 Found 58 hits with Last Name = 'masubuchi' and Initial = 'n'
Found 58 hits with Last Name = 'masubuchi' and Initial = 'n' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Renin
(Homo sapiens (Human)) | BDBM50392953
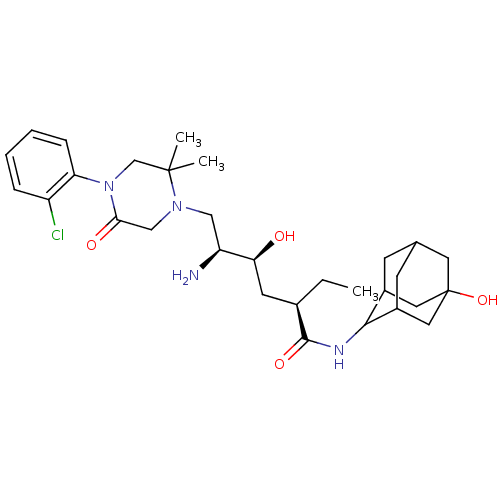
(CHEMBL2152353)Show SMILES CC[C@H](C[C@H](O)[C@@H](N)CN1CC(=O)N(CC1(C)C)c1ccccc1Cl)C(=O)NC1C2CC3CC1CC(O)(C3)C2 |r,wU:4.4,2.2,wD:6.6,TLB:38:35:28.29.30:32,36:35:28:30.31.32,27:28:34.38.35:30.31.32,27:28:32:34.35.37,THB:38:29:32:34.35.37,37:35:28:30.31.32,37:31:28:34.38.35,(13.51,-6.54,;12.17,-7.31,;12.17,-8.85,;10.84,-9.62,;9.51,-8.85,;9.51,-7.31,;8.17,-9.62,;6.84,-8.85,;8.17,-11.16,;6.84,-11.93,;5.5,-11.17,;4.16,-11.94,;2.83,-11.17,;4.18,-13.47,;5.51,-14.24,;6.84,-13.48,;7.61,-14.81,;8.38,-13.48,;2.85,-14.25,;1.51,-13.49,;.18,-14.26,;.18,-15.8,;1.52,-16.57,;2.85,-15.8,;4.19,-16.57,;13.51,-9.62,;13.51,-11.16,;14.84,-8.86,;16.17,-9.63,;17.67,-9.21,;17.67,-7.62,;18.71,-6.39,;17.36,-6.87,;17.37,-8.35,;18.7,-8.84,;20.09,-8.5,;21.42,-9.26,;20.1,-6.97,;19.08,-9.77,)| Show InChI InChI=1S/C30H45ClN4O4/c1-4-19(28(38)33-27-20-9-18-10-21(27)14-30(39,12-18)13-20)11-25(36)23(32)15-34-16-26(37)35(17-29(34,2)3)24-8-6-5-7-22(24)31/h5-8,18-21,23,25,27,36,39H,4,9-17,32H2,1-3H3,(H,33,38)/t18?,19-,20?,21?,23+,25+,27?,30?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin compound treated for 10 mins before substrate addition measured after 90 mins by fluorescence method |
ACS Med Chem Lett 3: 754-758 (2012)
Article DOI: 10.1021/ml300168e
BindingDB Entry DOI: 10.7270/Q29S1S41 |
More data for this
Ligand-Target Pair | |
Renin
(Homo sapiens (Human)) | BDBM17950
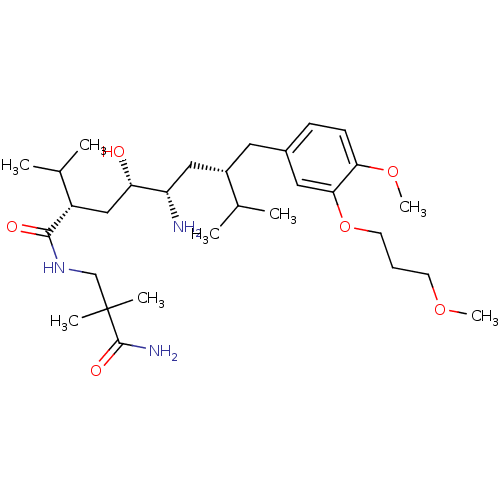
((2S,4S,5S,7S)-5-amino-N-(2-carbamoyl-2,2-dimethyle...)Show SMILES COCCCOc1cc(C[C@@H](C[C@H](N)[C@@H](O)C[C@@H](C(C)C)C(=O)NCC(C)(C)C(N)=O)C(C)C)ccc1OC |r| Show InChI InChI=1S/C30H53N3O6/c1-19(2)22(14-21-10-11-26(38-8)27(15-21)39-13-9-12-37-7)16-24(31)25(34)17-23(20(3)4)28(35)33-18-30(5,6)29(32)36/h10-11,15,19-20,22-25,34H,9,12-14,16-18,31H2,1-8H3,(H2,32,36)(H,33,35)/t22-,23-,24-,25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant renin compound treated for 10 mins before substrate addition measured after 90 mins by fluorescence method |
ACS Med Chem Lett 3: 754-758 (2012)
Article DOI: 10.1021/ml300168e
BindingDB Entry DOI: 10.7270/Q29S1S41 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Renin
(Homo sapiens (Human)) | BDBM50392953

(CHEMBL2152353)Show SMILES CC[C@H](C[C@H](O)[C@@H](N)CN1CC(=O)N(CC1(C)C)c1ccccc1Cl)C(=O)NC1C2CC3CC1CC(O)(C3)C2 |r,wU:4.4,2.2,wD:6.6,TLB:38:35:28.29.30:32,36:35:28:30.31.32,27:28:34.38.35:30.31.32,27:28:32:34.35.37,THB:38:29:32:34.35.37,37:35:28:30.31.32,37:31:28:34.38.35,(13.51,-6.54,;12.17,-7.31,;12.17,-8.85,;10.84,-9.62,;9.51,-8.85,;9.51,-7.31,;8.17,-9.62,;6.84,-8.85,;8.17,-11.16,;6.84,-11.93,;5.5,-11.17,;4.16,-11.94,;2.83,-11.17,;4.18,-13.47,;5.51,-14.24,;6.84,-13.48,;7.61,-14.81,;8.38,-13.48,;2.85,-14.25,;1.51,-13.49,;.18,-14.26,;.18,-15.8,;1.52,-16.57,;2.85,-15.8,;4.19,-16.57,;13.51,-9.62,;13.51,-11.16,;14.84,-8.86,;16.17,-9.63,;17.67,-9.21,;17.67,-7.62,;18.71,-6.39,;17.36,-6.87,;17.37,-8.35,;18.7,-8.84,;20.09,-8.5,;21.42,-9.26,;20.1,-6.97,;19.08,-9.77,)| Show InChI InChI=1S/C30H45ClN4O4/c1-4-19(28(38)33-27-20-9-18-10-21(27)14-30(39,12-18)13-20)11-25(36)23(32)15-34-16-26(37)35(17-29(34,2)3)24-8-6-5-7-22(24)31/h5-8,18-21,23,25,27,36,39H,4,9-17,32H2,1-3H3,(H,33,38)/t18?,19-,20?,21?,23+,25+,27?,30?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human plasma renin activity after 60 mins by competitive RIA |
ACS Med Chem Lett 3: 754-758 (2012)
Article DOI: 10.1021/ml300168e
BindingDB Entry DOI: 10.7270/Q29S1S41 |
More data for this
Ligand-Target Pair | |
Renin
(Homo sapiens (Human)) | BDBM17950

