

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537072 (CHEMBL440072) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537063 (CHEMBL4590517) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537069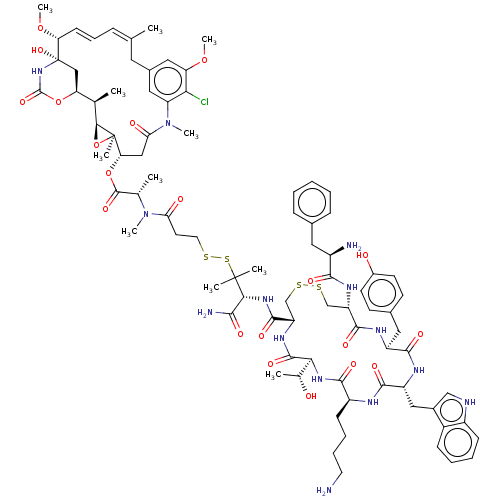 (CHEMBL4584764) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 0.0150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537077 (CHEMBL4550617) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 0.0150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537066 (CHEMBL4541310) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 0.0150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537076 (CHEMBL4564727) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 0.0180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537061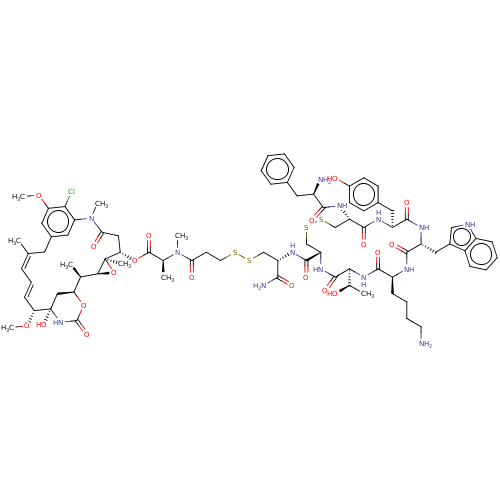 (CHEMBL4527856) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 0.0220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537068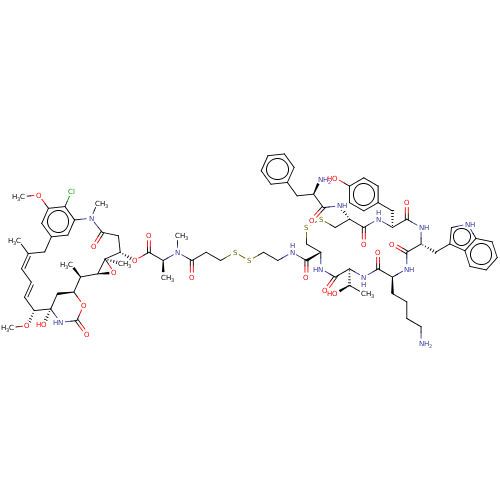 (CHEMBL4592483) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 0.0610 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50278495 (4-chloro-1-(4-(3-(piperidin-1-yl)propoxy)benzyl)pi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0680 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johann Wolfgang Goethe-Universität Curated by ChEMBL | Assay Description Displacement of [125I]iodoproxyfan from human histamine H3 receptor expressed in HEL293 cells | Bioorg Med Chem Lett 19: 2172-5 (2009) Article DOI: 10.1016/j.bmcl.2009.02.110 BindingDB Entry DOI: 10.7270/Q2H41RBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537070 (CHEMBL4581874) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 0.0750 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50278350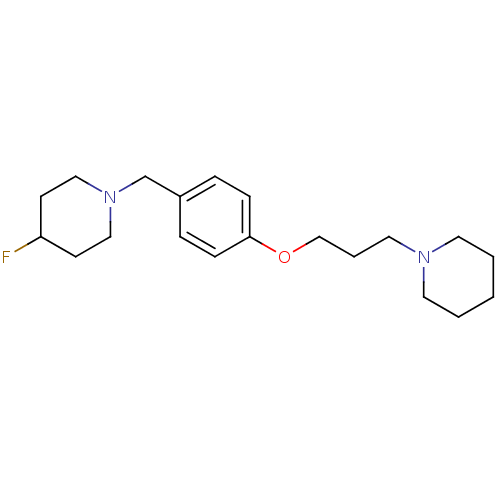 (4-fluoro-1-(4-(3-(piperidin-1-yl)propoxy)benzyl)pi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0940 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johann Wolfgang Goethe-Universität Curated by ChEMBL | Assay Description Displacement of [125I]iodoproxyfan from human histamine H3 receptor expressed in HEL293 cells | Bioorg Med Chem Lett 19: 2172-5 (2009) Article DOI: 10.1016/j.bmcl.2009.02.110 BindingDB Entry DOI: 10.7270/Q2H41RBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537074 (CHEMBL4556000) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537067 (CHEMBL4532058) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537062 (CHEMBL4549303) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537064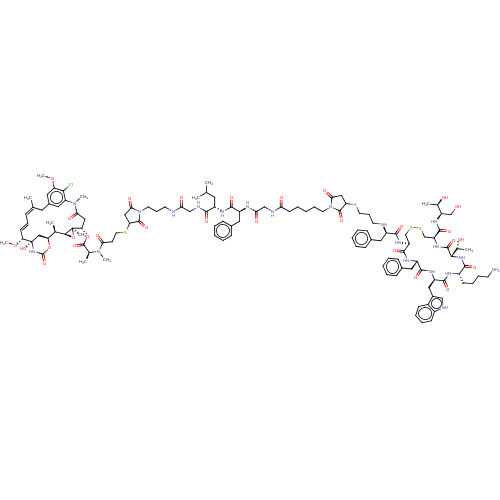 (CHEMBL4563111) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537075 (CHEMBL4548228) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (GUINEA PIG) | BDBM50247053 (1-(3-(3-(4-chlorophenyl)propoxy)propyl)piperidine ...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Jagiellonian University Medical College Curated by ChEMBL | Assay Description Antagonist activity at human histamine H3 receptor expressed in HEK293 cells by [35S]GTPgammaS binding assay | Bioorg Med Chem 17: 3037-42 (2009) Article DOI: 10.1016/j.bmc.2009.03.014 BindingDB Entry DOI: 10.7270/Q2SF2X3H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Thymidylate synthase (Mus musculus) | BDBM50403879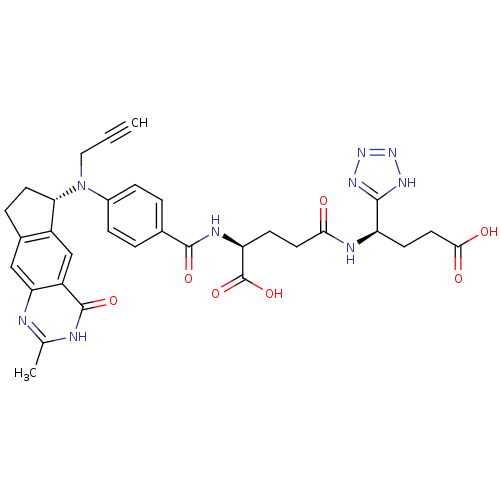 (CHEMBL320217) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CRC Laboratory Curated by ChEMBL | Assay Description Tested for inhibitory activity against thymidylate synthase of L1210 cells | Bioorg Med Chem Lett 11: 3015-7 (2001) BindingDB Entry DOI: 10.7270/Q2NK3G7F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537071 (CHEMBL4581646) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537065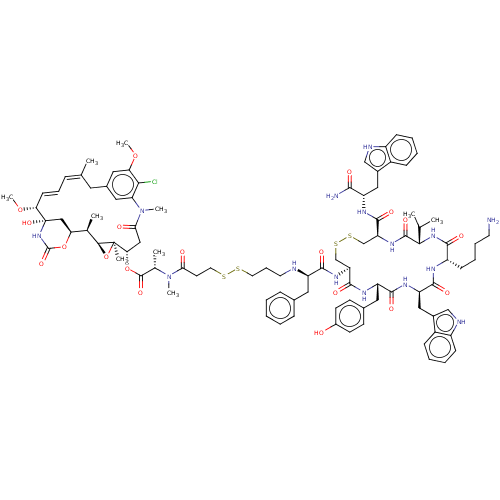 (CHEMBL4537192) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50278449 (4-bromo-1-(4-(3-(piperidin-1-yl)propoxy)benzyl)pip...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johann Wolfgang Goethe-Universität Curated by ChEMBL | Assay Description Displacement of [125I]iodoproxyfan from human histamine H3 receptor expressed in HEL293 cells | Bioorg Med Chem Lett 19: 2172-5 (2009) Article DOI: 10.1016/j.bmcl.2009.02.110 BindingDB Entry DOI: 10.7270/Q2H41RBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Thymidylate synthase (Mus musculus) | BDBM50088159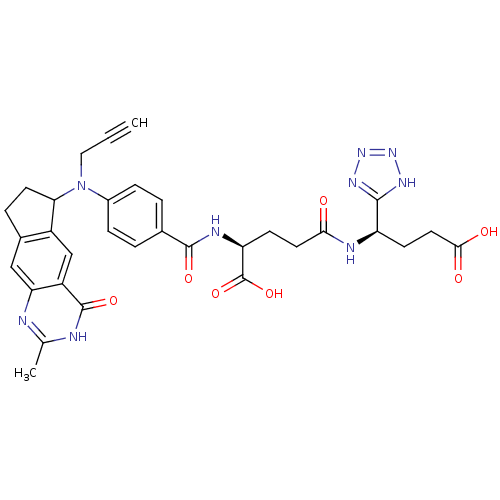 (4-[3-Carboxy-1-(1H-tetrazol-5-yl)-propylcarbamoyl]...