI: 10.7270/Q2WD446P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50546874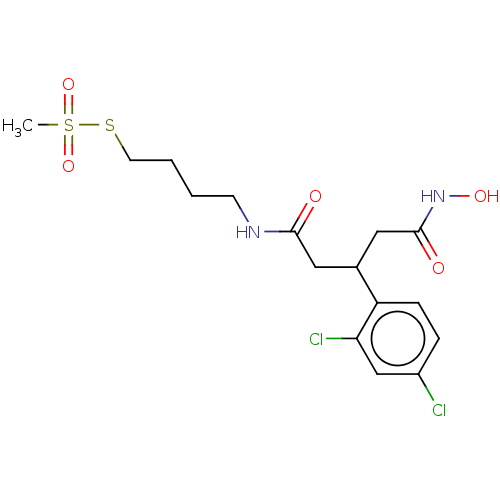 (CHEMBL4795025) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 3.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Time dependent inhibition of Clostridium botulinum BoNT/A light chain expressed in Escherichia coli BL21 (DE3) using SNAPtide flp6 as substrate by me... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01006 BindingDB Entry DOI: 10.7270/Q2WD446P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50546876 (CHEMBL4787837) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 3.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Time dependent inhibition of Clostridium botulinum BoNT/A light chain expressed in Escherichia coli BL21 (DE3) using SNAPtide flp6 as substrate by me... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01006 BindingDB Entry DOI: 10.7270/Q2WD446P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50546880 (CHEMBL4799810) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 4.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Irreversible inhibition of Clostridium botulinum BoNT/A light chain expressed in Escherichia coli BL21 (DE3) using SNAPtide as substrate preincubated... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01006 BindingDB Entry DOI: 10.7270/Q2WD446P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50546875 (CHEMBL4746123) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 4.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Time dependent inhibition of Clostridium botulinum BoNT/A light chain expressed in Escherichia coli BL21 (DE3) using SNAPtide flp6 as substrate by me... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01006 BindingDB Entry DOI: 10.7270/Q2WD446P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50048539 (CHEMBL3309328) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem | Article PubMed | 5.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Covalent inhibition of Clostridium botulinum BoNT/A light chain expressed in Escherichia coli BL21 (DE3) using SNAPtide flp6 as substrate preincubate... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01006 BindingDB Entry DOI: 10.7270/Q2WD446P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50546878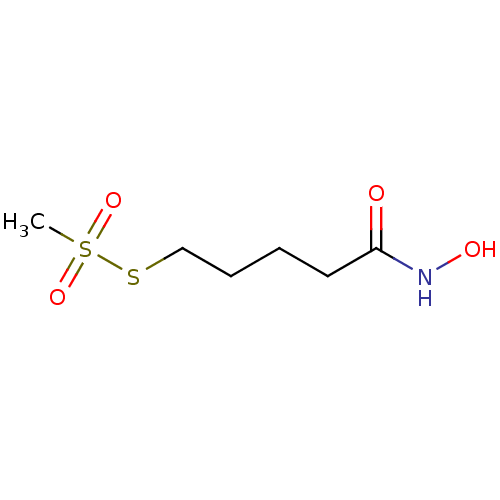 (CHEMBL4790780) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 6.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Irreversible inhibition of Clostridium botulinum BoNT/A light chain expressed in Escherichia coli BL21 (DE3) using SNAPtide as substrate preincubated... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01006 BindingDB Entry DOI: 10.7270/Q2WD446P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50546877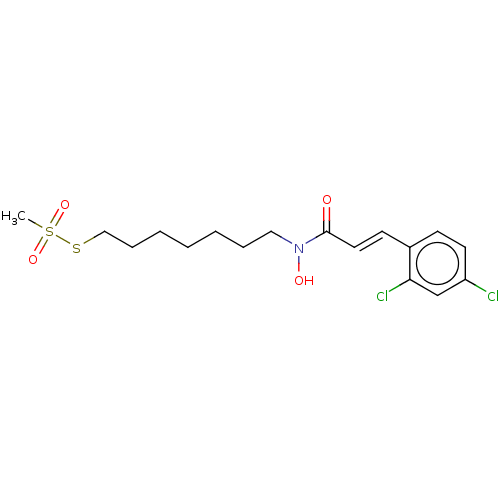 (CHEMBL4743480) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 8.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Irreversible inhibition of Clostridium botulinum BoNT/A light chain expressed in Escherichia coli BL21 (DE3) using SNAPtide flp6 as substrate preincu... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01006 BindingDB Entry DOI: 10.7270/Q2WD446P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50546879 (CHEMBL4787587) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 8.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Irreversible inhibition of Clostridium botulinum BoNT/A light chain expressed in Escherichia coli BL21 (DE3) using SNAPtide as substrate preincubated... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01006 BindingDB Entry DOI: 10.7270/Q2WD446P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50546881 (CHEMBL4761785) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 9.98E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Irreversible inhibition of Clostridium botulinum BoNT/A light chain expressed in Escherichia coli BL21 (DE3) using SNAPtide as substrate preincubated... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01006 BindingDB Entry DOI: 10.7270/Q2WD446P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429171 (CHEMBL2336714) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAPtide as substrate by FRET assay | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429170 (CHEMBL2336715) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAPtide as substrate by FRET assay | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429175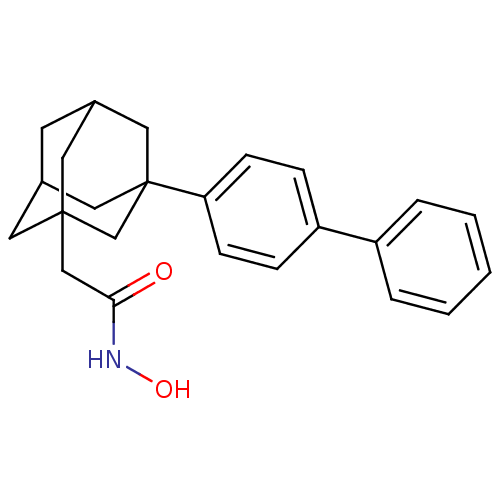 (CHEMBL2336718) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAPtide as substrate by FRET assay | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429172 (CHEMBL2336713) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 250 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAPtide as substrate by FRET assay | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429178 (CHEMBL2336712) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 250 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAPtide as substrate by FRET assay | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429180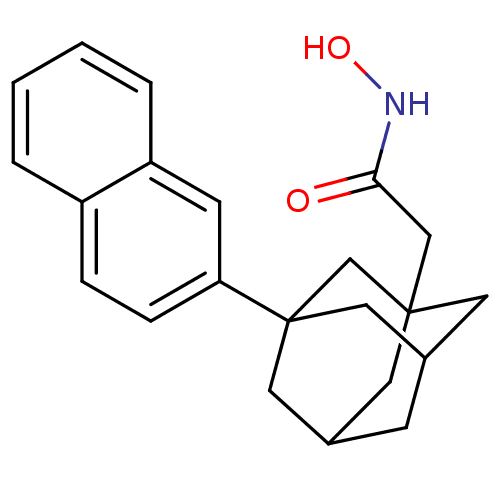 (CHEMBL2336710) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAPtide as substrate by FRET assay | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429176 (CHEMBL2336717) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAPtide as substrate by FRET assay | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM23274 ((2E)-3-(2,4-dichlorophenyl)-N-hydroxyprop-2-enamid...) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | DrugBank MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 410 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of Clostridium botulinum BoNT/A | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c01006 BindingDB Entry DOI: 10.7270/Q2WD446P | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429173 (CHEMBL2336709) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAPtide as substrate by FRET assay | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429177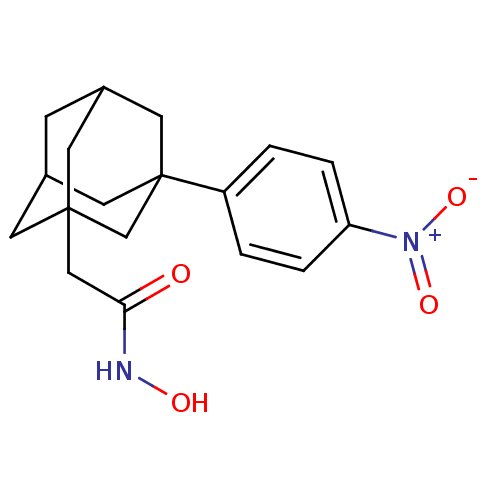 (CHEMBL2336716) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAPtide as substrate by FRET assay | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM23289 ((2E)-3-(4-chloro-2-methylphenyl)-N-hydroxyprop-2-e...) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A | Bioorg Med Chem Lett 20: 206-8 (2010) Article DOI: 10.1016/j.bmcl.2009.10.129 BindingDB Entry DOI: 10.7270/Q2H41RJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM23274 ((2E)-3-(2,4-dichlorophenyl)-N-hydroxyprop-2-enamid...) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | DrugBank MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 900 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A | Bioorg Med Chem Lett 20: 206-8 (2010) Article DOI: 10.1016/j.bmcl.2009.10.129 BindingDB Entry DOI: 10.7270/Q2H41RJ9 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50445595 (CHEMBL3103453) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 970 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A light chain (1 to 425 amino acids) by FRET method | Bioorg Med Chem 22: 1208-17 (2014) Article DOI: 10.1016/j.bmc.2013.11.053 BindingDB Entry DOI: 10.7270/Q2W66N71 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429174 (1-Adamantyl N-Hydroxyacetamide | CHEMBL2336721) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem | PDB Article PubMed | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAPtide as substrate by FRET assay | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429179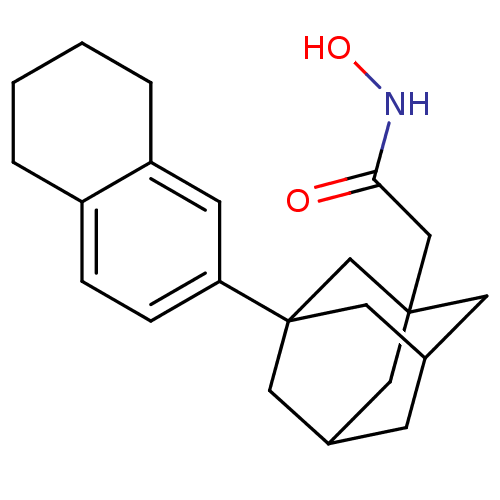 (CHEMBL2336711) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAPtide as substrate by FRET assay | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50445596 (CHEMBL3103447) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A light chain (1 to 425 amino acids) by FRET method | Bioorg Med Chem 22: 1208-17 (2014) Article DOI: 10.1016/j.bmc.2013.11.053 BindingDB Entry DOI: 