((2S,4S,5S,7S)-5-amino-N-(2-carbamoyl-2,2-dimethyle...)Show SMILES COCCCOc1cc(C[C@@H](C[C@H](N)[C@@H](O)C[C@@H](C(C)C)C(=O)NCC(C)(C)C(N)=O)C(C)C)ccc1OC |r| Show InChI InChI=1S/C30H53N3O6/c1-19(2)22(14-21-10-11-26(38-8)27(15-21)39-13-9-12-37-7)16-24(31)25(34)17-23(20(3)4)28(35)33-18-30(5,6)29(32)36/h10-11,15,19-20,22-25,34H,9,12-14,16-18,31H2,1-8H3,(H2,32,36)(H,33,35)/t22-,23-,24-,25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human plasma renin activity after 60 mins by competitive RIA |
ACS Med Chem Lett 3: 754-758 (2012)
Article DOI: 10.1021/ml300168e
BindingDB Entry DOI: 10.7270/Q29S1S41 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Renin
(Macaca fascicularis) | BDBM50392953

(CHEMBL2152353)Show SMILES CC[C@H](C[C@H](O)[C@@H](N)CN1CC(=O)N(CC1(C)C)c1ccccc1Cl)C(=O)NC1C2CC3CC1CC(O)(C3)C2 |r,wU:4.4,2.2,wD:6.6,TLB:38:35:28.29.30:32,36:35:28:30.31.32,27:28:34.38.35:30.31.32,27:28:32:34.35.37,THB:38:29:32:34.35.37,37:35:28:30.31.32,37:31:28:34.38.35,(13.51,-6.54,;12.17,-7.31,;12.17,-8.85,;10.84,-9.62,;9.51,-8.85,;9.51,-7.31,;8.17,-9.62,;6.84,-8.85,;8.17,-11.16,;6.84,-11.93,;5.5,-11.17,;4.16,-11.94,;2.83,-11.17,;4.18,-13.47,;5.51,-14.24,;6.84,-13.48,;7.61,-14.81,;8.38,-13.48,;2.85,-14.25,;1.51,-13.49,;.18,-14.26,;.18,-15.8,;1.52,-16.57,;2.85,-15.8,;4.19,-16.57,;13.51,-9.62,;13.51,-11.16,;14.84,-8.86,;16.17,-9.63,;17.67,-9.21,;17.67,-7.62,;18.71,-6.39,;17.36,-6.87,;17.37,-8.35,;18.7,-8.84,;20.09,-8.5,;21.42,-9.26,;20.1,-6.97,;19.08,-9.77,)| Show InChI InChI=1S/C30H45ClN4O4/c1-4-19(28(38)33-27-20-9-18-10-21(27)14-30(39,12-18)13-20)11-25(36)23(32)15-34-16-26(37)35(17-29(34,2)3)24-8-6-5-7-22(24)31/h5-8,18-21,23,25,27,36,39H,4,9-17,32H2,1-3H3,(H,33,38)/t18?,19-,20?,21?,23+,25+,27?,30?/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of cynomolgus monkey plasma renin activity after 60 mins by competitive RIA |
ACS Med Chem Lett 3: 754-758 (2012)
Article DOI: 10.1021/ml300168e
BindingDB Entry DOI: 10.7270/Q29S1S41 |
More data for this
Ligand-Target Pair | |
Renin
(Macaca fascicularis) | BDBM17950

((2S,4S,5S,7S)-5-amino-N-(2-carbamoyl-2,2-dimethyle...)Show SMILES COCCCOc1cc(C[C@@H](C[C@H](N)[C@@H](O)C[C@@H](C(C)C)C(=O)NCC(C)(C)C(N)=O)C(C)C)ccc1OC |r| Show InChI InChI=1S/C30H53N3O6/c1-19(2)22(14-21-10-11-26(38-8)27(15-21)39-13-9-12-37-7)16-24(31)25(34)17-23(20(3)4)28(35)33-18-30(5,6)29(32)36/h10-11,15,19-20,22-25,34H,9,12-14,16-18,31H2,1-8H3,(H2,32,36)(H,33,35)/t22-,23-,24-,25-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of cynomolgus monkey plasma renin activity after 60 mins by competitive RIA |
ACS Med Chem Lett 3: 754-758 (2012)
Article DOI: 10.1021/ml300168e
BindingDB Entry DOI: 10.7270/Q29S1S41 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cathepsin D
(Homo sapiens (Human)) | BDBM50392953

(CHEMBL2152353)Show SMILES CC[C@H](C[C@H](O)[C@@H](N)CN1CC(=O)N(CC1(C)C)c1ccccc1Cl)C(=O)NC1C2CC3CC1CC(O)(C3)C2 |r,wU:4.4,2.2,wD:6.6,TLB:38:35:28.29.30:32,36:35:28:30.31.32,27:28:34.38.35:30.31.32,27:28:32:34.35.37,THB:38:29:32:34.35.37,37:35:28:30.31.32,37:31:28:34.38.35,(13.51,-6.54,;12.17,-7.31,;12.17,-8.85,;10.84,-9.62,;9.51,-8.85,;9.51,-7.31,;8.17,-9.62,;6.84,-8.85,;8.17,-11.16,;6.84,-11.93,;5.5,-11.17,;4.16,-11.94,;2.83,-11.17,;4.18,-13.47,;5.51,-14.24,;6.84,-13.48,;7.61,-14.81,;8.38,-13.48,;2.85,-14.25,;1.51,-13.49,;.18,-14.26,;.18,-15.8,;1.52,-16.57,;2.85,-15.8,;4.19,-16.57,;13.51,-9.62,;13.51,-11.16,;14.84,-8.86,;16.17,-9.63,;17.67,-9.21,;17.67,-7.62,;18.71,-6.39,;17.36,-6.87,;17.37,-8.35,;18.7,-8.84,;20.09,-8.5,;21.42,-9.26,;20.1,-6.97,;19.08,-9.77,)| Show InChI InChI=1S/C30H45ClN4O4/c1-4-19(28(38)33-27-20-9-18-10-21(27)14-30(39,12-18)13-20)11-25(36)23(32)15-34-16-26(37)35(17-29(34,2)3)24-8-6-5-7-22(24)31/h5-8,18-21,23,25,27,36,39H,4,9-17,32H2,1-3H3,(H,33,38)/t18?,19-,20?,21?,23+,25+,27?,30?/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin D |
ACS Med Chem Lett 3: 754-758 (2012)
Article DOI: 10.1021/ml300168e
BindingDB Entry DOI: 10.7270/Q29S1S41 |
More data for this
Ligand-Target Pair | |
Cathepsin E
(Homo sapiens (Human)) | BDBM50392953