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CRC Laboratory Curated by ChEMBL | Assay Description Tested for inhibitory activity against thymidylate synthase of L1210 cells | Bioorg Med Chem Lett 11: 3015-7 (2001) BindingDB Entry DOI: 10.7270/Q2NK3G7F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Thymidylate synthase (Mus musculus) | BDBM50088159 (4-[3-Carboxy-1-(1H-tetrazol-5-yl)-propylcarbamoyl]...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research Curated by ChEMBL | Assay Description Binding affinity against thymidylate synthase from L1210 mouse leukemia cells | J Med Chem 43: 1910-26 (2000) BindingDB Entry DOI: 10.7270/Q2KP81DM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-2 adrenergic receptor (Homo sapiens (Human)) | BDBM50292219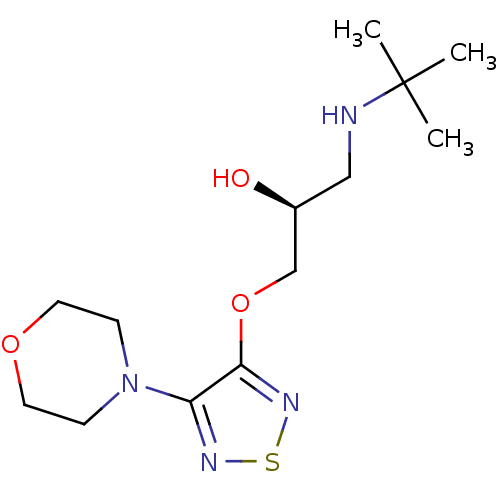 ((-)-3-morpholino-4-(3-tert-butylamino-2-hydroxypro...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 0.209 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Florence Curated by ChEMBL | Assay Description Displacement of [3H]-CGP12177 from human beta2 ADR expressed in HEK293T cell membrane after 90 mins by scintillation counting | J Med Chem 61: 5380-5394 (2018) Article DOI: 10.1021/acs.jmedchem.8b00625 BindingDB Entry DOI: 10.7270/Q2XS5XX3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50278448 (4,4-difluoro-1-(4-(3-(piperidin-1-yl)propoxy)benzy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johann Wolfgang Goethe-Universität Curated by ChEMBL | Assay Description Displacement of [125I]iodoproxyfan from human histamine H3 receptor expressed in HEL293 cells | Bioorg Med Chem Lett 19: 2172-5 (2009) Article DOI: 10.1016/j.bmcl.2009.02.110 BindingDB Entry DOI: 10.7270/Q2H41RBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Thymidylate synthase (Mus musculus) | BDBM50088168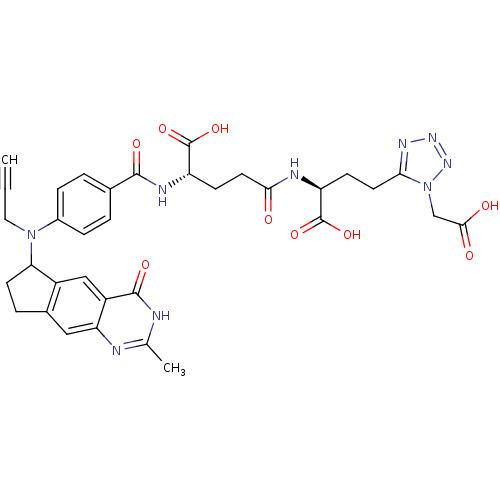 (2-(4-Carboxy-4-{4-[(2-methyl-4-oxo-4,6,7,8-tetrahy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research Curated by ChEMBL | Assay Description Binding affinity against thymidylate synthase from L1210 mouse leukemia cells | J Med Chem 43: 1910-26 (2000) BindingDB Entry DOI: 10.7270/Q2KP81DM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50278445 (4-fluoro-1-(4-(3-(pyrrolidin-1-yl)propoxy)benzyl)p...