 Found 262 hits with Last Name = 'mcdevitt' and Initial = 'j'
Found 262 hits with Last Name = 'mcdevitt' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cyclin-A2/Cyclin-dependent kinase 1
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 1 | -50.9 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
GlaxoSmithKline
| Assay Description
In vitro CDK enzymatic assay using purified CDK mixed with cyclin A, was incubated with substrate, and test compounds in the presence of 1.4 uM ATP/ ... |
J Biol Chem 277: 46609-15 (2002)
Article DOI: 10.1074/jbc.M201233200
BindingDB Entry DOI: 10.7270/Q2D798PF |
More data for this
Ligand-Target Pair | |
Cyclin-A2/Cyclin-dependent kinase 2
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 2.90 | -48.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
GlaxoSmithKline
| Assay Description
In vitro CDK enzymatic assay using purified CDK mixed with cyclin A, was incubated with substrate, and test compounds in the presence of 1.4 uM ATP/ ... |
J Biol Chem 277: 46609-15 (2002)
Article DOI: 10.1074/jbc.M201233200
BindingDB Entry DOI: 10.7270/Q2D798PF |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Serine/threonine-protein kinase Chk1
(Homo sapiens (Human)) | BDBM17054
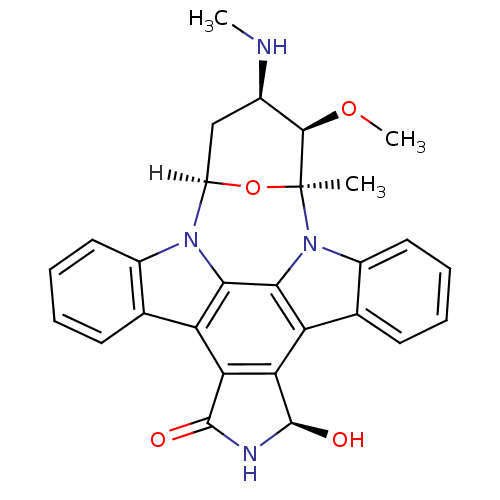
((2S,3R,4R,6R,18R)-18-hydroxy-3-methoxy-2-methyl-4-...)Show SMILES [H][C@]12C[C@@H](NC)[C@@H](OC)[C@](C)(O1)n1c3ccccc3c3c4[C@@H](O)NC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O4/c1-28-25(35-3)15(29-2)12-18(36-28)31-16-10-6-4-8-13(16)19-21-22(27(34)30-26(21)33)20-14-9-5-7-11-17(14)32(28)24(20)23(19)31/h4-11,15,18,25,27,29,34H,12H2,1-3H3,(H,30,33)/t15-,18-,25-,27-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 5.60 | -49.0 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
GlaxoSmithKline
| Assay Description
In vitro Chk1 enzymatic assay using purified enzyme, was incubated with substrate, and test compounds in the presence of 10 uM ATP/ [gamma-32P] ATP. ... |
J Biol Chem 277: 46609-15 (2002)
Article DOI: 10.1074/jbc.M201233200
BindingDB Entry DOI: 10.7270/Q2D798PF |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-A2/Cyclin-dependent kinase 2
(Homo sapiens (Human)) | BDBM17140
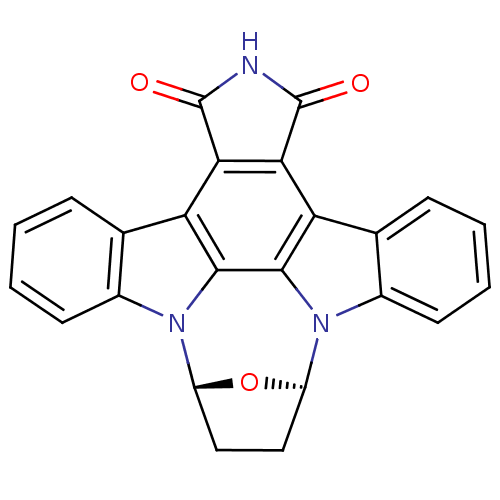
((15R,18R)-28-oxa-4,14,19-triazaoctacyclo[12.11.2.1...)Show SMILES O=C1NC(=O)c2c1c1c3ccccc3n3[C@H]4CC[C@@H](O4)n4c5ccccc5c2c4c13 |r| Show InChI InChI=1S/C24H15N3O3/c28-23-19-17-11-5-1-3-7-13(11)26-15-9-10-16(30-15)27-14-8-4-2-6-12(14)18(22(27)21(17)26)20(19)24(29)25-23/h1-8,15-16H,9-10H2,(H,25,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 5.60 | -46.6 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
GlaxoSmithKline
| Assay Description
In vitro CDK enzymatic assay using purified CDK mixed with cyclin A, was incubated with substrate, and test compounds in the presence of 1.4 uM ATP/ ... |
J Biol Chem 277: 46609-15 (2002)
Article DOI: 10.1074/jbc.M201233200
BindingDB Entry DOI: 10.7270/Q2D798PF |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Serine/threonine-protein kinase Chk1
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 7.80 | -48.1 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
GlaxoSmithKline
| Assay Description
In vitro Chk1 enzymatic assay using purified enzyme, was incubated with substrate, and test compounds in the presence of 10 uM ATP/ [gamma-32P] ATP. ... |
J Biol Chem 277: 46609-15 (2002)
Article DOI: 10.1074/jbc.M201233200
BindingDB Entry DOI: 10.7270/Q2D798PF |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Serine/threonine-protein kinase Chk1
(Homo sapiens (Human)) | BDBM17140

((15R,18R)-28-oxa-4,14,19-triazaoctacyclo[12.11.2.1...)Show SMILES O=C1NC(=O)c2c1c1c3ccccc3n3[C@H]4CC[C@@H](O4)n4c5ccccc5c2c4c13 |r| Show InChI InChI=1S/C24H15N3O3/c28-23-19-17-11-5-1-3-7-13(11)26-15-9-10-16(30-15)27-14-8-4-2-6-12(14)18(22(27)21(17)26)20(19)24(29)25-23/h1-8,15-16H,9-10H2,(H,25,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 15 | -46.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
GlaxoSmithKline
| Assay Description
In vitro Chk1 enzymatic assay using purified enzyme, was incubated with substrate, and test compounds in the presence of 10 uM ATP/ [gamma-32P] ATP. ... |
J Biol Chem 277: 46609-15 (2002)
Article DOI: 10.1074/jbc.M201233200
BindingDB Entry DOI: 10.7270/Q2D798PF |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-dependent kinase 4/G1/S-specific cyclin-D1
(Homo sapiens (Human)) | BDBM17140