(CHEMBL2152353)Show SMILES CC[C@H](C[C@H](O)[C@@H](N)CN1CC(=O)N(CC1(C)C)c1ccccc1Cl)C(=O)NC1C2CC3CC1CC(O)(C3)C2 |r,wU:4.4,2.2,wD:6.6,TLB:38:35:28.29.30:32,36:35:28:30.31.32,27:28:34.38.35:30.31.32,27:28:32:34.35.37,THB:38:29:32:34.35.37,37:35:28:30.31.32,37:31:28:34.38.35,(13.51,-6.54,;12.17,-7.31,;12.17,-8.85,;10.84,-9.62,;9.51,-8.85,;9.51,-7.31,;8.17,-9.62,;6.84,-8.85,;8.17,-11.16,;6.84,-11.93,;5.5,-11.17,;4.16,-11.94,;2.83,-11.17,;4.18,-13.47,;5.51,-14.24,;6.84,-13.48,;7.61,-14.81,;8.38,-13.48,;2.85,-14.25,;1.51,-13.49,;.18,-14.26,;.18,-15.8,;1.52,-16.57,;2.85,-15.8,;4.19,-16.57,;13.51,-9.62,;13.51,-11.16,;14.84,-8.86,;16.17,-9.63,;17.67,-9.21,;17.67,-7.62,;18.71,-6.39,;17.36,-6.87,;17.37,-8.35,;18.7,-8.84,;20.09,-8.5,;21.42,-9.26,;20.1,-6.97,;19.08,-9.77,)| Show InChI InChI=1S/C30H45ClN4O4/c1-4-19(28(38)33-27-20-9-18-10-21(27)14-30(39,12-18)13-20)11-25(36)23(32)15-34-16-26(37)35(17-29(34,2)3)24-8-6-5-7-22(24)31/h5-8,18-21,23,25,27,36,39H,4,9-17,32H2,1-3H3,(H,33,38)/t18?,19-,20?,21?,23+,25+,27?,30?/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin E |
ACS Med Chem Lett 3: 754-758 (2012)
Article DOI: 10.1021/ml300168e
BindingDB Entry DOI: 10.7270/Q29S1S41 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50381899
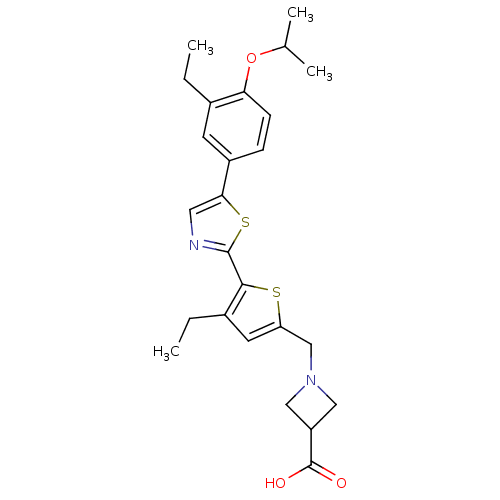
(CHEMBL2022904)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1ncc(s1)-c1ccc(OC(C)C)c(CC)c1 Show InChI InChI=1S/C25H30N2O3S2/c1-5-16-9-18(7-8-21(16)30-15(3)4)22-11-26-24(32-22)23-17(6-2)10-20(31-23)14-27-12-19(13-27)25(28)29/h7-11,15,19H,5-6,12-14H2,1-4H3,(H,28,29) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50381900
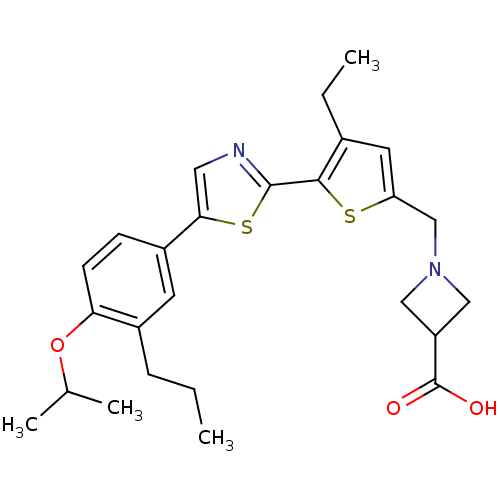
(CHEMBL2022905)Show SMILES CCCc1cc(ccc1OC(C)C)-c1cnc(s1)-c1sc(CN2CC(C2)C(O)=O)cc1CC Show InChI InChI=1S/C26H32N2O3S2/c1-5-7-18-10-19(8-9-22(18)31-16(3)4)23-12-27-25(33-23)24-17(6-2)11-21(32-24)15-28-13-20(14-28)26(29)30/h8-12,16,20H,5-7,13-15H2,1-4H3,(H,29,30) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 400 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50381901
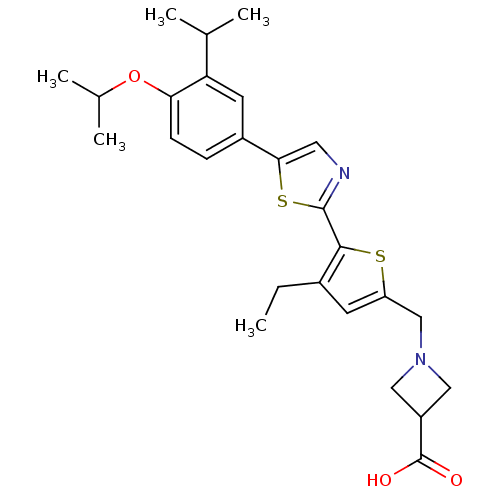
(CHEMBL2022906)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1ncc(s1)-c1ccc(OC(C)C)c(c1)C(C)C Show InChI InChI=1S/C26H32N2O3S2/c1-6-17-9-20(14-28-12-19(13-28)26(29)30)32-24(17)25-27-11-23(33-25)18-7-8-22(31-16(4)5)21(10-18)15(2)3/h7-11,15-16,19H,6,12-14H2,1-5H3,(H,29,30) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50381902
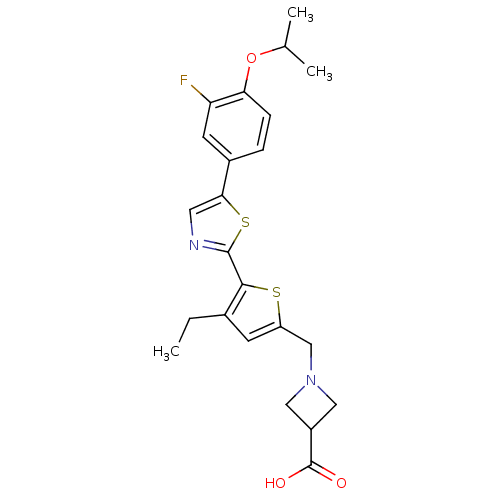
(CHEMBL2022907)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1ncc(s1)-c1ccc(OC(C)C)c(F)c1 Show InChI InChI=1S/C23H25FN2O3S2/c1-4-14-7-17(12-26-10-16(11-26)23(27)28)30-21(14)22-25-9-20(31-22)15-5-6-19(18(24)8-15)29-13(2)3/h5-9,13,16H,4,10-12H2,1-3H3,(H,27,28) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50381903