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johann Wolfgang Goethe-Universität Curated by ChEMBL | Assay Description Displacement of [125I]iodoproxyfan from human histamine H3 receptor expressed in HEL293 cells | Bioorg Med Chem Lett 19: 2172-5 (2009) Article DOI: 10.1016/j.bmcl.2009.02.110 BindingDB Entry DOI: 10.7270/Q2H41RBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537078 (CHEMBL4577466) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50278444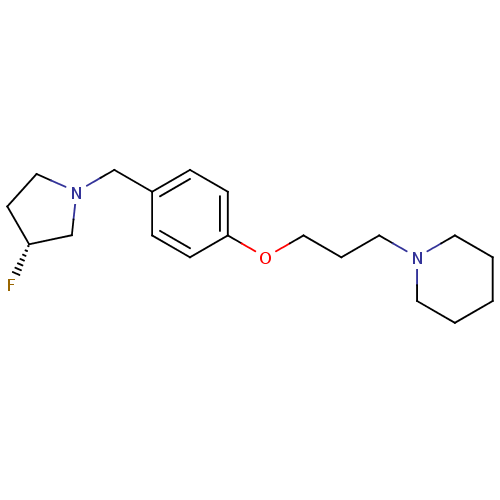 ((R)-1-(3-(4-((3-fluoropyrrolidin-1-yl)methyl)pheno...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johann Wolfgang Goethe-Universität Curated by ChEMBL | Assay Description Displacement of [125I]iodoproxyfan from human histamine H3 receptor expressed in HEL293 cells | Bioorg Med Chem Lett 19: 2172-5 (2009) Article DOI: 10.1016/j.bmcl.2009.02.110 BindingDB Entry DOI: 10.7270/Q2H41RBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50278400 ((S)-1-(3-(4-((3-fluoropyrrolidin-1-yl)methyl)pheno...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johann Wolfgang Goethe-Universität Curated by ChEMBL | Assay Description Displacement of [125I]iodoproxyfan from human histamine H3 receptor expressed in HEL293 cells | Bioorg Med Chem Lett 19: 2172-5 (2009) Article DOI: 10.1016/j.bmcl.2009.02.110 BindingDB Entry DOI: 10.7270/Q2H41RBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50278446 ((R)-3-fluoro-1-(4-(3-(pyrrolidin-1-yl)propoxy)benz...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johann Wolfgang Goethe-Universität Curated by ChEMBL | Assay Description Displacement of [125I]iodoproxyfan from human histamine H3 receptor expressed in HEL293 cells | Bioorg Med Chem Lett 19: 2172-5 (2009) Article DOI: 10.1016/j.bmcl.2009.02.110 BindingDB Entry DOI: 10.7270/Q2H41RBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Thymidylate synthase (Mus