

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Botulinum neurotoxin type A (Clostridium botulinum (strain Hall / ATCC 3502 / N...) | BDBM50242333 ((S)-2-{(S)-2-[(S)-6-Amino-2-((2S,3R)-2-{(S)-2-[(S)...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SAIC-Frederick, Inc. Curated by ChEMBL | Assay Description Inhibition of BoNT/A light chain metalloprotease activity | J Biol Chem 282: 5004-14 (2007) Article DOI: 10.1074/jbc.M608166200 BindingDB Entry DOI: 10.7270/Q20C4VJF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum (strain Hall / ATCC 3502 / N...) | BDBM50260295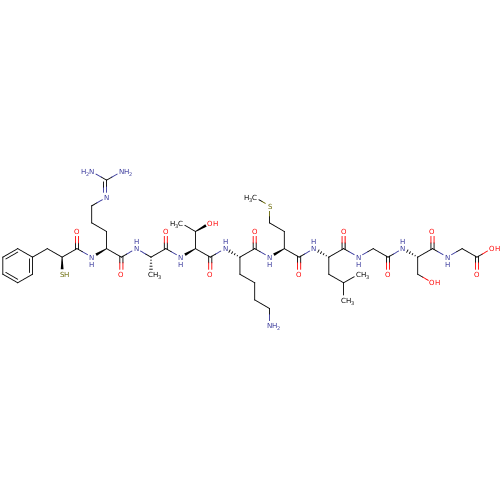 (2-((S)-2-(2-((S)-2-((S)-2-((S)-6-amino-2-((2S,3R)-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SAIC-Frederick, Inc. Curated by ChEMBL | Assay Description Inhibition of BoNT/A light chain metalloprotease activity | J Biol Chem 282: 5004-14 (2007) Article DOI: 10.1074/jbc.M608166200 BindingDB Entry DOI: 10.7270/Q20C4VJF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum (strain Hall / ATCC 3502 / N...) | BDBM50260294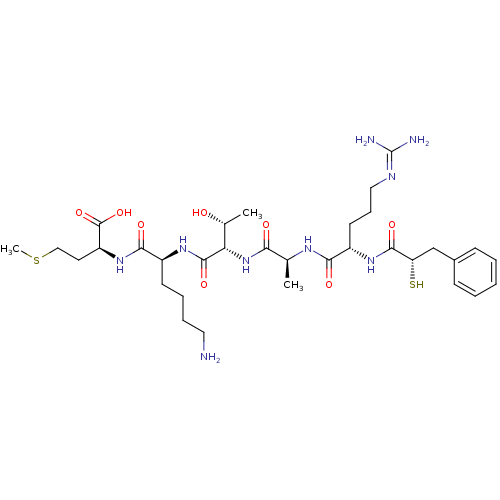 ((S)-2-((S)-6-amino-2-((2S,3R)-2-((S)-2-((S)-5-guan...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SAIC-Frederick, Inc. Curated by ChEMBL | Assay Description Inhibition of BoNT/A light chain metalloprotease activity | J Biol Chem 282: 5004-14 (2007) Article DOI: 10.1074/jbc.M608166200 BindingDB Entry DOI: 10.7270/Q20C4VJF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum (strain Hall / ATCC 3502 / N...) | BDBM50242339 ((S)-2-{(S)-2-[(S)-6-Amino-2-((2S,3R)-2-{(S)-2-[(S)...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SAIC-Frederick, Inc. Curated by ChEMBL | Assay Description Inhibition of BoNT/A light chain metalloprotease activity | J Biol Chem 282: 5004-14 (2007) Article DOI: 10.1074/jbc.M608166200 BindingDB Entry DOI: 10.7270/Q20C4VJF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum (strain Hall / ATCC 3502 / N...) | BDBM50242337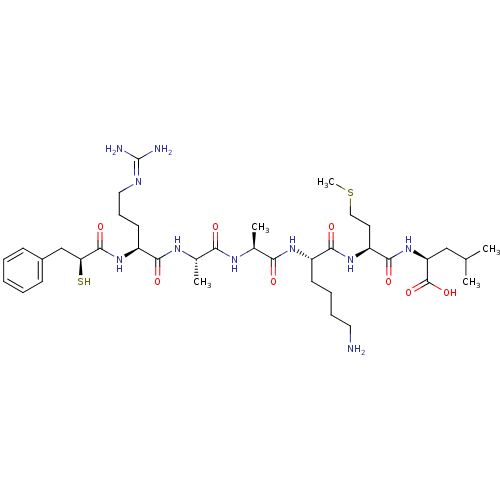 ((S)-2-{(S)-2-[(S)-6-Amino-2-((S)-2-{(S)-2-[(S)-5-g...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SAIC-Frederick, Inc. Curated by ChEMBL | Assay Description Inhibition of BoNT/A light chain metalloprotease activity | J Biol Chem 282: 5004-14 (2007) Article DOI: 10.1074/jbc.M608166200 BindingDB Entry DOI: 10.7270/Q20C4VJF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum (strain Hall / ATCC 3502 / N...) | BDBM50260296 (CHEMBL501525 | CRATKML) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SAIC-Frederick, Inc. Curated by ChEMBL | Assay Description Inhibition of BoNT/A light chain metalloprotease activity | J Biol Chem 282: 5004-14 (2007) Article DOI: 10.1074/jbc.M608166200 BindingDB Entry DOI: 10.7270/Q20C4VJF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum (strain Hall / ATCC 3502 / N...) | BDBM50242336 ((S)-2-{(S)-2-[(S)-6-Amino-2-((2S,3R)-2-{(S)-2-[(S)...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SAIC-Frederick, Inc. Curated by ChEMBL | Assay Description Inhibition of BoNT/A light chain metalloprotease activity | J Biol Chem 282: 5004-14 (2007) Article DOI: 10.1074/jbc.M608166200 BindingDB Entry DOI: 10.7270/Q20C4VJF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum (strain Hall / ATCC 3502 / N...) | BDBM50240902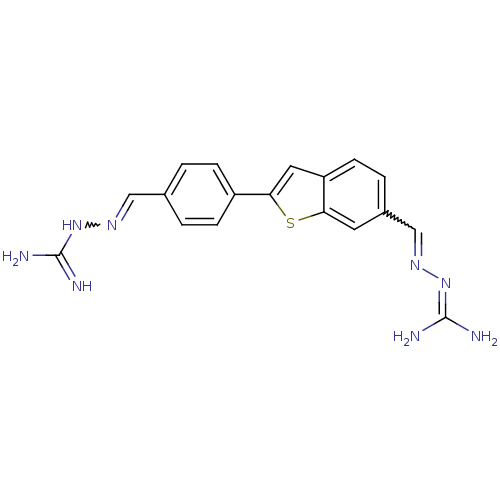 ((2E)-2-{4-[6-((E)-{[(E)-amino(imino)methyl]hydrazo...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SAIC-Frederick, Inc. Curated by ChEMBL | Assay Description Inhibition of BoNT/A light chain metalloprotease activity | J Biol Chem 282: 5004-14 (2007) Article DOI: 10.1074/jbc.M608166200 BindingDB Entry DOI: 10.7270/Q20C4VJF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum (strain Hall / ATCC 3502 / N...) | BDBM50242338 ((S)-2-{(S)-2-[(S)-2-((2S,3R)-2-{(S)-2-[(S)-5-Guani...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SAIC-Frederick, Inc. Curated by ChEMBL | Assay Description Inhibition of BoNT/A light chain metalloprotease activity | J Biol Chem 282: 5004-14 (2007) Article DOI: 10.1074/jbc.M608166200 BindingDB Entry DOI: 10.7270/Q20C4VJF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum (strain Hall / ATCC 3502 / N...) | BDBM50260293 ((S)-6-amino-2-((2S,3R)-2-((S)-2-((S)-5-guanidino-2...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SAIC-Frederick, Inc. Curated by ChEMBL | Assay Description Inhibition of BoNT/A light chain metalloprotease activity | J Biol Chem 282: 5004-14 (2007) Article DOI: 10.1074/jbc.M608166200 BindingDB Entry DOI: 10.7270/Q20C4VJF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum (strain Hall / ATCC 3502 / N...) | BDBM50240901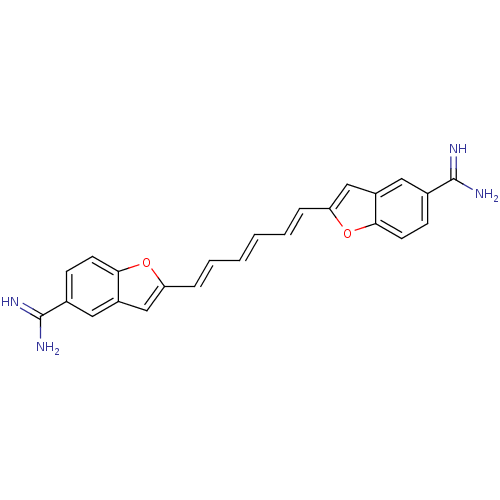 (2-((1E,3E,5E)-6-{5-[(E)-amino(imino)methyl]-1-benz...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 6.