((15R,18R)-28-oxa-4,14,19-triazaoctacyclo[12.11.2.1...)Show SMILES O=C1NC(=O)c2c1c1c3ccccc3n3[C@H]4CC[C@@H](O4)n4c5ccccc5c2c4c13 |r| Show InChI InChI=1S/C24H15N3O3/c28-23-19-17-11-5-1-3-7-13(11)26-15-9-10-16(30-15)27-14-8-4-2-6-12(14)18(22(27)21(17)26)20(19)24(29)25-23/h1-8,15-16H,9-10H2,(H,25,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 16 | -45.2 | n/a | n/a | n/a | n/a | n/a | 7.5 | 30 |
GlaxoSmithKline
| Assay Description
CDK4 and cyclin D1 were expressed in a baculovirus expression system and subsequently purified. The catalytic activity of the CDK4 protein was assaye... |
J Biol Chem 277: 46609-15 (2002)
Article DOI: 10.1074/jbc.M201233200
BindingDB Entry DOI: 10.7270/Q2D798PF |
More data for this
Ligand-Target Pair | |
Cyclin-A2/Cyclin-dependent kinase 1
(Homo sapiens (Human)) | BDBM17140

((15R,18R)-28-oxa-4,14,19-triazaoctacyclo[12.11.2.1...)Show SMILES O=C1NC(=O)c2c1c1c3ccccc3n3[C@H]4CC[C@@H](O4)n4c5ccccc5c2c4c13 |r| Show InChI InChI=1S/C24H15N3O3/c28-23-19-17-11-5-1-3-7-13(11)26-15-9-10-16(30-15)27-14-8-4-2-6-12(14)18(22(27)21(17)26)20(19)24(29)25-23/h1-8,15-16H,9-10H2,(H,25,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 23 | -43.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
GlaxoSmithKline
| Assay Description
In vitro CDK enzymatic assay using purified CDK mixed with cyclin A, was incubated with substrate, and test compounds in the presence of 1.4 uM ATP/ ... |
J Biol Chem 277: 46609-15 (2002)
Article DOI: 10.1074/jbc.M201233200
BindingDB Entry DOI: 10.7270/Q2D798PF |
More data for this
Ligand-Target Pair | |
Cyclin-A2/Cyclin-dependent kinase 2
(Homo sapiens (Human)) | BDBM17054

((2S,3R,4R,6R,18R)-18-hydroxy-3-methoxy-2-methyl-4-...)Show SMILES [H][C@]12C[C@@H](NC)[C@@H](OC)[C@](C)(O1)n1c3ccccc3c3c4[C@@H](O)NC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O4/c1-28-25(35-3)15(29-2)12-18(36-28)31-16-10-6-4-8-13(16)19-21-22(27(34)30-26(21)33)20-14-9-5-7-11-17(14)32(28)24(20)23(19)31/h4-11,15,18,25,27,29,34H,12H2,1-3H3,(H,30,33)/t15-,18-,25-,27-,28+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 30 | -42.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
GlaxoSmithKline
| Assay Description
In vitro CDK enzymatic assay using purified CDK mixed with cyclin A, was incubated with substrate, and test compounds in the presence of 1.4 uM ATP/ ... |
J Biol Chem 277: 46609-15 (2002)
Article DOI: 10.1074/jbc.M201233200
BindingDB Entry DOI: 10.7270/Q2D798PF |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cyclin-dependent kinase 4/G1/S-specific cyclin-D1
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 41 | -42.9 | n/a | n/a | n/a | n/a | n/a | 7.5 | 30 |
GlaxoSmithKline
| Assay Description
CDK4 and cyclin D1 were expressed in a baculovirus expression system and subsequently purified. The catalytic activity of the CDK4 protein was assaye... |
J Biol Chem 277: 46609-15 (2002)
Article DOI: 10.1074/jbc.M201233200
BindingDB Entry DOI: 10.7270/Q2D798PF |
More data for this
Ligand-Target Pair | |
Cyclin-A2/Cyclin-dependent kinase 1
(Homo sapiens (Human)) | BDBM17054

((2S,3R,4R,6R,18R)-18-hydroxy-3-methoxy-2-methyl-4-...)Show SMILES [H][C@]12C[C@@H](NC)[C@@H](OC)[C@](C)(O1)n1c3ccccc3c3c4[C@@H](O)NC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O4/c1-28-25(35-3)15(29-2)12-18(36-28)31-16-10-6-4-8-13(16)19-21-22(27(34)30-26(21)33)20-14-9-5-7-11-17(14)32(28)24(20)23(19)31/h4-11,15,18,25,27,29,34H,12H2,1-3H3,(H,30,33)/t15-,18-,25-,27-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 95 | -39.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
GlaxoSmithKline
| Assay Description
In vitro CDK enzymatic assay using purified CDK mixed with cyclin A, was incubated with substrate, and test compounds in the presence of 1.4 uM ATP/ ... |
J Biol Chem 277: 46609-15 (2002)
Article DOI: 10.1074/jbc.M201233200
BindingDB Entry DOI: 10.7270/Q2D798PF |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hexokinase-2
(Homo sapiens (Human)) | BDBM50169013
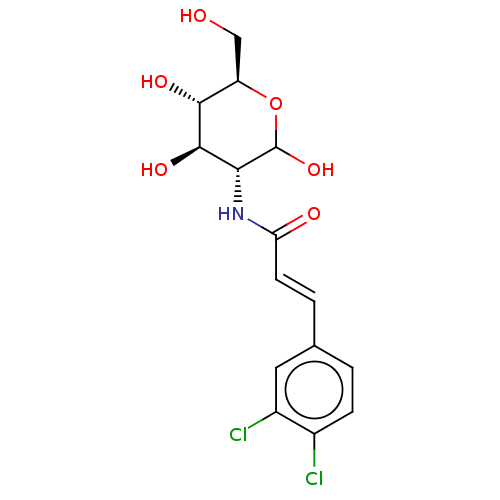
(CHEMBL3805703)Show SMILES OC[C@H]1OC(O)[C@H](NC(=O)\C=C\c2ccc(Cl)c(Cl)c2)[C@@H](O)[C@@H]1O |r| Show InChI InChI=1S/C15H17Cl2NO6/c16-8-3-1-7(5-9(8)17)2-4-11(20)18-12-14(22)13(21)10(6-19)24-15(12)23/h1-5,10,12-15,19,21-23H,6H2,(H,18,20)/b4-2+/t10-,12-,13-,14-,15?/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Competitive inhibition of His-tagged human HK2 (17 to 916 residues) expressed in Escherichia coli BL21(DE3) assessed as formation of G6P by continuou... |
ACS Med Chem Lett 7: 217-22 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00214
BindingDB Entry DOI: 10.7270/Q2PR7XWW |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 4/G1/S-specific cyclin-D1
(Homo sapiens (Human)) | BDBM17054