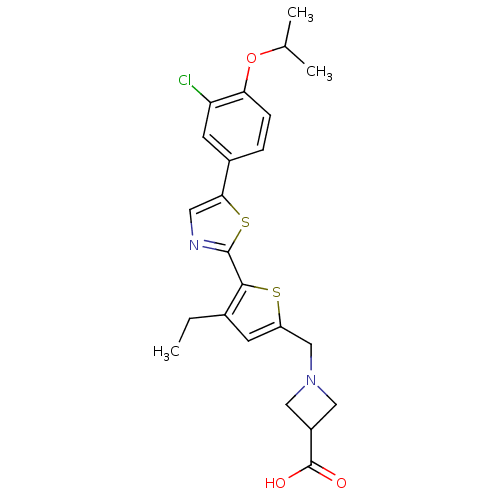
(CHEMBL2022908)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1ncc(s1)-c1ccc(OC(C)C)c(Cl)c1 Show InChI InChI=1S/C23H25ClN2O3S2/c1-4-14-7-17(12-26-10-16(11-26)23(27)28)30-21(14)22-25-9-20(31-22)15-5-6-19(18(24)8-15)29-13(2)3/h5-9,13,16H,4,10-12H2,1-3H3,(H,27,28) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50381904
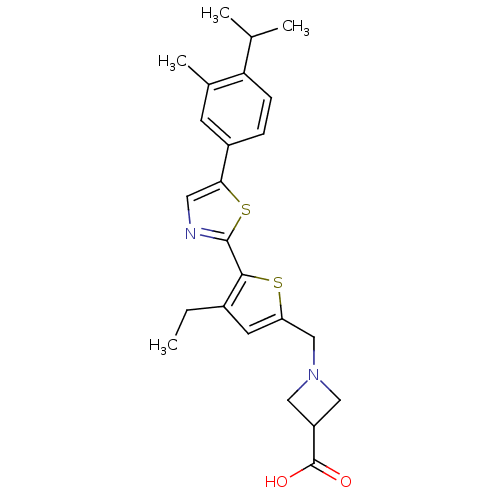
(CHEMBL2022909)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1ncc(s1)-c1ccc(C(C)C)c(C)c1 Show InChI InChI=1S/C24H28N2O2S2/c1-5-16-9-19(13-26-11-18(12-26)24(27)28)29-22(16)23-25-10-21(30-23)17-6-7-20(14(2)3)15(4)8-17/h6-10,14,18H,5,11-13H2,1-4H3,(H,27,28) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50381905
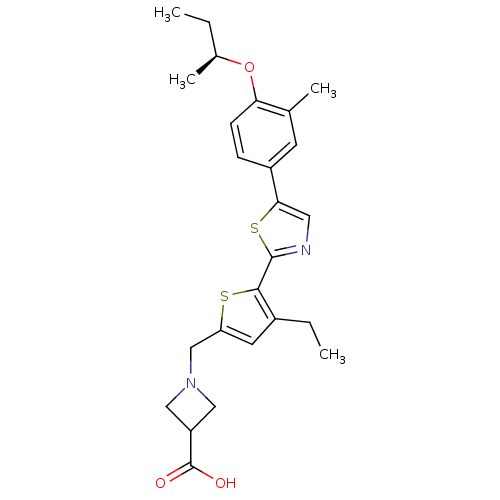
(CHEMBL2022910)Show SMILES CC[C@H](C)Oc1ccc(cc1C)-c1cnc(s1)-c1sc(CN2CC(C2)C(O)=O)cc1CC |r| Show InChI InChI=1S/C25H30N2O3S2/c1-5-16(4)30-21-8-7-18(9-15(21)3)22-11-26-24(32-22)23-17(6-2)10-20(31-23)14-27-12-19(13-27)25(28)29/h7-11,16,19H,5-6,12-14H2,1-4H3,(H,28,29)/t16-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50364607
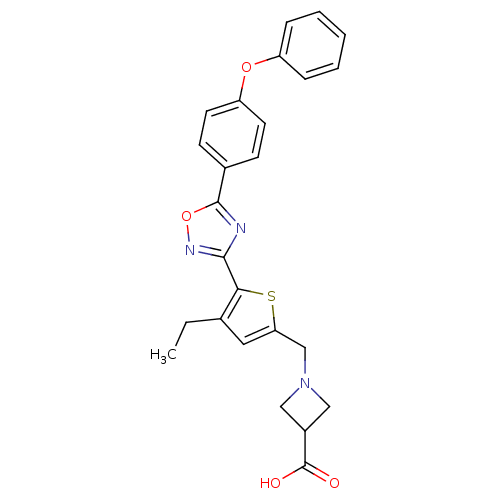
(CHEMBL1951304)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1noc(n1)-c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C25H23N3O4S/c1-2-16-12-21(15-28-13-18(14-28)25(29)30)33-22(16)23-26-24(32-27-23)17-8-10-20(11-9-17)31-19-6-4-3-5-7-19/h3-12,18H,2,13-15H2,1H3,(H,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381906
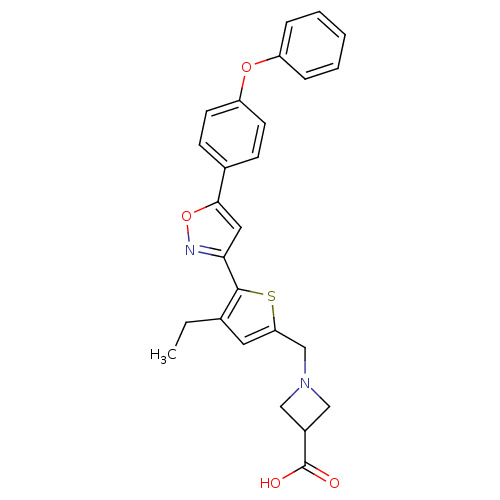
(CHEMBL2022706)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1cc(on1)-c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C26H24N2O4S/c1-2-17-12-22(16-28-14-19(15-28)26(29)30)33-25(17)23-13-24(32-27-23)18-8-10-21(11-9-18)31-20-6-4-3-5-7-20/h3-13,19H,2,14-16H2,1H3,(H,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 22 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381907
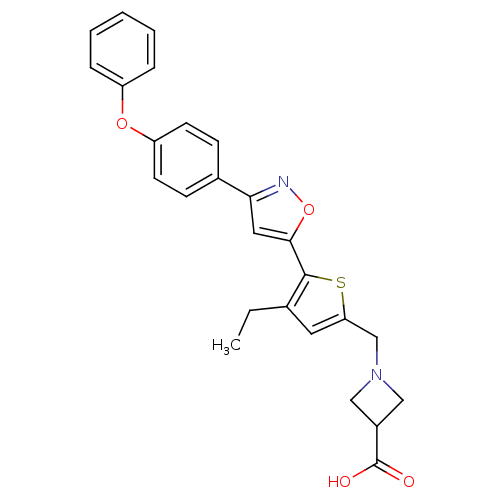
(CHEMBL2022705)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1cc(no1)-c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C26H24N2O4S/c1-2-17-12-22(16-28-14-19(15-28)26(29)30)33-25(17)24-13-23(27-32-24)18-8-10-21(11-9-18)31-20-6-4-3-5-7-20/h3-13,19H,2,14-16H2,1H3,(H,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 15 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381908
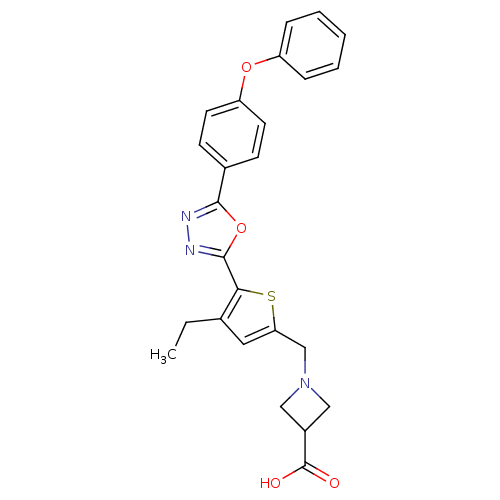
(CHEMBL2022704)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1nnc(o1)-c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C25H23N3O4S/c1-2-16-12-21(15-28-13-18(14-28)25(29)30)33-22(16)24-27-26-23(32-24)17-8-10-20(11-9-17)31-19-6-4-3-5-7-19/h3-12,18H,2,13-15H2,1H3,(H,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 54 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381891