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SAIC-Frederick, Inc. Curated by ChEMBL | Assay Description Inhibition of BoNT/A light chain metalloprotease activity | J Biol Chem 282: 5004-14 (2007) Article DOI: 10.1074/jbc.M608166200 BindingDB Entry DOI: 10.7270/Q20C4VJF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum (strain Hall / ATCC 3502 / N...) | BDBM50242334 ((S)-2-{(S)-2-[(S)-6-Amino-2-((2S,3R)-2-{(S)-2-[(S)...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 8.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SAIC-Frederick, Inc. Curated by ChEMBL | Assay Description Inhibition of BoNT/A light chain metalloprotease activity | J Biol Chem 282: 5004-14 (2007) Article DOI: 10.1074/jbc.M608166200 BindingDB Entry DOI: 10.7270/Q20C4VJF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum (strain Hall / ATCC 3502 / N...) | BDBM50260300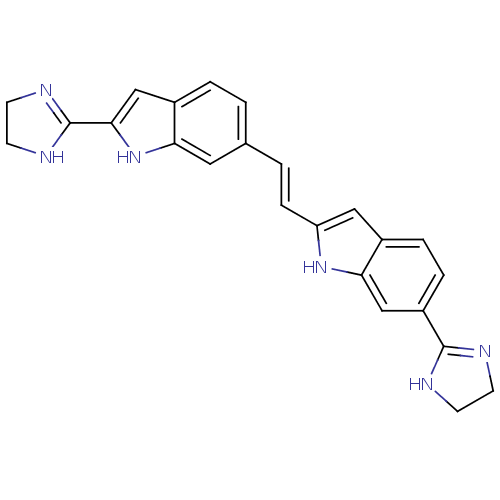 (6-(4,5-dihydro-1H-imidazol-2-yl)-2-(2-(2-(4,5-dihy...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SAIC-Frederick, Inc. Curated by ChEMBL | Assay Description Inhibition of BoNT/A light chain metalloprotease activity | J Biol Chem 282: 5004-14 (2007) Article DOI: 10.1074/jbc.M608166200 BindingDB Entry DOI: 10.7270/Q20C4VJF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum (strain Hall / ATCC 3502 / N...) | BDBM50260292 ((2S,3R)-2-{(S)-2-[(S)-5-Guanidino-2-((S)-2-mercapt...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SAIC-Frederick, Inc. Curated by ChEMBL | Assay Description Inhibition of BoNT/A light chain metalloprotease activity | J Biol Chem 282: 5004-14 (2007) Article DOI: 10.1074/jbc.M608166200 BindingDB Entry DOI: 10.7270/Q20C4VJF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum (strain Hall / ATCC 3502 / N...) | BDBM50260291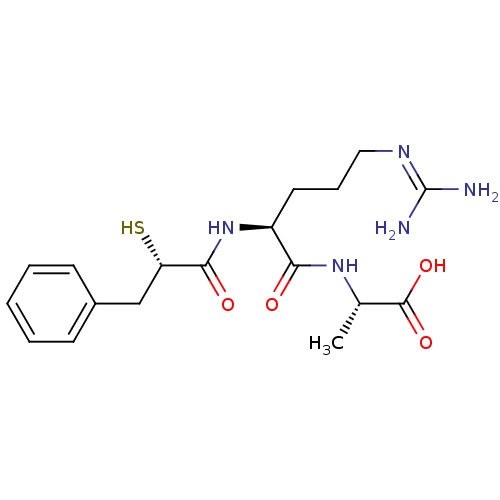 ((S)-2-[(S)-5-Guanidino-2-((S)-2-mercapto-3-phenyl-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 6.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SAIC-Frederick, Inc. Curated by ChEMBL | Assay Description Inhibition of BoNT/A light chain metalloprotease activity | J Biol Chem 282: 5004-14 (2007) Article DOI: 10.1074/jbc.M608166200 BindingDB Entry DOI: 10.7270/Q20C4VJF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum (strain Hall / ATCC 3502 / N...) | BDBM50242311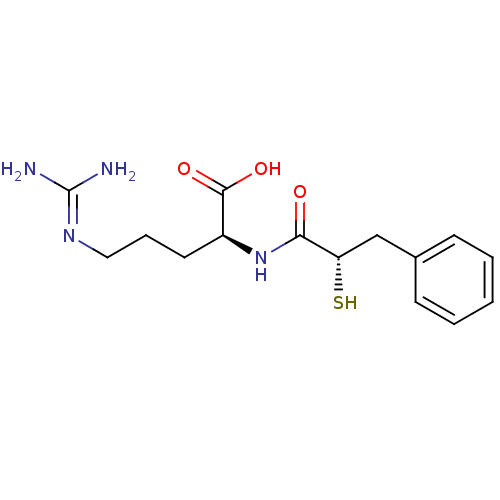 ((S)-5-Guanidino-2-((S)-2-mercapto-3-phenyl-propion...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 6.