((2S,3R,4R,6R,18R)-18-hydroxy-3-methoxy-2-methyl-4-...)Show SMILES [H][C@]12C[C@@H](NC)[C@@H](OC)[C@](C)(O1)n1c3ccccc3c3c4[C@@H](O)NC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O4/c1-28-25(35-3)15(29-2)12-18(36-28)31-16-10-6-4-8-13(16)19-21-22(27(34)30-26(21)33)20-14-9-5-7-11-17(14)32(28)24(20)23(19)31/h4-11,15,18,25,27,29,34H,12H2,1-3H3,(H,30,33)/t15-,18-,25-,27-,28+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 3.60E+3 | -31.6 | n/a | n/a | n/a | n/a | n/a | 7.5 | 30 |
GlaxoSmithKline
| Assay Description
CDK4 and cyclin D1 were expressed in a baculovirus expression system and subsequently purified. The catalytic activity of the CDK4 protein was assaye... |
J Biol Chem 277: 46609-15 (2002)
Article DOI: 10.1074/jbc.M201233200
BindingDB Entry DOI: 10.7270/Q2D798PF |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128673
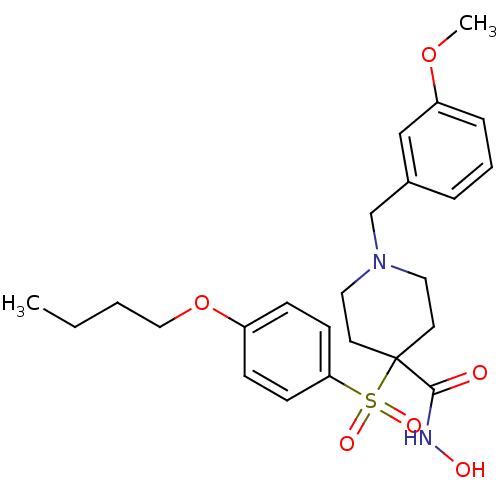
(1-(3-methoxybenzyl)-4-(4-butoxyphenylsulfonyl)-N-h...)Show SMILES CCCCOc1ccc(cc1)S(=O)(=O)C1(CCN(Cc2cccc(OC)c2)CC1)C(=O)NO Show InChI InChI=1S/C24H32N2O6S/c1-3-4-16-32-20-8-10-22(11-9-20)33(29,30)24(23(27)25-28)12-14-26(15-13-24)18-19-6-5-7-21(17-19)31-2/h5-11,17,28H,3-4,12-16,18H2,1-2H3,(H,25,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128649
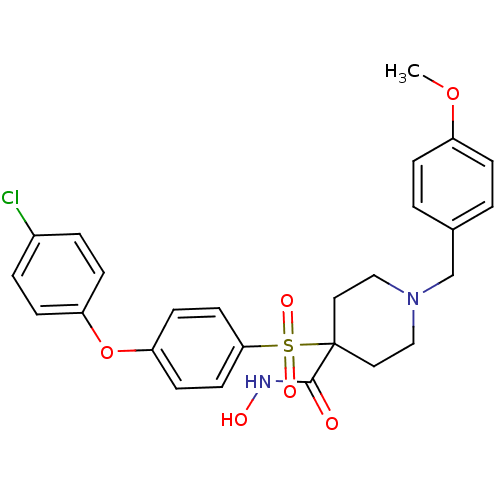
(1-(4-methoxybenzyl)-4-(4-(4-chlorophenoxy)phenylsu...)Show SMILES COc1ccc(CN2CCC(CC2)(C(=O)NO)S(=O)(=O)c2ccc(Oc3ccc(Cl)cc3)cc2)cc1 Show InChI InChI=1S/C26H27ClN2O6S/c1-34-21-6-2-19(3-7-21)18-29-16-14-26(15-17-29,25(30)28-31)36(32,33)24-12-10-23(11-13-24)35-22-8-4-20(27)5-9-22/h2-13,31H,14-18H2,1H3,(H,28,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128668
(1-Butyl-4-[4-(4-chloro-phenoxy)-benzenesulfonyl]-p...)Show SMILES CCCCN1CCC(CC1)(C(=O)NO)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C22H27ClN2O5S/c1-2-3-14-25-15-12-22(13-16-25,21(26)24-27)31(28,29)20-10-8-19(9-11-20)30-18-6-4-17(23)5-7-18/h4-11,27H,2-3,12-16H2,1H3,(H,24,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Neutrophil collagenase
(Homo sapiens (Human)) | BDBM50128648
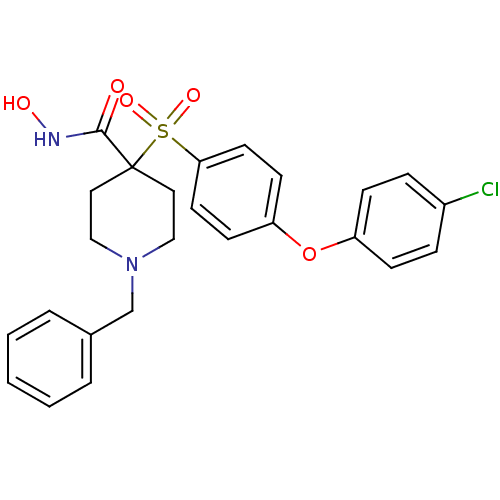
(1-Benzyl-4-[4-(4-chloro-phenoxy)-benzenesulfonyl]-...)Show SMILES ONC(=O)C1(CCN(Cc2ccccc2)CC1)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C25H25ClN2O5S/c26-20-6-8-21(9-7-20)33-22-10-12-23(13-11-22)34(31,32)25(24(29)27-30)14-16-28(17-15-25)18-19-4-2-1-3-5-19/h1-13,30H,14-18H2,(H,27,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of Matrix metalloprotease-8 |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128648