musculus) | BDBM50403876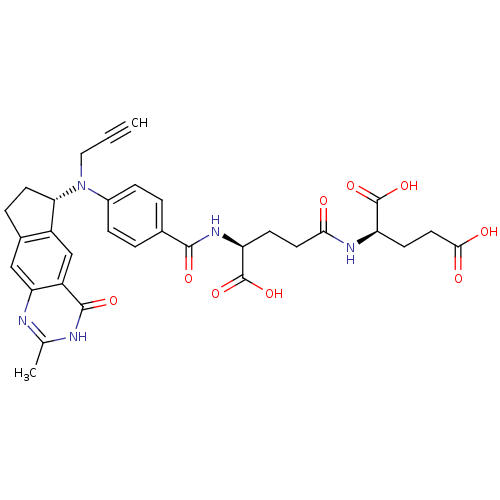 (CHEMBL323098) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | 0.330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CRC Laboratory Curated by ChEMBL | Assay Description Tested for inhibitory activity against thymidylate synthase of L1210 cells | Bioorg Med Chem Lett 11: 3015-7 (2001) BindingDB Entry DOI: 10.7270/Q2NK3G7F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Thymidylate synthase (Mus musculus) | BDBM50088161 (2-(4-Carboxy-4-{4-[(2-methyl-4-oxo-4,6,7,8-tetrahy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | PubMed | 0.420 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research Curated by ChEMBL | Assay Description Binding affinity against thymidylate synthase from L1210 mouse leukemia cells | J Med Chem 43: 1910-26 (2000) BindingDB Entry DOI: 10.7270/Q2KP81DM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Thymidylate synthase (Mus musculus) | BDBM50088161 (2-(4-Carboxy-4-{4-[(2-methyl-4-oxo-4,6,7,8-tetrahy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | PubMed | 0.420 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CRC Laboratory Curated by ChEMBL | Assay Description Tested for inhibitory activity against thymidylate synthase of L1210 cells | Bioorg Med Chem Lett 11: 3015-7 (2001) BindingDB Entry DOI: 10.7270/Q2NK3G7F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 9 (Homo sapiens (Human)) | BDBM50564139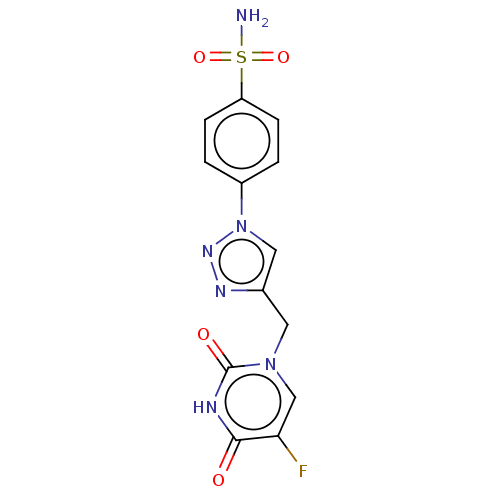 (CHEMBL4785560) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human CA9 pre-incubated for 15 mins measured by phenol red dye based stopped flow CO2 hydration assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112112 BindingDB Entry DOI: 10.7270/Q2J96B4W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 9 (Homo sapiens (Human)) | BDBM50334361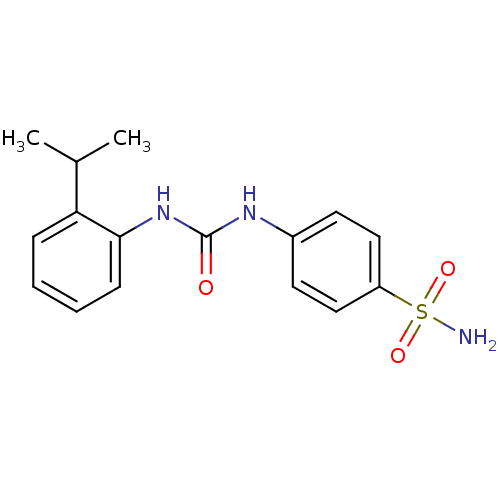 (4-(3-(2-isopropylphenyl)ureido)benzenesulfonamide ...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Florida College of Medicine Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 9 for 15 mins by stopped flow CO2 hydration assay | Bioorg Med Chem 24: 976-81 (2016) Article DOI: 10.1016/j.bmc.2016.01.019 BindingDB Entry DOI: 10.7270/Q2445P9F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50159110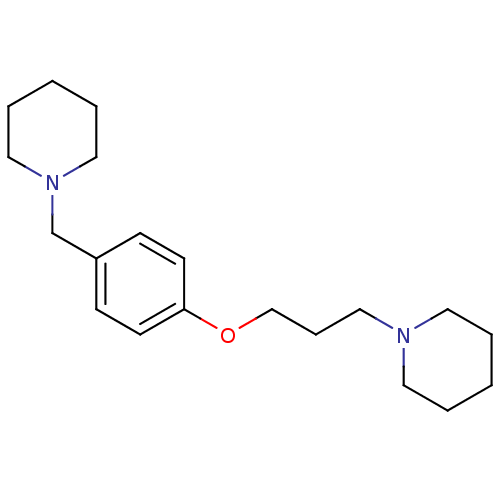 (1-(3-(4-(piperidin-1-ylmethyl)phenoxy)propyl)piper...