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SAIC-Frederick, Inc. Curated by ChEMBL | Assay Description Inhibition of BoNT/A light chain metalloprotease activity | J Biol Chem 282: 5004-14 (2007) Article DOI: 10.1074/jbc.M608166200 BindingDB Entry DOI: 10.7270/Q20C4VJF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Botulinum neurotoxin type A (Clostridium botulinum (strain Hall / ATCC 3502 / N...) | BDBM50242335 ((S)-2-{(S)-2-[(S)-6-Amino-2-((2S,3R)-2-{(S)-2-[(S)...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | >3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SAIC-Frederick, Inc. Curated by ChEMBL | Assay Description Inhibition of BoNT/A light chain metalloprotease activity | J Biol Chem 282: 5004-14 (2007) Article DOI: 10.1074/jbc.M608166200 BindingDB Entry DOI: 10.7270/Q20C4VJF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50322823 ((S)-N-(4-(3-chloro-4-fluorophenylamino)-7-(tetrahy...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK as substrate preinc... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50322823 ((S)-N-(4-(3-chloro-4-fluorophenylamino)-7-(tetrahy...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR L858R mutant (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK as sub... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50161957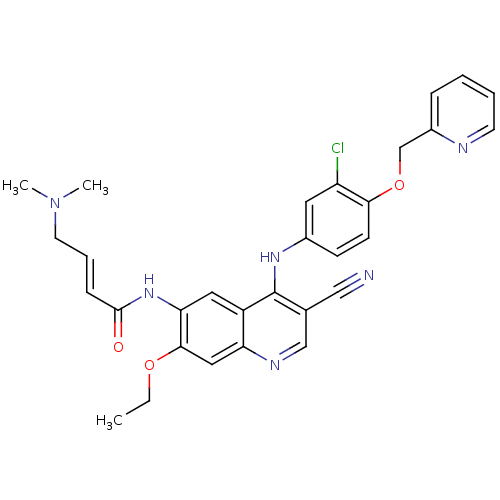 (4-Dimethylamino-but-2-enoic acid {4-[3-chloro-4-(p...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK as substrate preinc... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50161957 (4-Dimethylamino-but-2-enoic acid {4-[3-chloro-4-(p...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR L858R mutant (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK as sub... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50029668 (AZD-9291 | Osimertinib | US10085983, Compound AZD-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR L858R/T790M mutant (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK ... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM5445 (CHEMBL554 | GW572016 | LAPATINIB DITOSYLATE | Lapa...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB Article PubMed | n/a | n/a | 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK as substrate preinc... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50029668 (AZD-9291 | Osimertinib | US10085983, Compound AZD-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR L858R mutant (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK as sub... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50547250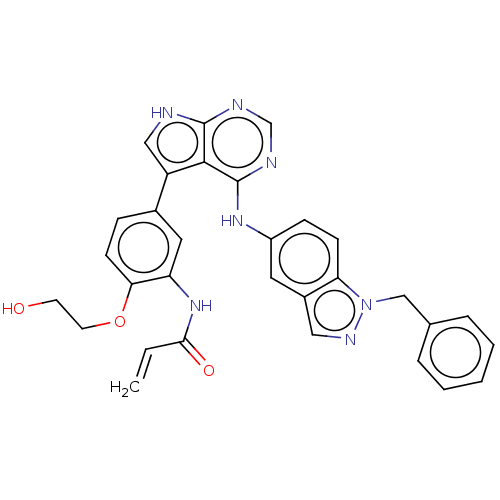 (CHEMBL4756368) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK as substrate preinc... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50547247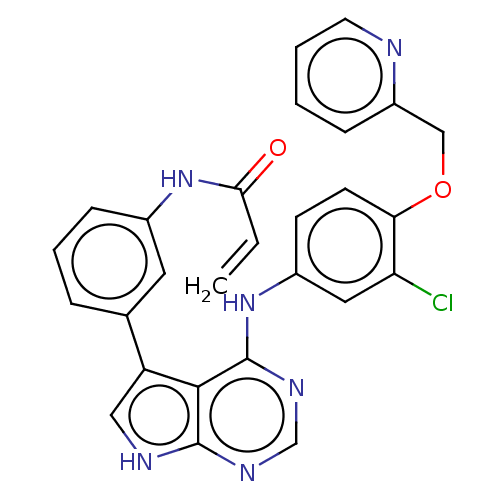 (CHEMBL4787483) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK as substrate preinc... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50547249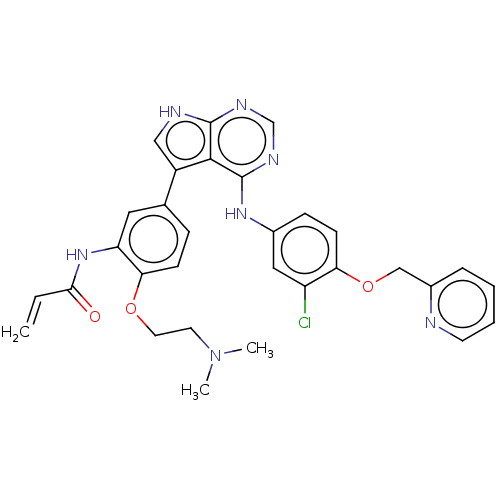 (CHEMBL4757145) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK as substrate preinc... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50547250 (CHEMBL4756368) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR L858R mutant (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK as sub... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50547247 (CHEMBL4787483) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR L858R mutant (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK as sub... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50547249 (CHEMBL4757145) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR L858R mutant (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK as sub... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50547248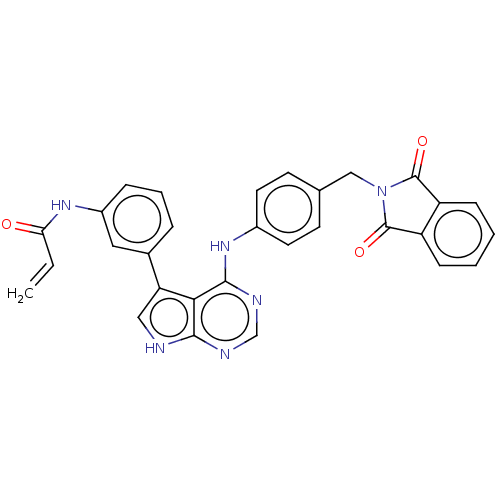 (CHEMBL4795165) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR L858R mutant (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK as sub... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM5445 (CHEMBL554 | GW572016 | LAPATINIB DITOSYLATE | Lapa...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB Article PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR L858R mutant (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK as sub... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50029668 (AZD-9291 | Osimertinib | US10085983, Compound AZD-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK as substrate preinc... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50322823 ((S)-N-(4-(3-chloro-4-fluorophenylamino)-7-(tetrahy...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR L858R/T790M mutant (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK ... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50547248 (CHEMBL4795165) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK as substrate preinc... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50161957 (4-Dimethylamino-but-2-enoic acid {4-[3-chloro-4-(p...