(1-Benzyl-4-[4-(4-chloro-phenoxy)-benzenesulfonyl]-...)Show SMILES ONC(=O)C1(CCN(Cc2ccccc2)CC1)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C25H25ClN2O5S/c26-20-6-8-21(9-7-20)33-22-10-12-23(13-11-22)34(31,32)25(24(29)27-30)14-16-28(17-15-25)18-19-4-2-1-3-5-19/h1-13,30H,14-18H2,(H,27,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
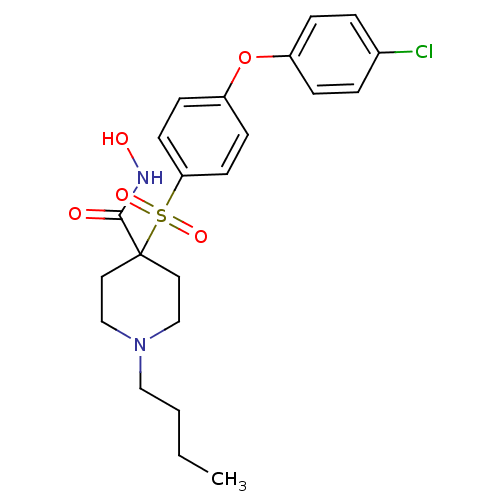
Collagenase 3
(Homo sapiens (Human)) | BDBM50128631
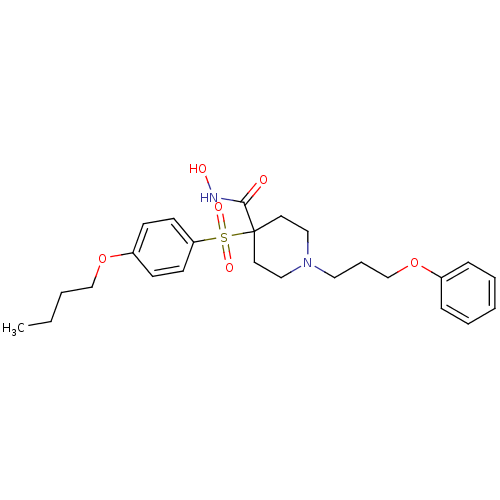
(4-(4-Butoxy-benzenesulfonyl)-1-(3-phenoxy-propyl)-...)Show SMILES CCCCOc1ccc(cc1)S(=O)(=O)C1(CCN(CCCOc2ccccc2)CC1)C(=O)NO Show InChI InChI=1S/C25H34N2O6S/c1-2-3-19-32-22-10-12-23(13-11-22)34(30,31)25(24(28)26-29)14-17-27(18-15-25)16-7-20-33-21-8-5-4-6-9-21/h4-6,8-13,29H,2-3,7,14-20H2,1H3,(H,26,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128645
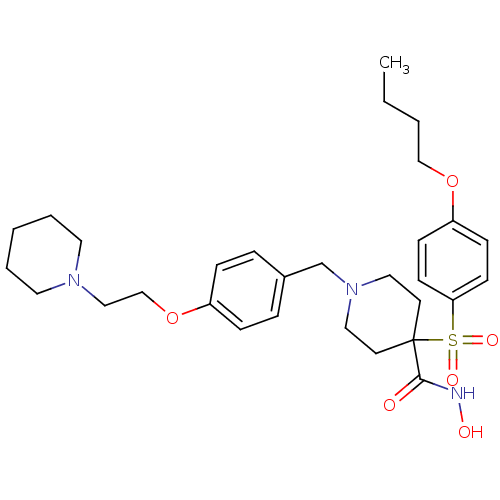
(1-(4-(2-(piperidin-1-yl)ethoxy)benzyl)-4-(4-butoxy...)Show SMILES CCCCOc1ccc(cc1)S(=O)(=O)C1(CCN(Cc2ccc(OCCN3CCCCC3)cc2)CC1)C(=O)NO Show InChI InChI=1S/C30H43N3O6S/c1-2-3-22-38-27-11-13-28(14-12-27)40(36,37)30(29(34)31-35)15-19-33(20-16-30)24-25-7-9-26(10-8-25)39-23-21-32-17-5-4-6-18-32/h7-14,35H,2-6,15-24H2,1H3,(H,31,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128643
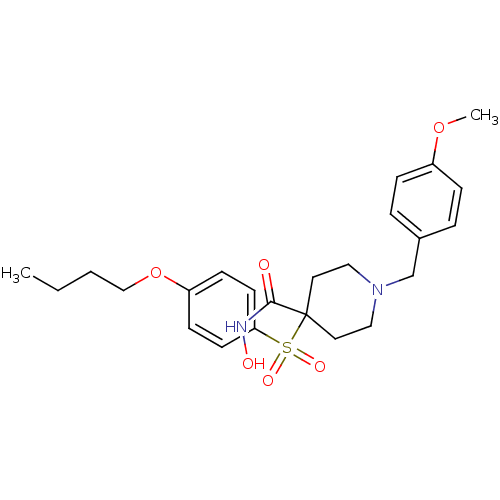
(1-(4-methoxybenzyl)-4-(4-butoxyphenylsulfonyl)-N-h...)Show SMILES CCCCOc1ccc(cc1)S(=O)(=O)C1(CCN(Cc2ccc(OC)cc2)CC1)C(=O)NO Show InChI InChI=1S/C24H32N2O6S/c1-3-4-17-32-21-9-11-22(12-10-21)33(29,30)24(23(27)25-28)13-15-26(16-14-24)18-19-5-7-20(31-2)8-6-19/h5-12,28H,3-4,13-18H2,1-2H3,(H,25,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50128649

(1-(4-methoxybenzyl)-4-(4-(4-chlorophenoxy)phenylsu...)Show SMILES COc1ccc(CN2CCC(CC2)(C(=O)NO)S(=O)(=O)c2ccc(Oc3ccc(Cl)cc3)cc2)cc1 Show InChI InChI=1S/C26H27ClN2O6S/c1-34-21-6-2-19(3-7-21)18-29-16-14-26(15-17-29,25(30)28-31)36(32,33)24-12-10-23(11-13-24)35-22-8-4-20(27)5-9-22/h2-13,31H,14-18H2,1H3,(H,28,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-9. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128651
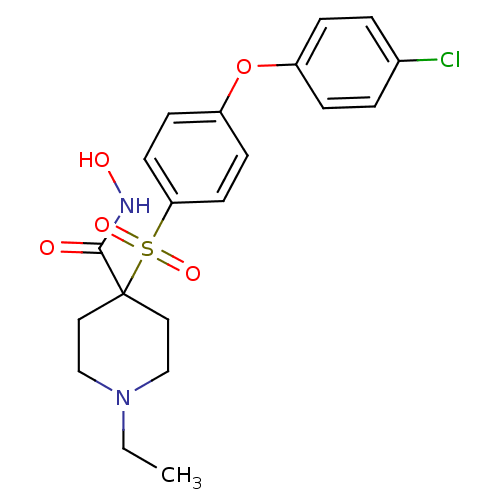
(4-(4-(4-chlorophenoxy)phenylsulfonyl)-1-ethyl-N-hy...)Show SMILES CCN1CCC(CC1)(C(=O)NO)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C20H23ClN2O5S/c1-2-23-13-11-20(12-14-23,19(24)22-25)29(26,27)18-9-7-17(8-10-18)28-16-5-3-15(21)4-6-16/h3-10,25H,2,11-14H2,1H3,(H,22,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128597
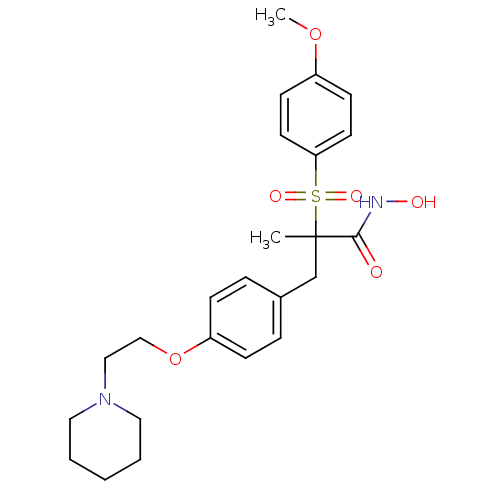
((R)-N-hydroxy-2-(4-methoxyphenylsulfonyl)-2-methyl...)Show SMILES COc1ccc(cc1)S(=O)(=O)C(C)(Cc1ccc(OCCN2CCCCC2)cc1)C(=O)NO Show InChI InChI=1S/C24H32N2O6S/c1-24(23(27)25-28,33(29,30)22-12-10-20(31-2)11-13-22)18-19-6-8-21(9-7-19)32-17-16-26-14-4-3-5-15-26/h6-13,28H,3-5,14-18H2,1-2H3,(H,25,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50128651