(CHEMBL2022707)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1nnc(s1)-c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C25H23N3O3S2/c1-2-16-12-21(15-28-13-18(14-28)25(29)30)32-22(16)24-27-26-23(33-24)17-8-10-20(11-9-17)31-19-6-4-3-5-7-19/h3-12,18H,2,13-15H2,1H3,(H,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | 25 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381909
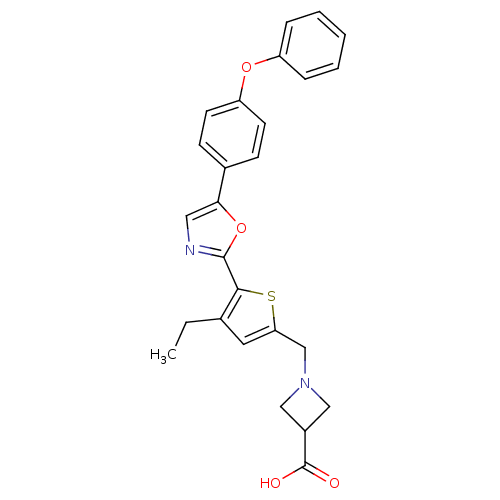
(CHEMBL2022708)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1ncc(o1)-c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C26H24N2O4S/c1-2-17-12-22(16-28-14-19(15-28)26(29)30)33-24(17)25-27-13-23(32-25)18-8-10-21(11-9-18)31-20-6-4-3-5-7-20/h3-13,19H,2,14-16H2,1H3,(H,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 90 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381910
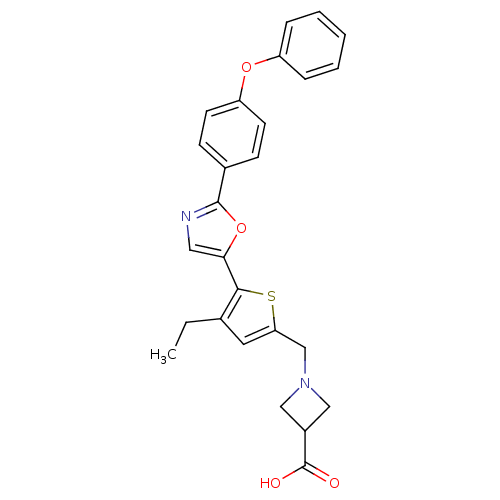
(CHEMBL2022899)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1cnc(o1)-c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C26H24N2O4S/c1-2-17-12-22(16-28-14-19(15-28)26(29)30)33-24(17)23-13-27-25(32-23)18-8-10-21(11-9-18)31-20-6-4-3-5-7-20/h3-13,19H,2,14-16H2,1H3,(H,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 300 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381911
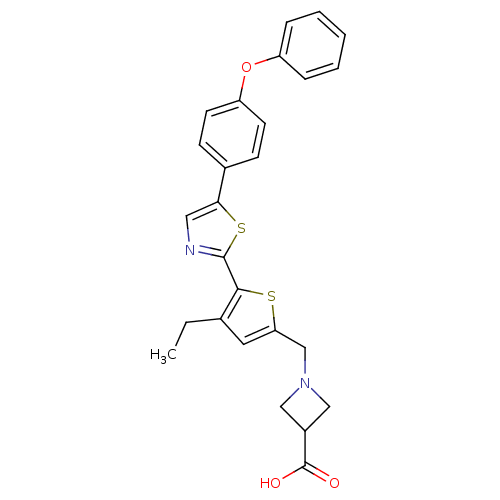
(CHEMBL2022900)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1ncc(s1)-c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C26H24N2O3S2/c1-2-17-12-22(16-28-14-19(15-28)26(29)30)32-24(17)25-27-13-23(33-25)18-8-10-21(11-9-18)31-20-6-4-3-5-7-20/h3-13,19H,2,14-16H2,1H3,(H,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 4.5 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381912
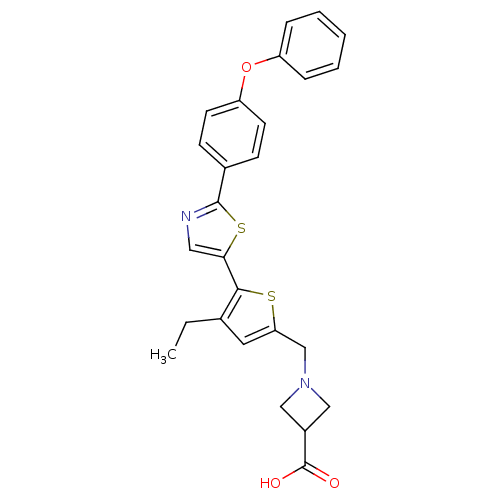
(CHEMBL2022901)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1cnc(s1)-c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C26H24N2O3S2/c1-2-17-12-22(16-28-14-19(15-28)26(29)30)32-24(17)23-13-27-25(33-23)18-8-10-21(11-9-18)31-20-6-4-3-5-7-20/h3-13,19H,2,14-16H2,1H3,(H,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 35 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381913
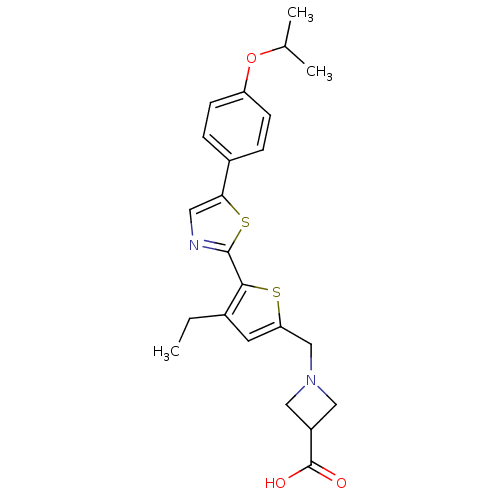
(CHEMBL2022902)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1ncc(s1)-c1ccc(OC(C)C)cc1 Show InChI InChI=1S/C23H26N2O3S2/c1-4-15-9-19(13-25-11-17(12-25)23(26)27)29-21(15)22-24-10-20(30-22)16-5-7-18(8-6-16)28-14(2)3/h5-10,14,17H,4,11-13H2,1-3H3,(H,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 10 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381899