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR L858R/T790M mutant (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK ... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proto-oncogene tyrosine-protein kinase Src (Gallus gallus (Chicken)) | BDBM50322535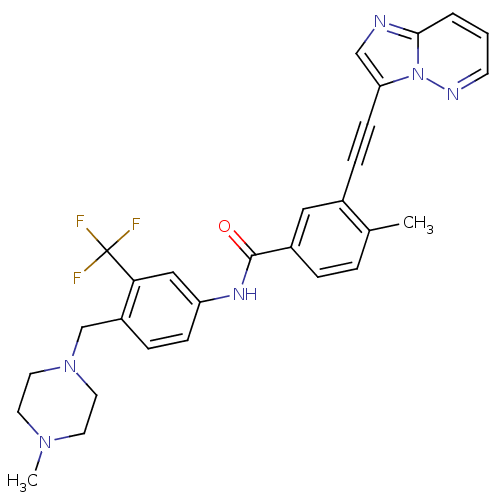 (3-(imidazo[1,2-b]pyridazin-3-ylethynyl)-4-methyl-N...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
Technical University of Dortmund Curated by ChEMBL | Assay Description Inhibition of chicken wild-type cSRC (251 to 533)-mediated phosphorylation of biotinylated poly-Glu-Tyr expressed in Escherichia coli BL21(DE3) prein... | J Med Chem 56: 5757-72 (2014) Article DOI: 10.1021/jm4004076 BindingDB Entry DOI: 10.7270/Q2W37XPH | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50258974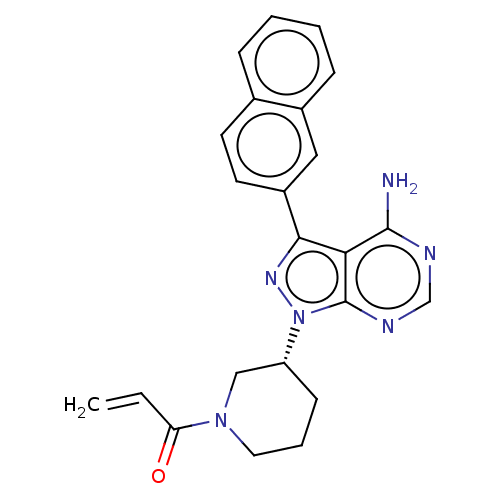 (CHEMBL4090601) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
TU Dortmund University Curated by ChEMBL | Assay Description Inhibition of human recombinant GST-tagged EGFR L858R mutant expressed in baculovirus expression system preincubated for 30 mins followed by ATP and ... | J Med Chem 60: 7725-7744 (2017) Article DOI: 10.1021/acs.jmedchem.7b00515 BindingDB Entry DOI: 10.7270/Q28W3GS9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50029668 (AZD-9291 | Osimertinib | US10085983, Compound AZD-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TU Dortmund University Curated by ChEMBL | Assay Description Inhibition of recombinant human GST-tagged EGFR L858R/T790M double mutant expressed in baculovirus expression system preincubated for 30 mins followe... | J Med Chem 60: 7725-7744 (2017) Article DOI: 10.1021/acs.jmedchem.7b00515 BindingDB Entry DOI: 10.7270/Q28W3GS9 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM5447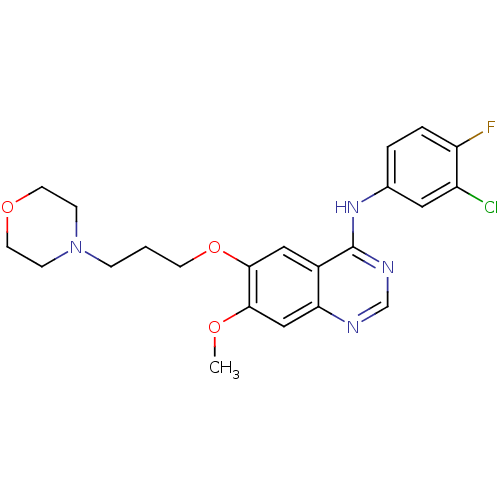 (CHEMBL939 | GEFITINIB | Iressa | N-(3-Chloro-4-flu...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB Article PubMed | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TU Dortmund University Curated by ChEMBL | Assay Description Inhibition of human recombinant GST-tagged EGFR L858R mutant expressed in baculovirus expression system preincubated for 30 mins followed by ATP and ... | J Med Chem 60: 7725-7744 (2017) Article DOI: 10.1021/acs.jmedchem.7b00515 BindingDB Entry DOI: 10.7270/Q28W3GS9 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50383274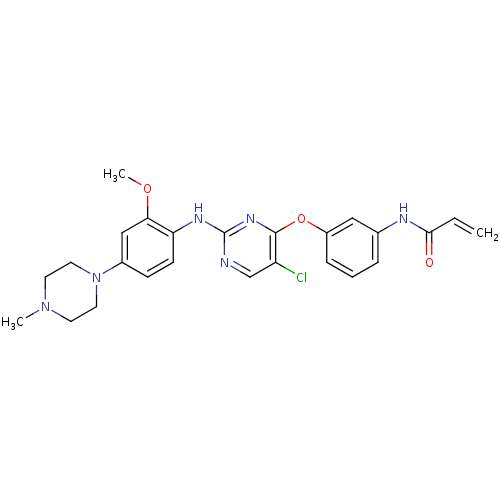 (CHEMBL1229592 | US10167264, WZ4002 | US9670213, WZ...