(4-(4-(4-chlorophenoxy)phenylsulfonyl)-1-ethyl-N-hy...)Show SMILES CCN1CCC(CC1)(C(=O)NO)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C20H23ClN2O5S/c1-2-23-13-11-20(12-14-23,19(24)22-25)29(26,27)18-9-7-17(8-10-18)28-16-5-3-15(21)4-6-16/h3-10,25H,2,11-14H2,1H3,(H,22,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-9. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128658
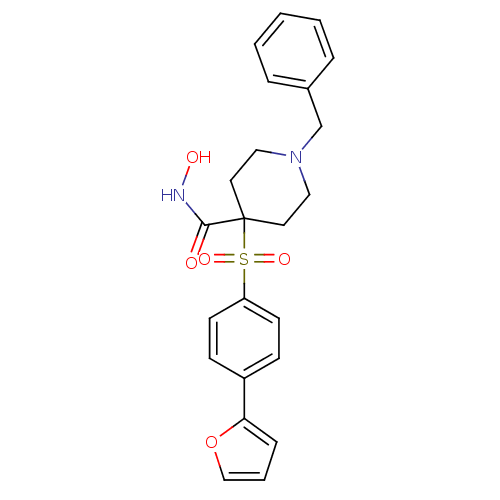
(1-Benzyl-4-(4-furan-2-yl-benzenesulfonyl)-piperidi...)Show SMILES ONC(=O)C1(CCN(Cc2ccccc2)CC1)S(=O)(=O)c1ccc(cc1)-c1ccco1 Show InChI InChI=1S/C23H24N2O5S/c26-22(24-27)23(12-14-25(15-13-23)17-18-5-2-1-3-6-18)31(28,29)20-10-8-19(9-11-20)21-7-4-16-30-21/h1-11,16,27H,12-15,17H2,(H,24,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128654
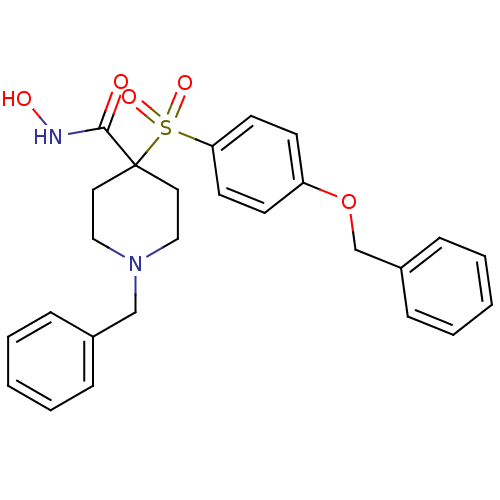
(1-Benzyl-4-(4-benzyloxy-benzenesulfonyl)-piperidin...)Show SMILES ONC(=O)C1(CCN(Cc2ccccc2)CC1)S(=O)(=O)c1ccc(OCc2ccccc2)cc1 Show InChI InChI=1S/C26H28N2O5S/c29-25(27-30)26(15-17-28(18-16-26)19-21-7-3-1-4-8-21)34(31,32)24-13-11-23(12-14-24)33-20-22-9-5-2-6-10-22/h1-14,30H,15-20H2,(H,27,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50128648

(1-Benzyl-4-[4-(4-chloro-phenoxy)-benzenesulfonyl]-...)Show SMILES ONC(=O)C1(CCN(Cc2ccccc2)CC1)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C25H25ClN2O5S/c26-20-6-8-21(9-7-20)33-22-10-12-23(13-11-22)34(31,32)25(24(29)27-30)14-16-28(17-15-25)18-19-4-2-1-3-5-19/h1-13,30H,14-18H2,(H,27,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-9. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50128668

(1-Butyl-4-[4-(4-chloro-phenoxy)-benzenesulfonyl]-p...)Show SMILES CCCCN1CCC(CC1)(C(=O)NO)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C22H27ClN2O5S/c1-2-3-14-25-15-12-22(13-16-25,21(26)24-27)31(28,29)20-10-8-19(9-11-20)30-18-6-4-17(23)5-7-18/h4-11,27H,2-3,12-16H2,1H3,(H,24,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-9. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50128648

(1-Benzyl-4-[4-(4-chloro-phenoxy)-benzenesulfonyl]-...)Show SMILES ONC(=O)C1(CCN(Cc2ccccc2)CC1)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C25H25ClN2O5S/c26-20-6-8-21(9-7-20)33-22-10-12-23(13-11-22)34(31,32)25(24(29)27-30)14-16-28(17-15-25)18-19-4-2-1-3-5-19/h1-13,30H,14-18H2,(H,27,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of Matrix metalloprotease-2 |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128667
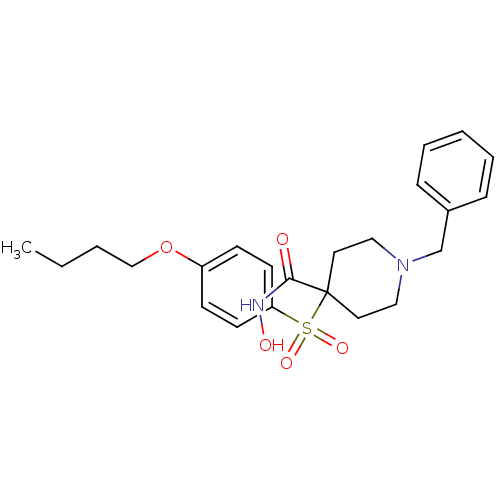
(1-Benzyl-4-(4-butoxy-benzenesulfonyl)-piperidine-4...)Show SMILES CCCCOc1ccc(cc1)S(=O)(=O)C1(CCN(Cc2ccccc2)CC1)C(=O)NO Show InChI InChI=1S/C23H30N2O5S/c1-2-3-17-30-20-9-11-21(12-10-20)31(28,29)23(22(26)24-27)13-15-25(16-14-23)18-19-7-5-4-6-8-19/h4-12,27H,2-3,13-18H2,1H3,(H,24,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128654