(CHEMBL2022904)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1ncc(s1)-c1ccc(OC(C)C)c(CC)c1 Show InChI InChI=1S/C25H30N2O3S2/c1-5-16-9-18(7-8-21(16)30-15(3)4)22-11-26-24(32-22)23-17(6-2)10-20(31-23)14-27-12-19(13-27)25(28)29/h7-11,15,19H,5-6,12-14H2,1-4H3,(H,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 3.40 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381900

(CHEMBL2022905)Show SMILES CCCc1cc(ccc1OC(C)C)-c1cnc(s1)-c1sc(CN2CC(C2)C(O)=O)cc1CC Show InChI InChI=1S/C26H32N2O3S2/c1-5-7-18-10-19(8-9-22(18)31-16(3)4)23-12-27-25(33-23)24-17(6-2)11-21(32-24)15-28-13-20(14-28)26(29)30/h8-12,16,20H,5-7,13-15H2,1-4H3,(H,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381901

(CHEMBL2022906)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1ncc(s1)-c1ccc(OC(C)C)c(c1)C(C)C Show InChI InChI=1S/C26H32N2O3S2/c1-6-17-9-20(14-28-12-19(13-28)26(29)30)32-24(17)25-27-11-23(33-25)18-7-8-22(31-16(4)5)21(10-18)15(2)3/h7-11,15-16,19H,6,12-14H2,1-5H3,(H,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.30 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381902

(CHEMBL2022907)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1ncc(s1)-c1ccc(OC(C)C)c(F)c1 Show InChI InChI=1S/C23H25FN2O3S2/c1-4-14-7-17(12-26-10-16(11-26)23(27)28)30-21(14)22-25-9-20(31-22)15-5-6-19(18(24)8-15)29-13(2)3/h5-9,13,16H,4,10-12H2,1-3H3,(H,27,28) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 5.80 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381903

(CHEMBL2022908)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1ncc(s1)-c1ccc(OC(C)C)c(Cl)c1 Show InChI InChI=1S/C23H25ClN2O3S2/c1-4-14-7-17(12-26-10-16(11-26)23(27)28)30-21(14)22-25-9-20(31-22)15-5-6-19(18(24)8-15)29-13(2)3/h5-9,13,16H,4,10-12H2,1-3H3,(H,27,28) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 3.20 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381904

(CHEMBL2022909)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1ncc(s1)-c1ccc(C(C)C)c(C)c1 Show InChI InChI=1S/C24H28N2O2S2/c1-5-16-9-19(13-26-11-18(12-26)24(27)28)29-22(16)23-25-10-21(30-23)17-6-7-20(14(2)3)15(4)8-17/h6-10,14,18H,5,11-13H2,1-4H3,(H,27,28) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 3.5 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381905