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TU Dortmund University Curated by ChEMBL | Assay Description Inhibition of human recombinant GST-tagged EGFR L858R mutant expressed in baculovirus expression system preincubated for 30 mins followed by ATP and ... | J Med Chem 60: 7725-7744 (2017) Article DOI: 10.1021/acs.jmedchem.7b00515 BindingDB Entry DOI: 10.7270/Q28W3GS9 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50383274 (CHEMBL1229592 | US10167264, WZ4002 | US9670213, WZ...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TU Dortmund University Curated by ChEMBL | Assay Description Inhibition of wild type N-terminal GST-fused human EGFR cytoplasmic domain expressed in baculovirus expression system preincubated for 30 mins follow... | J Med Chem 60: 7725-7744 (2017) Article DOI: 10.1021/acs.jmedchem.7b00515 BindingDB Entry DOI: 10.7270/Q28W3GS9 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM5447 (CHEMBL939 | GEFITINIB | Iressa | N-(3-Chloro-4-flu...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB Article PubMed | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TU Dortmund University Curated by ChEMBL | Assay Description Inhibition of wild type N-terminal GST-fused human EGFR cytoplasmic domain expressed in baculovirus expression system preincubated for 30 mins follow... | J Med Chem 60: 7725-7744 (2017) Article DOI: 10.1021/acs.jmedchem.7b00515 BindingDB Entry DOI: 10.7270/Q28W3GS9 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50258974 (CHEMBL4090601) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TU Dortmund University Curated by ChEMBL | Assay Description Inhibition of wild type N-terminal GST-fused human EGFR cytoplasmic domain expressed in baculovirus expression system preincubated for 30 mins follow... | J Med Chem 60: 7725-7744 (2017) Article DOI: 10.1021/acs.jmedchem.7b00515 BindingDB Entry DOI: 10.7270/Q28W3GS9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Proto-oncogene tyrosine-protein kinase Src (Gallus gallus (Chicken)) | BDBM50437150 (CHEMBL2403820) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Technical University of Dortmund Curated by ChEMBL | Assay Description Inhibition of chicken wild-type cSRC (251 to 533)-mediated phosphorylation of biotinylated poly-Glu-Tyr expressed in Escherichia coli BL21(DE3) prein... | J Med Chem 56: 5757-72 (2014) Article DOI: 10.1021/jm4004076 