(1-Benzyl-4-(4-benzyloxy-benzenesulfonyl)-piperidin...)Show SMILES ONC(=O)C1(CCN(Cc2ccccc2)CC1)S(=O)(=O)c1ccc(OCc2ccccc2)cc1 Show InChI InChI=1S/C26H28N2O5S/c29-25(27-30)26(15-17-28(18-16-26)19-21-7-3-1-4-8-21)34(31,32)24-13-11-23(12-14-24)33-20-22-9-5-2-6-10-22/h1-14,30H,15-20H2,(H,27,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128659
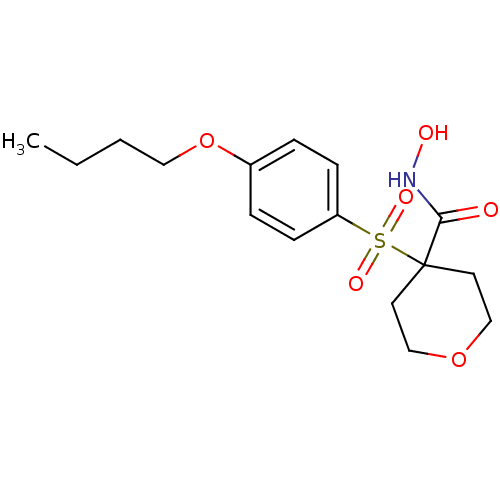
(4-(4-Butoxy-benzenesulfonyl)-tetrahydro-pyran-4-ca...)Show InChI InChI=1S/C16H23NO6S/c1-2-3-10-23-13-4-6-14(7-5-13)24(20,21)16(15(18)17-19)8-11-22-12-9-16/h4-7,19H,2-3,8-12H2,1H3,(H,17,18) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128640
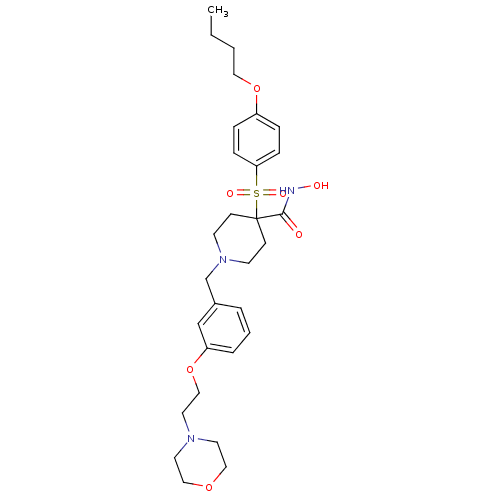
(4-(4-Butoxy-benzenesulfonyl)-1-[3-(2-morpholin-4-y...)Show SMILES CCCCOc1ccc(cc1)S(=O)(=O)C1(CCN(Cc2cccc(OCCN3CCOCC3)c2)CC1)C(=O)NO Show InChI InChI=1S/C29H41N3O7S/c1-2-3-18-38-25-7-9-27(10-8-25)40(35,36)29(28(33)30-34)11-13-32(14-12-29)23-24-5-4-6-26(22-24)39-21-17-31-15-19-37-20-16-31/h4-10,22,34H,2-3,11-21,23H2,1H3,(H,30,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50128645

(1-(4-(2-(piperidin-1-yl)ethoxy)benzyl)-4-(4-butoxy...)Show SMILES CCCCOc1ccc(cc1)S(=O)(=O)C1(CCN(Cc2ccc(OCCN3CCCCC3)cc2)CC1)C(=O)NO Show InChI InChI=1S/C30H43N3O6S/c1-2-3-22-38-27-11-13-28(14-12-27)40(36,37)30(29(34)31-35)15-19-33(20-16-30)24-25-7-9-26(10-8-25)39-23-21-32-17-5-4-6-18-32/h7-14,35H,2-6,15-24H2,1H3,(H,31,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-9. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128638
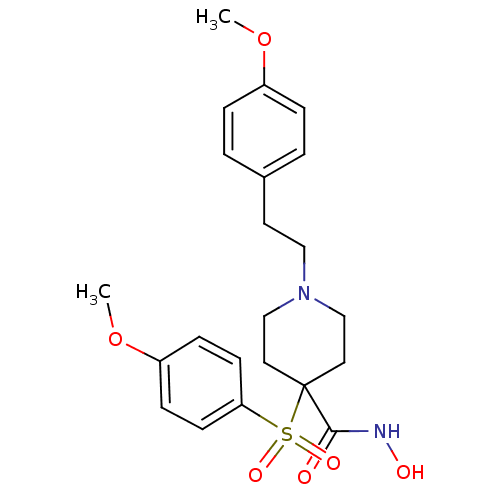
(1-(4-methoxyphenethyl)-N-hydroxy-4-(4-methoxypheny...)Show SMILES COc1ccc(CCN2CCC(CC2)(C(=O)NO)S(=O)(=O)c2ccc(OC)cc2)cc1 Show InChI InChI=1S/C22H28N2O6S/c1-29-18-5-3-17(4-6-18)11-14-24-15-12-22(13-16-24,21(25)23-26)31(27,28)20-9-7-19(30-2)8-10-20/h3-10,26H,11-16H2,1-2H3,(H,23,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128671
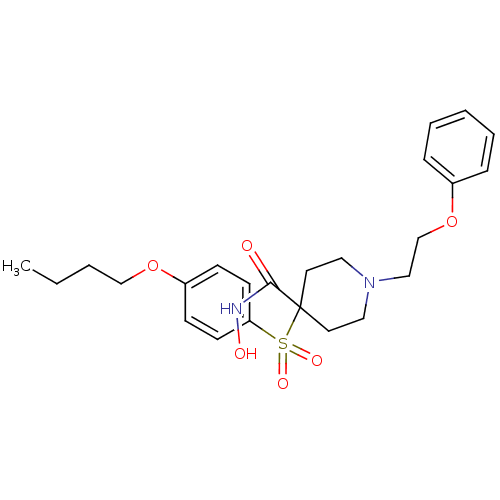
(4-(4-Butoxy-benzenesulfonyl)-1-(2-phenoxy-ethyl)-p...)Show SMILES CCCCOc1ccc(cc1)S(=O)(=O)C1(CCN(CCOc2ccccc2)CC1)C(=O)NO Show InChI InChI=1S/C24H32N2O6S/c1-2-3-18-31-21-9-11-22(12-10-21)33(29,30)24(23(27)25-28)13-15-26(16-14-24)17-19-32-20-7-5-4-6-8-20/h4-12,28H,2-3,13-19H2,1H3,(H,25,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128652
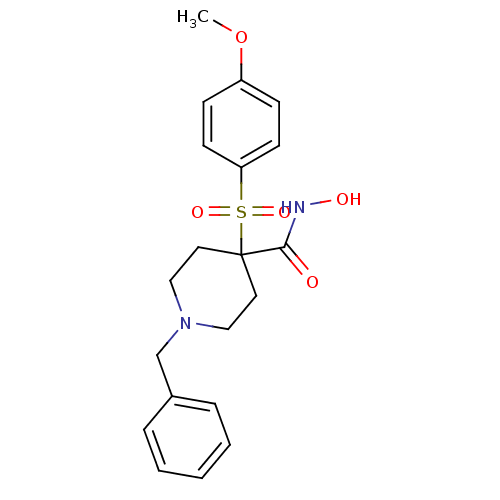
(1-Benzyl-4-(4-methoxy-benzenesulfonyl)-piperidine-...)Show SMILES COc1ccc(cc1)S(=O)(=O)C1(CCN(Cc2ccccc2)CC1)C(=O)NO Show InChI InChI=1S/C20H24N2O5S/c1-27-17-7-9-18(10-8-17)28(25,26)20(19(23)21-24)11-13-22(14-12-20)15-16-5-3-2-4-6-16/h2-10,24H,11-15H2,1H3,(H,21,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128666
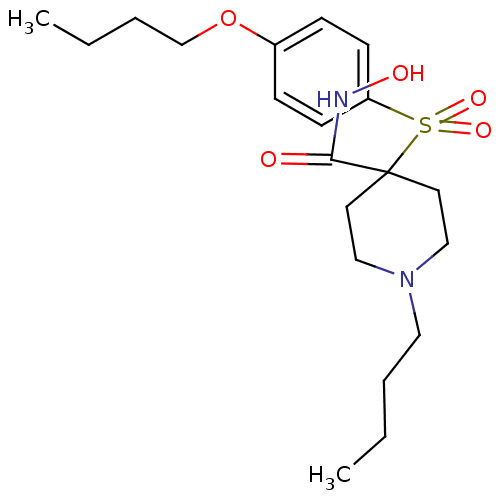
(4-(4-Butoxy-benzenesulfonyl)-1-butyl-piperidine-4-...)Show SMILES CCCCOc1ccc(cc1)S(=O)(=O)C1(CCN(CCCC)CC1)C(=O)NO Show InChI InChI=1S/C20H32N2O5S/c1-3-5-13-22-14-11-20(12-15-22,19(23)21-24)28(25,26)18-9-7-17(8-10-18)27-16-6-4-2/h7-10,24H,3-6,11-16H2,1-2H3,(H,21,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128635
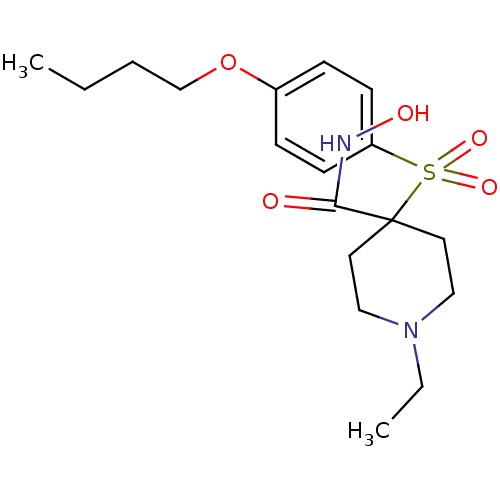
(4-(4-Butoxy-benzenesulfonyl)-1-ethyl-piperidine-4-...)Show SMILES CCCCOc1ccc(cc1)S(=O)(=O)C1(CCN(CC)CC1)C(=O)NO Show InChI InChI=1S/C18H28N2O5S/c1-3-5-14-25-15-6-8-16(9-7-15)26(23,24)18(17(21)19-22)10-12-20(4-2)13-11-18/h6-9,22H,3-5,10-14H2,1-2H3,(H,19,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128641