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 0.580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johann Wolfgang Goethe-Universität Curated by ChEMBL | Assay Description Displacement of [125I]iodoproxyfan from human histamine H3 receptor expressed in HEL293 cells | Bioorg Med Chem Lett 19: 2172-5 (2009) Article DOI: 10.1016/j.bmcl.2009.02.110 BindingDB Entry DOI: 10.7270/Q2H41RBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM50564133 (CHEMBL4786026) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.590 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human CA2 pre-incubated for 15 mins measured by phenol red dye based stopped flow CO2 hydration assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112112 BindingDB Entry DOI: 10.7270/Q2J96B4W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 12 (Homo sapiens (Human)) | BDBM50540944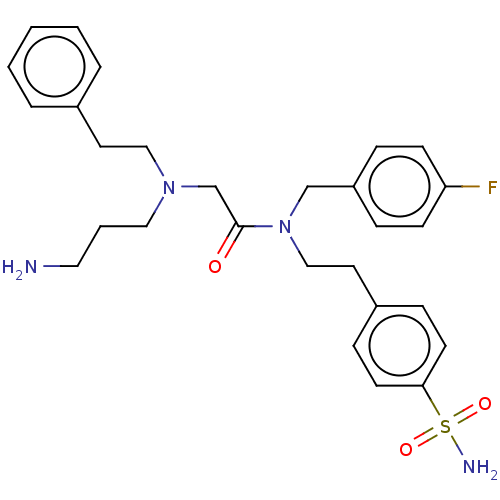 (CHEMBL4637053) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid PDB UniChem | Article PubMed | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Firenze Curated by ChEMBL | Assay Description Inhibition of human CA12 preincubated for 15 mins by stopped-flow CO2 hydration kinetic assay based Cheng-Prusoff equation analysis | J Med Chem 63: 7422-7444 (2020) Article DOI: 10.1021/acs.jmedchem.0c00733 BindingDB Entry DOI: 10.7270/Q2BV7M64 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM50564139 (CHEMBL4785560) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.670 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human CA2 pre-incubated for 15 mins measured by phenol red dye based stopped flow CO2 hydration assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112112 BindingDB Entry DOI: 10.7270/Q2J96B4W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM50540941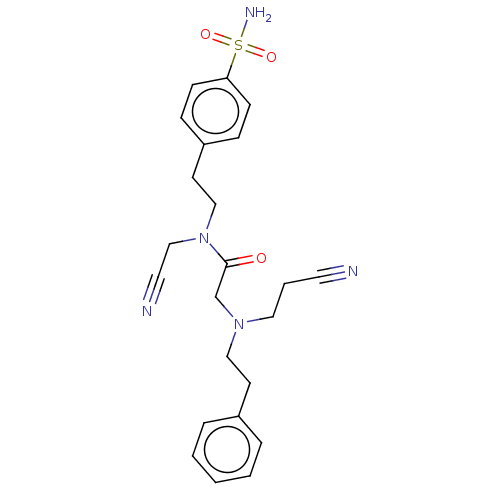 (CHEMBL4633228) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Firenze Curated by