(CHEMBL2022910)Show SMILES CC[C@H](C)Oc1ccc(cc1C)-c1cnc(s1)-c1sc(CN2CC(C2)C(O)=O)cc1CC |r| Show InChI InChI=1S/C25H30N2O3S2/c1-5-16(4)30-21-8-7-18(9-15(21)3)22-11-26-24(32-22)23-17(6-2)10-20(31-23)14-27-12-19(13-27)25(28)29/h7-11,16,19H,5-6,12-14H2,1-4H3,(H,28,29)/t16-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 6 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
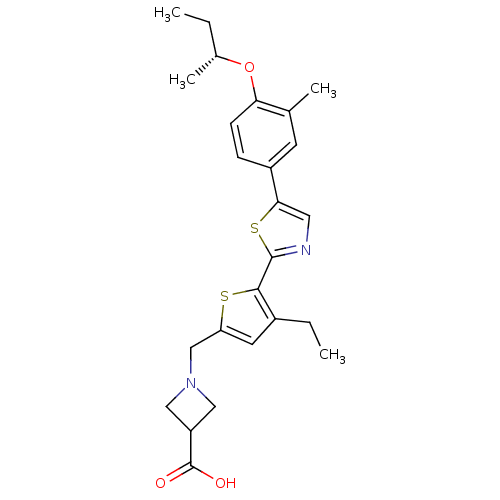
(Homo sapiens (Human)) | BDBM50381893
(CHEMBL2022911)Show SMILES CC[C@@H](C)Oc1ccc(cc1C)-c1cnc(s1)-c1sc(CN2CC(C2)C(O)=O)cc1CC |r| Show InChI InChI=1S/C25H30N2O3S2/c1-5-16(4)30-21-8-7-18(9-15(21)3)22-11-26-24(32-22)23-17(6-2)10-20(31-23)14-27-12-19(13-27)25(28)29/h7-11,16,19H,5-6,12-14H2,1-4H3,(H,28,29)/t16-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 8 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381894

(CHEMBL2022912)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1cnc(s1)-c1ccc(OC(C)C)c(C)c1 Show InChI InChI=1S/C24H28N2O3S2/c1-5-16-9-19(13-26-11-18(12-26)24(27)28)30-22(16)21-10-25-23(31-21)17-6-7-20(15(4)8-17)29-14(2)3/h6-10,14,18H,5,11-13H2,1-4H3,(H,27,28) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 11 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381895
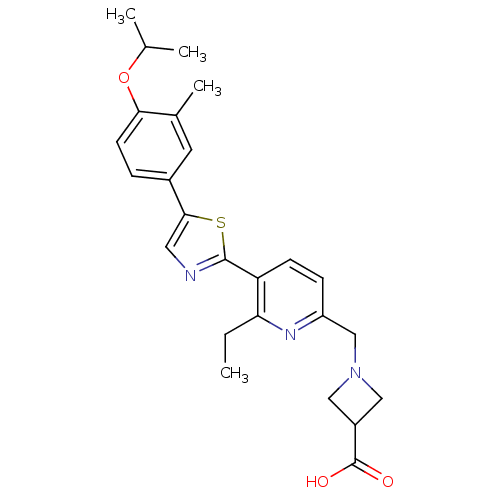
(CHEMBL2022913)Show SMILES CCc1nc(CN2CC(C2)C(O)=O)ccc1-c1ncc(s1)-c1ccc(OC(C)C)c(C)c1 Show InChI InChI=1S/C25H29N3O3S/c1-5-21-20(8-7-19(27-21)14-28-12-18(13-28)25(29)30)24-26-11-23(32-24)17-6-9-22(16(4)10-17)31-15(2)3/h6-11,15,18H,5,12-14H2,1-4H3,(H,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 5.5 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381896
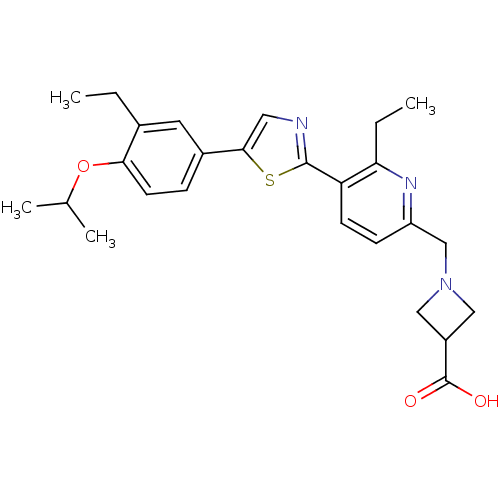
(CHEMBL2022914)Show SMILES CCc1cc(ccc1OC(C)C)-c1cnc(s1)-c1ccc(CN2CC(C2)C(O)=O)nc1CC Show InChI InChI=1S/C26H31N3O3S/c1-5-17-11-18(7-10-23(17)32-16(3)4)24-12-27-25(33-24)21-9-8-20(28-22(21)6-2)15-29-13-19(14-29)26(30)31/h7-12,16,19H,5-6,13-15H2,1-4H3,(H,30,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381892
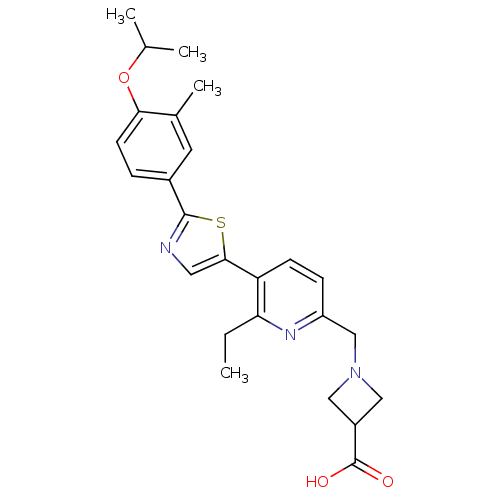
(CHEMBL2022915)Show SMILES CCc1nc(CN2CC(C2)C(O)=O)ccc1-c1cnc(s1)-c1ccc(OC(C)C)c(C)c1 Show InChI InChI=1S/C25H29N3O3S/c1-5-21-20(8-7-19(27-21)14-28-12-18(13-28)25(29)30)23-11-26-24(32-23)17-6-9-22(16(4)10-17)31-15(2)3/h6-11,15,18H,5,12-14H2,1-4H3,(H,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.80 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381897
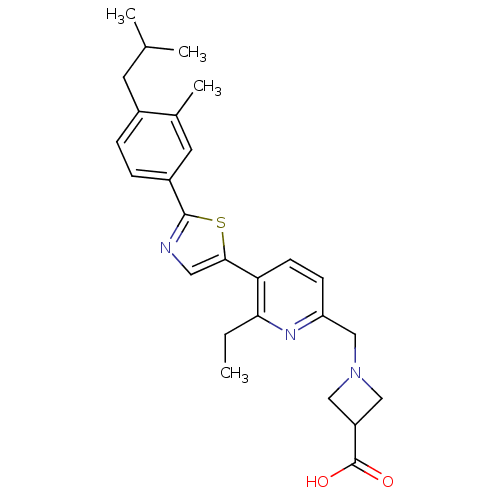
(CHEMBL2022916)Show SMILES CCc1nc(CN2CC(C2)C(O)=O)ccc1-c1cnc(s1)-c1ccc(CC(C)C)c(C)c1 Show InChI InChI=1S/C26H31N3O2S/c1-5-23-22(9-8-21(28-23)15-29-13-20(14-29)26(30)31)24-12-27-25(32-24)19-7-6-18(10-16(2)3)17(4)11-19/h6-9,11-12,16,20H,5,10,13-15H2,1-4H3,(H,30,31) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 4.10 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50381898