(1-(4-(pyridin-2-yl)benzyl)-N-hydroxy-4-(4-methoxyp...)Show SMILES COc1ccc(cc1)S(=O)(=O)C1(CCN(Cc2ccc(cc2)-c2ccccn2)CC1)C(=O)NO Show InChI InChI=1S/C25H27N3O5S/c1-33-21-9-11-22(12-10-21)34(31,32)25(24(29)27-30)13-16-28(17-14-25)18-19-5-7-20(8-6-19)23-4-2-3-15-26-23/h2-12,15,30H,13-14,16-18H2,1H3,(H,27,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128637
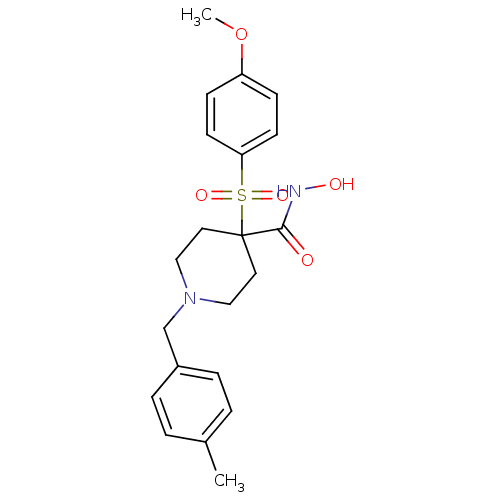
(4-(4-Methoxy-benzenesulfonyl)-1-(4-methyl-benzyl)-...)Show SMILES COc1ccc(cc1)S(=O)(=O)C1(CCN(Cc2ccc(C)cc2)CC1)C(=O)NO Show InChI InChI=1S/C21H26N2O5S/c1-16-3-5-17(6-4-16)15-23-13-11-21(12-14-23,20(24)22-25)29(26,27)19-9-7-18(28-2)8-10-19/h3-10,25H,11-15H2,1-2H3,(H,22,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128630
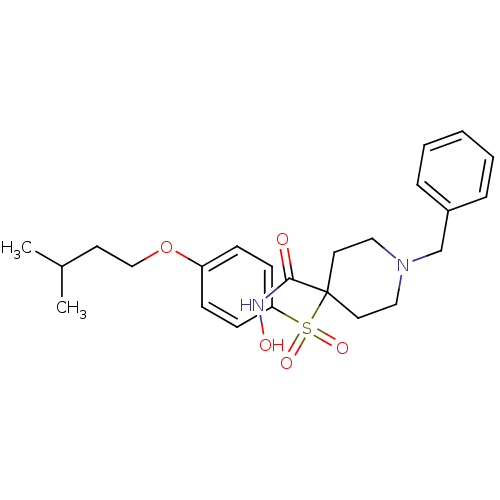
(1-Benzyl-4-[4-(3-methyl-butoxy)-benzenesulfonyl]-p...)Show SMILES CC(C)CCOc1ccc(cc1)S(=O)(=O)C1(CCN(Cc2ccccc2)CC1)C(=O)NO Show InChI InChI=1S/C24H32N2O5S/c1-19(2)12-17-31-21-8-10-22(11-9-21)32(29,30)24(23(27)25-28)13-15-26(16-14-24)18-20-6-4-3-5-7-20/h3-11,19,28H,12-18H2,1-2H3,(H,25,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128657
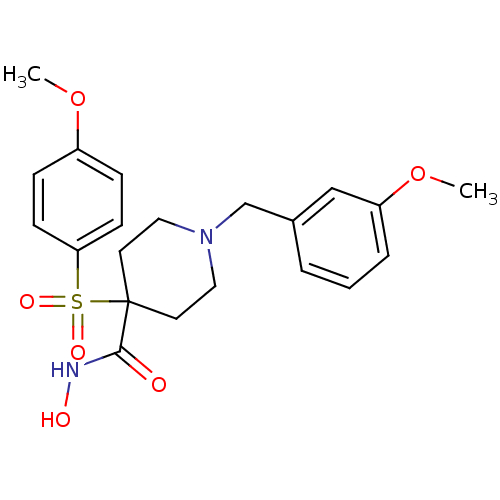
(1-(3-methoxybenzyl)-N-hydroxy-4-(4-methoxyphenylsu...)Show SMILES COc1ccc(cc1)S(=O)(=O)C1(CCN(Cc2cccc(OC)c2)CC1)C(=O)NO Show InChI InChI=1S/C21H26N2O6S/c1-28-17-6-8-19(9-7-17)30(26,27)21(20(24)22-25)10-12-23(13-11-21)15-16-4-3-5-18(14-16)29-2/h3-9,14,25H,10-13,15H2,1-2H3,(H,22,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50128640