10.7270/Q2W66N71 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429183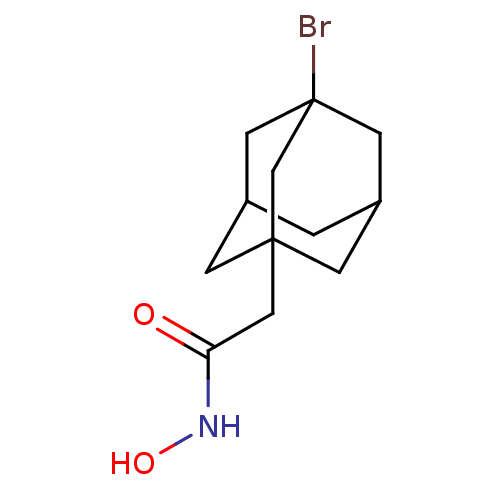 (CHEMBL2336727) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAPtide as substrate by FRET assay | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429184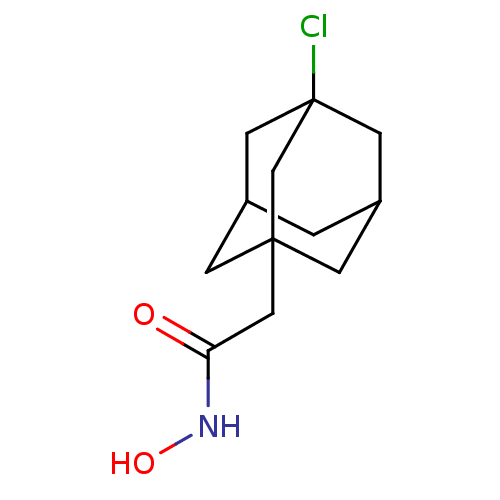 (CHEMBL2336726) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAPtide as substrate by FRET assay | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50445604 (CHEMBL3103448) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A light chain (1 to 425 amino acids) by FRET method | Bioorg Med Chem 22: 1208-17 (2014) Article DOI: 10.1016/j.bmc.2013.11.053 BindingDB Entry DOI: 10.7270/Q2W66N71 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50445602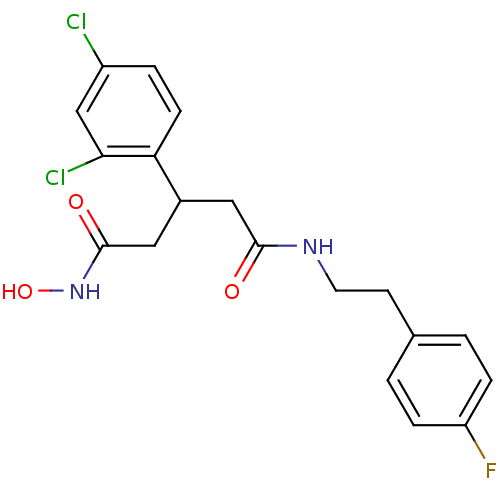 (CHEMBL3103450) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A light chain (1 to 425 amino acids) by FRET method | Bioorg Med Chem 22: 1208-17 (2014) Article DOI: 10.1016/j.bmc.2013.11.053 BindingDB Entry DOI: 10.7270/Q2W66N71 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50445599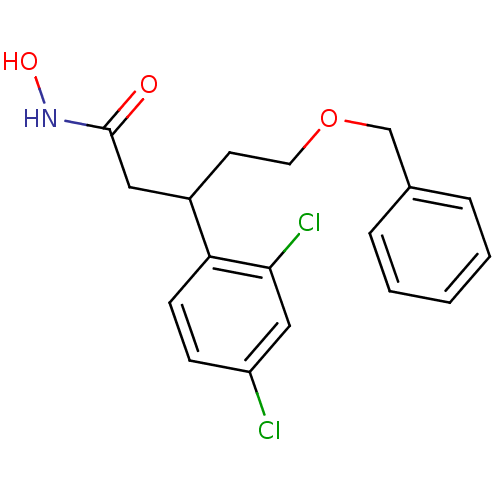 (CHEMBL3103454) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A light chain (1 to 425 amino acids) by FRET method | Bioorg Med Chem 22: 1208-17 (2014) Article DOI: 10.1016/j.bmc.2013.11.053 BindingDB Entry DOI: 10.7270/Q2W66N71 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429185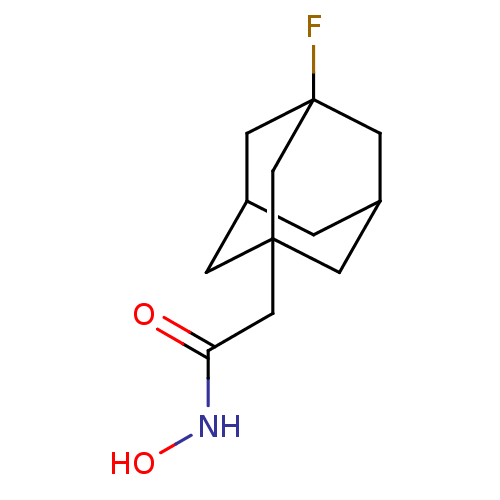 (CHEMBL2336725) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAPtide as substrate by FRET assay | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429181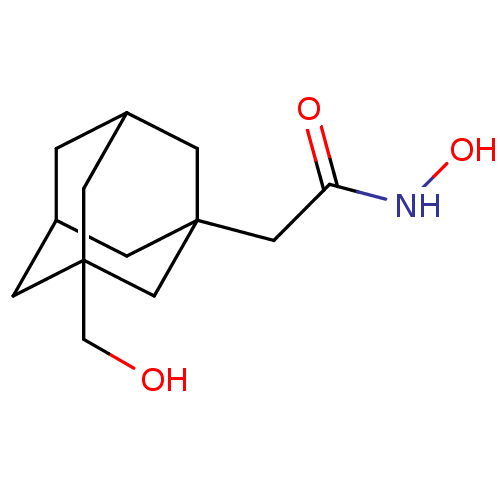 (CHEMBL2336708) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAPtide as substrate by FRET assay | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50445598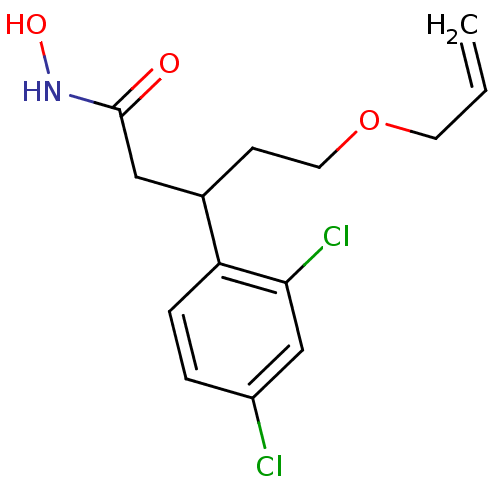 (CHEMBL3103455) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A light chain (1 to 425 amino acids) by FRET method | Bioorg Med Chem 22: 1208-17 (2014) Article DOI: 10.1016/j.bmc.2013.11.053 BindingDB Entry DOI: 10.7270/Q2W66N71 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum) | BDBM50429187 (CHEMBL2336723) | PDB MMDB NCI pathway Reactome pathway UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Curated by ChEMBL | Assay Description Inhibition of Clostridium botulinum BoNT/A Hall A hyper protease light chain (1-425aa) using SNAPtide as substrate by FRET assay | Bioorg Med Chem 21: 1344-8 (2013) Article DOI: 10.1016/j.bmc.2012.12.001 BindingDB Entry DOI: 10.7270/Q2N58NQJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 90 total ) | Next | Last >> |