BindingDB Entry DOI: 10.7270/Q2W37XPH | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Proto-oncogene tyrosine-protein kinase Src (Gallus gallus (Chicken)) | BDBM92348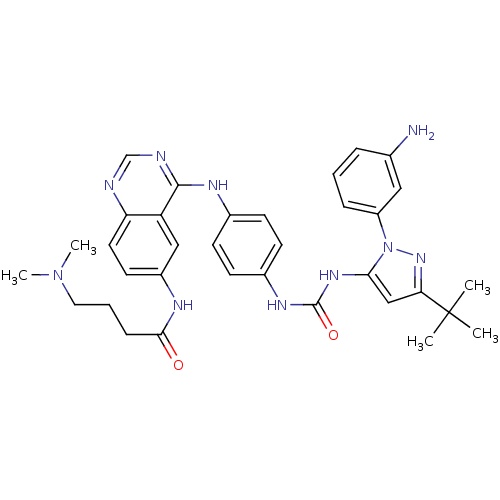 (1,4-Hybrid, 6) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Technical University of Dortmund Curated by ChEMBL | Assay Description Inhibition of chicken wild-type cSRC (251 to 533)-mediated phosphorylation of biotinylated poly-Glu-Tyr expressed in Escherichia coli BL21(DE3) prein... | J Med Chem 56: 5757-72 (2014) Article DOI: 10.1021/jm4004076 BindingDB Entry DOI: 10.7270/Q2W37XPH | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Proto-oncogene tyrosine-protein kinase Src (Gallus gallus (Chicken)) | BDBM92347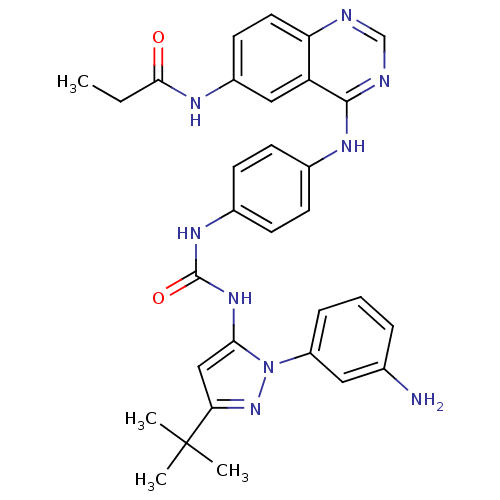 (1,4-Hybrid, 5) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Technical University of Dortmund Curated by ChEMBL | Assay Description Inhibition of chicken wild-type cSRC (251 to 533)-mediated phosphorylation of biotinylated poly-Glu-Tyr expressed in Escherichia coli BL21(DE3) prein... | J Med Chem 56: 5757-72 (2014) Article DOI: 10.1021/jm4004076 BindingDB Entry DOI: 10.7270/Q2W37XPH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Receptor tyrosine-protein kinase erbB-2 (Homo sapiens (Human)) | BDBM50547276 (CHEMBL4786257) | PDB MMDB UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of C-terminal His6-tagged HER2 (703 to 1029 residues) (unknown origin) expressed in Sf9 insect cells using TK as substrate preincubated fo... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50547249 (CHEMBL4757145) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of human N-terminal GST-tagged EGFR L858R/T790M mutant (669 to 1210 residues) expressed in baculovirus infected Sf9 insect cells using TK ... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00870 BindingDB Entry DOI: 10.7270/Q2VQ368T | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50258986 (CHEMBL4103201) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
TU Dortmund University Curated by ChEMBL | Assay Description Inhibition of human recombinant GST-tagged EGFR L858R mutant expressed in baculovirus expression system preincubated for 30 mins followed by ATP and ... | J Med Chem 60: 7725-7744 (2017) Article DOI: 10.1021/acs.jmedchem.7b00515 BindingDB Entry DOI: 10.7270/Q28W3GS9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 293 total ) | Next | Last >> |