ChEMBL | Assay Description Inhibition of human CA2 preincubated for 15 mins by stopped-flow CO2 hydration kinetic assay based Cheng-Prusoff equation analysis | J Med Chem 63: 7422-7444 (2020) Article DOI: 10.1021/acs.jmedchem.0c00733 BindingDB Entry DOI: 10.7270/Q2BV7M64 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Thymidylate synthase (Mus musculus) | BDBM50403877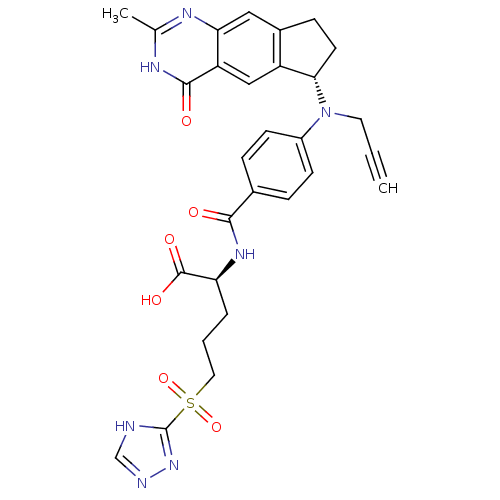 (CHEMBL103105) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.710 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CRC Laboratory Curated by ChEMBL | Assay Description Tested for inhibitory activity against thymidylate synthase of L1210 cells | Bioorg Med Chem Lett 11: 3015-7 (2001) BindingDB Entry DOI: 10.7270/Q2NK3G7F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM50564137 (CHEMBL4796488) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.710 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human CA2 pre-incubated for 15 mins measured by phenol red dye based stopped flow CO2 hydration assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112112 BindingDB Entry DOI: 10.7270/Q2J96B4W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM50537073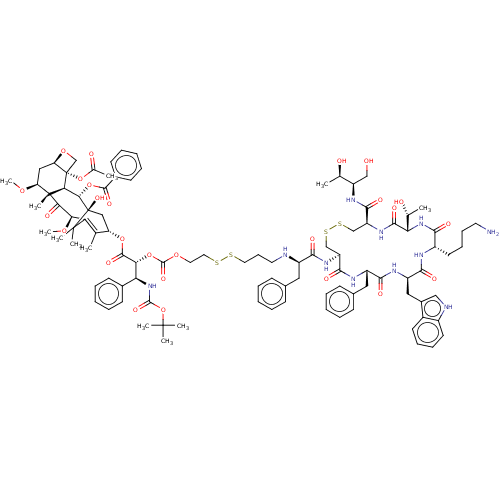 (CHEMBL4534477) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | Article PubMed | 0.770 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tarveda Therapeutics Curated by ChEMBL | Assay Description Displacement of [125I]somatostatin from human SSTR2 expressed in CHO-K1 cell membranes after 240 mins | J Med Chem 62: 2708-2719 (2019) Article DOI: 10.1021/acs.jmedchem.8b02036 BindingDB Entry DOI: 10.7270/Q2NK3JJ9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Thymidylate synthase (Mus musculus) | BDBM50408876 (CHEMBL434602) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research Curated by ChEMBL | Assay Description Binding affinity against thymidylate synthase from L1210 mouse leukemia cells | J Med Chem 43: 1910-26 (2000) BindingDB Entry DOI: 10.7270/Q2KP81DM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Thymidylate synthase (Mus musculus) | BDBM50088164 ((S)-2-{4-[(2-Methyl-4-oxo-4,6,7,8-tetrahydro-3H-cy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institute of Cancer Research Curated by ChEMBL | Assay Description Binding affinity against thymidylate synthase from L1210 mouse leukemia cells | J Med Chem 43: 1910-26 (2000) BindingDB Entry DOI: 10.7270/Q2KP81DM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Thymidylate synthase (Mus musculus) | BDBM50088164 ((S)-2-{4-[(2-Methyl-4-oxo-4,6,7,8-tetrahydro-3H-cy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CRC Laboratory Curated by ChEMBL | Assay Description Tested for inhibitory activity against thymidylate synthase of L1210 cells | Bioorg Med Chem Lett 11: 3015-7 (2001) BindingDB Entry DOI: 10.7270/Q2NK3G7F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM50564142 (CHEMBL4782608) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.810 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human CA2 pre-incubated for 15 mins measured by phenol red dye based stopped flow CO2 hydration assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112112 BindingDB Entry DOI: 10.7270/Q2J96B4W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50278496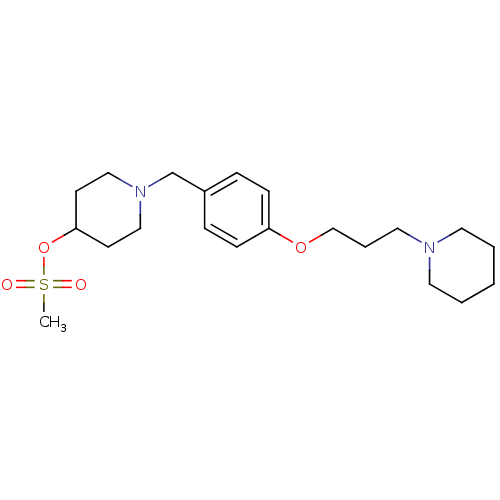 (1-(4-(3-(piperidin-1-yl)propoxy)benzyl)piperidin-4...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.840 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Johann Wolfgang Goethe-Universität Curated by ChEMBL | Assay Description Displacement of [125I]iodoproxyfan from human histamine H3 receptor expressed in HEL293 cells | Bioorg Med Chem Lett 19: 2172-5 (2009) Article DOI: 10.1016/j.bmcl.2009.02.110 BindingDB Entry DOI: 10.7270/Q2H41RBM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM50280336 (CHEMBL4165727) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 0.850 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Florence Curated by ChEMBL | Assay Description Effect on plasma amine oxidase (0.1 uM) after 60 min of incubation at pH 7.2 | ACS Med Chem Lett 8: 1314-1319 (2017) Article DOI: 10.1021/acsmedchemlett.7b00399 BindingDB Entry DOI: 10.7270/Q29P345B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 1049 total ) | Next | Last >> |