(CHEMBL2022917)Show SMILES CCc1cc(ccc1CC(C)C)-c1ncc(s1)-c1ccc(CN2CC(C2)C(O)=O)nc1CC Show InChI InChI=1S/C27H33N3O2S/c1-5-18-12-20(8-7-19(18)11-17(3)4)26-28-13-25(33-26)23-10-9-22(29-24(23)6-2)16-30-14-21(15-30)27(31)32/h7-10,12-13,17,21H,5-6,11,14-16H2,1-4H3,(H,31,32) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 5.40 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50364607

(CHEMBL1951304)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1noc(n1)-c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C25H23N3O4S/c1-2-16-12-21(15-28-13-18(14-28)25(29)30)33-22(16)23-26-24(32-27-23)17-8-10-20(11-9-17)31-19-6-4-3-5-7-19/h3-12,18H,2,13-15H2,1H3,(H,29,30) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50381906

(CHEMBL2022706)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1cc(on1)-c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C26H24N2O4S/c1-2-17-12-22(16-28-14-19(15-28)26(29)30)33-25(17)23-13-24(32-27-23)18-8-10-21(11-9-18)31-20-6-4-3-5-7-20/h3-13,19H,2,14-16H2,1H3,(H,29,30) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50381907

(CHEMBL2022705)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1cc(no1)-c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C26H24N2O4S/c1-2-17-12-22(16-28-14-19(15-28)26(29)30)33-25(17)24-13-23(27-32-24)18-8-10-21(11-9-18)31-20-6-4-3-5-7-20/h3-13,19H,2,14-16H2,1H3,(H,29,30) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50381908

(CHEMBL2022704)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1nnc(o1)-c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C25H23N3O4S/c1-2-16-12-21(15-28-13-18(14-28)25(29)30)33-22(16)24-27-26-23(32-24)17-8-10-20(11-9-17)31-19-6-4-3-5-7-19/h3-12,18H,2,13-15H2,1H3,(H,29,30) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50381909

(CHEMBL2022708)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1ncc(o1)-c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C26H24N2O4S/c1-2-17-12-22(16-28-14-19(15-28)26(29)30)33-24(17)25-27-13-23(32-25)18-8-10-21(11-9-18)31-20-6-4-3-5-7-20/h3-13,19H,2,14-16H2,1H3,(H,29,30) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50381910

(CHEMBL2022899)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1cnc(o1)-c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C26H24N2O4S/c1-2-17-12-22(16-28-14-19(15-28)26(29)30)33-24(17)23-13-27-25(32-23)18-8-10-21(11-9-18)31-20-6-4-3-5-7-20/h3-13,19H,2,14-16H2,1H3,(H,29,30) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50381911

(CHEMBL2022900)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1ncc(s1)-c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C26H24N2O3S2/c1-2-17-12-22(16-28-14-19(15-28)26(29)30)32-24(17)25-27-13-23(33-25)18-8-10-21(11-9-18)31-20-6-4-3-5-7-20/h3-13,19H,2,14-16H2,1H3,(H,29,30) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50381912

(CHEMBL2022901)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1cnc(s1)-c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C26H24N2O3S2/c1-2-17-12-22(16-28-14-19(15-28)26(29)30)32-24(17)23-13-27-25(33-23)18-8-10-21(11-9-18)31-20-6-4-3-5-7-20/h3-13,19H,2,14-16H2,1H3,(H,29,30) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50381913

(CHEMBL2022902)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1ncc(s1)-c1ccc(OC(C)C)cc1 Show InChI InChI=1S/C23H26N2O3S2/c1-4-15-9-19(13-25-11-17(12-25)23(26)27)29-21(15)22-24-10-20(30-22)16-5-7-18(8-6-16)28-14(2)3/h5-10,14,17H,4,11-13H2,1-3H3,(H,26,27) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50381890
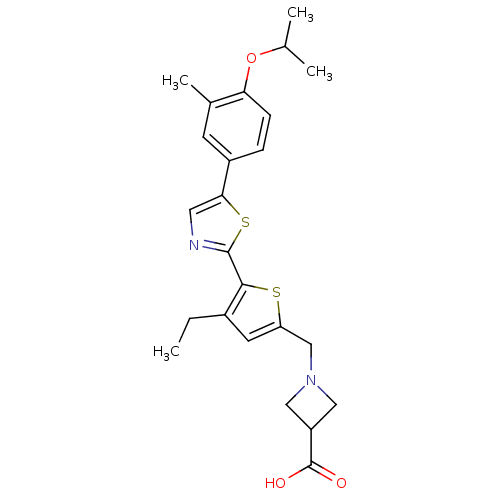
(CHEMBL2022903)Show SMILES CCc1cc(CN2CC(C2)C(O)=O)sc1-c1ncc(s1)-c1ccc(OC(C)C)c(C)c1 Show InChI InChI=1S/C24H28N2O3S2/c1-5-16-9-19(13-26-11-18(12-26)24(27)28)30-22(16)23-25-10-21(31-23)17-6-7-20(15(4)8-17)29-14(2)3/h6-10,14,18H,5,11-13H2,1-4H3,(H,27,28) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 3
(Homo sapiens (Human)) | BDBM50381898

(CHEMBL2022917)Show SMILES CCc1cc(ccc1CC(C)C)-c1ncc(s1)-c1ccc(CN2CC(C2)C(O)=O)nc1CC Show InChI InChI=1S/C27H33N3O2S/c1-5-18-12-20(8-7-19(18)11-17(3)4)26-28-13-25(33-26)23-10-9-22(29-24(23)6-2)16-30-14-21(15-30)27(31)32/h7-10,12-13,17,21H,5-6,11,14-16H2,1-4H3,(H,31,32) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
Daiichi Sankyo Co., Ltd
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P3 receptor by [S35]GTPgammaS binding assay |
Bioorg Med Chem Lett 22: 3083-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.067
BindingDB Entry DOI: 10.7270/Q2JS9RG9 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data