(4-(4-Butoxy-benzenesulfonyl)-1-[3-(2-morpholin-4-y...)Show SMILES CCCCOc1ccc(cc1)S(=O)(=O)C1(CCN(Cc2cccc(OCCN3CCOCC3)c2)CC1)C(=O)NO Show InChI InChI=1S/C29H41N3O7S/c1-2-3-18-38-25-7-9-27(10-8-25)40(35,36)29(28(33)30-34)11-13-32(14-12-29)23-24-5-4-6-26(22-24)39-21-17-31-15-19-37-20-16-31/h4-10,22,34H,2-3,11-21,23H2,1H3,(H,30,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-9. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128656
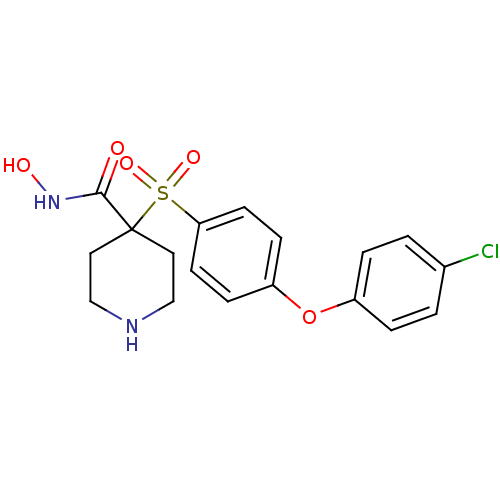
(4-(4-(4-chlorophenoxy)phenylsulfonyl)-N-hydroxypip...)Show SMILES ONC(=O)C1(CCNCC1)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C18H19ClN2O5S/c19-13-1-3-14(4-2-13)26-15-5-7-16(8-6-15)27(24,25)18(17(22)21-23)9-11-20-12-10-18/h1-8,20,23H,9-12H2,(H,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50128656

(4-(4-(4-chlorophenoxy)phenylsulfonyl)-N-hydroxypip...)Show SMILES ONC(=O)C1(CCNCC1)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C18H19ClN2O5S/c19-13-1-3-14(4-2-13)26-15-5-7-16(8-6-15)27(24,25)18(17(22)21-23)9-11-20-12-10-18/h1-8,20,23H,9-12H2,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-9. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128669
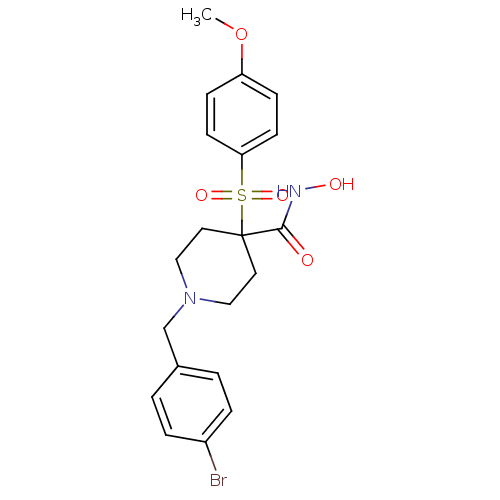
(1-(4-Bromo-benzyl)-4-(4-methoxy-benzenesulfonyl)-p...)Show SMILES COc1ccc(cc1)S(=O)(=O)C1(CCN(Cc2ccc(Br)cc2)CC1)C(=O)NO Show InChI InChI=1S/C20H23BrN2O5S/c1-28-17-6-8-18(9-7-17)29(26,27)20(19(24)22-25)10-12-23(13-11-20)14-15-2-4-16(21)5-3-15/h2-9,25H,10-14H2,1H3,(H,22,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128674
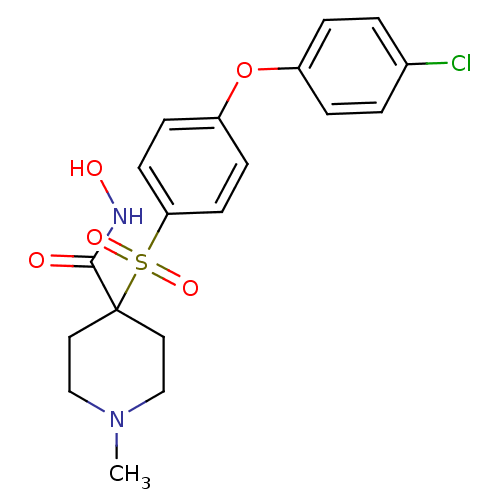
(4-(4-(4-chlorophenoxy)phenylsulfonyl)-N-hydroxy-1-...)Show SMILES CN1CCC(CC1)(C(=O)NO)S(=O)(=O)c1ccc(Oc2ccc(Cl)cc2)cc1 Show InChI InChI=1S/C19H21ClN2O5S/c1-22-12-10-19(11-13-22,18(23)21-24)28(25,26)17-8-6-16(7-9-17)27-15-4-2-14(20)3-5-15/h2-9,24H,10-13H2,1H3,(H,21,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |
Collagenase 3
(Homo sapiens (Human)) | BDBM50128632
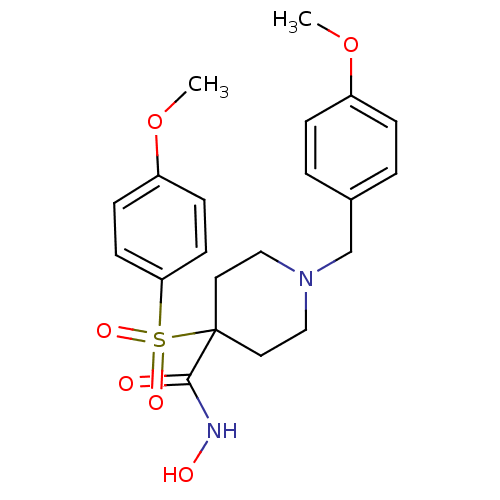
(1-(4-methoxybenzyl)-N-hydroxy-4-(4-methoxyphenylsu...)Show SMILES COc1ccc(CN2CCC(CC2)(C(=O)NO)S(=O)(=O)c2ccc(OC)cc2)cc1 Show InChI InChI=1S/C21H26N2O6S/c1-28-17-5-3-16(4-6-17)15-23-13-11-21(12-14-23,20(24)22-25)30(26,27)19-9-7-18(29-2)8-10-19/h3-10,25H,11-15H2,1-2H3,(H,22,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against matrix metalloprotease-13. |
J Med Chem 46: 2376-96 (2003)
Article DOI: 10.1021/jm0205550
BindingDB Entry DOI: 10.7270/Q2N0178C |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data